Abstract
A total of 1,103 inshore notothenioid fish were caught by means of trammel-nets in 4 sites surrounding Cierva Point (Moss Island 1; Moss Island 2; Sterneck Island; Leopardo Island), Danco Coast, West Antarctic Peninsula, during February and March 2000. The families Nototheniidae, Channichthyidae and Bathydraconidae were represented in the samples, Notothenia coriiceps being the dominant fish of the area. Gobionotothen gibberifrons and Trematomus newnesi followed in importance. In general, the fish sampled agreed in terms of number and mass with those of the South Shetland Islands area, except for a marked higher occurrence of G. gibberifrons in the Danco Coast. This supports the hypothesis that the commercial fishery around the South Shetland Islands at the end of the 1970s was responsible for the decrease in the inshore population of G. gibberifrons in that area during the last 17 years. Information on morphometry, reproduction and diet of the fish species caught is provided.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The role of fish in the Antarctic marine ecosystem is a very important ecological subject and, hence, has been the object of several studies during the last three decades. The ecological studies on coastal demersal fish from the South Shetland Islands and west Antarctic Peninsula waters (FAO Statistical Subarea 48.1) were done mainly in the first area (Bellisio 1967; Moreno and Bahamonde 1975; Tarverdiyeva and Pinskaya 1980; Linkowski et al. 1983; Kock 1989; Casaux et al. 1990; Gröhsler 1994; Kulesz 1994; Barrera-Oro 1996; among others) and a fewer number in the second area (Moreno et al. 1977; Daniels 1982; Daniels and Lipps 1982). In line with this, in 1983 the Ichthyology Project of the Instituto Antártico Argentino implemented a long-term research program focused on monitoring and ecological aspects of demersal fish in inshore sites of the South Shetland Islands, mainly in Potter Cove, King George Island, using trammel-nets (Barrera-Oro et al. 2000). This research program allowed the development of studies about the ecology of fish (e.g. populational aspects, trophic position, age and growth, predator-prey interactions), as well as about the impact of the offshore commercial fishery on inshore fish of the area (summarised in Barrera-Oro and Casaux 1998).
The substantial commercial activities in subarea 48.1 occurred throughout the South Shetland Islands offshore (>100 m depth) from 1977/1978 to 1989/1990, prior to closure by the Commission for the Conservation of Antarctic Marine Living Resources (CCAMLR) primarily west and northwest of Elephant Island and north of King George Island (Kock 1998). Besides the serious depletion of the target species in offshore waters, a diminution in the stocks of juvenile Gobionotothen gibberifrons and Notothenia rossii in inshore waters was demonstrated and explained as a reduction in recruitment due to heavy fishing suffered by the reproductive stock (Barrera-Oro and Marschoff 1991).
In 1997/1998, the scope of our research project was extended to the Danco Coast, a less investigated area of the Antarctic Peninsula, which has remained outside the influence of the commercial fishery. Thus, the aim of this study is to provide new data on occurrence, morphometry, reproduction and diet of demersal fish from this last region, by the analysis of samples obtained by trammel-nets at four sites during February and March 2000.
Materials and methods
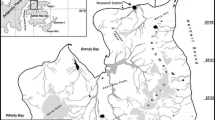
A total of 1,103 fish were caught by means of trammel-nets in 4 sites surrounding Cierva Point (64º09'S; 60º57'W; Fig. 1) (Moss Island 1; Moss Island 2; Sterneck Island; Leopardo Island; Fig. 2), Danco Coast, Antarctic Peninsula, between 2 February and 31 March 2000. The distance between the farthest sampling sites, Leopardo Island and Moss Island 1, is 7.5 km. The nets (length 25 m; width 1.5 m; inner mesh 2.5 cm, outer mesh 12 cm) were fastened to rocks at the coast and laid on the bottom at depths from 20 to 70 m (Table 1). Stones and algae were often found in the nets at the four sampling sites. Although we have no information on the seabed topography in the sampling area, rocky bottoms with algae beds are likely constituents.
