Abstract
To study vertebral fracture (VF) prevalence and the scanographic bone attenuation coefficient of the first lumbar vertebra (SBAC-L1) on computed tomography scans (CT-scans) in systemic sclerosis patients. This monocentric retrospective study included patients followed from 2000 to 2014 and fulfilling ACR/EULAR 2013 criteria for systemic sclerosis and who underwent a thoracic or thoraco-abdomino-pelvic CT-scan during their follow-up. Clinical characteristics for sclerosis and osteoporosis risk factors were collected. For CT-scan, the VFs were determined according to Genant’s classification, the SBAC-L1 was measured in Hounsfield Units (HU), and a SBAC-L1 ≤ 145 HU (fracture threshold) defined patients at VF risk. Predictive factors for SBAC-L1 ≤ 145 HU were studied. A total of 70 patients were included [mean age, 62.3 (± 15.6) years, women 88.5%, diffuse sclerosis 22.9% (n = 16)]. On CT-scans, three VFs were detected in three patients (4.3%). The mean SBAC-L1 was 157.26 HU (± 52.1), and 35 patients (50%) presented a SBAC-L1 ≤ 145 HU. In multivariate analysis, only age (especially patients older than 63 years, OR = 1.08, CI 95% 1.04–1.13, p = 0.001) and calcinosis (OR = 6.04, CI 95% 1.27–28.70, p = 0.02) were independently associated with a SBAC-L1 ≤ 145 HU. On a large sample of patients with systemic sclerosis, the VF prevalence on CT-scan was low (4.3%) while 50% of the patients presented a SBAC-L1 ≤ 145 HU. Interestingly, the presence of calcinosis, periarticular calcifications or acro-osteolysis was linked with low SBAC-L1 and should lead to an osteoporosis screening, especially for patients under 63 years old.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Systemic sclerosis is a multi-systemic disease and its prognosis depends on cardiac, pulmonary and vascular impairments. The improvement of life expectancy (survival at 10 years, 80–90% for the limited cutaneous disease and 60–80% for the systemic disease) [1,2,3] requires better management of comorbidities [4], especially for osteoporosis in this population, which is mainly composed of women (3–8 women for every man) with a disease beginning between 45 and 64 years. Until now, osteoporosis screening in sclerosis was conducted in a subgroup of patients for whom Dual Energy X-ray Absorptiometry (DEXA) and /or radiographic assessment were available. The studies based on DEXA found a large range of osteoporosis prevalence varying from 3–60% [5,6,7]. In comparison with the general population, some authors found an increased risk for osteoporosis, but other authors did not confirm this increased risk [5,6,7,8,9,10,11,12,13,14,15,16,17,18,19,20,21,22]. Some studies comparing patients with rheumatoid arthritis and with systemic sclerosis (adjusted for age and sex or without differences between the two groups), demonstrated a similar risk of osteoporosis [5, 11, 21]. Concerning the vertebral fractures (VFs) on radiography, studies showed also a varying prevalence of VF ranging from 0 to 38% [5, 7, 18, 20, 23] with a relative risk of 1.78 compared to healthy paired patients [23]. Osteoporosis in this population was multifactorial and the proposed physiopathological mechanisms were: loss of mobility and strains on bone in severe systemic sclerosis [6, 10, 11, 15, 19,20,21,22], displacement of calcium stocked in bone towards vascular and soft tissues (which is responsible for calcinosis), periarticular calcifications and ectopic calcifications [11, 19], and finally, digestive malabsorption related or not to a loss of vitamin D activity [6, 10, 11, 15, 19, 21, 22].
Thoracic or thoraco-abdomino-pelvic (TAP) computed tomography scan (CT-scan), classically performed in the follow-up of sclerosis and mainly to evaluate lung involvement, allowed both an evaluation of VF and of the first lumbar vertebra bone attenuation coefficient (SBAC-L1) [24]. Recently, Pickhardt et al. showed that a measurement of the SBAC-L1 was a good alternative to identify patients at risk of VF in general population. These authors demonstrated that a SBAC-L1 ≤ 145 HU (Hounsfield Units) permitted to identify 96.6% of the patients with VF, whereas DEXA [with a T score ≤ − 2.5 standard deviation (SD)] identified only 39% of them. The primary objective of this study was to evaluate the prevalence of VF on CT-scan and to measure the SBAC-L1 and its reliability (intra- and inter-reader) and to determine the proportion of patients at the fracture threshold (≤ 145 HU) in a large sample of sclerosis patients who were assessed or not by DEXA. The secondary objectives were to determine the prevalence of osteoporotic risk factors and to identify factors associated with VF and SBAC-L1 ≤ 145 HU in this population.
