Abstract
A protocol for Agrobacterium-mediated transformation with mannose selection was developed for cotyledon petiole, hypocotyl and leaf explants of tomato (Lycopersicon esculentum L. Mill). More than 400 transgenic plants from three tomato varieties were selected with 1% mannose in combination with 0.1–0.5% glucose. Average transformation frequencies ranged from 2.0 to 15.5% depending on the construct, genotype and type of tissue used for transformation. The highest transformation rate was obtained for hypocotyl explants from tomato variety SG048. The ploidy levels of 264 independent transgenic events and 233 non-transgenic plants regenerated from tissue culture were assessed by flow cytometry. The incidence of polyploids within the total population of transgenic plants varied from 10 to 78% and was not significantly different from the non-transgenic population. The greatest variation in the proportion of polyploids was observed in plants derived from different explant types, both in transgenic and non-transgenic regenerants, across three studied genotypes. Transgenic and non-transgenic plants regenerated from leaves included the highest number of normal diploid plants (82–100%), followed by cotyledon petiole-derived plants (63–78%). Transgenic plants produced from hypocotyls contained 22–58% diploids depending on the genotype used in transformation. Results described in this study demonstrate that, although transformation frequencies for leaf tissue are still lower under current protocols, the high percentage of diploids obtained make leaf tissue an attractive transformation target.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Tomato transformation using Agrobacterium tumefaciens is a well-established technique first described by McCormick et al. (1986) for leaf explants. Since then numerous reports have been published on the recovery of transgenic tomato plants with various transformation rates from different genotypes and different types of explants. Cotyledons have been the favorite explant source in many studies (Shahin et al. 1986; Fillati et al. 1987; Hamza and Chupeau 1993; van Roekel et al. 1993; Ultzen et al. 1995; Frary and Earle 1996; Ling et al. 1998; Hu and Phillips 2001; Ellul et al. 2003; Park et al. 2003), but transgenic plants were also recovered from stem segments (Chyi and Phillips 1987), hypocotyls (Frary and Earle 1996; Park et al. 2003) and leaves (Koornneef et al. 1986; Jacobs and Yoder 1989; Park et al. 2003).
In the majority of experiments, the neomycin phosphotransferase II gene, which confers resistance to the antibiotic kanamycin, was used for selection of stable transformants. Due to decreasing public acceptance of antibiotic and herbicide resistance genes in food crops—although there is no scientific support for this concern—alternative selectable markers and even complete removal of marker genes (Hohn et al. 2001; Penna et al. 2002) are subjects of much research. One alternative approach utilizes the carbohydrate mannose as a selective agent. Many plants cannot utilize mannose as a carbohydrate source, and such cells will not grow when cultured on mannose-containing media. However, when cells are transformed with the phosphomannose isomerase (PMI) gene, they are able to survive by utilizing mannose as a carbohydrate source. Unlike standard antibiotic or herbicide selection, mannose selection does not result in a direct, acute toxic effect, but rather provides a physiological advantage to transformed cells over non-transformed cells (hence the term “positive selection”). Efficient mannose selection has been developed for several monocot crops such as maize, rice and wheat (Negrotto et al. 2000; Wang et al. 2000; Lucca et al. 2001; Wright et al. 2001), and dicot crops such as sugar beet, cassava and orange (Joesrbo et al. 1998; Zhang and Puonti-Kaerlas 2000; Boscariol et al. 2003). For each new crop, mannose selection must be optimized for efficient transgenic plant production (Reed et al. 2001).
No routine positive selection system has previously been developed for tomato. Haldrup et al. (1998) produced transgenic tomato with xylose selection in one experiment and as proof of concept only. Development of an efficient and reproducible system for tomato transformation with mannose selection would present a step forward in recovery of high quality tomato transformants.
A further issue in tomato transformation is the production of plants with an abnormal ploidy level (Jacobs and Yoder 1989; van Roekel et al. 1993; Ellul et al. 2003). The number of these abnormal plants in the transgenic population can significantly influence the efficiency of obtaining “useful” transformants, as a large proportion of the total transformed population can be polyploid. There is no clear understanding of what causes polyploidization in transgenic plants. However, the source tissue for culture appears to be one influencing factor (van den Bulk et al. 1990). Another factor may be prolonged exposure of tissue cultures to plant growth regulators (Joubes and Chevalier 2000).
The aim of this study was to develop an efficient Agrobacterium-mediated transformation technique with mannose selection for different types of tomato explants. We also investigated whether prolonged exposure to tissue culture conditions with our mannose selection scheme would contribute to polyploidization of transgenic plants. Here, we describe the successful application of mannose selection to produce 360 transgenic plants from cotyledon petiole and hypocotyl explants of three elite tomato genotypes. Sixty-six additional transgenic lines were recovered from leaf explants after modifications to our transformation and selection protocol. The ploidy level of transgenic plants selected on mannose was analyzed and compared with the ploidy level of non-transgenic regenerants. We found that the mannose selection procedure described here did not increase the incidence of polyploid plants. Overall, the rate of polyploids among transgenic and non-transgenic plants, produced in vitro, depended mainly on the source of tissue used for regeneration and/or transformation.
Materials and methods
Plant regeneration
Tomato seeds from the elite lines SG048, 00-5223-1 and 00-0498-B were provided by Syngenta Seeds. SG048 is an inbred line of an indeterminate type used in the production of hybrid varieties for the southern European fresh tomato market. 00-5223-1 (red line) is a determinate, fresh market beef type cultivar with large red fruits (>220 g) suitable for growing in the fields in the eastern United States and Mexico. 00-0498-B (golden line) is similar to the red line and is used in both commercial and experimental hybrids in the United States and Mexico.