From the fish, the following data were obtained: total (TL) and standard (SL) lengths to 0.1 cm (in the text, size measurements are expressed as TL), weight (in grammes) and sex. The otoliths were removed and the gonad stage was determined according to the scale in Kock and Kellermann (1991). In this scale, stage I indicates immature ovaries and testis, stage II ovaries maturing virgin or resting and testis developing or resting, stage III ovaries developing and testis developed, stage IV ovaries gravid and testis ripe, and stage V ovaries and testis spent.
Nine hundred and twenty-five stomach contents were examined according to the mixed method of Hureau (1970). Data are expressed in terms of the dietary coefficient ( Q), which is the product of the percentage by number and the percentage by mass of each prey type. According to this index, the prey items are separated into the following categories: Q >200 main preys, 200> Q >20 secondary preys and 20> Q occasional preys. To estimate the percentage by number of algae, the number of algae species present in each stomach content was considered as the number of specimens represented in the sample. The stomach fullness was evaluated according to a 5-point scale: 0 (empty), 1 (1/4 full), 2 (1/2 full), 3 (3/4 full), 4 (full).
The fish species are identified following the nomenclature given in Gon and Heemstra (1990).
Results
There were no statistical differences in the number of fish caught per net between sampling sites (ANOVA, F 3,1103=0.23; ns) (Table 1).
Nototheniid species (471 N. coriiceps; 265 Trematomus newnesi; 215 G. gibberifrons; 45 T. bernacchii; 28 Lepidonotothen nudifrons; 3 N. rossii and 3 Trematomus hansoni) dominated in the samples whereas the families Bathydraconidae (12 Parachaenichthys charcoti) and Channichthyidae (8 Chaenocephalus aceratus) were scarcely represented.
Several specimens were not assigned to species because their soft tissues were eaten by amphipods and only unidentifiable parts were recovered. Most of these fish belonged to the genus Trematomus, since they exhibited the scapular foramen enclosed within scapula.
Overall, N. coriiceps was always present in the catches and was the dominant fish by number and mass in all of the sampling sites, except at Sterneck Island where G. gibberifrons was the most abundant fish (Table 2). In the other sites, this last species and T. newnesi followed in importance. The relative abundance of N. coriiceps decreased (Spearman test, r =−0.90, P <0.01) and that of G. gibberifrons increased with depth ( r =0.93, P <0.001).
The total length ranges observed in N. coriiceps, T. newnesi and G. gibberifrons are plotted in Fig. 3. The size frequency distribution of T. newnesi was unimodal whereas those of N. coriiceps and G. gibberifrons showed polymodality. The size of N. coriiceps (ANOVA, F 3,471=25.6; P <0.00001), T. newnesi (ANOVA, F 3,265=5.7; P <0.001) and G. gibberifrons (ANOVA, F 3,215=5.5; P <0.01) specimens caught at the different sites differed statistically (Table 3). The largest (mean length) N. coriiceps, T. newnesi and G. gibberifrons specimens were caught at Leopardo Island, Moss Island 2 and Sterneck Island, respectively. The sizes of G. gibberifrons ( r =−0.96, P <0.001) and N. coriiceps (Spearman test, r =0.70, P <0.05) decreased and increased along the depth sampling range, respectively.
For the study of the predator-prey interactions between seabirds/seals and fish of the area, based on the analysis of otoliths recovered from faeces, regurgitated casts and stomach contents, we have estimated the otolith length-standard/total fish length relationships according to the equations presented in Table 4.
The total length-standard length relationship was estimated applying the equation TL= a *SL, where a is a constant. The results are presented in Table 5.
Length-weight relationships were estimated applying the equation W = a *TL k where W is the weight in grammes (the mass of the stomach contents was excluded), TL is the total length in centimetres, and a and k are constants (Table 6). Except in T. newnesi, at similar lengths males were heavier than females.