Materials and methods
Population
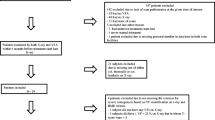
This descriptive analytical and retrospective study was conducted in patients who were followed at Nancy University Hospital between January 2000 and April 2014 for diffuse or limited systemic sclerosis. We chose medical records with codes M34.0, M34.1, M34.8 and M34.9 of the International Classification of Diseases and corresponding to the classification of ACR/EULAR (American College of Rheumatology/European League Against Rheumatism) 2013 for systemic sclerosis. Patients were included if they had a thoracic TAP CT-scan during the follow-up. Patients were excluded if they did not satisfy ACR/EULAR criteria for systemic sclerosis and without thoracic or TAP CT-scan during their follow-up.
Demographic (age, gender, and disease duration), clinical (medical history, arthralgia, modified Rodnan score, and Raynaud Syndrome) and biological [C-reactive protein (CRP), erythrocyte sedimentation rate, antinuclear antibodies (ANA), phosphoremia and calcemia] characteristics and treatments concerning sclerosis were collected on all of the medical records (anterior to the CT-scan). A biological inflammation was defined either a CRP ≥ 5 mg/L or a ESR ≥ age/2 for a men and (age + 10)/2 for women. The selected complementary exams (radiographs of the hands, Pulmonary Function Testing (PFT) and cardiac echography) had to be conducted one year before or after CT-scan evaluation. Hand structural damages were studied on the radiographs of the hands and according to the Erre Score [25] by a senior rheumatologist with much experience scoring. Pulmonary involvement was studied functionally with PFT and morphologically with thoracic or TAP CT-scan [non-specific interstitial pneumonia (NSIP), usual interstitial pneumonia (UIP) and interstitial lung disease (ILD)] by a senior radiologist (MD). Pulmonary arterial hypertension (PAH) was detected on cardiac echography and confirmed by right heart catheterization.
For the assessment of osteoporosis, clinical risk factors (gender, age, chronic biological inflammation, smoking, and corticosteroid therapy) were recorded during the entire follow-up. Data about DEXA and anti-osteoporotic treatments were recorded 2 years before or after the CT-scan. For DEXA, osteoporosis was retained for a T score ≤ − 2.5 SD on the spine and osteopenia with − 2.5 < T score ≤ − 1 SD.
Morphological and densitometric scanographic bone evaluation (scanographic bone attenuation coefficient of L1: SBAC-L1)
During the period of follow-up, all of the CT-scans were conducted in the same department of Radiology and thus 43 TAP, 26 thoracic and one lumbar CT-scans were selected. From the axial acquisitions, sagittal reconstructions in bone window permitted a study of the vertebral morphology from C7 to L1 for thoracic CT-scan and from C7 to S1 for TAP CT-scan based on OSIRIX software (v6.5.1–64 bits).
-
VF assessment The VFs were analysed manually, on sagittal sections of CT-scan, according to an adaptation of the Genant’s classification, which was usually used on spine radiographs [26]. The vertebrae were scored on visual inspection as normal (grade 0), mildly deformed (grade 1, 20–25% reduction of the anterior, middle or posterior height), moderately deformed (grade 2, 25–40% reduction in any height) or severe deformed (grade 3, over 40 reduction in any height). The grade was determined on the most severe lesion observed on one of the sections
-
SBAC-L1 measurement The SBAC-L1 study was conducted on L1 axial sections through the pedicles on the bone window. The largest elliptical Region of Interest (ROI) was drawn in trabecular bone and provided the average bone mineral density (in HU). If there was a VF on L1 vertebra, the measurement could be performed on the adjacent vertebrae (CT-scan performed similarly from T12 to L5 [24]). The intra-reader reliability was evaluated on two measurements performed with a delay of 2 weeks and using the same reader for all patients. The inter-reader reliability was evaluated on 30 CT-scans by two independent readers.
