Abstract
This study evaluated whether people with ankylosing spondylitis (AS) and spondyloarthritis are at higher risk of type 2 diabetes mellitus (T2DM). We used a sub-dataset of the National Health Insurance Research Database from 1996 to 2010 to established a AS cohort consisting new patients with AS or spondyloarthritis (N = 7,778) and a non-AS cohort without the diseases (N = 31,112). Incidences of T2DM in the two cohorts, hazard ratios (HRs) of risk of T2DM in association with AS, and cumulative probability of having T2DM were estimated by the end of 2010. The incidence of T2DM was 1.17-fold higher in the AS cohort than in the non-AS cohort (13.5 vs. 11.5, per 1,000 person-years), with an adjusted HR of 1.16 (95 % CI = 1.05–1.29). The T2DM incidence was higher for women than for men; while the Cox model measured sex-specific adjusted HR of T2DM was higher for men than for women. The incidence rate of T2DM increased with age in both cohorts, while the age-specific measures showed that the adjusted HR of T2DM was higher in young AS patients (≤50 years of age) than older ones, compared to their peers of non-AS group. The plot of Kaplan–Meier analysis showed that the overall probability of having T2DM was 2 % higher in the AS cohort than in the non-AS cohort (log-rank test: p < 0.0001). Patients with AS and spondyloarthritis have an increased risk of developing T2DM.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
A mass survey in Chinese military and community has shown that the prevalence of ankylosing spondylitis (AS) and spondyloarthritis in China is similar to that in Caucasians in the United States and Europe [1]. AS is a chronic inflammatory disease of the axial skeleton with approximately 0.2–0.9 % of the general populations affected [2]. Previous studies have suggested that patients with AS exhibit increased cardiovascular (CV) mortality and morbidity [3–6]. Another study has found that cerebrovascular diseases, such as stroke, are more common in patients with AS [7]. However, traditional CV risk factors fail to explain well the increased CV events for patients with AS [8]. Non-traditional CV risk factors such as C-reactive protein (CRP) and interlukine-6 (IL-6) are possible mechanisms in connection with CV events and AS. Previous studies have associated insulin resistance (IR) with both traditional or non-traditional factors of CV leading to the occurrence of CV events [9]. IR has been associated with not only CV disease but also rheumatoid arthritis [10, 11]. Studies also suggested that IR is one of the critical pathogeneses of type 2 diabetes mellitus (T2DM) [12, 13]. However, to the best of our knowledge, only one paper has addressed the association between AS and T2DM [14]. This study attempted to investigated whether patients with AS are at higher risk of T2DM.
Methods
Data sources
In 1995, Taiwan implemented a compulsory single-payer National Health Insurance (NHI) program, covering over 99 % of Taiwan’s 23.74 million residents [15]. With the authorization from the Taiwan Department of Health, the National Health Research Institutes (NHRI) has assisted the Bureau of NHI to establish several datasets for administrative use and research. This study used a sub-dataset consisting of longitudinal claims from 1996 to 2010 for a randomly sampled 1,000,000 insured people. We used the scrambled anonymous identification numbers of insured people to link files, including the registry of beneficiaries, inpatient claims, and ambulatory care claims. The International Classification of Disease, Ninth Revision of Clinical Modification (ICD-9-CM), is available in the claims data to define disease status. This study was exempted from full institutional review (CMU-REC-101-012).
Study subjects
From the database, we identified a cohort of AS, consisting of patients newly diagnosed with AS or spondyloarthritis from 2000 to 2010. Patients who had at least three medical visits for the diseases were selected from outpatient and/or inpatient claims base on the ICD-9 code of 720 in both principal/secondary diagnostic fields. The initial ambulatory or inpatient claims diagnosis date was defined as the index date. Patients with a history of T1DM or T2DM (ICD-9 code 250) before the index date or missing information on age or sex were excluded. From the insured population without the history of diabetes mellitus and AS, we randomly selected fourfold of subjects as the non-AS comparison cohort, frequency-matching for age [every 5 years], sex, index year and comorbidities of the AS cohort.
