Abstract
The extremely cold and infertile Antarctic is one of the harshest terrestrial ecosystems for the growth of vegetation, except for the grass species Deschampsia antarctica. We examined the main soil variables that determine the distribution of D. antarctica in King George Island by using Bayesian analysis of variance and regression methods. This study compared the density of D. antarctica between 2 sites; the density remained relatively stable at site 1, whereas it severely decreased in site 2 over a period of 3 years. Although site 2 showed better soil conditions for the growth of D. antarctica such as organic matter content, available phosphorus, NO3-N, and extractable cations, its poor drainage and low soil pH may affected the survival of D. antarctica by altering nutrition availability and inhibiting root respiration. Poisson analysis of covariance showed that the early melting of snow was also an important factor in the distribution of D. antarctica. The results also showed that seabirds and mammals might have greatly influenced the distribution of the grass species in King George Island by transferring nutrients from the sea onto land; thus, changing the chemical characteristics of the soil.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The terrestrial vegetation in Antarctica is largely dominated by bryophytes and lichens, with only two phanerogams occurring throughout Maritime Antarctica and not extending into continental Antarctica (Greene and Holtom 1971; Smith 1984). Deschampsia antarctica Desv. (Poaceae) is one of the two phanerogams and the only monocotyledonous species that is naturally adapted to the harsh environmental conditions of Antarctica (Alberdi et al. 2002; Smith 2003). Several surveys have documented that the distribution of D. antarctica expanded with rapid increases in temperature and lengthening of the growing seasons (Smith 1994; Day et al. 1999; Walther et al. 2002; Gerighausen et al. 2003; Robinson et al. 2003).
It has been suggested that environmental factors such as moisture availability, nutrients, temperature, salinity, and microtopography influence the distribution of Antarctic vegetation (Kappen 1985; Broady 1989; Inoue 1989; Kennedy 1993; Smith 1995; Leishman and Wild 2001). However, most of studies have focused on the bryophytes and lichens, with only a few studies relating to D. antarctica (Edwards 1972; Kim and Chung 2004; Vera 2011). Ice-free areas in Antarctica, known as polar deserts, are generally poor in nutrients. However, marine mammals and birds deposit large amounts of excrement, allowing fertilization of these terrestrial ecosystems (SmyKla et al. 2007). It is possible that these nutrients may play an important role in defining the soil chemical characters. These soil parameters can also determine the spatial distribution of D. antarctica, although information on these soil variables remains unknown.
This study analyzed the relationship among soil variables and D. antarctica distribution by using Bayesian statistical methods. The Bayesian methods have many applications in ecological studies. For example, relevant prior information can be easily incorporated into the analysis, and probabilistic predictions about the state of the world can be generated (McCarthy 2007). The application of Bayesian inference to ecological studies is increasing and has been extensively used to estimate species occurrences from logistically constrained samples or in response to expected environmental changes (Ellison 2004).
The main objectives of this study were as follows: (1) to ascertain the major soil variables that influence the distribution of D. antarctica and (2) to investigate factors that limit the growth and propagation of D. antarctica by using two selected study sites for the purpose of comparison.
Methods
Study area
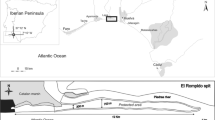
On King George Island, we conducted on-site surveys in February 2009, 2010, and 2011 at 3 study sites (site 1: 62° 14′ 11″ S, 58° 43′ 05″ W; site 2: 62° 14′ 28″ S, 58° 44′ 46″ W on Barton Peninsula; and site 3: 62° 12′ 11″ S, 58° 48′ 02″ W on Weaver Peninsula), where populations of D. antarctica were found (Fig. 1). Glaciers cover most of the surface of the island, but there is ice-free terrain in restricted areas along the shorelines. Soils on the peninsulas are subdivided into four suites according to bedrock type: granodiorite, basaltic andesite, lapilli tuff, and the Sejong formation (Lee et al. 2004). Permafrost was present 1 m below the surface (Jeong and Yoon 2001). On the ice-free terrain, the terrestrial vegetation includes 62 lichen species, 33 bryophyte species, and 2 phanerogam species (Kim et al. 2006; Kim et al. 2007). The mean temperature was 1.8 °C in January and −5.1 °C in July from 2000 to 2010, and the mean annual precipitation was 566 ± 216 mm (data obtained from the nearest meteorological station, King Sejong Station).