For regeneration and/or transformation experiments, seeds of each line were surface-sterilized with a 10% Clorox solution (0.525% sodium hypochlorite) for 20 min, rinsed three times with sterile water and then germinated on MSO medium (Murashige and Skoog 1962) containing half strength MS salts (Invitrogen, Grand Island, N.Y.), supplemented with 1% sucrose and solidified with 2 g/l Phytagel (Sigma, St. Louis, Mo.). The seeds were then grown at 25°C, 60 μE m−2 s−1 with a 16 h light/8 h dark photoperiod.
Cotyledon and hypocotyl explants were excised from 7-day-old plants. Leaves were excised from aseptically grown plants 4–6 weeks after subculture. For regeneration, intact cotyledons with petioles (CP), 5 mm hypocotyl segments (H), or 10×20 mm leaf discs (L) were plated onto 15×100 mm plates with regeneration medium 1 (R1; Table 1) and cultured as described above. After 3 weeks, explants were subcultured to regeneration medium 2 (R2; Table 1). After 3 weeks, emerging shoots were excised and transferred to Magenta boxes containing P medium for rooting (P major salts, MS minor salts, MS iron, 100 mg/l myo-inositol, 1 mg/l thiamine, 0.5 mg/l pyridoxine-HCl, 0.5 mg/l nicotinic acid, 1 mg/l glycine, 10 g/l sucrose, solidified with phytagar 7 g/l, pH adjusted to 5.8; Maliga 1982).
Mannose kill curves
To determine the optimum mannose concentration for inhibiting shoot formation, non-transformed CP and H explants were plated on R1 medium (without glucose) containing mannose at concentrations of 0, 2, 5 and 10 g/l in the presence of 0, 1 or 5 g/l sucrose. Ten explants were placed per plate and at least two plates were used for each mannose/sucrose concentration.
Plant transformation
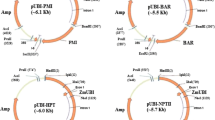
The binary vectors pNOV2147 and pNOV7100 (Fig. 1) were used for transformation. pNOV2147 was constructed by inserting the fluorescent protein gene AmCyan 1 from Anemonia majano (Matz et al. 1999) driven by the cestrum yellow leaf curling virus promoter (CMP; Stavolone et al. 2003) into a pHINK085-based binary vector containing the PMI gene (Miles and Guest 1984), which is also under control of the CMP promoter. pHINK085 is a binary vector with a pVS1 ORI for replication in Agrobacterium (Itoh et al. 1984), and the vector is maintained by spectinomycin selection conferred by the 0.93 kb gene from transposon Tn7 (Fling et al. 1985). pHINK085 also carries the ColE1 ORI for propagation in Escherichia coli. pNOV7100 was created by ligating the Medicago sativa chalcone isomerase (MsCHI) coding region into a plant expression cassette with the Arabidopsis thaliana UBQ3 promoter (AtUBQ3; Callis et al. 1990) driving expression of MsCHI, followed by the nopaline synthase terminator (tNOS). This cassette was inserted into a pHINK085-based backbone containing the PMI gene driven by the CMP promoter. The binary vectors were electroporated into the “disarmed” Agrobacterium strain LBA4404. Details on the cloning of the MsCHI gene will be published elsewhere.
Agrobacterium cultures were grown for 3 days on YP medium (5 g/l yeast extract, 10 g/l peptone, 5 g/l NaCl, 15 g/l agar, pH 6.8) supplemented with 100 mg/l spectinomycin. Before inoculation of plant tissue, Agrobacterium was resuspended in inoculation medium IM [MS salts, B5 vitamins (Gamborg et al. 1968), 0.5 mg/l benzylaminopurine (BAP), 20 g/l sucrose, 10 g/l glucose, pH 5.5] and induced for 3 h with 200 μM acetosyringone. The final bacterial density was adjusted to 5×108 cfu/ml.
CP and H explants were excised from 7-day-old seedlings and incubated in the bacterial suspension for 7 min. Explants were blotted on a sterile paper towel and transferred to co-cultivation medium CCM (half strength MS salts, B5 vitamins, 0.5 mg/l BAP, 20 g/l sucrose, 10 g/l glucose, 200 μM acetosyringone, 4 g/l Phytagel, pH 5.4) for 3 days at 22°C in the dark.
Determination of selection regime
For optimizing selection, CP and H explants were inoculated with an A. tumefaciens LBA4404 strain carrying the pNOV2147 plasmid, and plated onto selection/regeneration medium 1 (S/R1), containing 10 g/l mannose and 5 g/l glucose (Table 1). In other treatments, glucose in S/R1 was substituted with either 5 g/l sucrose, 5 g/l maltose or 5 g/l rhamnose; 20 CP and 20 H explants were plated for each treatment and the experiment was conducted in triplicate. The formation of blue fluorescing calli was assessed for each treatment 30 days after initial plating.
Standard selection protocol
For all further experiments, S/R1 medium (Table 1) supplemented with 5 g/l glucose was used. Explants were cultured on this medium for 6 weeks with 3-week subculture intervals before transfer to selection/regeneration medium 2 (S/R2; S/R1 without indoleacetic acid and glucose decreased to 1 g/l; Table 1). Green shoots were transferred to rooting P5M medium [P medium supplemented with 5 g/l mannose and 300 mg/l carbenicillin (Agrio-Bio, Fla.)].