-
Choice of the threshold Pickhardt et al. showed that a SBAC-L1 ≤ 145 HU identified 96.6% of the patients with VF in a general population, whereas DEXA (with T score ≤ − 2.5 SD) identified only 39% of the VF in the same population. A SBAC-L1 ≤ 145 HU allowed a good compromise between sensitivity and specificity to identify patients at risk of VF in a population with an unknown risk of osteoporosis. Other thresholds were also studied: 110 HU with moderate sensitivity (52.1%) and excellent specificity (91.3%) in a population without risk factors of osteoporosis and 160 HU with an excellent sensitivity (90%) but a moderate specificity (52.3%) in a population with risk of osteoporosis [24].
Ethics approval and consent to participate
All data were available from usual care in patients with sclerosis. The ethics committee of Nancy hospital agreed with this study (referral file number 166). This study was designed in accordance with the general ethical principles outlined in the Declaration of Helsinki, patients gave their consent to use their medical data when they were cared at the university hospital.
Statistical analysis
Both descriptive and comparative analyses were conducted by accounting for the nature and distribution of the variables. Qualitative variables were described with frequencies and the percentage; quantitative variables were evaluated with the mean ± SD (standard deviation) or with the median and interquartile range (IQR). The Kolmogorov–Smirnov test showed that among the continuous demographic and clinical variables, only age followed a normal distribution as a result the Student’s t test was used for the age and the Mann–Whitney U test for the other variables. For qualitative variables, the Chi-square test and/or the exact calculation of Fisher was used. The Spearman’s rho allowed for the analysis of the correlations for quantitative variables. The paired t test was used to compare the differences used in Bland–Altman analysis to 0 for intra-reader and inter-reader-reliability with concordance verified by the Cohen’s test. Significant results (univariate and multivariate analysis) are presented with the odds ratio (OR) and its 95% confidence interval (CI 95%). A logistic regression was performed for the variables significantly associated with the SBAC-L1 145 HU threshold and not correlated with each other (age, calcinosis and the binary outcome being SBAC-L1 145 HU). To identify the profiles of the subjects, a classification system displayed in a decision tree, i.e., the Chi-square automatic interaction detection (CHAID), was used to select the successive split variables associated with the SBAC-L1 145 HU threshold.
The risk α was established as 0.05, except for the inter-reader reliability study, where it was established at 0.01 given the repetition of the tests. IBM SPSS Statistics V22 was the software used for the data analysis.
Results
Demographic and clinical data
One hundred patients were selected from our request: 70 of them met the ACR/EULAR 2013 criteria and all of them were evaluated by a CT-scan during their follow-up. The population characteristics are presented in Table 1. Sixteen patients (22.9%) had diffuse cutaneous disease and 54 (77.1%) had limited cutaneous disease. Concerning the known clinical risk factors of osteoporosis: 47 women (67.1%) were menopausal (> 50 years), 17 (24.3%) patients smoked, 20 (28.6%) presented biological inflammation and 28 (40%) received corticosteroid treatment (average posology: 3.8 ± 6.6 mg/day). The differences between patients according to sclerosis clinical forms (diffuse or limited) are presented in Table 1.
Osteoporosis assessment
Sixty patients (85.7%) had at least one clinical risk factor for osteoporosis. The biology (calcemia and phosphoremia) was normal for all of the patients. During their follow-up, 30 patients (42.8%) had a DEXA: 5 (16.7%) were classified with osteoporosis, eight (26.7%) with osteopenia and 17 (56.7%) with normal bone density. Finally, for the 70 patients with systemic sclerosis, 18 patients (25.7%) received a supplementation with calcium and/or vitamin D and 10 (14.3%) a specific treatment for osteoporosis (antiresorptive therapy). For the 10 patients who received antiresorptive therapy, two had vertebral fracture and the others presented low T-score values on DEXA (≤ − 2.5 SD) or high risk of fracture at 10 years (FRAX).
Vertebral fracture (VF) evaluation on CT-scan
For CT-scan, three women presented a vertebral fracture (4.3%): two on dorsal localizations (T10 and T11) and one on the lumbar spine (L2). The 3 VFs were classified as Genant 2 (n = 2) and Genant 3 (n = 1). Patients with VF were older (71.7 years versus 62.3 years) and had a lower disease duration (7 years versus 10.2 years). DEXA was available for only one of these three patients and was normal, while her SBAC-L1 was measured at 102.5 HU.