Data collection and soil analysis
The line intercept method was used to determine the density of D. antarctica: Along the shoreline at sites 1 and 2, 16 transects with 20 quadrats (1 m2) per transect were established. The number of living D. antarctica within each quadrat was monitored for 3 years by using identical transect lines.
To document vegetation-dependent differences in soil characteristics, each study site had four vegetation plots (100 m2), and each vegetation plot was dominated by one of the four predominant vegetation types, that is, bare ground, moss (Sanionia spp.), D. antarctica, or lichen (Usnea spp.). Soil samples were collected from five random quadrats (0.5 m × 0.5 m) within each vegetation plot (n = 60). Soil electric conductivity and pH (soil: distilled water, 1:5) were measured using an electric conductivity meter and a pH meter, respectively. Water content was determined by measuring the amount of weight lost after drying the samples in a 105 °C oven for 48 h. Organic matter content was calculated according to the weight lost after the samples were kept in a muffle furnace at 550 °C for 4 h (John 2004). NO3-N was extracted with 2 M KCl and measured using an automatic Kjeldahl protein/nitrogen analyzer (Kjeltec Auto 1,035/1,038 System; Tecator AB, Sweden). Total nitrogen content was determined using an element analyzer (EA1110, CE Instruments, UK) at the National Center for Inter-University Research Facilities, Seoul National University. Bray No. 1 solution (Bray and Kurtz 1945) was used to extract PO4–P for a colorimetric assay performed using the ascorbic acid reduction method (Solorzano 1969). The minerals Mg, K, and Na were extracted with 1 N ammonium acetate and measured using an atomic absorption spectrometer (AA240FS, Varian, USA). Soil texture was determined using the hydrometer analysis method.
To describe the relationship between the abundance of D. antarctica and soil variables, we counted the seedlings and tufts of D. antarctica within 1 m2 quadrats separately, and soil samples were taken from three quadrat corners were mixed thoroughly to produce one sample for each quadrat. At site 1 (1 ha), 57 random quadrats were assessed in January 2010, and the next year, we selected 80 random quadrats from site 1 (2 ha). To reveal the effect of snow, we measured distance from the frozen snow to our random quadrats and assumed that closer quadrats from the snow have been severely influenced by the snow. We divided the site 1 into two groups in January 2010: late snow melting areas were <20 m from frozen snow (1), and early snow melting areas were more than 20 m from frozen snow (0). We took pictures to analyze moss cover and calculated percentage moss cover per quadrat (1 m2) by using image analysis software (ImageJ 1.34 s; Wayne Rasban, National Institutes of Health, USA). The physicochemical properties of the soil were analyzed using the same methods previously described.
Statistical analysis
Bayesian Poisson t test was applied to compare the density of D. antarctica between site 1 and site 2 over 3 years. Site 3 was excluded from the comparison because of the low abundance of the grass to conduct analysis (<20 turfs). Density values were calculated using the average density of 20 quadrats (1 m2) at each transect. The Poisson distribution is used for non-negative, discrete responses, making it more suitable than normal distributions to describe counts (Kéry 2010). The following model was used for D. antarctica count data:
where D. antarctica count (C i ) in transect (i) is distributed as a Poisson random variable with mean \( E(C_{i} ) = \lambda \), and α and β are unknown constants.
A multilevel two-way analysis of variance (ANOVA) model was applied to soil chemical data in order to compare the soil variable between sites and vegetation types. This model can be expressed as follows:
where Y is the soil variable, \( \beta 1_{i} \) is the vegetation plot effect, \( \beta 2_{i} \) is the site effect, and \( \beta 3_{ij} \) is the interaction effect. The residual term \( \varepsilon_{ijk} \) is assumed to have a normal distribution with mean 0 and a constant variance, where k = 1, 2 is the index of individual observations within each site-vegetation plot (Qian and Shen 2007).
Binomial ANCOVA (logistic regression) was used to analyze the distribution of D. antarctica seedlings by adding discrete and continuous soil covariates. We recorded the presence or absence of seedlings and sampled soil at the random 57 quadrats in site 1 (1 ha). It is assumed that the occurrence of the seedlings is determined randomly by using a Bernoulli distribution. This model can be expressed as follows:
where i is the index of the quadrat, C i is the presence or absence of D. antarctica in the quadrats, the link function is the logit, as is customary for a binomial distribution (Kéry 2010). Explanatory variables have been centered by subtracting the mean values and standardized by dividing the standard deviation to improve the efficiency of the Markov chain Monte Carlo (MCMC) algorithm (Elith 2002).