The sex ratio of the fish species is presented in Table 7. Female L. nudifrons and female T. bernacchii predominated largely in the sampling sites where these species occurred. Except for N. rossii, all the species caught were represented in the samples by reproductive specimens (gonad stages III and IV) (Table 8). Fish with spent gonads (stage V) were absent from the samples.
All T. hansoni and C. aceratus specimens showed empty stomachs (Table 9). This stage predominated also in T. bernacchii and T. newnesi. In contrast, full stomachs was the commonest stage in the two most important species by mass, N. coriiceps and G. gibberifrons.
The diet composition was diverse in N. coriiceps and G. gibberifrons (mainly at Moss Island 2 and Sterneck Island) but was limited in the remaining species (Tables 10, 11). N. coriiceps ate mainly algae and gammarid amphipods. This last prey predominated in the diet of G. gibberifrons, L. nudifrons and T. bernacchii. Krill was the main prey of N. rossii and T. newnesi, whereas P. charcoti ingested mainly fish.
The separate diet analysis of fish from the different sampling sites showed the same pattern, except for N. coriiceps at Sterneck and Leopardo Islands where gammarid amphipods and krill were the main preys, respectively (Fig. 4a); G. gibberifrons at Leopardo Island where polychaetes dominated in the diet (Fig. 4b); and P. charcoti at Leopardo Island where decapods constituted the main food item in the only stomach analysed.
Thirty-three percent of the fish ingested were identified to species but only a few could be measured. N. coriiceps preyed on L. nudifrons (14 specimens; 2 of them of 8.7 and 13.0 cm), T. newnesi (4 specimens; 1 of 13 cm), G. gibberifrons (4 specimens; 1 of 11.0 cm), Electrona antarctica (3) and P. charcoti (1 specimen of 19.2 cm). P. charcoti ate T. newnesi (2 specimens of 14.5 and 15.0 cm) and G. gibberifrons (1 specimen of 24.0 cm).
Eighty-seven Euphausia superba specimens ingested by T. newnesi averaged 4.63 cm in length (SD 0.48) and ranged from 3.67 to 5.89 cm.
Discussion
The fish species caught at the Danco Coast in the present study have been previously reported for neritic waters of the Antarctic Peninsula, and agree in terms of composition with those sampled also with trammel-nets in the South Shetland Islands area (Daniels and Lipps 1982; Fischer and Hureau 1985; Casaux et al. 1990; Barrera-Oro 1996). N. coriiceps was the dominant fish in number and mass in the sampling sites, except at Sterneck Island where G. gibberifrons was the most abundant. Given that the relative abundance of N. coriiceps and G. gibberifrons decreased and increased with depth respectively, this might be related to the fact that at Sterneck Island the nets were laid down deeper than at the remaining sites.
The total length ranges observed in N. coriiceps, T. newnesi and G. gibberifrons (Table 3, Fig. 3) coincide with those reported by Casaux et al. (1990) for fish caught with similar trammel-nets at Potter Cove, South Shetland Islands. The size-frequency distribution of T. newnesi was unimodal indicating the presence of one age class. In contrast, the size-frequency distributions of N. coriiceps and G. gibberifrons showed polymodality, reflecting the existence of different age classes. Although it is likely that several age classes are masked in the length distribution, N. coriiceps and G. gibberifrons specimens of 3–13 and 5–12 years of age (Casaux et al. 1990), respectively, seem to be represented in the whole sample.
It is known that G. gibberifrons displays a positive length stratification as a function of depth (Barrera-Oro 1989; Casaux et al. 1990; Kulesz 1994). However, the analysis of the sizes of G. gibberifrons at the sampling sites in the Danco Coast showed a decrease with the increment of depth. We have no conclusive explanation for this finding but one possibility is that the size distribution with depth of G. gibberifrons in the sampling area may have been altered by the intensive foraging activity of the Antarctic shag on this fish species (Casaux et al. 2002). Alteration in inshore waters of the structure of fish populations by cormorant predation has been documented in other ecosystems (Birt et al. 1987; Leopold et al. 1998).