Reliability of the SBAC-L1 measurement
Intra-reader (n = 70) and inter-reader (n = 30) reliabilities are represented in Fig. 1a, b, respectively. For each of the analyses, the mean difference between the two measurements did not differ from 0 and were − 0.79 HU (t = − 1.07, p = 0.29) and 0.20 HU (t = 0.11, p = 0.91). In each analysis, the Bland–Altman method showed 95% confidence intervals of the mean of the differences with limits that could be considered acceptable in the context under study. We do not observe a trend in the differences as these differences seem to be constant according to the increasing values of the x-axis.
The Bland–Altman method graph for intra- (a) and inter-reader (b) reliability. The difference between the two measurements for each observation is on the y-axis (HU), the mean of the two measurements for each observation is on the x-axis (HU), and the mean of the differences between the two measurements with the 95% confidence interval is represented by the solid lines; 95% confidences intervals corresponding to these three values (average of the differences and its two associated limits) are the dotted lines
Considering the threshold at 145 HU, Cohen’s Kappa resulted in an intra-reader value of 0.94 (p = 0.0001) with 68 concordant observations out of 70 and an inter-reader value of 0.93 (p = 0.0001) with 29 concordant observations out of 34. For the measurements available for the three readings (n = 30), the intra-class correlation coefficient was equal to 0.99 (CI 95% 0.995–0.998, p < 0.0001).
Evaluation of the SBAC-L1
The mean of the two intra-reader values was used to calculate the SBAC-L1. In our patients, the mean SBAC-L1 was 157.3 ± 52.2 HU and 35 patients (50%) had an SBAC-L1 ≤ 145 HU. The SBAC-L1 of the three patients with VF were 102.5, 131.4 and 171 HU.
Risk factors associated with a SBAC-L1 ≤ 145 HU
For the univariate analysis (Table 2), a diffuse form of sclerosis was not significantly associated with a SBAC-L1 ≤ 145 HU in comparison to the localized form. Other known osteoporotic risk factors such as gender, smoking or corticosteroid therapy were not significantly associated with a low SBAC-L1. However, age was significantly associated with a SBAC-L1 ≤ 145 HU (p = 0.0001). The mean age was 70 years for patients with SBAC-L1 ≤ 145HU versus 54.7 years for patients with SBAC-L1 > 145 HU. The odds ratio associated with age was 1.09 (CI 95% 1.40–1.13), which corresponded to a 9% increased risk of SBAC-L1 ≤ 145 HU for one additional year. Calcinosis and periarticular calcifications were also significantly associated with a SBAC-L1 ≤ 145 HU (p = 0.004 and 0.03, respectively). Their presence significantly increased the risk of SBAC-L1 ≤ 145 HU with an OR = 6.30 (CI 95% 1.61–24.75) and 3.22 (CI 95% 1.06–9.77), respectively.
The presence of a highly statistically significant relationship between calcinosis and periarticular calcifications (75% of those with calcinosis (12/16) also had periarticular calcifications (12/20) p = 0.0001) on one hand, and between periarticular calcifications and age on the other hand (mean 58.5 ± 14.2 years in the absence of periarticular calcifications versus 72.1 ± 14.9 years, p = 0.001), did not allow us to conduct a logistic regression association for them. Age and calcinosis showed no significant relationship (mean 60.9 ± 15.7 years in the absence of calcinosis versus 67.3 ± 14.8 years, p = 0.2). A logistic regression with age and calcinosis and the binary outcome being SBAC-L1 145 HU, showed an overall significant result (p = 0.0001, Hosmer–Lemeshow p = 0.3, 71.4% ranked) with age and calcinosis, which remained significant when considered together [age, OR = 1.08 (CI 95% 1.04–1.13, p = 0.001); calcinosis, OR = 6.04 (CI 95% 1.27–28.70, p = 0.02)]. In the obtained CHAID decision tree, age was the first split variable between the two groups defined by SBAC-L1 145 HU with a threshold at 63 years (83% (29/35) SBAC-L1 ≤ 145 HU over 63 years) (Online Resource 1 and 2). Among the 6 SBAC-L1 ≤ 145 HU under 63 years of age, 50% (3/6) presented periarticular calcifications and acro-osteolysis.
SBAC-L1 evaluation with 160 and 110 HU thresholds
For a SBAC-L1 ≤ 160 HU (a threshold with excellent sensitivity in a population with osteoporotic risk factors), 41 patients (58.6%) and 2 of the 3 patients with VF were under this threshold. For a SBAC-L1 ≤ 110 HU (a threshold with excellent specificity), 13 patients (18.6%) were under this threshold. Figure 2 shows the distribution of the SBAC-L1 as a function of the three thresholds. When we look at the coefficient distribution according to age, we noted a decrease of SBAC-L1 with age.
