Abstract
Epiphytic diatom communities on macroalgae from Iceland coastal waters were investigated during July 2005. Ten species of seaweeds have been collected belonging to brown, red and green algae. The analysis of epiphytic diatom community was carried out under scanning electron microscopy. The epiphytic diatom abundances varied from 7 ± 5 to 7524 ± 3491 cells mm−2. Erect growth forms were the most abundant, representing on average 50% of the total diatoms (Achnanthes cf. brevipes var. parvula, Tabularia investiens, T. fasciculata, Hyalosira cf. delicatula, Gomphoseptatum aestuarii, Pseudogomphonema plinskii), followed by adnate (29%) (Cocconeis stauroneiformis, C. scutellum) and motile forms (21%) (Nitzschia cf. amphibia and Navicula perminuta). Highly branched seaweeds with articulated thallus surface, offering a number of microenvironments to be occupied by the epiphytes, showed a high level of colonization, mainly due to erect and motile diatoms. Flat thalli with smooth surface allowed for the growth of mainly erect diatoms.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Benthic microalgae colonize a wide variety of substrata in shallow marine environments, ranging from soft sediments (epipelic and endopelic communities), sand grains (epipsammic), rock and artificial surfaces (epilithic and endolithic) to marine plants (epiphytic) and animals (epizoic) (Round et al. 1990). Living substrata represent a highly suitable habitat for the settlement and growth of a number of microorganisms, particularly in aquatic environments, due to the variety of interactions that can establish between the epibionts and their animal or plant hosts (Di Camillo et al. 2005; Pinckney and Micheli 1998; Romagnoli et al. 2007; Wahl 1989).
Most studies dealt with the colonization and distribution of microscopic epiphytes on seagrass hosts in marine and brackish environments (Frankovich et al. 2006; Moncreiff and Sullivan 2001; Novak 1984; Pinckney and Micheli 1998; Ruesink 1998; Wear et al. 1999), while there are fewer studies focusing on epiphytic microalgae on seaweeds (Al-Handal and Wulff 2008; Karsten et al. 2006; Snoeijs 1994; Steinberg and De Nys 2002; Sutherland 2008; Worm and Sommer 2000). Regardless of the type of substrata they are associated with, benthic diatoms are represented by different growth forms, which are composed of motile forms consisting of biraphid taxa freely moving over soft sediments or hard substrata, adnate forms comprising biraphid and monoraphid taxa living closely associated with the substratum and rarely colonial (e.g., Amphora Ehrenberg, Cocconeis Ehrenberg, Epithemia Kützing, Rhopalodia O. Müller), erect forms encompassing cells attached to substrata by mucilaginous stalks (e.g., Grammatophora Ehrenberg, Licmophora Agardh) or pads (e.g., Cyclophora Castracane, Synedra Ehrenberg) and tube-dwelling colonies consisting mostly of motile naviculoid or nitzschioid taxa living in mucilaginous tubes (Hudon and Legendre 1987; Romagnoli et al. 2007; Tuji 2000).
The distribution of epiphytic microalgae on host plants and, more specifically, the composition of benthic diatom communities in terms of cell abundance and relative importance of the growth forms, is affected by a number of environmental variables such as light availability, hydrodynamic regime, salinity and nutrients (Coleman and Burkholder 1994; Frankovich et al. 2006; Hillebrand and Sommer 2000; Marks and Power 2001; Snoeijs 1994, 1995; Wear et al. 1999), and biological processes such as grazing (Hillebrand et al. 2000; Nelson 1997; Worm and Sommer 2000), adhesive strength of diatoms (Tanaka 1986), chemical interactions with the host (Amsler et al. 2005; Gross et al. 2003; Steinberg and de Nys 2002) and physiological responses (Ruesink 1998). The role of the macroalgal thallus morphology on the composition of epiphytic diatom communities was studied by Thomas and Jiang (1986) from the subtidal and intertidal zones off the Australian Davis Station in Antarctica. They reported that foliose thalli hosted mainly epiphytic diatoms with a strong attachment mode, i.e. adnate forms, while filamentous thalli supported a higher number of taxa, increasing in highly branched thalli. Snoeijs (1994, 1995) reported from the Baltic Sea that, although the microepiphytic community was more affected by season and salinity gradient than by host preference, the plant hosts somehow affected the epiphytic taxonomic composition because they supported different abundances of epiphytes. In Antarctic waters, Al-Handal and Wulff (2008) suggested the existence of a host effect because a different epiphytic diatom composition occurred on seaweed species from the same area and depth and Sutherland (2008) reported that the epiphytic diatom communities differed in terms of dominant taxa on frondose and coralline red algae from the same area.
Seaweeds represent important component of benthic communities of Icelandic coastal waters. The macroalgal flora around Iceland has been extensively investigated over the last 15 years (Espinosa and Guerra-Garcia 2005; Gunnarsson 2000; Gunnarsson and Ingólfsson 1995; Munda 1994, 1999; Tittley et al. 2005). However, at present no data exist about epiphytic diatom communities; the knowledge of the benthic diatoms from Iceland is scarce and limited to microphytobenthos associated to soft sediments (Jiang et al. 2001). In this study, we report for the first time the distribution of epiphytic diatoms which are associated with some common intertidal seaweeds from Iceland, considering the role of the host thallus morphology in structuring the epiphytic diatom communities.
