Abstract
Haulout behaviour of harbour seals, living at the northern limit of their distributional range on Svalbard, Norway, was investigated from June to August 2000 using a combination of low-tide counts performed during boat surveys, hourly counts through 12- or 24-h cycles at specific haulout sites, and telemetric data from 37 VHF-tagged seals. The largest aggregations of seals were found at Skarvnes, a site where numbers increased steadily through the summer, reaching a peak during the moulting period in August. At this site, season/date, time of day, tidal state and temperature all significantly influenced the number of animals ashore. At the second most frequented haulout site, at Sørøya, season/date, time of day, temperature and cloud cover significantly affected the number of seals using the site. Pups were found predominantly at Sørøya (7.8 pups±6.3 SD, N=53 counts); they were less common at Skarvnes (1.0 pups±0.2 SD, N=95 counts). Haulout patterns varied by age and sex class in accordance with the demands of lactation, mating and moult. Our limited data on mother-pup pairs suggest that they are closely associated during the nursing period, spending approximately 50% of their time hauled out together. Post lactation, most adult females left haulout areas for periods of up to several days. The haulout behaviour of adult males suggested that they adjusted their behaviour to follow female distribution and movement patterns during the breeding period. Most juveniles and adults of both sexes stayed ashore for prolonged periods during moulting, which took place first in juveniles, then in adult females and finally in adult males. The results of our study show that the basic haulout behaviour patterns of harbour seals at Svalbard are similar to this species’ behaviour at lower latitudes.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The harbour seal (Phoca vitulina) is the most widespread of all pinniped species, ranging from approximately 30°N–80°N (Wiig 1989; Jefferson et al. 1993). The northern limit for harbour seal breeding is the Norwegian Arctic archipelago of Svalbard (Prestrud and Gjertz 1990; Gjertz and Børset 1992). The majority of Svalbard’s harbour seal population is found on Prins Karls Forland and adjacent islands and skerries. Compared to other coastal areas of Svalbard, this location has little sea ice (Vinje 1985); the warming influence of a northern branch of the Gulf Stream undoubtedly plays a role in making this area a suitable harbour seal habitat in other ways as well.
Harbour seal haulout behaviour has been studied at a variety of temperate locations. The general trend is that they haul out in the greatest numbers in favourable weather, particularly synchronously and for long periods when they moult. Several studies have identified wind speed, or concomitant factors such as wave intensity or surf height as factors that negatively affect the number of seals hauled out (e.g. Sullivan 1980; Schneider and Payne 1983; Kovacs et al. 1990). Similarly, factors that affect temperature such as the time of day, the intensity of solar radiation, wind chill, cloud cover and precipitation have been found to be significantly correlated with the number of seals hauled out (e.g. Boulva and McLaren 1979; Schneider and Payne 1983; Yochem et al. 1987; Godsell 1988; Hind and Gurney 1998). However, relationships between meteorological conditions and haulout numbers remain the subject of speculation, and results tend to be quite variable between years and locations (e.g. Stewart 1984; Thompson and Miller 1990; Roen and Bjørge 1995; Grellier et al. 1996; Watts 1996). Clearly, meteorological variables alone cannot sufficiently explain the observed variation in haulout patterns. Other factors must also be taken into account.
Quality and location of feeding habitats are recognised to be factors that influence haulout patterns of harbour seals (Härkönen 1987; Thompson and Miller 1990; Thompson et al. 1991). Abundance of prey can fluctuate through the year and cause differences in diet or foraging behaviour in this species (e.g. Olsen and Bjørge 1995; Brown and Pierce 1998; Hall et al. 1998). Another possible influence on haulout behaviour of harbour seals is predation pressure. All North Atlantic phocid seals face the risk of being hunted by killer whales (Orcinus orca), and possibly also by Greenland sharks (Somniosus microcephalus); polar bears (Ursus maritimus) and walruses (Odobenus rosmarus) are additional, potential threats to Arctic seals (Lydersen and Kovacs 1999). Factors of an intrinsic nature such as body condition or reproductive state and age also contribute to differences in observed individual patterns of haulout behaviour in harbour seals. Haulout patterns change through the annual cycle in relation to reproductive status (pupping and breeding) and the process of moult, each of which require specific behavioural adjustments. For example, adult female harbour seals must return to land for parturition and subsequent suckling of their pups. Conversely, male advertisement to females and mating take place in the water (Sullivan 1981; Hanggi and Schusterman 1994; Bjørge et al. 1995; Coltman et al. 1997; Van Parijs et al. 1997, 1999, 2000). Moulting, which takes several weeks (Thompson and Rothery 1987; Daniel et al. 2003), is enhanced by warm, dry conditions, which are advantageous for skin growth, which means that harbour seals should haul out a lot at this time (Feltz and Fay 1966).
Previous studies have found haulout peaks during the midsummer pupping period (Thompson et al. 1989, 1994a) and during late summer/early autumn when harbour seals moult (Van Bemmel 1956; Boulva and McLaren 1979; Everitt and Braham 1980; Brown and Mate 1983; Stewart and Yochem 1984). However, very little information has been gathered for this largely temperate species for populations living in the Arctic, under more severe climatic conditions and an unusual light regime. The main objective of this study was to investigate local movement patterns and haulout behaviour of harbour seals living at the northern limit of their distribution during the reproductive and moulting periods.