Scanographic bone attenuation coefficient of L1 (SBAC-L1) according to age with the three thresholds. The thresholds are presented on a continuous line (a grey line for the normal population and a threshold at 110 HU, a red line for a threshold at 145 HU, and a purple line for a population at risk and a threshold of 160 HU). The three patients with VF appear as green dots
Correlation between DEXA and CT-scan (SBAC-L1)
For the 16 patients who performed a DEXA with a delay, which did not exceed 2 years with the CT-scan, the mean bone mineral density of the spine was measured at 0.86 ± 0.16 g/cm2, the mean SBAC-L1 was 137 ± 37.4 HU. Spearman’s Rho showed no correlation between both modalities (0.2, p = 0.5).
Discussion
In this study, we showed that the prevalence of VF on CT-scan was low (4.3%) whereas most of our patients (85.7%) presented at least one clinical risk factor for osteoporosis. We also showed that the presence of calcinosis, periarticular calcifications and acro-osteolysis on hand radiography was linked with low SBAC-L1, especially for patients under 63 years old.
We demonstrated in this study that we could both assess VF prevalence and measure trabecular bone mass (SBAC-L1 in HU) on CT-scans, with a highly reproducibility, in a population with systemic sclerosis [4, 27]. We showed that most of the patients presented at least one risk factor for osteoporosis (85.7%) but only 30 patients (42.8%) were screened by DEXA. This percentage was lower than those observed in a well-known population with osteoporotic risk, such as rheumatoid arthritis (58.2%) [28]. For these 30 patients, 16.7% (n = 5) were diagnosed as osteoporotic on DEXA. This result was in agreement with those described in the literature with values ranging from 3 to 60% [5,6,7]. We also observed that supplementation with calcium and/or vitamin D and antiresorptive treatment prescription (25.7 and 14.3%, respectively) were low in a population where most of the patients presented at least one osteoporotic risk factor. Not surprisingly, similar results were found in the literature with supplementation with calcium and/or vitamin D ranging from 0 to 69% and specific anti-osteoporotic treatment ranging from 6 to 36.4% [8, 10, 14, 18, 21].
On CT-scan, three patients (4.3%) presented a VF. This prevalence was in agreement with data found in the literature and with values ranging from 0 to 38% [5, 7, 18, 20, 23].
Concerning SBAC-L1 measurement, it was an easy method, performed with high reliability (both intra- and inter-reader). This measure could also be directly performed by radiologists on the CT-work-station after CT-scan acquisition. We showed, for the first time in a large population with systemic sclerosis, that 50% of the patients presented a SBAC-L1 at the VF threshold (SBAC-L1 ≤ 145 HU), which suggested an underestimated risk of VF in patients with systemic sclerosis using DEXA.
Similar to DEXA, the SBAC-L1 was strongly dependant on the age (OR = 1.09) and mainly in patients over 63 years old. Interestingly, we showed that SBAC-L1 ≤ 145 HU was associated with some severe patterns of disease. Therefore, in younger patients with systemic sclerosis (less than 63 years old), a low SBAC-L1 value was strongly associated with calcinosis (OR = 6.30), periarticular calcifications (OR = 3.22), and to a lesser degree, acro-osteolysis. Some authors proposed physiopathological mechanisms to explain calcic deposits in soft tissues and secondary to a displacement of the calcium stocked in bone towards vascular and soft tissues [11, 19]. In our population, calcic deposits were not associated with biologic abnormalities since all of the patients had normal calcemia and phosphoremia. Interestingly, an association between age, calcinosis periarticular calcifications and acro-osteolysis was also described with osteoporosis measured with DEXA in other populations with systemic sclerosis [7, 17, 19, 22, 29]. We did not find an association between SBAC-L1 and a diffuse form of sclerosis. The literature data with DEXA showed an inconsistent association: sometimes the diffuse form was considered as risk factor [7, 13, 15, 20,21,22] and sometimes it was not related [5, 6, 10, 14, 17, 19]. Due to a lack of power, many specific risk factors tended to be significant: arthralgia and abnormal capillaroscopy seemed to be protective, whereas disease duration, acro-osteolysis, PAH, gastroesophageal reflux and digital ulcers seemed to get worse the risk. In the literature, some of the following parameters, including pulmonary or cardiac involvement [7, 13, 20,21,22], digital ulcers [8, 14, 21], and anticentromere antibodies [8, 14], were considered as risk factors and sometimes they were not related [5, 6, 10, 13, 14, 17, 22, 30]. Gender, smoking, and corticosteroid treatment were not associated with a low SBAC-L1 but similar results were found with DEXA [5, 8,9,10, 18, 31].