Poisson ANCOVA was used to assess the effects of soil variables on the distribution of D. antarctica tufts by using informative priors, which were obtained from previous research on Poisson ANCOVA in 2010.
where i is the index of the quadrat and C i is the number of D. antarctica tufts in the 1 m2 quadrats. The log-transformation of λ i is assumed to be a linear function. Explanatory variables were centered by subtracting the mean values. The error term ∈ i is used to account for unstructured over-dispersion (OD) and assumed to have a normal distribution with mean 0 and a constant variance.
Parameter estimation was based on a sample of 5,000 values after a burn in of 2,500 iterations from three chains. Convergence was assessed using the CODA package (Plummer et al. 2006) for model selection using R. Model selection was performed using the deviance information criterion (DIC), where a lower DIC suggests a better trade-off between model fit and parsimony. We used WinBUGS version 1.4.3 (Spiegelhalter et al. 2003) via R2WinBUGS package (Sturtz et al. 2005) for R, which implements MCMC methods with a Gibbs sampler.
Principal component analysis (PCA) was applied to correlations in soil chemical data to identify major gradients in site-to-site variation in soil chemistry (Culver and Beattie 1983).
Results
Density of D. antarctica
We estimated the posterior mean of D. antarctica density and compared their densities between sites 1 and 2 by using the Bayesian Poisson t test. As shown in Fig. 2, the estimated posterior mean was lower than the field data mean, but the difference was small; the change in mean values was similar over the course of time. The density of D. antarctica was 4.8-fold lower in site 2 than in site 1 in 2009. In addition, over the course of 3 years, the density decreased much more dramatically (85 % based on the mean values) in site 2 than in site 1. These results suggest that site 2 might have crucial limiting factors that affect the growth and propagation of D. antarctica.
Posterior distribution of mean D. antarctica density in site 1 (top) and site 2 (bottom) for 3 years. Solid red lines indicate the estimated posterior mean, and dashed red line is the field data mean within the 1 m2 quadrats (n = 160). Numerals are posterior mean values and the 95 % credible intervals in brackets. X-axis means that expected D. antarctica count within 1 m2 quadrat (color figure online)
Soil variables
We used PCA to condense the information from the 12 soil variables into a smaller set of new composite dimensions. The PCA result is presented in a biplot (Fig. 3). Two principle components explained 68.5 % of the total observed variation. Component 1 with an eigenvalue of 5.39 had a high negative load on electric conductivity (−0.399), organic matter (−0.365), water content (−0.361), and a positive load on sand content (0.353). The D. antarctica plot at site 2 (S2_D) stands out with the highest electric conductivity (EC), organic matter (OM), and water content, followed by S2_M, and S2_B. Component 2 with an eigenvalue of 2.85 had a high negative load on Na+ (−0.558) and K+ (−0.482), and these variables showed a weak relationship with soil texture. The biplot also shows that the plots at site 2 have lower pH and sand content than the other plots.
The multilevel two-way ANOVA model showed that the differences in soil properties depend on vegetation type (Veg effect) and the site (Site effect). Water content in moss plots was higher than the plot average, D. antarctica plots were close to average, but bare ground and lichen plots were lower than average. Site effects were more obvious than vegetation effects. Water content in site 2 was higher than average; in addition, its 95 % posterior credible interval was 0–10.5 (Fig. 4), suggesting that water content in site 2 may be higher than that in the other sites. Sand content of the soils did not differ greatly in relation to the vegetation plot type, but site 2 had significantly lower sand content than site 1. We could predict that D. antarctica may require good drainage condition for root respiration.
Estimated vegetation effect (left) and site effect (right) for water content (top) and sand content (bottom). Diamonds are the estimated posterior mean values, short thick lines are the 50 % credible intervals, and the long thin lines are the 95 % credible intervals. Those intervals not overlapping the zero line are considered significantly different from the mean values
As shown in Fig. 5, available phosphorus was highest in the D. antarctica plots; the occurrence of D. antarctica is positively associated with available phosphorus, and site 2 showed significantly higher available phosphorus than the other sites. When we compared the pH among the vegetation plots, we observed that pH was lowest in the lichen plot; however, there were no meaningful differences between the pH values in the other plots. pH was significantly lower in site 2 than in the other sites.