Outcome definition
Subjects in both AS and non-AS cohorts were followed up until they were diagnosed with T2DM (ICD-9 code 250.x0 and 250.x2), or they were censored because of loss to follow-up, withdraw from the insurance, death, or until the end of 2010. The patient with at least 3 diagnoses of T2DM in outpatients and/or inpatients records during the follow-up period was considered as a diabetic case. Principal diagnosis and secondary diagnosis were applied equally. During the follow-up period, there were 812 and 3,811 persons censored (included death) in the AS and non-AS cohorts, respectively. Patients diagnosed with type 1 diabetes were excluded.
Sociodemographic characteristics and comorbidity
Sociodemographic characteristics including age, sex, and income were compared between the two cohorts. Comorbidities presented if the disease appeared in subjects were identified 3 or more visits in the principal and/or secondary diagnose. The selected baseline comorbidities included hypertension (ICD-9 codes 401-405), hyperlipidemia (ICD-9 code 272), stroke (ICD-9 code 430-438), and coronary artery disease (CAD) (ICD-9 code 410-414).
Statistical analysis
Data analyses involved first comparing the AS cohort with the non-AS cohort for the distribution of age, sex, monthly income, and baseline comorbidities. The data were then subjected to the Chi square test for categorical variables and the t test for continuous variables. We estimated the incidence rate using the number of identified T2DM events divided by the total follow-up person-years for each cohort by sex, age, monthly income and the comorbidity status (yes or no). Univariate and multivariate Cox proportional hazards regression analyses were conducted to calculate hazard ratios (HRs) and 95 % confidence intervals (CIs) for the risk of T2DM in association with AS. Multivariate analysis included variable to sex, age and comorbidity status. The probability of overall incident T2DM was assessed using the Kaplan–Meier method, and the difference of probabilities between the two cohorts was compared using the log-rank test. All analyses were performed using the SAS statistical package (version 9.2 for Windows; SAS institute, Inc., Cary, NC, USA) and the results were considered statistically significant when two-tailed p values were less than 0.05.
Results
Baseline characteristics of the study participants
Eligible study subjects comprised 5,818 persons in the AS cohort and 23,265 persons in the non-AS cohort (Table 1). The average follow-up periods were 5.80 ± 3.18 years for the AS cohort and 5.78 ± 3.21 years for the comparison cohort. There were more men (53.0 %) and subjects aged ≤50 years (61.7 %). The monthly income was lower for the AS cohort. Both cohorts had similar age, sex and comorbidities distributions.
End points
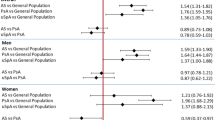
The overall incidence of T2DM was 1.17-fold greater in the AS cohort than in the non-AS cohort (13.5 vs. 11.5, per 1,000 person-years), with an adjusted HR of 1.16 (95 % CI = 1.05–1.29) (Table 2).
Subgroup analyses
The sex-specific analysis in Table 2 shows that incidence of diabetes was greater in women than in men in both cohorts. However, the analysis sex-specific HRs showed that men with AS had a higher relative hazard of diabetes than women with AS had. The incidence of diabetes increased with age in both cohorts. However, the age-specific AS to non-AS HRs showed that AS patients aged less than 50 years had the highest hazard of diabetes compared with their peers of same ages with an adjusted HR of 1.28 (95 % CI = 1.08–1.53). The incidence of T2DM was significantly higher in subjects presence of anyone comorbidity than in those without the comorbidity in both cohorts. Among subjects without comorbidity, the AS cohort had an adjusted HR of 1.32 (95 % CI = 1.10–1.58) compared with non-AS cohort.
The plot of the Kaplan–Meier analysis showed that, by the end of 11 follow-up years, the cumulative probability to develop T2DM was 2 % greater for the AS cohort than the non-AS cohort (13.3 vs. 11.3 %) (log-rank test: p = 0.0027) (Fig. 1).
Discussion
Using the Taiwan National Health Insurance data [16], our study showed that the risk of T2DM in the AS patients in Taiwan is 17.4 % higher than that in the general population. The relative risk is even 33.7 % greater for AS patients without the comorbidities, indicating patients with AS are at a higher risk of developing diabetes without the influence of other comorbid conditions. Our additional data analysis did show a higher comorbidity prevalence in the AS cohort than in the non-AS cohort, including hypertension, hyperlipidemia, stroke, and CVD. These comorbidities correspond to worldwide T2DM findings [14]. Patients with AS are thus at an increased risk of T2DM. Our study also showed that the incidence of T2DM in women with AS was higher than that in men with AS (14.8 vs. 12.3 per 1,000 person-years). This gap is slightly smaller than the gap between women and men in the general population, indicating the impact of AS is some what greater for men than for women. In addition, we found the incidence of T2DM increasing with age in both AS and non-AS cohorts. But, the age-specific analysis showed a higher relative risk of diabetes for younger patients with AS than for old patients with AS. This may indicate that the impact of AS is stronger for young patients than for old patients.