Limitations in this study were due to the studied segment of spine: for 26 patients (37.1%), the analysis was performed only on the thoracic segment (including L1). As a result, an underestimation of the lumbar VF prevalence should be considered. As described in the literature, two of the three VFs were localized in one of the regions of higher VF prevalence, e.g., the region from the T6 to T8 vertebrae and the region from the T11 to L1 vertebrae [31, 32]. For the patient with a VF and a SBAC-L1 at 171 HU, information concerning the circumstances of her fracture was unavailable since this patient was dead and the diagnosis of an osteoporotic fracture could not be confirmed.
To be relevant, the threshold depended on one hand on the characteristics of the population studied in terms of osteoporotic risk, and on the other hand, on the VF prevalence in this population. With only three VFs observed, we were unable to calculate the optimum threshold of SBAC-L1 in terms of sensitivity and specificity. Indeed, the 110 HU threshold only permitted the identification of one patient with VF, whereas the 145 and 160 HU thresholds identified two patients with VF. From our point of view, the threshold at 145 HU allowed the best compromise between sensitivity and specificity for an osteoporotic fracture risk screening with SBAC-L1, but further studies would be warranted.
In clinical practice, DEXA is the gold standard for osteoporosis screening, but according to other studies, our results suggest, in a population with systemic sclerosis including 40% of patients with corticosteroid treatment, that this exam is insufficiently performed. The aim was not to substitute the SBAC-L1 measurement to the DEXA, but to obtain information concerning the trabecular bone mineralization of the first lumbar vertebra in patients for whom CT-scan was performed during the follow-up of their disease (according to good clinical practice) whereas DEXA was unavailable. Thoracic and TAP CT-scans were usually performed on sclerosis to evaluate pulmonary involvement and were always performed in our population responding to ACR/EULAR criteria for sclerosis. We showed that CT-scan was an alternative method to exclusively explore the trabecular bone without the cortical bone and to avoid artefacts due to 2D projection as observed on DEXA (osteophyte, bone condensation or disc/vascular calcifications [33,34,35,36,37] or mesenteric adenopathy).
To conclude, we showed, on a large sample of patients with systemic sclerosis and with osteoporosis risk factors that only 42.8% were screened for osteoporosis by DEXA, which classified 16.7% of them as osteoporotic. The VF prevalence was measured at 4.3% in CT-scan and 50% of the population presented SBAC-L1 under the fracture threshold (≤ 145 HU). The presence of calcinosis, periarticular calcifications and acro-osteolysis on hand radiography was linked with low SBAC-L1 and should lead to an osteoporosis screening, especially for patients under 63 years old.