Estimated vegetation effect (left) and site effect (right) for available phosphorus (AP) (top) and pH (bottom). Diamonds are the estimated posterior mean values, short thick lines are 50 % credible intervals, and long thin lines are 95 % credible intervals. Those intervals not overlapping the zero line are considered significantly different from the mean values
Associations between distribution of D. antarctica and soil variables
The presence or absence of the seedlings of D. antarctica was applied to binomial ANCOVA by adding the categorical explanatory variable (Na) and the continuous explanatory variables [moss cover (moss), clay, water content (WC), organic matter (OM), and pH] to the linear predictor of a binomial generalized linear model (Fig. 6). We divided the Na concentration into three levels to estimate the effects of sea spray, and other continuous variables were assumed to have a normal distribution. The occurrence of seedlings of D. antarctica was positively associated with moss cover, organic matter, available phosphorus, and Na. The moss variable had the largest parameter mean value (mean, 2.92; CI, 0.84–6.07). Predicted positive association with Na showed that this grass was extremely tolerant of salinity. The 95 % credible interval of pH was −0.79 to 1.76; this suggests that the relationship between pH and the occurrence of seedlings might be weaker than other variables having positive 95 % credible interval. Binomial ANCOVA results also showed that the negative relationship between the occurrence of seedlings of D. antarctica and water content and clay.
Predictors of the occurrence of D. antarctica seedlings. Circles are the estimated posterior mean values, short thick lines are 50 % credible intervals, and long thin lines are 95 % credible intervals. Those intervals not overlapping the zero line are considered significantly different from zero. Parameter values (X-axis) are derived from the posterior distribution for the parameters
The Poisson distribution was used to analyze the relationship between the number of D. antarctica tufts and the continuous explanatory variables (moss, clay, water content, organic matter, and available phosphorus), except for snow (Fig. 7). To find our snow effect, random quadrats were subdivided into two groups based on the distance from the frozen snow in January 2010. The density of the tufts was positively associated with moss cover, organic matter, and available phosphorus. There was strong evidence for a relationship between moss and tuft density, but the predicted positive relationship with available phosphorus was relatively weaker than that with the other variables. Tuft density was negatively associated with clay, water content, and snow. The predicted negative relationship with snow and water content was significant, and the snow variable showed the largest absolute posterior mean value (mean, −2.54; CI, −3.58 to −1.55); we found fewer tufts near snow at the study site 1.
Predictors of the number of D. antarctica tufts. Circles are the estimated posterior mean values, short thick lines are the 50 % credible intervals, and the long thin lines are the 95 % credible intervals. Those intervals not overlapping the zero line are considered significantly different from zero. Parameter values (X-axis) are derived from the posterior distribution for the parameters
Discussion
It is likely that global warming would facilitate the expansion of the existing populations of D. antarctica (Walther et al. 2002; Gerighausen et al. 2003; Robinson et al. 2003; Kim et al. 2007; Vera 2011). However, the results of this study show that the density of D. antarctica decreased severely at site 2 over 3 years (Fig. 2), suggesting that site 2 might have critical factors that limit the growth of D. antarctica. A number of ecological variables might play a role in determining the survival of vegetation in maritime Antarctica, including low temperatures, high UV-B radiation, and high levels of salinity (Alberdi et al. 2002; Smith 2003). We focused our attention on the distribution of D. antarctica and its relationship with soil characteristics. Several soil variables were shown to vary between sites 1 and 2, namely soil nutrients, soil organic matter, drainage, and pH.