Modified selection protocol for leaf explants
For leaf transformation, we used leaves from 6-week-old aseptically grown plants. Bacterial inoculation was performed as described above, but the final bacterial concentration was reduced to 5×106 cfu. After 3 days of co-cultivation, explants were washed with a solution of 250 mg/l cefotaxime and 250 mg/l carbenicillin and plated onto modified selection/regeneration medium 1 (S/R1M, contained 2.5 g/l mannose supplemented with 5 g/l glucose; Table 1). After 3 weeks on this medium, explants were transferred to S/R1 medium followed by transfers to S/R2 and P5M according to the standard selection protocol.
Real-time PCR
Genomic DNA for TaqMan was prepared as described by Ingham et al. (2001). PCR primers and probes were designed using Primer Express software (Applied Biosystems, Foster City, Calif.). Primers and probes were purchased from Biosource International (Camarillo, Calif.). For the endogenous control, primers and probes specific to the tomato superoxide dismutase (GenBank accession No. AF034411) were designed. For the transgenes, primers and probes were designed specific to PMI (GenBank accession No. M15380), MsCHI (GenBank accession No. M91079.1) and AmCyan 1 (catalog no. K61001; Clontech, Palo Alto, Calif.). Probes were labeled at their 5′-end with a reporter fluorophore, tetrachloro-6-carboxyfluorescein (TET) for the endogenous control and fluorescein (FAM) for transgenes, and at the 3′-end with the quencher fluorophore tetramethylrhodamine (TAMRA). Sequences of primers and probes used in this work are shown in Table 2.
Polymerase chain reactions were carried out in 384-well reaction plates (Applied Biosystems). Reactions were multiplexed to simultaneously amplify the endogenous and transgene targets. For each sample, 3 μl extracted genomic DNA was combined with 7 μl 2×PCR Mastermix for quantitative PCR (product number P2893, Sigma-Aldrich, St. Louis, Mo.), 7.5 mM MgCl2, 150 nM Sulforhodamine 101 (product number S7635, Sigma-Aldrich), primers to a final concentration of 900 nM each, and probes to a final concentration of 100 nM each. Amplification plates were sealed with optical adhesive seals (product number 431197, Applied Biosystems) and PCR was run in the ABI Prism 7900HT instrument (Applied Biosystems), using the following amplification parameters: 5 min at 95°C followed by 40 cycles of 15 s at 95°C, and 1 min at 60°C. Post-run manipulations of data were performed according to the manufacturer’s instructions. Transgene copy number was calculated utilizing the exported cycle threshold (C t) values as described by Ingham et al. (2001). For simplicity, we estimated copy number as low (1–3 copies of the transgene) or high (>3 copies).
Southern blot analysis
DNA was isolated from leaves using a procedure from Dellaporta et al. (1983). The genomic DNA was digested with XmnI (New England Biolabs, Beverly, Mass.). XmnI cuts were made within the T-DNA on the 5′-side of MsCHI and just outside the T-DNA border sequence on the 3′-side of PMI. This digest excised both MsCHI and PMI genes on the same fragment. pNOV7100 cut with XmnI gives a single 3.8 kb band, while transformed plant tissues gave bands of variable sizes. Restriction fragments were separated by gel electrophoresis on a 0.8% agarose gel and blotted to Hybond-N+ nylon membrane (Amersham Biosciences, Piscataway, N.J.). Hybridization and washing were performed according to the method of Church and Gilbert (1984). DNA for an MsCHI probe was obtained by PCR amplification of the coding sequence and 32P-labeled probes were prepared using the RediPrime II DNA labeling system (Amersham Biosciences).
Determination of ploidy level
The ploidy level of regenerated plants was determined by flow cytometry. Nuclear DNA was isolated from leaves of transformed and seed-grown control plants using a High Resolution Plant Staining Kit and CellTrics disposable filters (Partec, Munster, Germany). Ploidy level of the transgenic plants was estimated by comparing the position of the nuclear DNA peaks on histograms from transgenic plants and seed grown controls. Plants were scored as diploid if their histograms matched those of diploid controls. Histograms of tetraploid plants had a major peak shift to the right as compared to diploid controls. The position of the major peak in tetraploids matched that of a second small peak that corresponded to a doubled DNA content on histograms of control plants.
Detection of cyan fluorescent protein
Transformed explants were examined under a fluorescence stereomicroscope (Leica MZ FLIII) equipped with a Leica cyan fluorescent protein (CFP) filter set: excitation filter, 436/20 nm; emission filter, 480/40 nm. No autofluorescence was detected with these filters.
Statistical analyses
Analysis on the significance of the effect of supplemental carbohydrates on transformation frequencies for CP and H explants was carried out using the SAS (version 8.2) PROC GENMOD procedure. The Wald X 2-test was used to compare the effect of supplemental polysaccharides on production of transgenic callus from each type of explants. X 2 analysis of the factors affecting transformation frequencies and ploidy levels of transgenic and non-transgenic plants was performed using the SAS PROC FREQ procedure. The Cochran-Mantel-Haenszel test was used to analyze the data stratified across three genotypes.
Results and discussion
Mannose selection
To develop a stepwise selection protocol, we assessed the effect of different combinations of mannose and sucrose on shoot formation from CP and H explants of the tomato variety SG048 in initial dose response experiments. Shoot development from both types of explant was totally inhibited on the medium with 5 g/l mannose (Fig. 2). However, in transformation experiments we were unable to recover transgenic shoots on this selection medium. It has been reported that an efficient mannose selection protocol requires the addition of a supplemental carbohydrate. In selection schemes developed for sugar beet, maize, rice and cassava, it was necessary to combine mannose and sucrose for better recovery of transgenic shoots (Joersbo et al. 1998; Negrotto et al. 2000; Zhang and Puonti-Kaerlas 2000; Lucca et al. 2001). We also found that the addition of sucrose to the selection medium modulated the inhibitory effect of mannose on shoot formation in tomato. With the addition of 5 g/l sucrose, a higher concentration of mannose (10 g/l) was required for total repression of shoot formation.