Although the information presented in Tables 4, 5 and 6 is not discussed in this work, it could be useful not only for analysis of fish morphological parameters but also for size and mass estimations of fish represented in food samples from top-predators, such as the seabirds and seals from the Antarctic Peninsula area.
The analysis of the N. coriiceps, T. newnesi, L. nudifrons, T. hansoni, C. aceratus and P. charcoti specimens at gonad stages III and IV indicates that the sampling time and the size of these fish (Table 8) agree with the spawning time and the length at first spawning reported for these species at other localities (see Bellisio 1967; Everson 1970; Hureau 1970; Hourigan and Radtke 1989; Kock 1989; Casaux et al. 1990; Kock and Kellermann 1991; Vacchi et al. 1996, among others).
A high proportion of the L. nudifrons (91.7%) and T. bernacchii (73.5%) specimens were females (Table 7). Except one stage III, all L. nudifrons ovaries were at stage IV, which suggests a pre-spawning female aggregation in the sampling area at that time.
Empty stomachs was the only stage in C. aceratus and T. hansoni and this stage predominated in T. bernacchii and T. newnesi; full stomachs predominated in the two most important fish by mass, N. coriiceps and G. gibberifrons. Fasting before spawning and a wider prey range in opportunistic feeders are factors that could be related to these results.
The analysis of the diet of the fish species sampled at the Danco Coast showed general agreement on the feeding types and feeding behaviour of these species in other areas. In general, the main preys reported here have been previously indicated in the diet of the same fish species considered in this study (see Moreno and Bahamonde 1975; Richardson 1975; Daniels 1982; Burchett 1983; Linkowski et al. 1983; Casaux et al. 1990; Vacchi et al. 1994; Barrera-Oro 1996; La Mesa et al. 2000). Although they seem to be primarily benthos ( N. coriiceps, G. gibberifrons, L. nudifrons and T. bernacchii) or plankton/water-column feeders ( T. newnesi, N. rossii and P. charcoti), all of them preyed on both benthic-demersal and pelagic organisms. Most of these fish species are specialised feeders (see Q index in Tables 10, 11). Gammarid amphipods were largely the main food of G. gibberifrons, L. nudifrons and T. bernacchii; krill was the main prey of N. rossii and T. newnesi whereas P. charcoti foraged mainly on fish. The diet composition of N. coriiceps was the most diverse and changed in the different sampling sites. This fish is an opportunistic feeder, and therefore its diet reflects the food availability of benthos at different sites and depths. Grazing is an important feeding strategy in some Antarctic demersal, shallow-water fish species. In this study, algae constituted a main food item for N. coriiceps and N. rossii. It has been demonstrated that algae are actively selected and consumed deliberately by fish (Barrera-Oro and Casaux 1990; Casaux et al. 1990; Iken et al. 1997).
Comparison of relative abundance of G. gibberifrons in inshore trammel-net catches between the present results from the Danco Coast and those from the South Shetland Islands area indicates high and very low values, respectively (this study; Barrera-Oro et al. 2000). In fact, this difference has been also reflected in the analysis of the diet of the Antarctic shag from both areas, in which the low predation by this bird on G. gibberifrons in inshore waters (<120 m depth) of the South Shetland Islands is evident (Casaux and Barrera-Oro 1993; Barrera-Oro and Casaux 1996; Casaux et al. 1997; Casaux et al. 2002). Barrera-Oro and Marschoff (1991) and later on Barrera-Oro et al. (2000) indicated that the effect of the commercial fishery around the South Shetland Islands at the end of the 1970s is the most likely reason for the decrease in the inshore population of G. gibberifrons in that area during the last 19 years. This explanation is supported by the data from this study, due to the fact that they were obtained in an area that has remained outside the influence of the commercial fishery. Likewise, present results validate the utility of the standard method implemented by the Ecosystem Monitoring and Management Working Group of CCAMLR, on the use of the Antarctic shag to monitor changes in the abundance of inshore demersal fish populations (SC-CAMLR-XVII/3, 1998).