Materials and methods
Study area and sampling
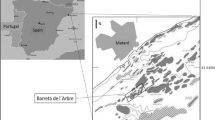
Sampling was carried out at one intertidal site in Sandgerdi (64°02′26″N; 22°42′49″W), in the Reykjanes Peninsula on the west coast of Iceland, on 24 July 2005 (Fig. 1). Study site is characterized by a gentle slope with a tidal range of 2.5–3.8 m at spring tides and water temperature about 10.0–11.5°C (Ingólfsson 1996).
Ten species of seaweeds were collected belonging to brown algae: Desmarestia aculeata (Linnaeus) Lamouroux (Desmarestiales), Fucus evanescens C. Agardh, F. vesiculosus Linnaeus (Fucales), Alaria esculenta (Linnaeus) Greville, Laminaria hyperborea (Gunnerus) Foslie, L. saccharina (Linnaeus) Lamouroux (Laminariales), red algae: Porphyra umbilicalis Kützing (Bangiales), Delesseria sanguinea (Hudson) Lamouroux, Polysiphonia lanosa (Linnaeus) Tandy (Ceramiales) and green algae: Ulva lactuca Linnaeus (Ulvales). Seaweeds were collected by hand during low tide, at a depth from the bottom of about 1–1.5 m. Small surfaces (1 cm2) of seaweed thalli were cut in triplicates, stored in Falcon tubes with 0.45 μm pore size filtered seawater and preserved with 4% formaldehyde and 1% glutaraldehyde. For Fucus evanescens, F. vesiculosus and Laminaria saccharina, the sampling was carried out separately from the apical (frond) and basal (stipe) parts.
Sample treatment and analysis of epiphytes
Seaweed samples were dehydrated individually in a serial alcohol gradient (10, 30, 50, 70, 80, 90, 95 and 100%) and processed through critical point drying (Polaron CPD7501) in order to preserve intact the diatom cells and their mode of attachment to the host plant. Dried seaweed samples were fixed on aluminium stubs with double adhesive carbon disc, coated with Au–Pd (Polaron SC7640), and examined in scanning electron microscopy (SEM, Philips 515) operating at 25 kV.
The enumeration of epiphytic diatoms were performed with the SEM by analysing between 80 and 400 visual fields, each having an area of 2976 μm2, at 1500 times magnification, depending on the number of the epiphytic diatom cells on the host plant. Abundances were expressed as the number of cells per mm2 of thallus surface and the relative abundance as their proportion to the total number of diatoms. The storage water in which samples were preserved has been examined for the identification and counting of detached taxa.
Epiphytic diatoms were subdivided into the following growth forms: erect for diatoms attached to seaweeds through mucus pads or peduncles, adnate for diatoms firmly attached to the substratum through their valve face, and motile for biraphid diatoms freely moving on the substratum.
The identification of diatoms to the lowest possible taxonomic rank was made possible through an oxidation of the organic component of their frustules. Epiphytic diatoms were scraped from seaweed surface and cleaned following the von Stosch’s method in Hasle and Syvertsen (1997). Few drops of cleaned diatom material were poured on a 0.2 μm pore size Nucleopore polycarbonate filter fixed to an aluminium stub with double adhesive carbon disc, left to air dry and coated with Au–Pd before examination in SEM. The identification of diatoms was made following Bérard-Therriault et al. (1986), Compère (1986), De Stefano and Romero (2005), De Stefano et al. (2000, 2008), Hendey (1964), Hustedt (1931–1959, 1961–1966), Kooistra et al. (2008), Poulin et al. (1984a, b), Snoeijs (1993), Witkowski et al. (2000).
Statistical analysis
Macroalgal thalli were subdivided in three groups defined on the basis of both thallus morphology and surface characteristics as follows: highly branched thalli with articulated surface (Fucus evanescens, F. vesiculosus, Polysiphonia lanosa, hereafter HBT), poorly branched thalli with flat surface (Alaria esculenta, Laminaria hyperborea, L. saccharina, hereafter PBT), sheet-like thalli with smooth surface (Delesseria sanguinea, Porphyra umbilicalis, Ulva lactuca, hereafter SST). Desmarestia aculeata characterized by a leathery thallus with cylindrical branches has not been included in any of the above three morphotypes.
The differences in the epiphytic diatom community structure between the 10 investigated seaweeds were tested by a distance-based permutational multivariate analysis of variance, PERMANOVA (Anderson 2001; McArdle and Anderson 2001). The analysis is based on Euclidean distances of not normalized data (Anderson and ter Braak 2003) and was run using FORTRAN-written PERMANOVA.exe program (Anderson 2005).
Differences in the abundance of the three growth forms of diatoms between (i) macroalgal species and (ii) between apical and basal parts of the same species were assessed through a one-way analysis of variance (ANOVA). Species and the thallus body parts (frond and stipe) were treated as fixed factors with ten and two levels, respectively. Differences between seaweed morphotypes in the abundance of the three growth forms of epiphytic diatoms were assessed through a two-way ANOVA, with morphotype (3 levels) and species (9 levels) as fixed and orthogonal factors. The homogeneity of variance was tested a priori through a Cochrans’ test and, when necessary, the data were log transformed. When significant differences were observed, a post hoc Student-Newman–Kuels (SNK) test was also performed. All ANOVA and SNK tests were carried out using the GMAV software (University of Sydney).