Previous studies have suggested that inflammation is one of factors leading to T2DM and subsequent diabetic vascular complications. C-reactive protein (CRP) is an inflammatory marker and the concentration is elevated among obese and diabetic patients [17, 18]. CRP is also a marker appearing in patients with atherosclerosis and acute-stage AS [19]. TNF alpha (TNF-α) is another inflammatory cytokine in patients with AS. TNF-α is involved in the IR pathway playing a primary role leading to abnormalities of glucose metabolism [20–22]. TNF-α can inhibit the tyrosine kinase activity of the insulin receptor through insulin receptor substrate-1 serine phosphorylation and causes hyperglycemia [23]. Decreased exercise is also a factor associated with developing T2DM [12, 13]. Patients with AS are more likely to be physically inactive and or immobile because of complications of spinal cord compression or spinal fracture. The restricted motion and/or decreased lung function appear in the late life or uncontrolled stage of AS may increase the diabetes risk. HLA-B27 has been found in certain patients with AS [24, 25]. Studies have also found patients with DM are more prevalent with the existence of HLA DR3 and DR4 [26–28]. Another two studies suggested that the HLA phenotype is associated with DM and AS [29, 30]. The medications for AS treatment may also explain of higher incidence of T2DM in AS patients. Corticosteroid therapy is a common cause of hyperglycemia [31]. Non-steroidal anti-inflammatory drugs (NSAIDs) are among the common prescriptions for pain control and anti-inflammation for patients with AS. Studies have also associated the risk of hypertension and CV events with the use of these drugs [32, 33]. Patients with hypertension are more likely to have T2DM through increased IR [34]. Anti-TNF therapy appears to have no effect on the control of glucose metabolism for patients with AS [35].
The typical age of onset of AS in the Western population is of young adults. A German study found near 80 % of patients of AS are diagnosed before 50 years old [36]. Bakland et al. [37] found in Northern Norway that the mean age was in 20 s years old in Northern Norway patients. On the other hand, in our study, approximately 62 % of the patients were diagnosed before the ages of 50 years and more than 17 % subjects were older than 65 years. Ethnic variation may explain partly of the difference. More importantly, the Taiwan National Health Insurance was lunched in 1995 and the claims data available for public uses began in 1996. Some patients who were prevalent before the availability of the insurance may have their clinic visits after 1996.
The strength of our study is the use of population data representative of the general population. However, certain limitations to our findings should be considered. First, the NHIRD does not contain detailed information regarding smoking habits, alcohol consumption, and family history of systemic diseases; all of which may have association with diabetes. Second, patients with AS were likely to have more physician visits than general population and increased the diagnosis of diabetes. However, this diagnosis bias has been minimized because our AS cohort and non-AS cohort are matched by comorbidities as well. Third, evidence derived from a retrospective cohort study is typically lower in statistical quality than that derived from randomized trials because of potential biases related to adjustments for confounding variables. In spite of our careful study design and control measures for confounding factors, bias resulting from unknown confounders may have affected our results. Fourth, all data in the NHIRD are anonymous. The relevant clinical variables, such as blood pressure, imaging results, pathology findings, and serum laboratory data were unavailable for this study. However, the data regarding AS and diabetes diagnoses were nonetheless reliable.
In conclusion, the nationwide population-based cohort study found AS and other inflammatory spondyloarthritis increase the risk of developing type 2 diabetes in an Asian population. However, because of the study limitations, additional unbiased studies and randomized control trials on the biological mechanisms are required before any conclusions can be confirmed.