In our study, the type of supplemental polysaccharide added with mannose had an even more significant effect on transformation efficiency (Fig. 3). We compared the effects of sucrose, maltose, glucose and rhamnose on transformation efficiency, based on the number of explants expressing CFP per total number of plated explants. The Wald X 2-test showed that efficiency for hypocotyls was significantly higher in treatments containing glucose, with sucrose being less efficient and maltose and rhamnose being almost non-effective (sucrose: P=0.028; maltose: P<0.001; rhamnose: P<0.001). However, for cotyledon petioles glucose treatments were significantly higher compared to maltose (P<0.0001) and rhamnose (P<0.0001), but not for sucrose (P=0.073). Glucose was chosen for all further experiments. In contrast, for sugar beet, Joersbo et al. (1999) found that a combination of mannose and sucrose provided the optimal selection regime, while the addition of glucose, maltose or fructose entirely eliminated the selective effect of mannose. Sucrose was also used as a supplemental carbohydrate in experiments with other crops (Negrotto et al. 2000; Zhang and Puonti-Kaerlas 2000; Lucca et al. 2001; Boscariol et al. 2003).
In our study, CP explants developed organogenic callus with shoots approximately 3 weeks after culture on S/R1 medium with 10 g/l mannose and 5 g/l glucose. H explants were slower by 1 week in the development of callus. Shoots did not develop from H explants until at least 6 weeks after initiation of selection. Typically, callus clumps with shoots were cut into smaller pieces when transferred to fresh medium, at 3-week intervals, but the origin of all callus lines was tracked in order to avoid producing multiple clones of the same event. Transformed shoots were easily detectable by CFP expression in the leaves and vascular tissue (Fig. 4). In most cases, shoots excised after less than 8 weeks of selection were escapes that did not express CFP. To increase selection stringency, the glucose concentration in the selection medium was reduced to 1 g/l (S/R2) for an additional 8-week round of selection, resulting in approximately 50% of all excised shoots expressing CFP (Fig. 5). The addition of 5 g/l mannose to rooting medium efficiently prevented non-transformed shoots from rooting and further improved selection stringency. Only shoots that developed roots on this medium were transgenics. Transformed tomato plants were ready for analysis 4.5–6 months after initiation of experiments.
We assessed regenerability from CP, H and L explants of two additional genotypes, 00-5223-1 and 00-0498-B, and compared it with regenerabilty of SG048 (Table 3). The regeneration rate from CP and H was very high for all three genotypes, but L explants were highly regenerable only for SG048. The mannose selection scheme developed for CP and H of SG048 was applicable to these other tomato genotypes and we successfully recovered transgenic plants from CP and H explants without any modifications to the selection protocol. However, explants from these lines responded to mannose selection in a different manner than explants from SG048. Organogenic callus developed from both CP and H of 00-5223-1 was less sensitive to mannose, compared to SG048. Only 30% of all 00-5223-1 shoots that developed on S/R2 medium and transferred to rooting on P5M medium were rooted, and only 89% of those rooted were transgenics according to TaqMan analysis (Fig. 5). In contrast, explants of 00-0498-B were very sensitive to mannose. Almost 70% of shoots transferred to P5M medium developed roots and all of these were transgenic (Fig. 5). However, transformation efficiencies for this genotype were low (Table 4). This indicates that although the mannose selection scheme developed for genotype SG048 is applicable to additional tomato genotypes without modifications, optimization may enable recovery of transgenic events at higher frequencies.
TaqMan analysis/copy numbers
DNA samples taken from young rooted plants grown in vitro were analyzed by TaqMan for the presence of the AmCyan and PMI genes in experiments with construct pNOV2147 or for the presence of MsCHI and PMI genes in experiments with construct pNOV7100 (Fig. 1). All 114 putative transgenic lines of SG048 obtained with construct pNOV2147 contained both the AmCyan and PMI genes. In experiments with pNOV7100, 129 putative transformed lines of SG048 and 30 lines of genotype 00-0498-B contained both MsCHI and PMI genes. Only 91 lines of 100 tested from genotype 00-5223-B transformed with pNOV7100 were positive for the presence of the MsCHI and PMI genes, while nine plants recovered after rooting on 5 g/l mannose turned out to be non-transformed escapes. As expected for an Agrobacterium-mediated transformation system, the majority of events were simple transformants with >70% of them containing low copy numbers (1–3) of the transgenes.