Results
Epiphytic diatom communities
Diatoms represented the dominant group of epiphytes on the investigated seaweeds, with some occasional occurrences of metaphyton and filamentous cyanobacteria, which were not included in the community analysis. All epiphytic diatom taxa observed on the surface thallus of the 10 species of seaweeds are reported in Table 1. Diatom abundance varied from 7 ± 5 cells mm−2 on Laminaria saccharina to the maximum value of 7524 ± 3491 cells mm−2 on Fucus vesiculosus (Fig. 2). The highest cell numbers were usually measured on the damaged parts of the macroalgal thalli. Erect diatoms were generally the most abundant among the three diatom growth forms observed on the macroalgal thalli accounting, on average, to 50% of the total diatom abundance, followed by adnate (29%) and motile (21%) forms (Fig. 3).
Abundance (cells mm−2) of total epiphytic diatoms on the investigated seaweeds: F eva F. evanescens, F ves Fucus vesiculosus, P lan Polysiphonia lanosa, A esc Alaria esculenta, L hyp Laminaria hyperborea, L sac L. saccharina, D san Delesseria sanguinea, P umb Porphyra umbilicalis, U lac Ulva lactuca, D acu Desmarestia aculeata. Error bars represent SD
Cumulative relative abundance of different diatom growth forms on 10 intertidal seaweeds from Iceland. Adnate growth form: grey bar, erect growth form: black bar, motile growth form: ruled bar. F eva Fucus evanescens, F ves F .vesiculosus, P lan Polysiphonia lanosa, A esc Alaria esculenta, L hyp Laminaria hyperborea, L sac L. saccharina, D san Delesseria sanguinea, P umb Porphyra umbilicalis, U lac Ulva lactuca, D acu Desmarestia aculeata
The epiphytic diatom community on the highly branched thalli with articulated surface (Fucus evanescens, F. vesiculosus and Polysiphonia lanosa) is presented in Fig. 4a–g. In F. vesiculosus, motile diatoms, mainly represented by Nitzschia cf. amphibia (Fig. 5a), with Navicula perminuta (Fig. 5b) and other Navicula spp., were the most abundant growth forms observed in the apical part of the thallus, accounting for 57% of the total diatom community (Fig. 3). In both Fucus taxa, the erect growth forms were very frequent, with the most abundant diatoms, Achnanthes cf. brevipes var. parvula (Fig. 5c), Gomphoseptatum aestuarii (Fig. 5d), Hyalosira cf. delicatula (Fig. 5e) and Tabularia investiens (Fig. 5f), which represented 36% of the total diatom community in F. vesiculosus, whereas T. investiens alone accounted for 59% of the total diatom abundance in F. evanescens. The adnate diatom Cocconeis stauroneiformis (Fig. 5j) represented only 12 and 7% of the total diatom community in F. evanescens and F. vesiculosus, respectively. In P. lanosa, adnate diatoms were the most frequent growth form (48%) and were represented mainly by C. stauroneiformis, followed by erect forms (30%) with Hyalosira cf. delicatula, Tabularia fasciculata (Fig. 5g) and T. investiens, and motile forms (22%) with Navicula perminuta and Nitzschia cf. amphibia.
Seaweed morphotypes with epiphytic diatom growth forms in scanning electron microscopy. a–g Seaweeds with highly branched thalli with articulated surface: a Fucus vesiculosus showing a rich colonization of adnate (Cocconeis) and erect forms (Grammatophora), the latter attached to the substratum by the basal cell of the zig-zag colony; b covering of Cocconeis and Rhoicosphenia marina on F. vesiculosus, note that the erect R. marina is attached to the thallus by a mucus pad; c Fucus evanescens showing a rich colonization of Tabularia spp. attached to the substratum by mucilaginous pad; d F. evanescens covered by bacteria and erect diatoms; e portion of F. evanescens thallus without algal colonization, indicating a patchy distribution of the epiphytic community; f Polysiphonia lanosa with high covering of erect diatoms and filamentous cyanobacteria; g P. lanosa with Cocconeis and Tabularia settled in folds of branched thallus; h–k seaweeds with poorly branched thalli with flat surface: h Alaria esculenta with scattered Cocconeis and filamentous cyanobacteria; i–j Laminaria hyperborea with a low number of epiphytic diatoms in the most part of thallus surface (i), while a rich colonization of diatoms occurs in damaged parts (j); k L. saccharina showing a low colonization with scarce occurrence of diatoms. l–n Seaweeds with soft and sheet-like thalli with smooth surface: l Delesseria sanguinea with a low number of epiphytic diatoms; a mat consisting of bacterial and cyanobacterial filaments and fungal hyphae is visible; m–n scattered diatom coverage on Porphyra umbilicalis (m) and Ulva lactuca (n); o Desmarestia aculeata, part of branch with low number of epiphytic diatoms. Scale bars: b–d, g, i, l = 10 μm; f, j = 20 μm; a, h, k = 50 μm; m, n = 100 μm; e, o = 200 μm
Most abundant epiphytic diatoms on seaweeds from Iceland in scanning electron microscopy. a–b Motile forms: a Nitzschia cf. amphibia; b Navicula perminuta. c–i Erect forms: c Achnanthes cf. brevipes var. parvula; d Gomphoseptatum aestuarii attached to the substratum by a mucilaginous stalk; e Hyalosira cf. delicatula; f Tabularia investiens; g colony of Tabularia fasciculata attached to the substratum by a mucus pad; h Pseudogomphonema plinskii attached to seaweed surface by a mucus pad; i Rhoicosphenia marina in girdle view attached to the substratum by mucus pad. j–k Adnate forms: j Cocconeis stauroneiformis; k Cocconeis scutellum var. scutellum. Scale bars: a, b, e, h = 2 μm; c, d, f, i-k = 5 μm; g = 10 μm
Diatoms epiphytic on poorly branched thalli with flat surface (Alaria esculenta, Laminaria hyperborea and L. saccharina) are presented in Fig. 4h–k. In A. esculenta, the dominant adnate growth form was represented by Cocconeis scutellum (Fig. 5k), which accounted for >90% of the total epiphytic diatom community and mainly settling near the frond rib, whereas erect diatoms were only occasionally recorded and the motile growth form was completely absent (Fig. 3). In L. hyperborea, the epiphytic diatoms showed low abundances and a distinct patchy distribution (Fig. 2). The erect growth form was the most frequent (81%), mainly represented by Pseudogomphonema plinskii (Fig. 5h), a small Rhoicosphaenia sp. and R. marina (Fig. 5i), with adnate diatoms, Amphora sp. and Cocconeis scutellum, and few occasional motile forms. Laminaria saccharina showed the minimum diatom abundance, and the most abundant species was the adnate Cocconeis scutellum.