The D. antarctica plots of site 2 were shown to have higher soil nutrient levels for NO3-N, available phosphorus, and available cations, as compared to site 1. Comparison of soil nutrient levels between vegetation-type plots by using multilevel Bayesian 2-way ANOVA showed that organic matter, NO3-N, and available phosphorus were highest in the D. antarctica plots, indicating the preference of this plant for fertile soil. This suggestion is also confirmed by the seedling occurrence and the tuft density results obtained using Bayesian binomial ANCOVA and Bayesian Poisson ANCOVA, respectively. Several studies have reported high abundances of tufts near bird nests in terms of the seed dispersal by birds (Edwards 1972; Kim et al. 2007; Parnikoza et al. 2007). These finding were also supported by supplement of soil nutrient through bird excrements. Most available phosphorus and NO3-N would be supplied by seabird or mammal excrement, and these nutrients enhance plant vitality in nutrient-poor Antarctic soils (Smith 1978). Smykla et al. (2007) had described zonation patterns in vegetation influenced by penguin rookeries on King George Island. Data from the study showed that D. antarctica has a wide ecological range, and its abundance decreased with low nutrient availability, especially in sites distant to the rookeries. Tatur et al. (1997) also found that nutrient-rich soils at abandoned rookeries served as optimum habitats for grass growth.
The distribution of seedlings and tufts showed a strong positive association with organic matter. It is well-known that the abundance of organic matter in soil affects plant production and plays a key role in the control of soil fertility (Jenny 1941; Tiessen et al. 1994). Kim et al. (2007) observed that most grass tufts were formed on ground covered by Sanionia spp. (carpet moss). Our results also show a statistically significant association between moss cover and D. antarctica distribution. This association may occur because of an overlap in environment preference, and moss covering could improve soil properties for D. antarctica by maintaining optimal water levels and supplying organic matter. Previous studies suggested multiple sources of potential organic matter in polar deserts, such as moss, lichen, terrestrial cyanobacteria and algae, microbial communities, and a legacy of paleolake sediments (Burkins et al. 2000; Hopkins et al. 2006). Although the origin of organic matter in Antarctic soil is complex, Hopkins et al. (2009) suggested that moss could serve as one of the principal sources of organic matter at productive sites. On the basis of the high moss cover and debris at study sites, this study suggests that the organic matter at the examined site was mainly derived from bryophytes.
Drainage is also an important environmental feature that influences the distribution of D. antarctica, as mentioned by Edwards (1972) and Kim and Chung (2004) in their field surveys. Soil drainage conditions, which are determined by water and sand clay content, greatly affect root respiration, pH, redox potential, and mineral transformations in the soil (Levine et al. 1994). For another, we found low soil pH in site 2; this could be unfavorable for the survival of D. antarctica because it can alter soil chemical characteristics such as phosphorus availability, cation availability, and the presence of heavy metals, which may be toxic to plants (U.S. Department of Agriculture 1983). Increased soil acidity might be attributable to seabird excretions (Hogg and Morton 1983; Wait et al. 2005); a resting pond of skuas was detected near site 2 (the distance <100 m). Although pH may vary in response to other factors such as soil type and presence of sea spray (Ellis 2005), it is possible that the poor drainage conditions in site 2 may have facilitated the accumulation of seabird droppings, resulting in the acidification of the soil by uric acid and the mineralization of the seabird-added organic matter (Garcia et al. 2002).
Early snow melting in the growing season has been identified as an important factor that affects the distribution of D. antarctica. Edwards (1972) and Vera (2011) also observed that D. antarctica commonly thrives in snow-free sites in the early austral summer. Early snow melting is more important than high vulnerability to cold periods during the spring and autumn, which are devoid of snow cover, whereas late snow cover in autumn may facilitate seed formation and landing on soil.
In summary, the present study showed the preference of D. antarctica for fertile soil and that poor drainage, low pH, and snow accumulation during the growing season could be critical factors for its survival in Maritime Antarctica. Furthermore, seabirds and mammals are important determinants of the distribution of the plants on King George Island on the basis of their role in nutrient transfer from the sea, thus changing the chemical characteristics of the soil. More extensive spatiotemporal studies are necessary to determine the impact of other factors on the distribution of D. antarctica.