Materials and methods
Study area
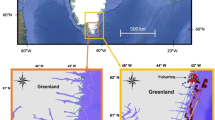
This study was conducted at Prins Karls Forland (78°30′N), an elongated island situated on the west coast of the Svalbard Archipelago, Norway (Fig. 1). Fieldwork was concentrated along the 55-km shoreline between Krykkjeskjera and Kobbebukta and took place from June to August 2000. The study area experienced 24 h daylight, with the position of the sun relative to the horizon being 35.0° at 12 noon and 11.8° at midnight at the beginning of the study (16 June), and 21.9° and −0.9°, respectively, at these times by the end of the study (26 August). Prins Karls Forland is surrounded by shallow water in the immediate vicinity of the harbour seal haulout areas (1–6 m), and the coast is encompassed by shelves that slope down to 200 m depth within a few kilometres of shore (Gjertz et al. 2001). The western coast of Prins Karls Forland is exposed to severe weather arriving from the open sea towards Greenland. Northerly (53%) and southerly (25%) winds predominated during the study period. Tidal amplitudes at Svalbard are small; they did not exceed 1.5 m during the study period (Fig. 2). Haulout sites in the study area included inter-tidal ledges, as well as rocky outcrops and sandbanks which were accessible throughout the tidal cycle.
Boat surveys
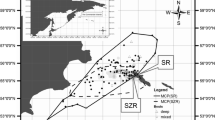
Haulout site use was monitored via boat surveys conducted every 2–3 days between Krykkjeskjera and Kobbebukta (Fig. 2). The region was divided into areas (A, B, C) (Fig. 1) based on the number of haulout sites that could be surveyed in the 3- to 4-h period around low tide. Boat surveys included most of the shoreline and all known haulout sites, which were identified from previous studies (Prestrud and Gjertz 1990; C. Lydersen and K.M. Kovacs, unpublished data; personal observations). Small aggregations of seals were counted from an inflatable boat using 10×30 binoculars. Counts of small groups performed from the boat resulted in fewer animals being classified as to age and sex, compared to land-based counts (see below) because the boat was kept at a distance to avoid scaring the animals into the water. If groups consisted of more than a few individuals, the boat was put ashore nearby and the animals were counted and classified from shore using a 30×75 telescope. Whenever possible, harbour seals were assigned to one of the following age/sex categories: adult male, adult female, or adult of unknown sex, juvenile (small-sized animals, which did not resemble pups) or pup (young of the year). Five meteorological parameters were measured at the observation sites: (1) wind speed (using a hand-held anemometer at approximately 1.5 m above the ground; m/s); (2) ambient temperature (°C, measured in shade); (3) cloud cover (percentage of covered sky); (4) irradiation (sun presence or absence), and (5) precipitation (presence or absence). Bearded seals (Erignathus barbatus) were seen hauled out alone or with harbour seals, but were easily distinguished from the harbour seals.
12-h and 24-h counts
The impact of meteorological, circadian and tidal influences, as well as seasonal patterns, on haulout behaviour and distribution patterns were investigated by making hourly counts every few days from 12 July until 26 August 2000 at the two most heavily utilised harbour seal haulout sites in the study area, Sørøya and Skarvnes (Fig. 1). At Sørøya, large aggregations of seals were found predominantly at a beach on the northeast corner of the island; 12-h counts (N=16), from 0830 to 1930 hours, were conducted at this site using a 30×75 telescope every 2nd or 3rd day (Fig. 2). Seals haulingout at Skarvnes were counted every 5th day on a 24-h basis (N=9; Fig. 2). Observations were made from ground-level hides with 10×30 binoculars, from a distance of approximately 100 m. Human-related disturbance occurred twice and data from these interrupted sessions were excluded from the analyses. Seals were classified in the age/sex categories given above and meteorological parameters were noted concurrent with hourly counts (N=148). Data on times of low and high tide were taken from “Tide tables from the Norwegian Coast and Svalbard” (Norwegian Mapping Authority 2000).
VHF telemetry
Individual haulout patterns of 37 seals were studied using VHF telemetry. A total of 139 seals were captured in tangle nets set close to haulout sites during the summer of 2000 for this and additional studies. All captured animals were weighed and measured, which helped facilitate classifying animals into age groupings in this study. Captured animals were also outfitted with roto-tags placed through the webbing of each hind flipper. During the period from 17 June to 6 July, radio transmitters (Televilt Positioning, Lindesberg, Sweden) weighing approximately 50 g and operating in the 150 MHz range were glued to the dorsal pelage, low on the back, of 10 pups, 10 juveniles, 10 adult females and 7 adult males using quick-setting epoxy. The erratic signals of animals swimming in the water were easily distinguished from the steady signals received when animals were hauled out. All adult females that were tagged had milk in their mammary glands, so were accompanied by pups, but not all mothers and pups were caught as pairs. Maximum transmission range of the transmitters was 10–15 km. Data on the duration and timing of haulout bouts were collected using automatic recording stations (RX-900, Televilt, Sweden) set up near three principal harbour seal haulout locations (Fig. 1). Data loggers ran 24 h/day and scanned each frequency for 23.5 s every 15 min from June to August until tags were moulted off. Each station used an omni-directional aerial antenna with a receiver system powered by a 12-V battery. Stations were visited intermittently to recharge batteries and service the recorders. Tests to verify that receivers were working, and to check areas of surveillance for each station were performed using a reference transmitter.