Transformation efficiencies
Based on the results of TaqMan analysis, we calculated transformation efficiencies for each experiment. Transformation efficiency was assessed as the percentage of transgenic lines per total number of plated explants. In experiments with tomato variety SG048 and construct pNOV2147, a total of 61 transgenic lines from H and 53 lines from CP explants were recovered with an average transformation efficiency of 15.5 and 13.2%, respectively (Table 4). The transformation efficiency of SG048 was significantly lower in experiments with construct pNOV7100 (P=0.0011). We recovered 96 transgenic lines from H explants and 33 lines from CP explants with average transformation efficiencies of 10.9% and 7.4%, respectively (Table 4). The lower transformation efficiency in these experiments may be explained by overexpression of MsCHI, an enzyme involved in flavonol biosynthesis that could produce detrimental effects on a small number of transgenic plants. Overall, for tomato variety SG048 we recovered more transgenic plants from H explants than from CP. When we transformed genotype 00-5223-1 with pNOV7100, transformation efficiencies were significantly higher for CP explants than for H (10.9 and 5.5%, respectively; P=0.0006) (Table 4). For 00-0498-B, however, average transformation efficiencies were not significantly different for CP and H explants (2.9 and 2.0%, respectively; P=0.3027) (Table 4). Transformation rates were highly influenced by genotype. Although all three varieties were highly regenerable from both CP and H explants, we produced more transgenic plants at a higher transformation rate from SG048 and 00-5223-1 than from 00-0498-B (Table 4). Statistical analysis showed a significant difference in recovery of transgenic plants from the three genotypes tested (P<0.0001). Ellul et al. (2003) also observed different competencies of tomato genotypes for transformation and regeneration, with highly regenerable lines being more recalcitrant to transformation.
Many studies report higher transformation efficiencies with mannose selection compared to selection with antibiotic resistance genes (Joersbo et al. 1998; Wright et al. 2001; Lucca et al. 2001), but for cassava, hygromycin selection was twice as efficient as mannose selection (Zhang and Puonti-Kaerlas 2000). In our research, we found that for all types of explants transformation efficiencies with mannose selection were lower than with hygromycin selection (on average 20%, unpublished data). However, direct comparison was not feasible because of the use of different types of vectors. As suggested by Joersbo et al. (1998), the good results with mannose selection for many crops (maize, rice, wheat) can be explained by the inhibitory, but non-toxic, effect of mannose on non-transformed cells. In many cases, regeneration of transformed cells is inhibited by kanamycin or hygromycin due to the formation of necrotic tissues in adjacent areas. Such deleterious effects of dying cells are not a problem in mannose selection, thus resulting in higher transformation frequencies. For tomatoes, however, this permissive effect of mannose was the main difficulty in optimizing the stringency of selection. We found that tomato explants in the presence of light had the ability to form shoots even on medium lacking carbohydrates (Fig. 2). Perhaps, it is for this reason that non-transformed shoots were easily formed on low stringency selection medium, which was optimal for high frequencies of transformation. To increase stringency of selection, multiple rounds of subcultures were required for 4–6 months, thus increasing the total timeline for production of transgenic tomato plants.
Southern blot analysis
Selected TaqMan-positive plants transformed with pNOV7100 were analyzed by Southern hybridization using XmnI-digested genomic DNA and an MsCHI probe (Fig. 6). This analysis confirmed the accuracy of the TaqMan assay in 100% of the plants. No signal was detected in the DNA from non-transgenic plants used as negative controls. As there is only one XmnI site in the T-DNA, the second XmnI site should come from the tomato genome, releasing a fragment containing both the PMI and MsCHI genes. Thus the number of hybridizing bands indicates the number of integration sites in the nuclear genome of transgenic plants. Since for Southern analysis we selected only the lines with low copy number according to the TaqMan assay, 15 of the 16 lines we tested had only one integration site and 1 line had two copies of the transgene.
Southern blot analysis of genomic DNAs from T0 transgenic tomato plants hybridized with an MsCHI cDNA probe: Lanes: P plasmid DNA, 1 genomic DNA from non-transformed tomato plant (control), 2–17 genomic DNA from independent tomato lines transformed with pNOV7100. Numbers on right indicate size standards (kb)
Transgenic plants that were diploid and had low copies of the PMI gene were selfed to obtain T1 progeny. A total of six independently transformed lines were analyzed by TaqMan analysis for segregation of the MsCHI gene. All lines tested had a typical Mendelian segregation ratio of 3:1, thus confirming stable inheritance of the transgenes in the progeny (data not shown).
Ploidy level of transgenic plants
All transgenic plants were analyzed by flow cytometry to evaluate their ploidy level. We found that a higher proportion of transgenic plants recovered from CP explants were diploids—68.2% of SG048, 72.9% of 00-5223-1 and 63.6% of 00-0498-B—compared to 34.9, 22.4 and 57.9% from H explants (Table 5). We did not estimate the proportion of polysomatic cells in different explant types of parental lines used for transformation, although parental plants were diploids based on analysis of leaf samples by flow cytometry.
To ascertain whether mannose selection contributes to polyploidization of resulting transgenic plants, we compared the ploidy levels of transgenic plants with the ploidy of non-transgenic plants regenerated from CP and H explants of all three genotypes used for transformation. For two of the genotypes, SG048 and 00-0498-B, we recovered more transgenic diploids from CP and H explants as compared to non-transgenic, indicating that our mannose protocol did not contribute to an increase in recovery of polyploid plants for these genotypes (Table 5). On the other hand, for 00-5223-1 we observed a slightly higher rate of diploid plants among non-transgenic regenerants compared to transformed plants both for CP and H explants (78.1% vs 72.9 and 36.2% vs 22.4%, respectively). However, statistical analysis demonstrated that there were no significant differences in the proportion of diploids between transgenic and non-transgenic regenerants for each genotype tested (SG048: P=0.66; 00-5223-1: P=0.25; 00-0498-B: P=0.23). Over all three genotypes, the Cochran-Mantel-Haenszel tests shows no significant differences in the proportion of diploids between transgenic and non-transgenic populations (P=0.505).