References
Scussel-Lonzetti L, Joyal F, Raynauld J-P, Roussin A, Rich E, Goulet J-R et al (2002) Predicting mortality in systemic sclerosis: analysis of a cohort of 309 French Canadian patients with emphasis on features at diagnosis as predictive factors for survival. Medicine (Baltimore) 81:154–167
Allanore Y, Avouac J, Kahan A (2008) Systemic sclerosis: an update in 2008. Jt Bone Spine 75:6505
Tyndall AJ, Bannert B, Vonk M, Airò P, Cozzi F, Carreira PE et al (2010) Causes and risk factors for death in systemic sclerosis: a study from the EULAR Sclerosis Trials and Research (EUSTAR) database. Ann Rheum Dis 69:180915
Haute Autorité de Santé - ALD no 21—Sclérodermie généralisée évolutive. https://www.has-sante.fr/portail/jcms/c_717292/fr/ald-n-21-sclerodermie-generalisee-evolutive. Accessed 9 July 2017
Avouac J, Koumakis E, Toth E, Meunier M, Maury E, Kahan A et al (2012) Increased risk of osteoporosis and fracture in women with systemic sclerosis: a comparative study with rheumatoid arthritis. Arthritis Care Res (Hoboken) 64:1871–1878
Ibn Yacoub Y, Amine B, Laatiris A, Wafki F, Znat F, Hajjaj-Hassouni N (2012) Bone density in Moroccan women with systemic sclerosis and its relationships with disease-related parameters and vitamin D status. Rheumatol Int 32:31438
Omair MA, Pagnoux C, McDonald-Blumer H, Johnson SR (2013) Low bone density in systemic sclerosis. A systematic review. J Rheumatol 40:188190
Marot M, Valéry A, Esteve E, Bens G, Müller A, Rist S et al (2015) Prevalence and predictive factors of osteoporosis in systemic sclerosis patients: a case-control study. Oncotarget 6:1486573
Omair MA, Pagnoux C, McDonald-Blumer H, Johnson SR (2013) Low bone density in systemic sclerosis. A systematic review. J Rheumatol 40:1881–1890
Mok CC, Chan PT, Chan KL, Ma KM (2013) Prevalence and risk factors of low bone mineral density in Chinese patients with systemic sclerosis: a case-control study. Rheumatology 52:296–303
Yuen SY, Rochwerg B, Ouimet J, Pope JE (2008) Patients with sclerosis may have increased risk of osteoporosis. A comparison to rheumatoid arthritis and noninflammatory musculoskeletal conditions. J Rheumatol 35:1073–1078
Souza RBC, Borges CTL, Takayama L, Aldrighi JM, Pereira RMR (2006) Systemic sclerosis and bone loss: the role of the disease and body composition. Scand J Rheumatol 35:3847
Frediani B, Baldi F, Falsetti P, Acciai C, Filippou G, Spreafico A et al (2004) Bone mineral density in patients with systemic sclerosis. Ann Rheum Dis 63:3267
Marot M (2013) Etude de la prévalence et des facteurs prédictifs d’ostéoporose chez des patientes atteintes de sclérodermie systémique: une étude cas–témoins. Thesis, University of Tours (France)
Di Munno O, Mazzantini M, Massei P, Ferdeghini M, Pitaro N, Latorraca A et al (1995) Reduced bone mass and normal calcium metabolism in systemic sclerosis with and without calcinosis. Clin Rheumatol 14:40712
Carbone L, Tylavsky F, Wan J, McKown K, Cheng S (1999) Bone mineral density in sclerosis. Rheumatology 38:3712
Da Silva HC, Szejnfeld VL, Assis LS, Sato EI (1997) Study of bone density in systemic sclerosis. Rev Assoc Med Bras 43:406
Atteritano M, Sorbara S, Bagnato G, Miceli G, Sangari D, Morgante S et al (2013) Bone mineral density, bone turnover markers and fractures in patients with systemic sclerosis: a case control study. PLoS One 8:e66991
Loucks J, Pope JE (2005) Osteoporosis in sclerosis. Semin Arthritis Rheum 34:67882
Wan Y-N, Zhang L, Wang Y-J, Yan J-W, Wang B-X, Wang J (2014) The association between systemic sclerosis and bone mineral density—a meta-analysis of observational studies. Int J Rheum Dis 17:84555
Kilic G, Kilic E, Akgul O, Ozgocmen S (2016) Increased risk for bone loss in women with systemic sclerosis: a comparative study with rheumatoid arthritis. Int J Rheum Dis 19:40511
Frediani B, Baldi F, Falsetti P, Acciai C, Filippou G, Spreafico A et al (2004) Clinical determinants of bone mass and bone ultrasonometry in patients with systemic sclerosis. Clin Exp Rheumatol 22:3138
Lai C-C, Wang S-H, Chen W-S, Liu C-J, Chen T-J, Lee P-C et al (2015) Increased risk of osteoporotic fractures in patients with systemic sclerosis: a nationwide population-based study. Ann Rheum Dis 74:134752
Pickhardt PJ, Pooler BD, Lauder T, del Rio AM, Bruce RJ, Binkley N (2013) Opportunistic screening for osteoporosis using abdominal computed tomography scans obtained for other indications. Ann Intern Med 158:58895
Erre GL, Marongiu A, Fenu P, Faedda R, Masala A, Sanna M et al (2009) The “sclerodermic hand”: a radiological and clinical study. J Bone Spine 75:426–431
Genant HK, Wu CY, van Kuijk C, Nevitt MC (1993) Vertebral fracture assessment using a semiquantitative technique. J Bone Miner Res 8:113748
Ould AB (2015) Sclérodermie systémique: prévalence et suivi de la pneumopathie interstitielle chronique d’une cohorte de 180 patients. Dissertation, University of Moroco