Statistical analyses
An Analysis of Covariance, with date, time of day and tidal state entered as fixed factors and meteorological parameters entered as covariates was used to examine the influence of these parameters on the number of seals hauled out (GLM, SPSS 2002). Square-root transformation was employed to normalise haulout counts prior to statistical analyses. Two-sample t-tests were used to explore seasonal trends between July and August within the different age categories at each of the two haulout sites. P<0.05 was considered to be statistically significant.
Results
Boat surveys
A total of three A, four B and seven C area surveys were completed during this study (Figs. 1, 2 and Table 1). The surveyed coast contained seven haulout sites (Krykkjeskjera, Skarvnes, Hornnes, Midtøya, Sørøya, Forlandsletta and adjacent skerries, and Kobbebukta), where harbour seal aggregations were found on most survey days. The coast between these sites was rarely used for haulout, but single animals or mother-pup pairs were occasionally seen away from groups early in the breeding season. These sightings, at intermediate places, decreased over the study period. Krykkjeskjera, the northernmost haulout site of the defined study area, was only visited three times during boat surveys because the number of animals declined markedly as the season progressed, and it was logistically difficult to get to the site. Skarvnes and Sørøya accounted for most of the hauled out seals in most surveys performed (Table 1). Low-tide counts made during boat surveys, as well as counts during observation periods, showed an increase in the number of harbour seals at Skarvnes through the study period (Fig. 3a). On average, 38.4±26.2 (N=12) seals hauled out between 1 and 2 h after low tide in July, while in August 122.8±43.5 (N=17) seals were present at this site. Haulout behaviour was more erratic at Sørøya and no seasonal trend in the total number of animals hauled out at low tide could be detected at this site (Fig. 3b). On average, 64.6±41.7 (N=14) seals hauled out in July at Sørøya and 61.8±24.9 (N=20) in August. The maximum count made during boat surveys occurred during a central survey (area C) on 10 August, when 398 animals were counted (Table 1).
12-h and 24-h counts
The two most frequented haulout sites were used for intensive observations, Skarvnes and Sørøya. Most pups were born at Sørøya whereas few, if any, were born at Skarvnes (Table 2). The number of pups that hauled out at Skarvnes did not change significantly between July and August (t=0.310, P=0.766), whereas a significant decrease was seen at the Sørøya site (t=2.891; P=0.012). Juvenile and adult haulout numbers increased significantly at Skarvnes (juveniles: t=−3.948, P=0.006; adults: t=−4.150; P=0.004), while there was no significant change at Sørøya between July and August (juveniles: t=−1.130, P=0.277; adults: t=−1.253; P=0.231). Seasonal patterns in haulout behaviour were not analysed for males and females separately using the count data because the percentage of animals classified was low and also highly variable between days (Skarvnes: 17.5%±15.6; Sørøya: 12.3%±17.4).
Harbour seal numbers at Skarvnes (Fig. 4) were influenced by the time of low tide in July, with slightly higher numbers during afternoon and early-evening low tides compared to those earlier in the day. In August, the harbour seal haulout pattern at Skarvnes was predominantly related to the time of day. Haulout numbers gradually increased towards midday, and then remained quite constant through the afternoon and evening, and began to decline again during the night. Significant effects of season/date (P<0.0001), time of day (P=0.02), tidal state (P=0.022) and temperature (P<0.0001) were detected via Analysis of Covariance at the Skarvnes site (Table 3). Haulout numbers at Sørøya followed somewhat different patterns (Fig. 5). At this site, season/date (P<0.0001), time of day (P<0.0001), temperature (P=0.002) and cloud cover (P=0.017) had a significant influence on the number of animals hauled out (Table 3).
Haulout numbers of pups (black), subadults (light grey), adult (hatched) and unspecified harbour seals (dark grey on top of the stacked bar) at the Skarvnes site with respect to time of day and tidal cycle; x marks cancelled observations or periods following disturbance. Thin vertical bars indicate high tides and thick bars indicate low tides
Haulout numbers of pups (black), subadults (light grey), adult (hatched) and unspecified harbour seals (dark grey on top of the stacked bar) at the Sørøya site with respect to time of day and tidal cycle; x marks cancelled observations or periods following disturbance. Thin vertical bars indicate high tides and thick bars indicate low tides
On several occasions during the study, seals stampeded into the water. During most of these instances, the seals tended to stay in the area and haul out again after several minutes. However, towards the end of the study period, the seals often left the haulout sites with the onset of dusk and, when agitated, they did not return to the haulout site.
VHF telemetry
Haulout patterns of radio-tagged seals in the study area are summarised in Fig. 6. Five individuals (two juveniles, two females and one male) were only occasionally detected by the stations and are assumed to have either been out of range much of the time, or their transmitters were not functioning properly; these individuals were excluded from further analyses. Capture activities were undertaken at various sites in the study area on a daily basis until 7 July, so the study area did have some human-induced disturbance in the first weeks in which telemetry data were collected.