In the genotype SG048 it was easy to distinguish diploid from tetraploid plants grown in vitro based on leaf morphology. Tetraploids resembled diploid plants but with larger and darker green leaves. The differences were more pronounced in plants grown in the greenhouse. Tetraploid plants were shorter and with more succulent dark green leaves compared to the normal diploid plants. Several hexaploid plants regenerated from all three genotypes showed severe abnormalities when grown in vitro. None of these were transferred to the greenhouse.
Variation in ploidy levels of tomato plants obtained in transformation and regeneration experiments has been observed previously (Jacobs and Yoder 1989; van Roekel et al. 1993; Ellul et al. 2003). Recent studies link polyploidization of plants coming from in vitro culture to the phenomenon of endoreduplication (repeated cycles of DNA synthesis without mitosis; Joubes and Chevalier 2000). It is important to understand the role of endoreduplication in cellular competence for regeneration and transformation in order to reduce somaclonal variation and polyploidization in tissue cultures. A number of studies have investigated the occurrence and distribution of polysomatic cells resulting from the process of endoreduplication in different types of tissues and different organs of tomato (van den Bulk et al. 1990; Smulders et al. 1994, 1995) and in other plants (Mishiba and Mii 2000; Kudo and Kimura 2001a, b). Studies by van den Bulk et al. (1990) and Smulders et al. (1994) have shown that tomato plants of cv. Moneymaker contained a mixture of diploid and polyploid cells, with leaves having the highest proportion of 2C nuclei (80–90%), followed by cotyledons (50–60%) and hypocotyls (30%). Van den Bulk et al. (1990) also demonstrated that explants with a polysomatic nature generated polyploid plants. There was a correlation between the degree of polyploidization of explant tissue and the proportion of polyploid regenerants. The van den Bulk study found the highest proportion of polyploid regenerants being produced from H explants, an observation supported by our research. Across all three genotypes tested, regenerants from H explants contained the highest proportion of tetraploids (Table 5; >60%). Statistical analysis of non-transformed regenerants derived from CP, H or L explants showed significant difference in the ratio of diploid vs tetraploids for each genotype tested (SG048: P<0.0001; 00-5223-1: P<0.0001; 00-0498-B: P=0.0023). Over all three genotypes, the Cochran-Mantel-Haenszel tests showed highly significant differences in the proportion of diploids between different types of explants (P<0.0001). Ellul et al. (2003), on the other hand, observed genotype dependency in ploidy variation in cotyledons of five tomato cultivars with the proportion of nuclei corresponding to 2C level ranging from 40 to 60%.
Prolonged exposure to plant growth regulators may be another factor linked to polyploidization of in vitro cultures. Several studies demonstrated that hormonal treatments of cultured cells were able to modify the status of dividing cells and induce extra rounds of DNA replications (Joubes and Chevalier 2000, and references therein). Van Roekel et al. (1993) hypothesized that prolonged exposure to tissue culture conditions could result in changes in ploidy levels in tomato transformants. Ellul et al. (2003) reported that the rate of tetraploid plants recovered from Agrobacterium-mediated transformation experiments was procedure dependent, and more tetraploid plants were recovered from explants inoculated with lower bacterial concentration though at overall improved transformation rate. Our study showed that neither prolonged exposure to tissue culture conditions (6 months), nor infection with Agrobacterium contributed to polyploidization of transformed plants for three tomato genotypes tested.
Leaf tissue was reported to have the highest rate of 2C nuclei, and regenerants produced from L explants contained the highest proportion of normal diploid plants (van den Bulk et al. 1990). When we tested regenerability from several tomato genotypes we found that for many genotypes (Table 3—data presented for only three genotypes) L explants were the least regenerable. Flow cytometry analysis of these leaf non-transgenic regenerants revealed the highest proportion of diploid plants, across all explant types tested, with 85.2% of SG048, 81.8% of 00-5223 and all four regenerated plants of 00-0498-B being diploids (Table 5). Therefore, we applied our transformation protocol with mannose selection to L explants.
Optimizing transformation and mannose selection for leaf explants
Since we observed high regenerability (>80%) in SG048 L explants, this line was chosen for transformation. However, no transgenic events were produced in ten experiments with our standard transformation and selection protocol, and we had to modify it to produce transformants. We observed high sensitivity of L explants both to Agrobacterium inoculation and to high mannose concentration at the initial selection step. Therefore, modifications were made to include lower bacterial density during inoculation, an antibiotic wash after co-cultivation and lower mannose concentration in the initial selection medium. With the modified protocol we recovered 66 transgenic events from 1,572 L explants with an average transformation rate of 4.2%. When the ploidy of 40 transformed plants produced after transformation of L explants was checked, we found that 90% of them were diploids (Table 5).
Leaf disk transformation with kanamycin selection has been reported previously (McCormick et al. 1986; Koornneef et al. 1986; Jacobs and Yoder 1989; Park et al. 2003). McCormick et al. (1986) also noticed variation in the regeneration response from leaf disks in different commercial tomato lines, with the best regenerable lines producing the highest number of transgenics. However, even from less regenerable lines they were able to recover transformed shoots after protocol modifications. Davis et al. (1991) found L explants formed transgenic callus as easily as cotyledons. Park et al. (2003) observed lower transformation rates for L explants compared to cotyledons and hypocotyls, though transgenic plants were recovered from all types of explants across five tested genotypes. Many recent studies on tomato transformation, however, have reported almost exclusively the use of cotyledon explants in their experiments (Ultzen et al. 1995; Ling et al. 1998; Hu and Phillips 2001; Ellul et al. 2003).