References
Barrera-Oro E (1989) Age determination of Notothenia gibberifrons from the South Shetland Islands, Antarctic Peninsula subarea (subarea 48.1). CCAMLR Sci Sel Pap 2:143–160
Barrera-Oro E (1996) Ecology of inshore demersal fish (Notothenioidei) from the South Shetland Islands. PhD Thesis, University of Bremen
Barrera-Oro E, Casaux R (1990) Feeding selectivity in Notothenia neglecta, Nybelin, from Potter Cove, South Shetland Islands, Antarctica. Antarct Sci 2:207–213
Barrera-Oro E, Casaux R (1996) Fish as diet of the blue-eyed shag Phalacrocorax atriceps bransfieldensis at Half-moon Island, South Shetland Islands. Cybium 20:37–45
Barrera-Oro E, Casaux R (1998) Ecology of demersal fish species from Potter Cove. In: Wiencke C, Ferreyra G, Arntz W, Rinaldi C (eds) The Potter Cove coastal ecosystem, Antarctica. Berichte 299, Bremerhaven, pp 156–167
Barrera-Oro E, Marschoff E (1991) A declining trend in the abundance of Notothenia rossii marmorata and Notothenia gibberifrons observed in fjords in two sites in the South Shetland Islands. Selected Scientific Papers 1990 (SC-CAMLR-SSP/7). CCAMLR, Hobart, Australia 263–274
Barrera-Oro E, Marschoff E, Casaux R (2000) Trends in relative abundance of fjord Notothenia rossii, Gobionotothen gibberifrons and Notothenia coriiceps at Potter Cove, South Shetland Islands, after the commercial fishing in the area. CCAMLR Sci 7:43–52
Bellisio N (1967) Peces antárticos del Sector Argentino (parte IV): Parachaenichthys charcoti, P. georgianus y Harpagifer bispinis antarcticus de Bahía Luna. Serv Hidrogr Nav 904:1–57
Birt V, Birt T, Goulet D, Lairus D, Montevecchi W (1987) Ashmole's halo: direct evidence for prey depletion by a seabird. Mar Ecol Prog Ser 40:205–208
Burchett M (1983) Food, feeding and behaviour of Notothenia rossii nearshore at South Georgia. Br Antarct Surv Bull 61:45–51
Casaux R, Barrera-Oro E (1993) The diet of the blue-eyed shag Phalacrocorax atriceps bransfieldensis feeding in the Bransfield Strait. Antarct Sci 5:335–338
Casaux R, Mazzotta A, Barrera-Oro E (1990) Seasonal aspects of the biology and diet of nearshore nototheniid fish at Potter Cove, South Shetland Islands, Antarctica. Polar Biol 11:63–72
Casaux R, Favero M, Coria N, Silva P (1997) Diet of the imperial cormorant Phalacrocorax atriceps: comparison of pellets and stomach contents. Mar Ornithol 25:1–4
Casaux R, Baroni A, Barrera-Oro E (2002) Fish in the diet of breeding Antarctic shags Phalacrocorax bransfieldensis at four colonies in the Danco Coast, Antarctic Peninsula. Antarct Sci (in press)
Daniels R (1982) Feeding ecology of some fishes of the Antarctic Peninsula. Fish Bull 80:575–588
Daniels R, Lipps J (1982) Distribution and ecology of fishes of the Antarctic Peninsula. J Biogeogr 9:1–9
Everson I (1970) Reproduction in Notothenia neglecta Nybelin. Br Antarct Surv Bull 23:81–92
Fischer W, Hureau J (1985) FAO species identification sheets for fishery purposes. Southern Ocean (CCAMLR Convention Area), vol 2. FAO, Rome
Gon O, Heemstra P (eds) (1990) Fishes of the Southern Ocean. J.L.B. Smith Institute of Ichthyology, Grahamstown