Average percent of time spent hauled out daily by radio-tagged harbour seals in the study area. The number of individuals equipped with VHF tags in different periods is provided above the horizontal brackets. Coverage of the study area by the three receiving stations was not complete; on several occasions one of two of the receivers failed. The periods when the stations failed are identified by grey hatching (Skarvnes station \\\, Sørøya station ///)
Pups hauled out nearly every day and were ashore more frequently than adult harbour seals. Failure of the Sørøya receiving station resulted in a marked decrease in the number of pups (and adult females) detected by the VHF stations, illustrating the significant usage of this site by mother-pup pairs (Fig. 6). Haulout behaviour of adult females changed through the study period in accordance with their maternal status. An activity profile of a transmitter-equipped mother-pup pair (seal nos. 155 and 465) is presented in Fig. 7. During early nursing, the adult female’s haulout periods correlated closely with those of her offspring, and their haulout pattern was independent of time of day or tidal state. Later in lactation, the haulout pattern of the adult female began to track a tidal rhythm. A switch from haulingout at late low tides to earlier low tides in the afternoon was also observed. The pup began to follow a similar tidal pattern, although it is less clear because the pup continued to spend more time on land than its mother. During moulting, the female hauled out most of the day and the haulout rhythm was again lost. In contrast, the pup’s haulout periods became much less frequent. In total, four females were in the area of an active receiver during the first weeks of recording, which coincides with the time of nursing. Two females (seal nos. 105 and 125) showed patterns similar to female no. 155 during the time of nursing. Female no. 165 had shorter haulout bouts centred on low tides during this time; she may have been separated from her offspring. Nursing was observed in the study area until 27 July. Some females’ activity profiles revealed a low-tide preference, while others displayed no discernible tidal pattern. There were no tidal or circadian patterns in female records during their period of peak moulting (Fig. 8).
Actograms of the haulout pattern of a mother-pup pair (female no. 155 and pup no. 465) throughout the study period. Vertical lines represent times of low tide. Black squares represent hours in which the animals were hauled out. White squares indicate when seals were detected, but were in the water and empty zones indicate the periods when one of the receiving stations was not working. The first 24-h cycle represents day 1 and is followed by day 2. Day 2 is repeated again in the next line, in combination with day 3 in order to enhance pattern recognition in the data that extend from one day to the next (over midnight)
Haulout behaviour of juveniles, adult females and adult males, in relation to the time of low tide and time of day for the selected period. Tidal stage is defined as hours before and after low tide with 0 referring to the time when the tide was at its lowest point. Low levels of haulout activity after tidal stage 6 and −6 are artefacts that arise because the period between tides is 12.4 h
Telemetry data did display both circadian and tidal influences on haulout behaviour for juveniles and adult males during some periods, while these influences were largely absent in other time periods. Juveniles had extensive haulout bouts that alternated with long periods at sea. During the time when juveniles were moulting, no tidal or temporal patterns were evident (Fig. 8). Among adult males, only seal no. 225 frequented the surveyed study area regularly in July. His activity profile followed the tidal cycle during early July, shifting from late evening low tides to afternoon low tides during the first month following tagging. The remaining five males in the data set were only occasionally hauled out for short bouts of 1–2 h in the study area during this time. In August, all of the adult males began haulingout in the study area regularly for long periods in the afternoon and evening hours, following a circadian pattern to a large extent (Fig. 8). Males were present on shore most of the day, leaving the haulout areas only for some hours during the late night and early morning. The number of radio-tagged juveniles and adult males declined when the receiver at either Sørøya or Skarvnes was not working, suggesting that these animals used both of these sites (Fig. 6).
VHF records showed clearly that the time of moult varied between the different age and sex classes (Fig. 6). Juveniles were the first to change their pelage, and hence lose their tags, moulting in mid–late July. They were followed by adult females which moulted early–mid-August. Adult males moulted last, during mid–late August. Two males had not lost their radio-tags at the end of the study period. Most individuals hauled out more frequently during their moult and spent prolonged periods on shore on a daily basis at this time.
Discussion
Time of day, tidal state and meteorological conditions are key elements influencing haulout patterns of harbour seals at various sites within their range (e.g. Calambokidis et al. 1987; Roen and Bjørge 1995; Thompson et al. 1997). Haulout behaviour of harbour seals at Svalbard showed both tidal and circadian cycles, and inclement meteorological conditions such as low temperatures and extensive cloud cover had significant adverse effects on haulout numbers. The impact of temperature on haulout behaviour was the most conspicuous of the weather parameters measured in influencing the number of animals hauled out. The temperature range experienced by harbour seals during the study extended from a minimum of −0.5°C to a maximum of 9.9°C. It was somewhat surprising that no correlation between haulout numbers and wind speed was evident in this study. Prins Karls Forland is located such that there is no physical obstruction impeding winds between Greenland and its coastline. Consequently, sea spray and waves during periods with strong wind can markedly impair the quality of haulout sites. The statistical results regarding the influence of wind on haulout numbers may have been influenced by a sampling bias. On five occasions, 12-h counting sessions at Sørøya had to be terminated because winds were too strong for the observers to remain on the island. However, the number of animals at the time of departure on these occasions includes sessions with both very high and very low numbers of seals and the statistical results were consistent with the Skarvnes site, where no sessions were terminated due to wind conditions. Another potential sampling bias is that the sites used for haulout by harbour seals tend to be the most sheltered bays or gullies, and thus microclimate conditions may be more favourable than wind speeds measured at 1.5 m above ground might indicate. In addition to the physical parameters mentioned above, on-shore versus at-sea behaviour in this study was also affected by seasonal patterns that were affiliated with ecologically important events, including giving birth to and nursing offspring, mating and moulting. Not surprisingly, these patterns differed according to age and sex of individuals.