The epiphytic diatom community on sheet-like thalli with smooth surface (Delesseria sanguinea, Porphyra umbilicalis and Ulva lactuca) is presented in Fig. 4l–n. These seaweeds are usually characterized by low epiphytic diatom coverage. In D. sanguinea, the epiphytic diatom community was dominated by the adnate growth form, represented by Cocconeis species (62%), with several erect forms, but without any motile forms observed. In P. umbilicalis, erect diatoms were the most abundant (52%) with Tabularia species. In U. lactuca, the epiphytic diatom community was dominated by erect forms, mainly Tabularia fasciculata and T. investiens, which altogether accounted for 85% of the total diatom abundance.
Finally, Desmarestia aculeata was characterized by the dominance of erect forms with Hyalosira cf. delicatula, Rhoicosphaenia marina and Tabularia investiens, which accounted for 50% of the total diatom abundance, and adnate diatoms such as Cocconeis scutellum and C. stauroneiformis, which represented 49% of the total diatom abundance.
The vertical distribution of epiphytic diatoms on macroalgal hosts has been only investigated in Fucus vesiculosus, F. evanescens and Laminaria saccharina. The diatom abundance significantly differed between the different thallus parts only for F. vesiculosus, irrespective of the growth form, with the number of epiphytic diatoms of the apical part significantly higher than in the basal one (one-way ANOVA; P < 0.001).
Differences in epiphyte composition between seaweed morphotypes
The composition of the epiphytic diatom community in terms of growth forms was significantly different between the three seaweed morphotypes (PERMANOVA; P < 0.05) (Table 2).
The abundances of motile forms were significantly different between morphotypes (two-way ANOVA; P < 0.01), while no significant difference was observed for adnate and erect forms (Table 3). In the HBT morphotype, erect diatoms showed the highest and the lowest abundance in Fucus vesiculosus and Polysiphonia lanosa, respectively (Table 3). The abundance of motile diatoms significantly differed both between morphotypes and between species belonging to the same morphotype (Table 3). In particular, the abundance of motile diatoms in HBT algae was higher than in the two other morphotypes (SNK test; P < 0.01). Among the HBT morphotype, F. vesiculosus hosted a significantly higher number of motile diatoms than F. evanescens and P. lanosa, while among the SST morphotype, Delesseria sanguinea had a significantly lower diatom abundance of motile forms than Ulva lactuca and Porphyra umbilicalis (Table 3).
Discussion
The number of epiphytic diatoms observed in this study showed a wide range of variation, from 7 ± 5 to 7524 ± 3491 cells mm−2 in Laminaria saccharina and Fucus vesiculosus, respectively. Moreover, the epiphytic communities were highly heterogeneous, showing a marked patchy distribution, with cell abundances varying up to two orders of magnitude among the three replicates of the same seaweed sample. The maximum number of epiphytic diatoms recorded in this study is comparable to those reported on macrophytes from various areas and depths (Coleman and Burkholder 1994; Neckles et al. 1994; Novak 1984; Thomas and Jiang 1986). However, quantitative data are reported only in a few studies and using different metrics, such as chlorophyll a biomass (Nelson 1997; Pinckney and Micheli 1998), biovolume (Snoeijs 1994, 1995), weight (Ruesink 1998; Worm and Sommer 2000), cells cm−2 (Coleman and Burkholder 1994; Neckles et al. 1994; Novak 1984; Thomas and Jiang 1986), cells mg−1 dry weight (Snoeijs 1994, 1995; Tanaka 1986) or relative abundance (Main and McIntire 1974; Sullivan 1977, 1979; Moncreiff et al. 1992; Siqueiros-Beltrones et al. 1985, 2005; Sutherland 2008), overall making data from different studies hardly comparable.