References
Alberdi M, Bravo LA, Gutierrez A, Gidekel M, Corcuera LJ (2002) Ecophysiology of Antarctic vascular plants. Physiol Plant 115:479–486
Bray RH, Kurtz L (1945) Determination of total, organic, and available forms of phosphorus in soils. Soil Sci 59:39–45
Broady P (1989) Broadscale patterns in the distribution of aquatic and terrestrial vegetation at three ice-free regions on Ross Island, Antarctica. Hydrobiologia 172:77–95
Burkins MB, Virginia RA, Chamberlain CP, Wall DH (2000) Origin and distribution of soil organic matter in Taylor Valley, Antarctica. Ecology 81:2377–2391
Culver DC, Beattie AJ (1983) Effects of ant mounds on soil chemistry and vegetation patterns in a Colorado montane meadow. Ecology 64:485–492
Day T, Ruhland C, Grobe C, Xiong F (1999) Growth and reproduction of Antarctic vascular plants in response to warming and UV radiation reductions in the field. Oecologia 119:24–35
Edwards J (1972) Studies in Colobanthus quitensis (Kunth) Bartl. and Deschampsia antarctica Desv.: V. Distribution, ecology and vegetative performance on Signy Island. Br Antarct Surv Bull 28:11–28
Elith RJ (2002) Predicting the distribution of plants. Dissertation. University of Melbourne, Parkville
Ellis JC (2005) Marine birds on land: a review of plant biomass, species richness, and community composition in seabird colonies. Plant Ecol 181:227–241
Ellison AM (2004) Bayesian inference in ecology. Ecol Lett 7:509–520
Garcia LV, Maranon T, Ojeda F, Clemente L, Redondo R (2002) Seagull influence on soil properties, chenopod shrub distribution, and leaf nutrient status in semi-arid Mediterranean Islands. Oikos 98:75–86
Gerighausen U, Bräutigam K, Mustafa O, Peter HU (2003) Expansion of vascular plants on an Antarctic island-a consequence of climate change?. Antarctic Biology in a Global Context Blackhuys Publishers, Leiden, pp 79–83
Greene DM, Holtom A (1971) Studies in Colobanthus quitensis (Kunth) Bartl. and Deschampsia antarctica Desv. III. Distribution, habitats and performance in the Antarctic botanical zone. Br Antarct Sur Bull 26:1–29
Hogg EH, Morton JK (1983) The effects of nesting gulls on the vegetation and soil of islands in the Great Lakes. Can J Bot 61:3240–3254
Hopkins DW, Sparrow AD, Novis PM, Gregorich EG, Elberling B, Greenfield LG (2006) Controls on the distribution of productivity and organic resources in Antarctic dry valley soils. Proc R Soc B 273:2687–2695
Hopkins DW, Sparrow AD, Gregorich EG, Greenfield LG, Novis P, Fraser F, Scrimgeour C, Dennis PG, Meier-Augenstein W, Elberling B (2009) Isotopic evidence for the provenance and turnover of organic carbon in Antarctic dry valley soils. Environ Microbiol 11:597–608
Inoue M (1989) Factors influencing the existence of lichens in the ice-free areas near Syowa station, East Antarctica. Proc NIPR Symp Polar Biol 4:167–180
Jenny H (1941) Factors of soil formation. McGraw-Hill, New York
Jeong G, Yoon H (2001) The origin of clay minerals in soils of King George Island, South Shetland Islands, West Antarctica, and its implications for the clay-mineral compositions of marine sediments. J Sediment Res 71:833–842
John B (2004) A comparison of two methods for estimating the organic matter content of sediments. J Paleolimnol 31:125–127
Kappen L (1985) Vegetation and ecology of ice-free areas of northern Victoria Land, Antarctica. 2. Ecological conditions in typical microhabitats of lichens at Birthday Ridge. Polar Bio 4:227–236
Kennedy AD (1993) Water as a limiting factor in the Antarctic terrestrial environment: a biogeographical synthesis. Arch Antarct Alp Res 25:308–315
Kéry M (2010) Introduction to WinBUGS for ecologists: bayesian approach to regression, ANOVA, mixed models and related analyses. Academic Press, Amsterdam, pp 167–236
Kim JH, Chung H (2004) Distribution pattern of Deschampsia antarctica, a flowering plant newly colonized around King Sejong Station in Antarctica. Ocean Polar Res 26:23–32
Kim JH, Ahn IY, Hong SG, Andreev M, Lim KM, Oh MJ, Koh YJ, Hur JS (2006) Lichen flora around the Korean Antarctic scientific station, King George Island, Antarctic. J Microbiol 44:480–491
Kim JH, Ahn IY, Lee KS, Chung H, Choi HG (2007) Vegetation of Barton Peninsula in the neighbourhood of King Sejong Station (King George Island, maritime Antarctic). Polar Biol 30:903–916
