Abstract
This population-based study aimed to determine the trend of incidence, prevalence, and mortality of systemic lupus erythematosus (SLE) in a 6-year period in Taiwan. Patients with international classification of diseases ninth revision (ICD-9) code 710.0 were retrieved from the Taiwanese National Health Insurance Research Database (NHIRD), which covered more than 96 % of the entire population, and from the Ministry of Interior between 2003 and 2008 in Taiwan. Patients with SLE registered as catastrophic illness were enrolled for analysis. The incidence rate, prevalence ratio, and mortality rate stratified by sex and age were analyzed. There were a total of 6,675 SLE patients (5,836 females and 839 in males) during the study period. The average annual incidence rate was 4.87 per 100,000 population, and the average female-to-male incidence ratio was 7.15. The ratio increased with age and peaked at the age of 40–49 years, then decreased thereafter. The incidence rate decreased by 4.2 % per year. The highest incidence rate was noted in the 20–29-year-old age group in females and the 70–79-year-old age group in males. The average prevalence and mortality rates were 97.5 and 1.2 per 100,000 population, respectively. Mortality was 3.2 % in patients diagnosed within 1 year and is more prevalent in young patients with average age of 15.6 years. Incidence rate of SLE has been declining in recent years but the prevalence rate has remained steady. The highest mortality rate is among younger patients diagnosed with SLE within 1 year.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Systemic lupus erythematosus (SLE) is a chronic, complex, and multi-system disease characterized by a wide variety of clinical and serologic manifestations. Its remarkably diverse clinical manifestations of the disease, which are remarkably diverse, include fever, erythematous rash, polyarthralgia and arthritis, polyserositis, anemia, thrombocytopenia, and renal, neurologic, and cardiac abnormalities. It usually occurs more primarily in women of child-bearing age [1]. In patients with childhood-onset SLE, the initial symptoms are reported to be more severe than those in adults [2]. The incidence of SLE in adults is estimated to be at 2.0–7.6 per 100,000 per year in western countries. However, few surveys are reported in children [3]. Using a sample questionnaire survey, SLE incidence was estimated to be 0.47 per 100,000 per year in Japan [4], while in Canada, it was approximately 0.36 per 100,000 based on a national registry [5]. The overall incidence was 2.8–3.1 per 100,000 between 2000 and 2006 in Southern China [6].
Survival has improved in the past few decades. The 5-year survival of SLE patients was below 50 % in the 1960s, but it has reached more than 90 % in most series reported in the 1990s [7]. This can probably be attributed to increased awareness of the condition and hence, early diagnosis and treatment, the availability of newer immuno-suppressive agents with less toxicity, and improvements in supportive care for disease and treatment-related complications like uremia, infection, and thromboembolism.
In Asia, SLE is not common. The epidemiologic data of SLE are important in helping physicians understand its impact and outcome on the entire country. It may also guide health policy and planning in the provision of adequate medical services to meet the specific needs of patients and health care providers. The implementation of the National Health Insurance (NHI) in Taiwan started in 1995, and its databases contained health care data from over 95 % of all the hospitals and clinics in Taiwan and more than 98 % of the population covered. Patients with SLE are registered as catastrophic disease that require government subsidy for medical expenses. Thus, it is essential to determine the trend of incidence, prevalence, and mortality of SLE in a particular time period in Taiwan based on this database.
Methods
Data source
The National Health Research Institutes (NHRI) obtained raw data from the National Health Insurance (NHI) to build the National Health Insurance Research Database (NHIRD). The raw data were taken from the data warehouse in the Bureau of National Health Insurance Information Office. The NHIRD had recently provided information for academic research on medicine and health care to enhance the quality of medical care for the general public. This study was based on continuous data from the Taiwanese NHIRD provided by the department of health statistics, which contained >98 % of registry files, and all medical benefits claim for about 22.60 million enrollees of the Taiwan NHI program. Data on the population composition and detailed age-specific population were disclosed in the Statistical Yearbook of Interior, the officially published materials by the Ministry of Interior in Taiwan (http://sowf.moi.gov.tw/stat/year/elist.htm). Each age group was categorized by 10 years. This study adhered to strict confidentiality guidelines, in accordance with regulations regarding personal electronic data protection, and was approved by the department of health statistics and NHIRD.