The differences observed in the distribution of the epiphytic diatoms on the investigated seaweeds can be partly explained considering the morphological differences of hosts, in terms of both thallus gross morphology and surface characteristics. Marine macroalgae have been categorized into functional form groups having an ecological meaning, considering factors such as production rates and resistance to grazing and hydrodynamism (Littler 1980; Littler and Littler 1980; Steneck and Watling 1982). However, in this study, we do not refer to such classic categories, as we considered a subdivision of seaweeds based on the kind of surface available for the microepiphyte colonization. In this sense, we grouped together Polysiphonia lanosa and Fucus spp., although they belong to different ecological groups. Algal thalli of Fucus and Polysiphonia, which offer articulated and diversified surfaces with a high number of microstructures (e.g., grooves, edges, rims) hosted more abundant and richer communities than thalli with flat and smooth surface, which due to their flexible nature let the epiphytic attachment very hard: this fact is stressed in a number of red algae, due to the slimy nature of their cell walls. Wuchter et al. (2008) already reported from the North Sea that diatoms were almost absent from the smooth and slimy surface of Laminaria digitata, which offers little protection against grazing and abrasion. Round (1981) found out that the physical nature of the host surface may affect the epiphyte selection, as comparing the epiphyte density on the stipes of two Laminaria species higher densities occurred on species having a rough surface than smooth.
The vertical distribution of epiphytic diatoms on Fucus and Laminaria species showed higher abundance on the frond than on the stipe because of the optimal light availability (Cebrián et al. 1999; Müller 1999). However, such concept cannot be generalized to all seaweeds, because in some red algae the younger parts (i.e. the apical ones) are the less colonized by epiphytes due to the different physical and/or chemical nature of outer cuticle (Perrone pers. obs.). Diatom taxa having higher adhesive strength (Cocconeis and Achnanthes species) were more abundant on fronds than on stipes because they can tolerate adverse hydrodynamic conditions such as the wave action (Tanaka 1986).
Regarding the species composition in the investigated macroalgae, although some taxa have been recorded only in selected hosts, as Achnanthes brevipes, which has been found only on Fucus spp., we did not point out any clear host specificity. Sullivan (1984) reported that the most abundant diatoms on seaweeds from jetty habitat did not appear on adjacent seagrass bed. Nevertheless, he suggested that the host specificity may be only apparent. Main and McIntire (1974) found in the Yaquina estuary an epiphytic microflora composed mainly of Cocconeis, Achnanthes, Licmophora, Melosira, Synedra and Gomphonema species and they stated that no evidence of host–epiphyte specificity may be pointed out. Similar conclusions have been reported by Siver (1980) in freshwater environment which observed a similar diatom composition in four macrophyte species.
The growth forms of epiphytic diatoms differed among the investigated seaweeds. Adnate diatoms (Cocconeis and Amphora) seem to be more markedly affected by the structure of host surface, with increasing cell abundances in thalli which offer a more complex microarchitecture for colonization, while they were absent in soft thalli with smooth surface. These results agree with those of Snoeijs (1994, 1995), who reported that adnate diatoms were clearly affected by the kind of available surface, showing a lower abundance in thin filamentous thalli. In this study, motile diatoms represented only a minor fraction of the epiphytic communities and they occurred almost only on highly branched thalli with articulated surface of Fucus spp. and Polysiphonia lanosa. The dominance of motile diatoms has been also observed in diatom communities epibiontic on the marine Mediterranean hydroid Eudendrium racemosum Gmelin (Romagnoli et al. 2007), which was explained in terms of a selective advantage by raphid diatom taxa in moving across the substratum for optimum nutrient supply and light availability (De Nicola and McIntire 1990; Hudon and Legendre 1987). Raphid diatoms are the most frequent earlier algal colonizers of natural and artificial substrata, where they make the construction of the primary biofilm through mucilage production (Higgins et al. 2003; Wetherbee et al. 1998). The dominance of erect growth forms (Achnanthes and Tabularia) on seaweeds from Iceland can be interpreted as an expression of mature and stable epiphytic community (Hameed 2003; Tanaka and Watanabe 1990; Tuji 2000).
In this study, we suggested that both the architecture of the macroalgal thallus and the surface characteristics may have a role in affecting the abundance and taxonomic composition of the epiphytic diatom communities. However, any host effect on epiphytic communities which may have been recognized in ecological studies may be explained invoking a number of more reasons, such as chemical interactions, different life cycles of hosts and also the different environmental conditions that may have been experienced by hosts, making difficult to isolate a single major factor.