Gröhsler T (1994) Feeding habits as indicators of ecological niches: investigations of Antarctic fish conducted near Elephant Island in late autumn/winter 1986. Arch Fish Mar Res 42:17–34
Hourigan T, Radtke R (1989) Reproduction of the Antarctic fish Nototheniops nudifrons. Mar Biol 100:277–283
Hureau J (1970) Biologie comparee de quelques poissons antarctiques (Nototheniidae). Bull Inst Océanogr Monaco 68:1–244
Iken K, Barrera-Oro E, Quartino M, Casaux R, Brey T (1997) Grazing by the Antarctic fish Notothenia coriiceps: evidence for selective feeding on macroalgae. Antarct Sci 9:386–391
Kock K (1989) Reproduction in fish around Elephant Island. Arch Fischereiwiss 39:171–210
Kock K (1998) Changes in fish biomass around Elephant Island (Subarea 48.1) from 1976 to 1996. CCAMLR Sci 5:165–189
Kock K, Kellermann A (1991) Reproduction in Antarctic notothenioid fish. Antarct Sci 3:125–150
Kulesz J (1994) Seasonal biology of Notothenia gibberifrons, N. rossii and Trematomus newnesi, as well as respiration of young fish from Admiralty Bay (King George, South Shetland Islands). Pol Arch Hydrobiol 41:79–102
La Mesa M, Vacchi M, Zunini Sertorio T (2000) Feeding plasticity of Trematomus newnesi (Pisces, Nototheniidae) in Terra Nova Bay, Ross Sea, in relation to environmental conditions. Polar Biol 23:38–45
Leopold M, Damme C van, Veer H van der (1998) Diet of cormorants and the impact of cormorant predation on juvenile flatfish in the Dutch Wadden Sea. J Sea Res 40:93–107
Linkowski T, Presler P, Zukowski C (1983) Food habits of nototheniid fishes (Nototheniidae) in Admiralty Bay (King George Island, South Shetland Islands). Pol Polar Res 4:79–95
Moreno C, Bahamonde N (1975) Nichos alimentarios y competencia por alimento entre Notothenia coriiceps neglecta Nybelin y Notothenia rossii marmorata Fischer en Shetland del Sur, Antarctica. Ser Cient Inst Antárt Chil 3:45–62
Moreno C, Zamorano J, Duarte W (1977) Distribución y segregación espacial de las poblaciones de peces en Bahía South (Isla Doumer, Antartica). Inst Antárt Chil Ser Cient 5:45–58
Richardson M (1975) The dietary composition of some Antarctic fish. Br Antarct Surv Bull 41, 42:113–120
SC-CAMLR (1998) Report of the Working Group on Ecosystem Monitoring and Management. Report of the Seventeenth Meeting of the Scientific Committee, Annex 4. CCAMLR, Hobart
Tarverdiyeva M, Pinskaya I (1980) The feeding of fishes of the families Nototheniidae and Channichthyidae on the shelves of the Antarctic Peninsula and the South Shetlands. J Ichthyol 20:50–60
Vacchi M, La Mesa M, Castelli A (1994) Diet of two coastal nototheniid fish from Terra Nova Bay, Ross Sea. Antarct Sci 6:61–65
Vacchi M, Williams R, La Mesa M (1996) Reproduction in three species of fish from the Ross Sea and Mawson Sea. Antarct Sci 8:185–192
Acknowledgements
We thank M. Favero and P. Silva for their help in the field and the staff of Primavera Station in summer 2000 for logistic support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Casaux, R., Barrera-Oro, E., Baroni, A. et al. Ecology of inshore notothenioid fish from the Danco Coast, Antarctic Peninsula. Polar Biol 26, 157–165 (2003). https://doi.org/10.1007/s00300-002-0463-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00300-002-0463-y