Study subjects
After making the diagnosis of SLE, physicians helped patients apply for the catastrophic illness certificate, which was verified and issued by the NHI. In addition to the obligatory health insurance, this certificate provided further decreases in medical expenses through government subsidies.
The SLE patients with code 710.0 of the International Classification of Disease, 9th Revision, Clinical Modification (ICD-9-CM) in catastrophic illness database were selected. Annual numbers of new SLE cases and deaths between January 2003 and December 2008 were retrieved from the NHIRD. Patients diagnosed with SLE in 2002 or earlier were excluded.
Statistical analysis
Incidence rates of SLE from 2003 to 2008 were calculated by dividing the number of new SLE cases every year by the population residing in Taiwan. Average age and sex-specific incidence rates were calculated by dividing the number of new cases in each age and sex group by age- and sex-specific populations, whereas prevalence was calculated by dividing the total numbers of cases by the corresponding population in a year.
Chi-square test was used to compare the incidence rates of SLE patients by sex. A p < 0.05 was considered statistically significant. Confidence intervals (CI) were calculated because these were national population data rather than sample data. The SAS 9.1 software was used for data management and data cleaning (SAS Institute, Cary, NC, USA) and the SPSS 17 software was used for data analysis (SPSS Inc., Chicago IL, USA).
Results
Incidence
In 2003, there were 1,208 new diagnosed SLE patients (1,058 females and 150 males). The cumulative patient numbers increased to 6,675 (5,836 females and 839 males) by 2008 in the NHIRD. The average incidence in this 6-year period was 4.87 per 100,000 population. However, the incidence rate decreased by 4.2 % annually. In terms of sex, the incidence was 8.7 and 1.2 per 100,000 population in females and males, respectively, such that SLE was 7.2 times more common in females than in males (Table 1).
In terms of age-specific incidence rates of SLE per 10 years of age (Table 2), the incidence rate was 38.4 % between 20 and 49 years of age, which declined with increasing age. However, the highest incidence rate was in males aged >70 years old. Female patients aged 40–49 years old had 9.06 times higher incidence than male patients of the same age. The female-to-male incidence ratio increased with age, peaked in the age group of 30–34 years old (6.94), and declined thereafter. In pediatric-onset SLE (age <15 years), there was a significant sex difference, with a ratio of 2.87. There was a marked increase in females after adolescence. The difference in sex ratio was less prominent in late-onset SLE.
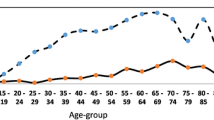
The SLE incidence rate increased with age in females. The increased rate was 70.8 % in the age group 5–10 years old and the peak incidence was in the 30 s. In males, the incidence rate increased gradually in each age group (Fig. 1a, b) and was similarly more prominent in the age group of 5–10 years old.
Trend of incidence rates (per 100,000) of systemic lupus erythematosus (SLE) in a males and b females in Taiwan based on NHIRD data from 2003 to 2008. The rates were similar for ages 0–19 years but started to diverge sharply thereafter, with females having significantly higher rates of SLE incidence until around the age of 60 years. The peak incidence among females occurred in the 30–39-year-old age group, which was in the 70–79-year-old age group in males
Prevalence
The cumulative number of SLE cases during the period studied was 133,488. The prevalence rate was 97.5 per 100,000 population. The prevalence rate decreased from 28.5 to 23.0 per 100,000 population in males, but increased from 164.3 to 179.4 per 100,000 population in females. The highest prevalence was in the 30–39-year-old group in females and the 70–79-year-old group in males (Fig. 2a, b).