The contrasts seen between the haulout patterns at the two most heavily used sites, in combination with VHF records for the different age and sex classes, exemplify many of the differences in the behaviour of different age and sex groups through the study period. Skarvnes was a site favoured during the post-breeding, moulting period whereas Sørøya was a nursery site. Preferences exhibited by harbour seals for specific haulout sites for pupping and moulting within local population ranges, and the concomitant tendency to have sex and age segregation, at least during some periods of the year, have also been noted in other harbour seal studies (e.g. Newby 1973; Fancher and Alcorn 1982; Slater and Markowitz 1983; Thompson 1989; Henry and Hammill 2001).
In Svalbard, females gave birth at Sørøya (and a few other sites) from mid June until early July; subsequently they use this island as a nursing and resting area for the following 3–4 weeks. The use of Sørøya as a pupping site may be related to the physical qualities of the area (Nordstrom 2002). Sørøya lies within a group of small islands off the Prins Karls Forland coast. It is a safe refuge from terrestrial predators, such as the arctic fox, while the small bays and waterways between skerries in front of the pupping beaches at this site are sheltered from most wind directions and may reduce the increased likelihood of mother-pup separations during storms (Boness et al. 1992). Haulout numbers around the time of low tide were erratic on a day-to-day basis and no seasonal trend in total numbers was observed at this site. The number of pups present at Sørøya declined from July to August, as pups were weaned and became more dispersed and aquatic, following the normal pattern of ontogeny in this species (Bowen et al. 1999; Jørgensen et al. 2001). Females and their dependent pups spent much of their time hauled out, and when they entered the water, they did so independent of tide levels, because females fast during early lactation (Renouf 1984; Lawson and Renouf 1985; Bowen et al. 1992, 1999). The tendency for lactating females to haul out at irregular times has also been noted in other studies (e.g. Allen et al. 1984). As the season progressed and pups became more independent, the mother-pup haulout patterns became less synchronous within pairs. Female harbour seals have insufficient body stores to completely support the energetic costs of lactation, and hence commence foraging in mid- or late lactation, some with their pup accompanying them and others not (Bowen et al. 1992; Boness et al. 1994; Thompson et al. 1994a; Bowen et al. 1999). The VHF data in this study showed that the adult females started following a tidally influenced haulout pattern before the time that they would have been expected to wean their pups, and post-weaning females began leaving the study area for several days at a time. Such extended trips following the lactation period have also been noted in studies from other localities (Thompson and Harwood 1990; Bjørge et al. 1995; Tollit et al. 1998; Lesage et al. 1999). Although most females in this study did haul out daily during the week prior to tag loss, they did not spend the extended periods of the day hauled out that juveniles and adult males did during their moult (Figs. 6, 8). This may be due to the females’ needs to replenish their body reserves following lactation. Most pups in this study spent large amounts of time at sea during August. Long-distance movements and high dispersal rates of harbour seal pups have been previously reported (e.g. Bonner and Witthames 1974; Boulva and McLaren 1979; Thompson et al. 1994b).
Relatively few animals used the Skarvnes site early in the study period, but numbers increased gradually toward a peak in August. Both tidal and circadian patterns were observed in the behaviour of harbour seal numbers at this location. Few pups were seen at this site. The total number of animals hauled out at Skarvnes was the result of a composite picture of the haulout patterns of juveniles, adult females and adult males. The peak numbers at this site occurred in the post-breeding period, when juveniles had resumed a normal low-tide haulout pattern following the completion of their moult (see Krafft et al. 2002), but when the end of the adult female moult overlapped with the start of the adult male moult. The use of Skarvnes for moulting may be related to the fact that it is a very sheltered site, composed of two adjoining bays each of which is sheltered from a somewhat different wind direction.
The data from Skarvnes, Sørøya and total-area boat surveys are all consistent with other reports of peak haulout numbers for harbour seals occurring during the time of the annual moult (e.g. Boulva and McLaren 1979; Everitt and Braham 1980; Brown and Mate 1983; Stewart and Yochem 1984).
Adult male harbour seals were rare in the study area early in the study period. But as females appoached the end of lactation, the number of adult males increased at both Skarvnes and Sørøya, and concentrated efforts to capture them were successful. Except for occasional short visits at haulout sites, all males except one were virtually absent from shore in the weeks after they were tagged. This is likely because the onset of female oestrus coincides with the time of weaning in phocid seals (e.g. Fisher 1954; Harrison 1960; Thompson 1988), and copulation in harbour seals takes place in the water (e.g. Coltman et al. 1997, 1998, 1999; Van Parijs et al. 1997). Recent evidence from acoustic surveys in Scotland suggests that reproductive males restrict their range to areas where they are likely to intercept females, such as on females’ foraging grounds, near haulout sites and along transit routes between the two (Van Parijs et al. 1997, 1999, 2000). In the present study, females spent quite a lot of time at sea after weaning their pups. It is very likely that the absence of the five radio-tagged males at haulout sites during this period reflects their response to the behaviour patterns of the adult females. However, some females stayed relatively close to haulout locations and adult male no. 225 was hauled out during afternoon and evening low tides. This individual might have been using nearby display areas. Walker and Bowen (1993) suggested that male harbour seals might have a variety of strategies during the breeding season. During mid-August, all radio-tagged males came ashore for long periods daily, particularly in the afternoons and evenings. Similar changes of haulout trends prior to and during the moult have been described for harbour seal males in Orkney (Thompson et al. 1989).