References
Al-Handal AY, Wulff A (2008) Marine epiphytic diatoms from the shallow sublittoral zone in Potter Cove, King George Island, Antarctica. Bot Mar 51:411–435. doi:10.1515/BOT.2008.053
Amsler CD, Okogbue IN, Landry DM, Amsler MO, McClintock JB, Baker BJ (2005) Potential chemical defenses against diatom fouling in Antarctic macroalgae. Bot Mar 48:318–322. doi:10.1515/BOT.2005.041
Anderson MJ (2001) A new method for non-parametric multivariate analysis of variance. Austral Ecol 26:32–46. doi:10.1046/j.1442-9993.2001.01070.x
Anderson MJ (2005) PERMANOVA: a FORTRAN computer program for permutational multivariate analysis of variance. Department of Statistics, University of Auckland, New Zealand
Anderson MJ, ter Braak CJF (2003) Permutation tests for multi-factorial analysis of variance. J Stat Comp Sim 73:85–113. doi:10.1080/00949650215733
Bérard-Therriault L, Cardinal A, Poulin M (1986) Les diatomées (Bacillariophyceae) benthiques de substrats durs des eaux marines et saumâtres du Québec. 6. Naviculales: Cymbellaceae et Gomphonemaceae. Naturaliste can 113:405–429
Cebrián J, Enríquez S, Fortes M, Agawin N, Vermaat JE, Duarte CM (1999) Epiphyte accrual on Posidonia oceanica (L.) Délile leaves: implication for light absorption. Bot Mar 42:123–128
Coleman VL, Burkholder JM (1994) Community structure and productivity of epiphytic microalgae on eelgrass (Zostera marina L.) under water-column nitrate enrichment. J Exp Mar Biol Ecol 179:29–48. doi:10.1016/0022-0981(94)90015-9
Compère P (1986) Algues récoltées par J. Léonard dans le désert de Libye. Bull Jard Bot Nat Belg 56:9–50. doi:10.2307/3667756
De Nicola DM, McIntire CD (1990) Effects of substrate relief on the distribution of periphyton in laboratory streams. I. Hydrology. J Phycol 26:624–633. doi:10.1111/j.0022-3646.1990.00624.x
De Stefano M, Marino D, Mazzella L (2000) Marine taxa of Cocconeis on leaves of Posidonia oceanica, including a new species and two new varieties. Eur J Phycol 35:225–242
De Stefano M, Romero O (2005) A survey of alveolate species of the diatom genus Cocconeis (Ehr.) with remarks on the new section Alveolatae. Bibl Diatomol 52:1–133
De Stefano M, Romero O, Totti C (2008) A comparative study of Cocconeis scutellum Ehrenberg and its varieties (Bacillariophyta). Bot Mar 51:506–536. doi:10.1515/BOT.2008.058
Di Camillo C, Puce S, Romagnoli T, Tazioli S, Totti C, Bavestrello G (2005) Relationships between benthic diatoms and hydrozoans (Cnidaria). J Mar Biol Assoc UK 85:1373–1380. doi:10.1017/S0025315405012555
Espinosa F, Guerra-Garcia JM (2005) Algae, macrofaunal assemblages and temperature: a quantitative approach to intertidal ecosystems of Iceland. Helgol Mar Res 59:273–285. doi:10.1007/s10152-005-0003-6
Frankovich TA, Gaiser EE, Zieman JC, Wachnicka AH (2006) Spatial and temporal distributions of epiphytic diatoms growing on Thalassia testudinum Banks ex König: relationships to water quality. Hydrobiologia 569:259–271. doi:10.1007/s10750-006-0136-x
Gross EM, Feldbaum C, Graf A (2003) Epiphyte biomass and elemental composition on submersed macrophytes in shallow eutrophic lakes. Hydrobiologia 506–509:559–565. doi:10.1023/B:HYDR.0000008538.68268.82
Gunnarsson K (2000) Benthic marine algal colonisation on the new lava at Heimaey, Vestmannaeyjar archipelago, southern Iceland. Surtsey Res 11:69–74
Gunnarsson K, Ingólfsson A (1995) Seasonal changes in the abundance of intertidal algae in Southwestern Iceland. Bot Mar 38:68–77
Hameed HA (2003) The colonization of periphytic diatom species on artificial substrates in the Ashar canal, Basrah, Iraq. Limnologica 33:54–61. doi:10.1016/S0075-9511(03)80007-5
Hasle GR, Syvertsen EE (1997) Marine diatoms. In: Tomas CR (ed) Identifying marine phytoplankton. Academic Press, San Diego, pp 5–385
Hendey NI (1964) An introductory account of the smaller algae of British coastal waters. Part V. Bacillariophyceae (Diatoms). Her Majesty’s Stationery Office, London, UK
Higgins MJ, Molino P, Mulvaney P, Wetherbee R (2003) The structure and nanomechanical properties of the adhesive mucilage that mediates diatom-substratum adhesion and motility. J Phycol 39:1181–1193. doi:10.1111/j.0022-3646.2003.03-027.x
Hillebrand H, Sommer U (2000) Effect of continuous nutrient enrichment on microalgae colonizing hard substrates. Hydrobiologia 426:185–192. doi:10.1023/A:1003943201178
Hillebrand H, Worm B, Lotze HK (2000) Marine microbenthic community structure regulated by nitrogen loading and grazing pressure. Mar Ecol Prog Ser 204:27–38. doi:10.3354/meps204027
Hudon C, Legendre P (1987) The ecological implications of growth forms in epibenthic diatoms. J Phycol 23:434–441. doi:10.1111/j.1529-8817.1987.tb02529.x