Lee YI, Lim HS, Yoon HI (2004) Geochemistry of soils of King George Island, South Shetland Islands, West Antarctica: implications for pedogenesis in cold polar regions. Geochim Cosmochim Acta 68:4319–4333
Leishman MR, Wild C (2001) Vegetation abundance and diversity in relation to soil nutrients and soil water content in Vestfold Hills, East Antarctica. Antarct Sci 13(2):126–134
Levine E, Knox R, Lawrence W (1994) Relationships between soil properties and vegetation at the northern experimental forest, Howland, Maine. Remote Sens Environ 47:231–241
McCarthy MA (2007) Bayesian methods for ecology. Cambridge University, New York, pp 119–157
Parnikoza IY, Maidanuk D, Kozeretska I (2007) Are Deschampsia antarctica Desv. and Colobanthus quitensis (Kunth) Bartl. migratory relicts? Cytol Genet 41:226–229
Plummer M, Best N, Cowles K, Vines K (2006) CODA: convergence diagnosis and output analysis for MCMC. R News 6:7–11
Qian SS, Shen Z (2007) Ecological applications of multilevel analysis of variance. Ecology 88:2489–2495
Robinson SA, Wasley J, Tobin AK (2003) Living on the edge–plants and global change in continental and maritime Antarctica. Glob Change Biol 9:1681–1717
Smith V (1978) Animal-plant-soil nutrient relationships on Marion Island (Subantarctic). Oecologia 32:239–253
Smith RIL (1984) Terrestrial plant biology of the sub-Antarctic and Antarctic. In: Laws RM (ed) Antarctic ecology, vol 1. Academic Press, London, pp 61–162
Smith RIL (1994) Vascular plants as bioindicators of regional warming in Antarctica. Oecologia 99:322–328
Smith RIL (1995) Colonization by lichens and the development of lichen-dominated communities in the maritime Antarctic. Lichenologist 27:473–483
Smith RIL (2003) The enigma of Colobanthus quitensis and Deschampsia antarctica in Antarctica. Antarctic biology in a global context. Backhuys Publishers, Leiden, pp 234–239
Smykla J, Wolek J, Barcikowski A (2007) Zonation of vegetation related to penguin rookeries on King George Island, Maritime Antarctic. Arch Antarct Alp Res 39:143–151
Solorzano L (1969) Determination of ammonia in natural waters by the phenolhypochlorite method. Limnol Oceanogr 14:799–801
Spiegelhalter DJ, Thomas A, Best NG, Lunn D (2003) WinBUGS Version 1.4. Imperial College & MRC Biostatistics Unit, UK
Sturtz S, Ligges U, Gelman A (2005) R2WinBUGS: a package for running WinBUGS from R. J Stat Softw 12:1–16
Tatur A, Myrcha A, Nieodzisz J (1997) Formation of abandoned penguin rookery ecosystems in the Maritime Antarctic. Polar Biol 17:405–417
Tiessen H, Cuevas E, Chacon P (1994) The role of soil organic matter in sustaining soil fertility. Nature 371:783–785
U.S. Department of Agriculture, Soil Conservation Service (1983) National soils handbook. U.S. Gov Printing Office, Washington, DC
Vera ML (2011) Colonization and demographic structure of Deschampsia antarctica and Colobanthus quitensis along an altitudinal gradient on Livingston Island, South Shetland Islands, Antarctica. Polar Res 30:1–10
Wait DA, Aubrey DP, Anderson WB (2005) Seabird guano influences on desert islands: oil chemistry and herbaceous species richness and productivity. J Arid Environ 60:681–695
Walther GR, Post E, Convey P, Menzel A, Parmesan C, Beebee TJC, Fromentin JM, Hoegh-Guldberg O, Bairlein F (2002) Ecological responses to recent climate change. Nature 416:389–395
Acknowledgments
We are very grateful to all the staff of the King Sejong Station for their cooperation and hospitality. We thank Jeong-Hoon Kim for his valuable comments about the distribution of D. antarctica. We are grateful to Namyi Chae and Bang Yong Lee for providing the weather information. Finally, we thank Dr. Dieter Piepenburg and anonymous referees for their helpful comments. This research was supported by Korea Polar Research Institute (Grant No. PE10040). The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Park, J.S., Ahn, IY. & Lee, E.J. Influence of soil properties on the distribution of Deschampsia antarctica on King George Island, Maritime Antarctica. Polar Biol 35, 1703–1711 (2012). https://doi.org/10.1007/s00300-012-1213-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00300-012-1213-4