Juvenile harbour seals used all haulout sites in the study area. They displayed haulout patterns that suggested that they were at sea for significant periods, followed by long resting periods ashore. In July, juveniles moulted; during this period, they were hauled out, on average, 36±7% of their time. Individual animals spent most of their time on land in the days prior to their VHF tags falling off. The longest haulout period documented in the study lasted for over 39 h, and 43% of juveniles’ haulout bouts exceeded 5 h. In this study, juveniles moulted first, followed by adult females, then adult males. This pattern is similar to harbour seals in Orkney, Scotland (Thompson and Rothery 1987) and harbour seals on Tugidak Island, Alaska (Daniel et al. 2003). The later moult of adults might be a result of poor body condition due to high energetic demands during the reproductive period (Ling 1970; Daniel et al. 2003). Thompson and Rothery (1987) suggested that the reproductive status of individuals might influence the timing of their moult, with high oestrogen and testosterone levels inhibiting hair growth (Ling 1970).
In conclusion, this study has found fine-scale differences in haulout activity patterns between different age and sex categories of harbour seals, as well as larger-scale seasonal differences in haulout site use. Harbour seal haulout patterns on Svalbard during summer showed a predictable circadian pattern, despite the presence of 24-h light, with a preference for afternoon and evening haulout in ecological periods when a preference was exhibited. Timing of haulout was also influenced by the occurrence of low tides, and to varying degrees between different haulout locations with other seasonal and meteorological influences. Generally, warm, dry, calm weather promoted harbour seals resting on land. Although age and sex segregation at haulout sites was not exclusive, particular sites were favoured by mother-pup pairs, while other sites were favoured by adult males and moulting aggregations. Behaviour of radio-tagged individuals showed that age and sex groups exhibited quite different behaviour patterns; adult animals were heavily influenced by their breeding status at a particular time in the breeding cycle, and juveniles and adults adjusted their haulout behaviour when moulting. High Arctic harbour seals appear to display haulout behaviour patterns that are similar to those of temperate populations of this species.
References
Allen SG, Ainley DG, Page GW, Ribic CA (1984) The effect of disturbance on harbor seal haul out patterns at Bolinas Lagoon, California. Fish Bull 82:493–500
Bjørge A, Thompson D, Hammond P, Fedak M, Bryant E, Aarefjord H, Roen R, Olsen M (1995) Habitat use and diving behaviour of harbour seals in a coastal archipelago in Norway. In: Blix AS, Walløe L, Ulltang Ø (eds) Whales, seals, fish and man. Elsevier, Amsterdam, pp 211–223
Boness DJ, Bowen WD, Iverson SJ, Oftedal OT (1992) Influence of storms and maternal size on mother-pup separations and fostering in the harbor seal, Phoca vitulina. Can J Zool 70:1640–1644
Boness DJ, Bowen D, Oftedal OT (1994) Evidence of a maternal foraging cycle resembling that of otariid seals in a small phocid, the harbor seal. Behav Ecol Sociobiol 34:95–104
Bonner WN, Witthames SR (1974) Dispersal of common seals (Phoca vitulina), tagged in the Wash, east Anglia. J Zool Lond 174:528–531
Boulva J, McLaren IA (1979) Biology of the harbor seal, Phoca vitulina, in eastern Canada. Bull Fish Res Board Can 200:1–24
Bowen WD, Oftedal OT, Boness DJ (1992) Mass and energy transfer during lactation in a small phocid, the harbor seal (Phoca vitulina). Physiol Zool 65:844–866
Bowen WD, Boness DJ, Iverson SJ (1999) Diving behaviour of lactating harbour seals and their pups during maternal foraging trips. Can J Zool 77:978–988
Brown EG, Pierce GJ (1998) Monthly variation in the diet of harbour seals in inshore waters along the southeast Shetland (UK) coastline. Mar Ecol Prog Ser 167:275–289
Brown RF, Mate BR (1983) Abundance, movements, and feeding habits of harbor seals, Phoca vitulina, at Netarts and Tillamook Bays, Oregon. Fish Bull 81:291–301
Calambokidis J, Taylor BL, Carter SD, Steiger GH, Dawson PK, Antrim LD (1987) Distribution and haul-out behavior of harbor seals in Glacier Bay, Alaska. Can J Zool 65:1391–1396
Coltman DW, Bowen WD, Boness DJ, Iverson SJ (1997) Balancing foraging and reproduction in the male harbour seal, an aquatically mating pinniped. Anim Behav 54:663–678
Coltman DW, Bowen WD, Wright JM (1998) Male mating success in an aquatically mating pinniped, the harbour seal (Phoca vitulina), assessed by microsatellite DNA markers. Mol Ecol 7:627–638