References
Ng SC, Liao Z, Yu DT, Chan ES, Zhao L, Gu J (2007) Epidemiology of spondyloarthritis in the People’s Republic of China: review of the literature and commentary. Semin Arthritis Rheum 37:39–47
Akkoc N, Khan MA (2006) Epidemiology of ankylosing spondylitis and related spondyloarthropathies. In: Weisman MHRJ, van der Heijde D (eds) Ankylosing spondylitis and the spondyloarthropathies: a companion to rheumatology. Mosby-Elsevier, London, pp 117–131
Radford EP, Doll R, Smith PG (1977) Mortality among patients with ankylosing spondylitis not given X-ray therapy. N Engl J Med 297:572–576
Lehtinen K (1993) Mortality and causes of death in 398 patients admitted to hospital with ankylosing spondylitis. Ann Rheum Dis 52:174–176
Mathieu S, Gossec L, Dougados M, Soubrier M (2011) Cardiovascular profile in ankylosing spondylitis: a systematic review and meta-analysis. Arthritis Care Res (Hoboken) 63:557–563
Goodson NJ, Wiles NJ, Lunt M, Barrett EM, Silman AJ, Symmons DP (2002) Mortality in early inflammatory polyarthritis: cardiovascular mortality is increased in seropositive patients. Arthritis Rheum 46:2010–2019
Han C, Robinson DW Jr, Hackett MV, Paramore LC, Fraeman KH, Bala MV (2006) Cardiovascular disease and risk factors in patients with rheumatoid arthritis, psoriatic arthritis, and ankylosing spondylitis. J Rheumatol 33:2167–2172
Peters MJ, van der Horst-Bruinsma IE, Dijkmans BA, Nurmohamed MT (2004) Cardiovascular risk profile of patients with spondylarthropathies, particularly ankylosing spondylitis and psoriatic arthritis. Semin Arthritis Rheum 34:585–592
Haffner SM (1999) Epidemiology of insulin resistance and its relation to coronary artery disease. Am J Cardiol 84:11J–14J
Svenson KL, Lundqvist G, Wide L, Hällgren R (1987) Impaired glucose handling in active rheumatoid arthritis: relationship to the secretion of insulin and counter-regulatory hormones. Metabolism 36:940–943
Svenson KL, Pollare T, Lithell H, Hällgren R (1988) Impaired glucose handling in active rheumatoid arthritis: relationship to peripheral insulin resistance. Metabolism 37:125–130
Ritchie LD, Ganapathy S, Woodward-Lopez G, Gerstein DE, Fleming SE (2003) Prevention of type 2 diabetes in youth: etiology, promising interventions and recommendations. Pediatr Diabetes 4:174–209
Freeman JS (2010) A physiologic and pharmacological basis for implementation of incretin hormones in the treatment of type 2 diabetes mellitus. Mayo Clin Proc 85:S5–S14
Brophy S, Cooksey R, Atkinson M, Zhou SM, Husain MJ, Macey S et al (2012) No increased rate of acute myocardial infarction or stroke among patients with ankylosing spondylitis-a retrospective cohort study using routine data. Semin Arthritis Rheum 42:140–145
Cheng TM (2009) Taiwan’s National Health Insurance system: high value for the dollar. In: Okma KGH, Crivelli L (eds) Six countries, six reform models: the health reform experience of Israel, the Netherlands, New Zealand, Singapore, Switzerland and Taiwan. World Scientific, New Jersey, pp 71–204
Jiang YD, Chang CH, Tai TY, Chen JF, Chuang LM (2012) Incidence and prevalence rates of diabetes mellitus in Taiwan: analysis of the 2000–2009 Nationwide Health Insurance database. J Formos Med Assoc 111:599–604
Ford ES (1999) Body mass index, diabetes, and C-reactive protein among U.S. adults. Diabetes Care 22:1971–1977
McMillan DE (1989) Increased levels of acute-phase serum proteins in diabetes. Metabolism 38:1042–1046
Libby P, Ridker PM, Maseri A (2002) Inflammation and atherosclerosis. Circulation 105:1135–1143
Hotamisligil GS, Shargill NS, Spiegelman BM (1993) Adipose expression of tumor necrosis factor-alpha: direct role in obesity-linked insulin resistance. Science 259:87–91
Gimeno RE, Klaman LD (2005) Adipose tissue as an active endocrine organ: recent advances. Curr Opin Pharmacol 5:122–128