Our study shows that it is worthwhile to reconsider L explants as alternative targets for tomato transformation even with lower organogenic and transformation competence compared to cotyledons, the more common transformation target. High rates of normal diploid plants among leaf-derived transgenics may compensate for lower transformation frequencies.
Conclusions
We have developed a reliable and efficient mannose selection protocol for various tomato explants: CP, H and L. Transformed tomato plants were recovered from mannose selection from three tomato genotypes, SG048, 00-5223-1 and 00-0498-B. We demonstrated that our mannose selection protocol did not increase polyploidization of tomato transformants compared to in vitro regeneration for two tomato genotypes, even though selection required a much longer (2–3×) period in tissue culture. The rate of polyploid plants among the total population of transgenics depended mostly on the source of tissue used for transformation. We considered leaves to be attractive targets for transformation experiments, even though the transformation rate was lower compared to CP and H explants, because over 90% of transgenic plants derived from leaves were diploid.
Abbreviations
- BAP:
-
Benzylaminopurine
- MS:
-
Murashige-Skoog
- MsCHI:
-
Medicago sativa chalcone isomerase
- PMI:
-
Phosphomannose isomerase
References
Boscariol RL, Almeida WAB, Derbyshire MTVC, Mourao Filho FAA, Mendes BMJ (2003) The use of the PMI/mannose selection system to recover transgenic sweet orange plants (Citrus sinensis L. Osbeck). Plant Cell Rep 22:122–128
Bulk RW van den, Loffler HJM, Lindhout WH, Koornneef M (1990) Somaclonal variation in tomato: effect of explant source a comparison with chemical mutagenesis. Theor Appl Genet 80:817–825
Callis J, Raasch JA, Vierstra RD (1990) Ubiquitin extension proteins of Arabidopsis thaliana: structure, localization and expression of their promoters in transgenic tobacco. J Biol Chem 265:12486–12493
Church GM, Gilbert W (1984) Genomic sequencing. Proc Natl Acad Sci USA 81:1991–1995
Chyi YS, Phillips GC (1987) High efficiency Agrobacterium-mediated transformation of Lycopersicon based on conditions favorable for regeneration. Plant Cell Rep 6:105–108
Davis ME, Lineberger RD, Miller AR (1991) Effects of tomato cultivar, leaf age, and bacterial strain on transformation by Agrobacterium tumefaciens. Plant Cell Tissue Organ Cult 24:115–121
Dellaporta SL, Wood J, Hicks JB (1983) A plant DNA minipreparation: version II. Plant Mol Biol Rep 1:19–21
Ellul P, Garcia-Sogo B, Pineda B, Rios G, Roig LA, Moreno V (2003) The ploidy level of transgenic plants in Agrobacterium-mediated transformation of tomato cotyledons (Lycopersicon esculentum L. Mill.) is genotype and procedure dependent. Theor Appl Genet 106:231–238
Fillati JJ, Kiser J, Rose R, Comai L (1987) Efficient transfer of a glyphosate tolerance gene into tomato using a binary Agrobacterium tumefaciens vector. Biotechnology 5:726–730
Fling ME, Kopf J, Richards C (1985) Nucleotide sequence of the transposon Tn7 gene encoding an aminoglycoside-modifying enzyme, 3″(9)-0-nucleotidyltransferase. Nucleic Acids Res 13:7095–7106
Frary A, Earle ED (1996) An examination of factors affecting the efficiency of Agrobacterium-mediated transformation of tomato. Plant Cell Rep 16:235–240
Gamborg OL, Miller RA, Ojima K (1968) Nutrient requirements of suspension cultures of soybean root cells. Exp Cell Res 50:150–158
Haldrup A, Petersen SG, Okkels FT (1998) The xylose isomerase gene from Thermoanaerobacterium thermosulfurogenes allows effective selection of transgenic plant cells using d-xylose as the selection agent. Plant Mol Biol 37:287–296
Hamza S, Chupeau Y (1993) Re-evaluation of conditions for plant regeneration and Agrobacterium-mediated transformation from tomato (Lycopersicon esculentum). J Exp Bot 44:1837–1845
Hohn B, Levy AA, Puchta H (2001) Elimination of selection markers from transgenic plants. Curr Opin Biotechnol 12:139–143
Hu W, Phillips GC (2001) A combination of overgrowth-control antibiotics improves Agrobacterium tumefaciens-mediated transformation efficiency for cultivated tomato (L. esculentum). In Vitro Cell Dev Biol Plant 37:12–18
Ingham DJ, Beer S, Money S, Hansen G (2001) Quantitative real-time PCR assay for determining transgene copy number in transformed plants. BioTechniques 31:132–140
Itoh Y, Watson JM, Haas D, Leisinger T (1984) Genetic and molecular characterization of the Pseudomonas plasmid pVS1. Plasmid 11:206–220
Jacobs JP, Yoder JI (1989) Ploidy levels in transgenic plants determined by chloroplast number. Plant Cell Rep 7:662–664
Joersbo M, Donaldson I, Kreiberg J, Petersen SG, Brunstedt J, Okkels FT (1998) Analysis of mannose selection used for transformation of sugar beet. Mol Breed 4:111–117
Joersbo M, Petersen SG, Okkels FT (1999) Parameters interacting with mannose selection employed for the production of transgenic sugar beet. Physiol Plant 105:109–115
Joubes J, Chevalier C (2000) Endoreduplication in higher plants. Plant Mol Biol 43:735–745