Coltman DW, Bowen WD, Wright JM (1999) A multivariate analysis of phenotype and paternity in male harbor seals, Phoca vitulina, at Sable Island, Nova Scotia. Behav Ecol 10:169–177
Daniel RG, Jemison LA, Pendleton GW, Crowley SM (2003) Molting phenology of harbor seals on Tugidak Island, Alaska. Mar Mammal Sci 19:128–140
Everitt RD, Braham HW (1980) Aerial survey of Pacific harbor seals in the southeastern Bering Sea. Northwest Sci 54:281–288
Fancher LE, Alcorn DJ (1982) Harbor seal census in south San Francisco Bay (1972–1977 and 1979–1980). Calif Fish Game 68:118–121
Feltz ET, Fay FH (1966) Thermal requirements in vitro of epidermal cells from seals. Cryobiology 3:261–264
Fisher HD (1954) Delayed implantation in the harbour seal, Phoca vitulina L. Nature 173:879–880
Gjertz I, Børset A (1992) Pupping in the most northerly harbor seal (Phoca vitulina). Mar Mammal Sci 8:103–109
Gjertz I, Lydersen C, Wiig Ø (2001) Distribution and diving of harbour seals (Phoca vitulina) in Svalbard. Polar Biol 24:209–214
Godsell J (1988) Herd formation and haul-out behaviour in harbour seals (Phoca vitulina). J Zool Lond 215:83–98
Grellier K, Thompson PM, Corpe HM (1996) The effect of weather conditions on harbour seal (Phoca vitulina) haulout behaviour in the Moray Firth, northeast Scotland. Can J Zool 74:1806–1811
Hall AJ, Watkins J, Hammond PS (1998) Seasonal variation in the diet of harbour seals in the south-western North Sea. Mar Ecol Prog Ser 170:269–281
Hanggi EB, Schusterman RJ (1994) Underwater acoustic displays and individual variation in male harbour seals, Phoca vitulina. Anim Behav 48:1275–1283
Härkönen TJ (1987) Influence of feeding on haul-out patterns and sizes of sub-populations in harbour seals. Neth J Sea Res 21:331–339
Harrison RJ (1960) Reproduction and reproductive organs in common seals (Phoca vitulina) in the Wash, East Anglia. Mammalia 24:372–385
Henry E, Hammill MO (2001) Impact of small boats on the haulout activity of harbour seals (Phoca vitulina) in Métis Bay, Saint Lawrence Estuary, Québec, Canada. Aquat Mamm 27:140–148
Hind AT, Gurney WSC (1998) Are there thermoregulatory constraints on the timing of pupping for harbour seals? Can J Zool 76:2245–2254
Jefferson TA, Leatherwood S, Webber MA (1993) Marine mammals of the world. FAO, Rome
Jørgensen C, Lydersen C, Brix O, Kovacs KM (2001) Diving development in nursing harbour seal pups. J Exp Biol 204:3993–4004
Kovacs KM, Jonas KM, Welke SE (1990) Sex and age segregation by Phoca vitulina concolor at haul-out sites during the breeding season in the Passamaquoddy Bay region, New Brunswick. Mar Mammal Sci 6:204–214
Krafft BA, Lydersen C, Gjertz I, Kovacs KM (2002) Diving behaviour of sub-adult harbour seals (Phoca vitulina) at Prins Karls Forland, Svalbard. Polar Biol 25:230–234
Lawson JW, Renouf D (1985) Parturition in the Atlantic harbor seal, Phoca vitulina concolor. J Mammal 66:395–398
Lesage V, Hammill MO, Kovacs KM (1999) Functional classification of harbor seal (Phoca vitulina) dives using depth profiles, swimming velocity, and an index of foraging success. Can J Zool 77:74–87
Ling JK (1970) Pelage and molting in wild mammals with special reference to aquatic forms. Q Rev Biol 45:16–54
Lydersen C, Kovacs KM (1999) Behaviour and energetics of ice-breeding, North Atlantic phocid seals during the lactation period. Mar Ecol Prog Ser 187:265–281
Newby TC (1973) Observations on the breeding behavior of the harbor seal in the State of Washington. J Mammal 54:540–543
Nordstrom CA (2002) Haul-out selection by Pacific harbor seals (Phoca vitulina richardii): isolation and perceived predation risk. Mar Mammal Sci 18:194–205
Norwegian Mapping Authority (2000) Tide tables from the Norwegian coast and Svalbard. Sjøkartverket, Stavanger
Olsen M, Bjørge A (1995) Seasonal and regional variations in the diet of harbour seal in Norwegian waters. In: Blix AS, Walløe L, Ulltang Ø (eds) Whales, seals, fish and man. Elsevier, Amsterdam, pp 271–285
Prestrud P, Gjertz I (1990) The most northerly harbor seal, Phoca vitulina, at Prins Karls Forland, Svalbard. Mar Mammal Sci 6:215–220
Renouf D (1984) The vocalization of the harbour seal pup (Phoca vitulina) and its role in the maintenance of contact with the mother. J Zool Lond 202:583–590
Roen R, Bjørge A (1995) Haul-out behaviour of the Norwegian harbour seal during summer. In: Blix AS, Walløe L, Ulltang Ø (eds) Whales, seals, fish and man. Elsevier, Amsterdam, pp 61–67
Schneider DC, Payne PM (1983) Factors affecting haul-out of harbor seals at a site in southeastern Massachusetts. J Mammal 64:518–520
Slater LM, Markowitz H (1983) Spring population trends in Phoca vitulina richardi in two central California coastal areas. Calif Fish Game 69:217–226