Prevalence rate by a gender and b different age groups from 2003 to 2008. The prevalence rate increased in females but decreased in males during this period. Prevalence was 5.8-fold higher in female SLE patients than in male patients in 2003 and increased to 7.8-fold in 2008. The highest prevalence rate by age was in the 30–39-year-old age group
Mortality
During the study period, the annual number of deaths increased from 220 to 313 and the total deaths were 1,611. The mortality rate with diagnosis of SLE was 117.6 per 10 million population. Female SLE patients had a higher mortality rate (194.5 per 10 million) than male SLE patients (42.8 per 10 million). Of all SLE deaths, the average age was 16.2 years and both sexes presented with similar age. The average rate of new-onset male SLE patients (disease duration less than 1 year) who died within 1 year was 7.24 % during the study period and was 2.58 % in female SLE patients. The fatality rate increased by 29.7 % per year in male SLE patients but remained steady in female SLE patients. The average standardized mortality rate during study period was 11.1 and was similar in both sexes (Table 3).
Discussion
The National Health Insurance Research Database (NHIRD) is well suited for the study of disease incidence because it provides medical records of nearly the entire population of Taiwan. It systematically includes diagnostic codes, but not all signs, symptoms, or laboratory findings leading to a diagnosis. The diagnosis of SLE is based on the revised American College of Rheumatology (ACR) criteria and is confirmed by rheumatologists. SLE is categorized as a catastrophic illness that must be registered in this database for government support. As such, the present population-based investigation using the NHIRD can reflect the true incidence of SLE.
Confidence intervals (CIs) are not reported because these are national population data rather than sample data. In the past, the incidence survey on SLE relied on hospital- or community-based records. However, the present study used a national registered database to reflect real-life conditions. In the past five decades until the 1990s, the average incidence of SLE was 3.06 per 100,000 population, with an increasing trend [8]. In a survey in the United Kingdom in the 1990s, the overall age-standardized incidence of SLE was 4.71 per 100,000 [9]. The mean annual incidence was estimated to be 3.1 per 100,000 in Hong Kong between 2000 and 2006, without an increasing trend [6]. In the present survey, the average incidence was 4.87 per 100,000 population, with gradual decrease. Early detection of low-avidity autoantibodies, probably combined with early use of protective cardiovascular measures, might have contributed to the reduced incidence in Norway [10].
As for the prevalence rate, variations show significant disparity in different countries. A survey in Shanghai showed a higher prevalence of 70 per 100,000 population, while surveys in India, Japan, and Saudi Arabia showed lower prevalences of 3.2–19.3 per 100,000 [11]. These variations cannot be ascribed only to genetic or ethnic factors but the sampling of enrolled populations. From the current survey, the study period revealed a prevalence of up to 97.5 per 100,000 population. Generally, the prevalence rate of pediatric-onset SLE is reportedly 3.3–8.8 per 100,000 children [12]. From a previous survey conducted in 1999, the prevalence rate of pediatric SLE in children aged <16 years old was 6.3 per 100,000 population, which was 1.8 per 100,000 in boys (males) [13]. However, prevalence was 5.3 for those younger than 10 years old, even 16.4 in children aged 10–19 years old in the current survey. The increasing proportion of pediatric-onset male SLE patients merits further investigation.
The disease predominantly affects women, especially those in their 20 and 30 s [14], with 10–15 % of patients diagnosed during childhood [2]. In the present study, SLE is nine times more common in females aged 20–50 years old compared to males. Even in the age group <10 years old, SLE is almost six times higher in females than in males and around three times higher after the age of 60 years. In the study period, incidence decreases prominently in female but not in male patients. The general trend of decreasing incidence is attributed to the female population. From this survey, the average male SLE incidence is 1.22 per 100,000 population, which remains steady each year.
In a European lupus cohort with 1,000 participants, female predominance is not as pronounced in the elderly onset group (female-to-male ratio 5:1) [15]. When dividing different age groups by 10 years of age, there is a high female-to-male ratio in the child-bearing age, followed by children and the elderly. Kariuki et al. [16] explained the association between rs9138 C and higher serum osteopontin and interferon-alpha as restricted to younger female SLE. Risk allele carriers showed a strong age-related genetic effect of the rs9138 genotype on both serum osteopontin and interferon-alpha. In the current survey, peak SLE incidence occurs at age 20–29 years, followed by 30–39 years old for females, but in 70–74 years old for males. The significant disparity in terms of sex is at the age of 40–49 years old.