Koornneef M, Hanhart C, Jongsma M, Toma I, Weide R, Zabel P, Hille J (1986) Breeding of a tomato genotype readily accessible to genetic manipulation. Plant Sci 45:201–208
Kudo N, Kimura Y (2001a) Flow cytometric evidence for endopolyploidy in seedlings of some Brassica species. Theor Appl Genet 102:104–110
Kudo N, Kimura Y (2001b) Patterns of endopolyploidy during seedling development in cabbage (Brassica oleracea L.). Ann Bot 87:275–281
Ling H-Q, Kriseleit D, Ganal MW (1998) Effect of ticarcillin/potassium clavulanate on callus growth and shoot regeneration in Agrobacterium-mediated transformation of tomato (Lycopersicon esculentum Mill.). Plant Cell Rep 17:843–847
Lucca P, Ye X, Potrykus I (2001) Effective selection and regeneration of transgenic rice plants with mannose as selective agent. Mol Breed 7:43–49
Maliga P (1982) Cell culture procedures for Nicotiana plumbaginifolia. Plant Mol Biol News 3:88–94
Matz MV, Fradkov AF, Labas YA, Savitsky AP, Zaraisky AG, Markelov ML, Lukyanov SA (1999) Fluorescent proteins from non bioluminescent Anthozoa species. Nat Biotechnol 17:969–973
McCormick S, Niedermeyer J, Fry J, Barnason A, Horsch R, Fraley R (1986) Leaf disc transformation of cultivated tomato (L. esculentum) using Agrobacterium tumefaciens. Plant Cell Rep 5:81–85
Miles JS, Guest JR (1984) Nucleotide sequence and transcriptional start point of the phosphomannose isomerase gene (manA) of Escherichia coli. Gene 32:41–48
Mishiba K, Mii M (2000) Polysomaty analysis in diploid and tetraploid Portulaca grandiflora. Plant Sci 156:213–219
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15:473–497
Negrotto D, Jolley M, Beer S, Wenck AR, Hansen G (2000) The use of phosphomannose-isomerase as a selectable marker to recover transgenic maize plants (Zea mays L.) via Agrobacterium transformation. Plant Cell Rep 19:798–803
Park SH, Morris JL, Park JE, Hirschi KD, Smith RH (2003) Efficient and genotype-independent Agrobacterium-mediated tomato transformation. J Plant Physiol 160:1253–1257
Penna S, Sagi L, Swennen R (2002) Positive selectable marker genes for routine plant transformation. In Vitro Cell Dev Biol Plant 38:125–128
Reed JN, Privalle LS, Powell MJ, Meghji M, Dawson J, Dunder EM, Suttie J, Wenck AR, Launis K, Kramer C, Chang Y-F, Hansen G, Wright M (2001) Phosphomannose isomerase: an efficient selectable marker for plant transformation. In Vitro Cell Dev Biol Plant 37:127–132
Roekel JSC van, Damm B, Melchers LS, Hoekema A (1993) Factors influencing transformation frequency of tomato (Lycopersicon esculentum). Plant Cell Rep 12:644–647
Shahin EA, Sukhapinda K, Simpson RB, Spivey R (1986) Transformation of cultivated tomato by a binary vector in Agrobacterium rhizogenes: transgenic plants with normal phenotypes harbor binary vector T-DNA, but not Ri-plasmid T-DNA. Theor Appl Genet 72:770–777
Smulders MJM, Rus-Kortekaas W, Gilissen LJW (1994) Development of polysomaty during differentiation in diploid and tetraploid tomato (Lycopersicon esculentum) plants. Plant Sci 97:53–60
Smulders MJM, Rus-Kortekaas W, Gilissen LJW (1995) Natural variation in patterns of polysomaty among individual tomato plants and their regenerated progeny. Plant Sci 106:129–139
Stavolone L, Kononova M, Pauli S, Ragozzino A, de Haan P, Milligan S, Lawton K, Hohn T (2003) Cestrum yellow leaf curling virus (CmYLCV) promoter: a new strong constitutive promoter for heterologous gene expression in a wide variety of crops. Plant Mol Biol 53:703–713
Ultzen T, Gielen J, Venema F, Westerbroek A, de Haan P, Tan M-L, Schram A, van Grinsven M, Goldbach R (1995) Resistance to tomato spotted wilt virus in transgenic tomato hybrids. Euphytica 85:159–168
Wang AS, Evans RA, Altendorf PR, Hanten JA, Doyle MC, Rosichan JL (2000) A mannose selection system for production of fertile transgenic maize plants from protoplasts. Plant Cell Rep 19:654–660
Wright M, Dawson J, Dunder E, Suttie J, Reed J, Kramer C, Chang Y, Novitzky R, Wang H, Artim-Moore L (2001) Efficient biolistic transformation of maize (Zea mays L.) and wheat (Triticum aestivum L.) using the phosphomannose isomerase gene, pmi, as the selectable marker. Plant Cell Rep 20:429–436
Zhang P, Puonti-Kaerlas J (2000) PIG-mediated cassava transformation using positive and negative selection. Plant Cell Rep 19:1041–1048
Acknowledgements
We are grateful to Natasha Kornegay for assisting with tomato transformation experiments, Yu-Yu Bai for helping with statistical analysis, Mary Fielder for performing the TaqMan assay and Brian Potter for taking care of plants in the greenhouse.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by E.D. Earle
Rights and permissions
About this article
Cite this article
Sigareva, M., Spivey, R., Willits, M.G. et al. An efficient mannose selection protocol for tomato that has no adverse effect on the ploidy level of transgenic plants. Plant Cell Rep 23, 236–245 (2004). https://doi.org/10.1007/s00299-004-0809-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-004-0809-8