SPSS (2002) SPSS release 11.0.0 for Windows. Statistical Package for the Social Sciences, Chicago, Ill
Stewart BS (1984) Diurnal hauling out patterns of harbor seals at San Miguel Island, California. J Wildl Manage 48:1459–1461
Stewart BS, Yochem PK (1984) Seasonal abundance of pinnipeds at San Nicolas Island, California, 1980–1982. Bull South Calif Acad Sci 83:121–132
Sullivan RM (1980) Seasonal occurrence and haul-out use in pinnipeds along Humboldt County, California. J Mammal 61:754–760
Sullivan RM (1981) Aquatic displays and interactions in harbor seals, Phoca vitulina, with comments on mating systems. J Mammal 62:825–831
Thompson P (1988) Timing of mating in the common seal (Phoca vitulina). Mammal Rev 18:105–112
Thompson PM (1989) Seasonal changes in the distribution and composition of common seal (Phoca vitulina) haul-out groups. J Zool Lond 217:281–294
Thompson PM, Harwood J (1990) Methods for estimating the population size of common seals, Phoca vitulina. J Appl Ecol 27:924–938
Thompson PM, Miller D (1990) Summer foraging activity and movements of radio-tagged common seals (Phoca vitulina. L.) in the Moray Firth, Scotland. J Appl Ecol 27:492–501
Thompson P, Rothery P (1987) Age and sex differences in the timing of moult in the common seal, Phoca vitulina. J Zool Lond 212:597–603
Thompson PM, Fedak MA, McConnell BJ, Nicholas KS (1989) Seasonal and sex-related variation in the activity patterns of common seals (Phoca vitulina). J Appl Ecol 26:521–535
Thompson PM, Pierce GJ, Hislop JRG, Miller D, Diack JSW (1991) Winter foraging by common seals (Phoca vitulina) in relation to food availability in the Inner Moray Firth, N.E. Scotland. J Anim Ecol 60:283–294
Thompson PM, Miller D, Cooper R, Hammond PS (1994a) Changes in the distribution and activity of female harbour seals during the breeding season: implications for their lactation strategy and mating patterns. J Anim Ecol 63:24–30
Thompson PM, Kovacs KM, McConnell BJ (1994b) Natal dispersal of harbour seals (Phoca vitulina) from breeding sites in Orkney, Scotland. J Zool Lond 234:668–673
Thompson PM, Tollit DJ, Wood D, Corpe HM, Hammond PS, Mackay A (1997) Estimating harbour seal abundance and status in an estuarine habitat in north-east Scotland. J Appl Ecol 34:43–52
Tollit DJ, Black AD, Thompson PM, Mackay A, Corpe HM, Wilson B, Van Parijs SM, Grellier K, Parlane S (1998) Variations in harbour seal Phoca vitulina diet and dive-depths in relation to foraging habitat. J Zool Lond 244:209–222
Van Bemmel ACV (1956) Planning a census of the harbour seal (Phoca vitulina L.) on the coasts of the Netherlands. Beaufortia 54:121–132
Van Parijs SM, Thompson PM, Tollit DJ, Mackay A (1997) Distribution and activity of male harbour seals during the mating season. Anim Behav 54:35–43
Van Parijs SM, Hastie GD, Thompson PM (1999) Geographical variation in temporal and spatial vocalization patterns of male harbour seals in the mating season. Anim Behav 58:1231–1239
Van Parijs SM, Janik VM, Thompson PM (2000) Display-area size, tenure length, and site fidelity in the aquatically mating male harbour seal, Phoca vitulina. Can J Zool 78:2209–2217
Vinje T (1985) Drift, composition, morphology and distribution of the sea ice fields in the Barents Sea. Nor Polarinst Skr 179:1–26
Walker BG, Bowen WD (1993) Behavioural differences among adult male harbour seals during the breeding season may provide evidence of reproductive strategies. Can J Zool 71:1585–1591
Watts P (1996) The diel hauling-out cycle of harbour seals in an open marine environment: correlates and constraints. J Zool Lond 240:175–200
Wiig Ø (1989) A description of common seals, Phoca vitulina L. 1758, from Svalbard. Mar Mammal Sci 5:149–158
Yochem PK, Stewart BS, Delong RL, Demaster DP (1987) Diel haul-out patterns and site fidelity of harbor seals (Phoca vitulina richardsi) on San Miguel Island, California, in autumn. Mar Mammal Sci 3:323–332
Acknowledgements
We thank Christian Jørgensen and Hans Lund for their help in the field, and Drs. Sofie Van Parijs, Peter Corkeron and Dierk Blomeier, as well as Christian Jørgensen, for helpful input on data handling and editorial work. Dr. Georg Elvebakk, Statistics Department, University of Tromsø provided valuable statistical advice. The Research Council of Norway (Arctic Light and Heat Program) and the Norwegian Polar Institute funded this research.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Reder, S., Lydersen, C., Arnold, W. et al. Haulout behaviour of High Arctic harbour seals (Phoca vitulina vitulina) in Svalbard, Norway. Polar Biol 27, 6–16 (2003). https://doi.org/10.1007/s00300-003-0557-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00300-003-0557-1