Hotamisligil GS, Spiegelman BM (1994) Tumor necrosis factor alpha: a key component of the obesity-diabetes link. Diabetes 43:1271–1278
Hotamisligil GS, Murray DL, Choy LN, Spiegelman BM (1994) Tumor necrosis factor alpha inhibits signaling from the insulin receptor. Proc Natl Acad Sci USA 91:4854–4858
Brewerton DA, Hart FD, Nicholls A, Caffrey M, James DC, Sturrock RD (1973) Ankylosing spondylitis and HL-A 27. Lancet 1:904–907
Schlosstein L, Terasaki PI, Bluestone R, Pearson CM (1973) High association of an HL-A antigen, W27, with ankylosing spondylitis. N Engl J Med 288:704–706
Platz P, Jakobsen BK, Morling N, Ryder LP, Svejgaard A, Thomsen M, Christy M, Kromann H, Benn J, Nerup J, Green A, Hauge M (1981) HLA-D and -DR antigens in genetic analysis of insulin dependent diabetes mellitus. Diabetologia 21:108–115
Reitnauer PJ, Roseman JM, Barger BO, Murphy CC, Kirk KA, Acton RT (1981) HLA associations with insulin-dependent diabetes mellitus in a sample of the American Black population. Tissue Antigens 17:286–293
Pittman WB, Acton RT, Barger BO, Bell DS, Go RC, Murphy CC et al (1982) HLA-A, -B, and -DR associations in type I diabetes mellitus with onset after age forty. Diabetes 31:122–125
Acton RT, Hodge TW, McDaniel DO, Reveille JD, Napier-Littrell M, Barger BO (1985) HLA restriction fragment length polymorphisms associated with insulin-dependent diabetes mellitus and the seronegative spondyloarthropathies. Biosci Rep 5:815–829
Sattar MA, Al-Sughyer AA, Siboo R (1988) Coexistence of rheumatoid arthritis, ankylosing spondylitis and dermatomyositis in a patient with diabetes mellitus and the associated linked HLA antigens. Br J Rheumatol 27:146–149
Clore JN, Thurby-Hay L (2009) Glucocorticoid-induced hyperglycemia. Endocr Pract 15:469–474
Mamdani M, Juurlink DN, Lee DS, Rochon PA, Kopp A, Naglie G et al (2004) Cyclo-oxygenase-2 inhibitors versus non-selective non-steroidal anti-inflammatory drugs and congestive heart failure outcomes in elderly patients: a population-based cohort study. Lancet 363:1751–1756
White WB, Kent J, Taylor A, Verburg KM, Lefkowith JB, Whelton A (2002) Effects of celecoxib on ambulatory blood pressure in hypertensive patients on ACE inhibitors. Hypertension 39:929–934
Matthaei S, Stumvoll M, Kellerer M, Häring HU (2000) Pathophysiology and pharmacological treatment of insulin resistance. Endocr Rev 21:585–618
da Silva BS, Bonfá E, de Moraes JC, Saad CG, Ribeiro AC, Gonçalves CR et al (2010) Effects of anti-TNF therapy on glucose metabolism in patients with ankylosing spondylitis, psoriatic arthritis or juvenile idiopathic arthritis. Biologicals 38:567–569
Feldtkeller E, Khan MA, van der Heijde D, van der Linden S, Braun J (2003) Age at disease onset and diagnosis delay in HLA-B27 negative vs. positive patients with ankylosing spondylitis. Rheumatol Int 23:61–66
Bakland G, Nossent HC, Gran JT (2005) Incidence and prevalence of ankylosing spondylitis in Northern Norway. Arthritis Rheum 53:850–855
Acknowledgments
The study was supported in part by the study projects (DMR-103-012) in our hospital and Taiwan Department of Health Clinical Trial and Research Center and for Excellence (DOH102-TD-B-111-004), and Taiwan Department of Health Cancer Research Center for Excellence (DOH102-TD-C-111-005).
Conflict of interest
All authors state that they have no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Hue-Yong Chen and Hsin-Hung Chen have contributed equally to this manuscript.
Rights and permissions
About this article
Cite this article
Chen, HH., Yeh, SY., Chen, HY. et al. Ankylosing spondylitis and other inflammatory spondyloarthritis increase the risk of developing type 2 diabetes in an Asian population. Rheumatol Int 34, 265–270 (2014). https://doi.org/10.1007/s00296-013-2927-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00296-013-2927-5