Hustedt F (1931–1959) Die Kieselalgen Deutschlands, Österreichs und der Schweiz. In: Rabenhorst L (ed) Dr. L. Rabenhorst’s Kryptogamen-Flora von Deutschland, Österreich und der Schweiz. vol 7, part 2, Akademische Verlagsgesellschaft, Leipzig
Hustedt F (1961–1966) Die Kieselalgen Deutschlands, Österreichs und der Schweiz. In: Rabenhorst L (ed) Dr. L. Rabenhorst’s Kryptogamen-Flora von Deutschland, Österreich und der Schweiz. vol 7, part 3. Akademische Verlagsgesellschaft, Leipzig
Ingólfsson A (1996) The distribution of intertidal macrofauna of the coasts of Iceland in relation to temperature. Sarsia 81:29–44
Jiang H, Seidenkrantz MS, Knudsen KL, Eiríksson J (2001) Diatom surface sediment assemblages around Iceland and their relationships to oceanic environmental variables. Mar Micropaleontol 41:73–96. doi:10.1016/S0377-8398(00)00053-0
Karsten U, Schumann R, Rothe S, Jung I, Medlin L (2006) Temperature and light requirements for growth of two diatom species (Bacillariophyceae) isolated from an Arctic macroalga. Polar Biol 29:476–486. doi:10.1007/s00300-005-0078-1
Kooistra WHCF, Forlani G, De Stefano M (2008) Adaptations of araphid pennate diatoms to a planktonic existence. Mar Ecol-Evol Persp 30:1–15. doi:10.1111/j.1439-0485.2008.00262.x
Littler M (1980) Morphological form and photosynthetic performances hypothesis. Bot Mar 22:161–165
Littler MM, Littler DS (1980) The evolution of thallus form and survival strategies in benthic marine macroalgae: field and laboratory tests of a functional form model. Am Nat 116:25–44. doi:10.1086/283610
Main SP, McIntire CD (1974) The distribution of epiphytic diatoms in Yaquina Estuary, Oregon (USA). Bot Mar 17:88–99
Marks JC, Power ME (2001) Nutrient induced changes in the species composition of epiphytes on Cladophora glomerata Kütz (Chlorophyta). Hydrobiologia 450:187–196. doi:10.1023/A:1017596927664
McArdle BH, Anderson MJ (2001) Fitting multivariate models to community data: a comment on distance-based redundancy analysis. Ecology 82:290–297
Moncreiff CA, Sullivan MJ (2001) Trophic importance of epiphytic algae in subtropical seagrass beds: evidence from multiple stable isotope analyses. Mar Ecol Prog Ser 215:93–106. doi:10.3354/meps215093
Moncreiff CA, Sullivan MJ, Daehnick AE (1992) Primary production dynamics in seagrass beds of Mississippi Sound: the contributions of seagrass, epiphytic algae, sand microflora, and phytoplankton. Mar Ecol Prog Ser 87:161–171
Müller U (1999) The vertical zonation of adpressed diatoms and other epiphytic algae on Phragmites australis. Eur J Phycol 34:487–496
Munda IM (1994) The benthic algal vegetation of the Mjóifjörđur, Eastern Iceland. Neth J Aquat Ecol 28:35–49. doi:10.1007/BF02334243
Munda IM (1999) The benthic algal vegetation of land-locked fjords in southeastern Iceland. Hydrobiologia 393:169–180. doi:10.1023/A:1003565309759
Neckles HA, Koepfler ET, Haas LW, Wetzel RL, Orth RJ (1994) Dynamics of epiphytic photoautotrophs and heterotrophs in Zostera marina L (eelgrass) microcosms: responses to nutrient enrichment and grazing. Estuaries 17:597–605. doi:10.2307/1352407
Nelson TA (1997) Epiphyte-grazer interactions on Zostera marina (Anthophyta: Monocotyledons): effects of density on community function. J Phycol 33:743–752. doi:10.1111/j.0022-3646.1997.00743.x
Novak R (1984) A study in ultra-ecology: microorganisms on the seagrass Posidonia oceanica (L) Delile, PSZNI. Mar Ecol 85:143–190. doi:10.1111/j.1439-0485.1984.tb00313.x
Pinckney JL, Micheli F (1998) Microalgae on seagrass mimics: does epiphyte community structure differ from live seagrasses? J Exp Mar Biol Ecol 221:59–70. doi:10.1016/S0022-0981(97)00115-9
Poulin M, Bérard-Therriault L, Cardinal A (1984a) Les diatomées benthiques de substrats durs des eaux marines et saumâtres du Québec. 1. Cocconeioideae (Achnanthales, Achnanthaceae). Naturaliste can 111:45–61
Poulin M, Bérard-Therriault L, Cardinal A (1984b) Les diatomées benthiques de substrats durs des eaux marines et saumâtres du Québec. 2. Tabellarioideae et Diatomoideae (Fragilariales, Fragilariaceae). Naturaliste can 111:275–295
Romagnoli T, Bavestrello G, Cucchiari E, De Stefano M, Di Camillo C, Pennesi C, Puce S, Totti C (2007) Microalgal communities epibiontic on the marine hydroid Eudendrium racemosum in the Ligurian Sea, during an annual cycle. Mar Biol 151:537–552. doi:10.1007/s00227-006-0487-x
Round FE (1981) The ecology of algae. Cambridge University Press, Cambridge, UK
Round FE, Crawford RM, Mann DG (1990) The diatoms. Biology and morphology of the genera. Cambridge University Press, Cambridge, UK
Ruesink JL (1998) Diatom epiphytes on Odonthalia floccosa: the importance of extent and timing. J Phycol 34:29–38. doi:10.1046/j.1529-8817.1998.340029.x
Siqueiros-Beltrones DA, Ibarra-Obando SE, Loya-Salinas DH (1985) An approximation to the floristic structure of the epiphytic diatoms of Zostera marina and its temporal variations in Bahia Falsa, San Quintin, BC. Cienc Mar (Méx) 11:69–88