Dougados M, Soubrier M, Antunez A, Balint P, Balsa A, Buch MH et al (2014) Prevalence of comorbidities in rheumatoid arthritis and evaluation of their monitoring: results of an international, cross-sectional study (COMORA). Ann Rheum Dis 73:628
Valenzuela A, Baron M, Herrick Al, Proudman S, Stevens W, Rodriguez-Reyna TS et al (2016) Calcinosis is associated with digital ulcers and osteoporosis in patients with systemic sclerosis: A Sclerosis Clinical Trials Consortium study. Semin Arthritis Rheum 46:3449
Sampaio-Barros PD, Costa-Paiva L, Filardi S, Sachetto Z, Samara AM, Marques-Neto JF (2005) Prognostic factors of low bone mineral density in systemic sclerosis. Clin Exp Rheumatol 23:1804
Burns JE, Yao J, Summers RM (2017) Vertebral body compression fractures and bone density: automated detection and classification on CT images. Radiology 284:788–797
Rajasekaran S, Kanna RM, Schnake KJ, Vaccaro AR, Schroeder GD, Sadiqi S et al (2017) Osteoporotic thoracolumbar fractures—how are they different? Classification and treatment algorithm. J Orthop Trauma 31:S4956
Bassett LW, Blocka KL, Furst DE, Clements PJ, Gold RH (1981) Skeletal findings in progressive systemic sclerosis (sclerosis). AJR Am J Roentgenol 136:11216
Ogawa T, Ogura T, Hayashi N, Hirata A (2009) Tumoral calcinosis of thoracic spine associated with systemic sclerosis. J Rheumatol 36:25523
Ward M, Curé J, Schabel S, Smith EA, Schumacher HR Jr, Silver RM (1997) Symptomatic spinal calcinosis in systemic sclerosis (sclerosis). Arthritis Rheum 40:18925
Durant C, Farge-Bancel D (2011) Clinical images: voluminous ectopic tumoral calcinosis of the spine in systemic sclerosis. Arthritis Rheum 63:411
Weerakoon A, Sharp D, Chapman J, Clunie G (2011) Lumbar canal spinal stenosis due to axial skeletal calcinosis and heterotopic ossification in limited cutaneous systemic sclerosis: successful spinal decompression. Rheumatol Oxf Engl 50:21446
Author information
Authors and Affiliations
Contributions
FM: corresponding author. Substantial contributions to the study conception and design and to acquisition, analysis and interpretation of the data; CT lectures with VF and SBAC-L1; intra-reader reproducibility; drafting the manuscript. BE: Substantial contributions to the acquisition of clinical data; revising the article critically for important intellectual content; final approval of the submitted version of the article to be published; agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. AE: substantial contributions to analysis and interpretation of the data; revising the article critically for important intellectual content; final approval of the submitted version of the article to be published; agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. PCJ: substantial contributions to acquisition of the data: inter-reader reproducibility; revising the article critically for important intellectual content; final approval of the submitted version of the article to be published; agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. DJ: substantial contributions to acquisition of the clinical data; final approval of the version of the article to be published. CF: substantial contributions to acquisition of the clinical data; study of right heart catheterization; final approval of the submitted version of the article to be published. MD: substantial contributions to acquisition of the data; study of pulmonary involvement on all CT to classify the sub-type of involvement; final approval of the submitted version of the article to be published. HO: substantial contributions to acquisition of the data; final approval of the submitted version of the article to be published. CVI: substantial contributions to the study conception and design; revising the article critically for important intellectual content; final approval of the submitted version of the article to be published; agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. LD: substantial contributions to the study conception and design and to analysis and interpretation of the data; scoring of hand structural damages on radiography; drafting and revising the article critically for important intellectual content; final approval of the submitted version of the article to be published; agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
All of the authors declare that they have no conflict of interest.
Research involving human participants
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
For this type of study formal consent is not required.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Fauny, M., Bauer, E., Albuisson, E. et al. Vertebral fracture prevalence and measurement of the scanographic bone attenuation coefficient on CT-scan in patients with systemic sclerosis. Rheumatol Int 38, 1901–1910 (2018). https://doi.org/10.1007/s00296-018-4139-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00296-018-4139-5