Siqueiros-Beltrones D, López-Fuerte FO, Gárate-Lizárraga I (2005) Structure of diatom assemblages living on prop roots of the red mangrove (Rhizophora mangle) from the west coast of Baja California Sur, Mexico. Pac Sci 59:79–96. doi:10.1353/psc.2005.0014
Siver PA (1980) Microattachment patterns of diatoms on leaves of Potamogeton robbinsii oakes. Trans Am Microsc Soc 99:217–220. doi:10.2307/3225710
Snoeijs P (1993) Intercalibration and distribution of diatom species in the Baltic Sea, vol 1. Opulus Press, Uppsala
Snoeijs P (1994) Distribution of epiphytic diatom species composition, diversity and biomass on different macroalgal host along seasonal and salinity gradients in the Baltic Sea. Diatom Res 9:189–211
Snoeijs P (1995) Effects of salinity on epiphytic diatom communities on Pilayella littoralis (Phaeophyceae) in the Baltic Sea. Ecoscience 2:382–394
Steinberg PD, de Nys R (2002) Chemical mediation of colonization of seaweed surfaces. J Phycol 38:621–629. doi:10.1046/j.1529-8817.2002.02042.x
Steneck RS, Watling L (1982) Feeding capabilities and limitation of herbivorous molluscs: a functional group approach. Mar Biol 68:299–319. doi:10.1007/BF00409596
Sullivan MJ (1977) Structural characteristics of a diatom community epiphytic on Ruppia marittima. Hydrobiologia 53:81–86. doi:10.1007/BF00021236
Sullivan MJ (1979) Epiphytic diatoms of three seagrass species in Mississippi Sound. Bull Mar Sci 29:459–464
Sullivan MJ (1984) Community structure of epiphytic diatoms from the Gulf coast of Florida, USA In: Mann DG (ed) Proceedings of the 7th diatom symposium 1982. Philadelphia, Königstein, O Koeltz, pp 373–384
Sutherland DL (2008) Surface-associated diatoms from marine habitats at Cape Evans, Antarctica, including the first record of living Eunotogramma marginopunctatum. Polar Biol 31:879–888. doi:10.1007/s00300-008-0426-z
Tanaka N (1986) Adhesive strength of epiphytic diatoms on various seaweeds. Bull Jpn Soc Sci Fish 52:817–821
Tanaka S, Watanabe T (1990) The colonization process of a typical epilithic algal community Homoeothrix janthina-Achnanthes japonica community in a less polluted river in Japan. Jpn J Phycol 38:167–177. doi:10.2170/jjphysiol.38.167
Thomas DP, Jiang J (1986) Epiphytic diatoms of the inshore marine area near Davis Station. Hydrobiologia 140:193–198. doi:10.1007/BF00007435
Tittley I, Nielsen R, Gunnarsson K (2005) Relationships of algal floras in North Atlantic Islands (Iceland, the Faroes, the Shetlands, the Orkneys). BIOFAR Proc 2005:33–52
Tuji A (2000) Observation of developmental processes in loosely attached diatom (Bacillariophyceae) communities. Phycol Res 48:75–84. doi:10.1111/j.1440-1835.2000.tb00199.x
Wahl M (1989) Marine epibiosis. I. Fouling and antifouling: some basic aspects. Mar Ecol Prog Ser 58:175–189. doi:10.3354/meps058175
Wear DJ, Sullivan MJ, Moore AD, Millie DF (1999) Effects of water-column enrichment on the production dynamics of three seagrass species and their epiphytic algae. Mar Ecol Prog Ser 179:201–213. doi:10.3354/meps179201
Wetherbee R, Lind JL, Burke J, Quatrano RS (1998) The first kiss: establishment and control of initial adhesion by raphid diatoms. J Phycol 34:9–15. doi:10.1046/j.1529-8817.1998.340009.x
Witkowski A, Lange-Bertalot H, Metzeltin D (2000) Diatom flora of marine coasts. I. Iconogr Diatomol 7:1–925
Worm B, Sommer U (2000) Rapid direct and indirect effects of a single nutrient pulse in a seaweed-epiphyte-grazer system. Mar Ecol Prog Ser 202:283–288. doi:10.3354/meps202283
Wuchter C, Marquardt J, Krumbein WE (2008) The epizoic diatom community on four bryozoan species from Helgoland (German Bight, North Sea). Helgol Mar Res 57:13–19
Acknowledgments
We gratefully acknowledge Gudmundur Vidir Helgason for his kind hospitality and for providing us all the laboratory support in the Sandgerdi Marine Centre (SMC). A special thanks to Ian Tittley of the Natural History Museum in London and Ruth Nielsen of the Natural History Museum at the University of Copenhagen, which we met at SMC for their help in identifying the seaweed species. Thanks to Claudia Ciniglia who shared with us the experience in Iceland and helped us during sampling. We are indebted to Antonio Pusceddu for his useful suggestions on an early draft of the manuscript. The stay at the SMC was supported by the program “Improving Human Potential: Access to Research Infrastructure” (IHP/ARI) provided by the European Community.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Totti, C., Poulin, M., Romagnoli, T. et al. Epiphytic diatom communities on intertidal seaweeds from Iceland. Polar Biol 32, 1681–1691 (2009). https://doi.org/10.1007/s00300-009-0668-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00300-009-0668-4