SLE is a complicated disease with various presentations of severe events. Asian patients reportedly have more organ involvement and higher mortality [17]. Other studies have found that mortality is not particularly different between Asian and Caucasian lupus patients residing in the same countries [18]. The influences of socioeconomic factors like poverty and cultural practices on health care utilization and disease outcome have also been increasingly recognized. The risk of high mortality has been associated with female sex, younger age, and shorter SLE duration. The risk for certain types of death related to renal disease has decreased, while the risk of death due to circulatory disease does not appear to have diminished [19]. Most causes of death or shortened survival rate are infection, bacteremia, and renal diseases, without any difference between sexes [20, 21]. The cause of death related to SLE in each patient cannot be retrieved from the NHIRD. However, the NHIRD reveals that male SLE patients diagnosed within 1 year have higher mortality rates than female patients.
The mortality rate varies in both sexes during the period studied. New therapies have been developed to improve survival from this disease and even though the incidence has decreased over the years, the prevalence has increased gradually. The female-to-male ratio of incidence is 7.14 and the sex ratio of mortality is 4.6. As such, male patients have worse outcome. A small retrospective analysis covering the period 1987–1996 shows that male SLE patients have higher mortality at 1 year [22]. Male SLE patients may have different clinical or immunologic features compared to female patients. A retrospective analysis of mortality reveals that male sex is really a risk factor [23]. The accurate cause of death cannot be retrieved is the limitation of this study.
In conclusion, this population-based survey shows a declining trend in incidence rates for SLE in recent years, while prevalence rates remain steady. There is also a trend of increasing mortality rate in younger male SLE patients diagnosed within 1 year.
References
Pons-Estel GJ, Alarcón GS, Scofield L, Reinlib L, Cooper GS (2010) Understanding the epidemiology and progression of systemic lupus erythematosus. Semin Arthr Rheum 39:257–268
Moradinejad MH, Zamani GR, Kiani AR, Esfahani T (2008) Clinical features of juvenile lupus erythematosus in Iranian children. Acta Rheumatol Port 33:63–67
Fessel WJ (1988) Epidemiology of systemic lupus erythematosus. Rheum Dis Clin N Am 14:15–23
Fujukawa S, Okuni M (1997) A national study of rheumatic diseases among Japanese children. Acta Paediatr Jpn 39:242–244
Malleson PJ, Fung MY, Rosenberg AM (1996) The incidence of pediatric rheumatic diseases: results from the Canadian pediatric rheumatology association disease registry. J Rheumatol 23:1981–1987
Mok CC, To CH, Ho LY, Yu KL (2008) Incidence and mortality of systemic lupus erythematosus in a Southern Chinese Population, 2000–2006. J Rheumatol 35:1978–1982
Mok CC (2011) Epidemiology and survival of systemic lupus erythematosus in Hong Kong Chinese. Lupus 20:767–771
Uramoto KM, Michet J, Thumboo J, Sunku J, O’Fallon WM, Gabriel SE (1999) Trends in the incidence and mortality of systemic lupus erythematosus, 1950–1992. Arthr Rheum 42:46–50
Somers EC, Thomas SL, Smeeth L, Schoonen WM, Hall AJ (2007) Incidence of systemic lupus erythematosus in the United Kingdom, 1990–1999. Arthr Rheum 57:612–618
Eilertsen GØ, Fismen S, Hanssen TA, Nossent JC (2011) Decreased incidence of lupus nephritis in northern Norway is linked to increased use of antihypertensive and anticoagulant therapy. Nephrol Dial Transplant 26:620–627
Osio-Salido E, Manapat-Reyes H (2010) Epidemiology of systemic lupus erythematosus in Asia. Lupus 19:1365–1373
Kamphuis S, Silverman ED (2010) Prevalence and burden of pediatric-onset systemic lupus erythematosus. Nat Rev Rheumatol 6:538–546
Huang JL, Yao TC, See LC (2004) Prevalence of pediatric systemic lupus erythematosus and juvenile chronic arthritis in a Chinese population: a nationwide prospective population-based study in Taiwan. Clin Exp Rheumatol 22:776–780
Chiu YM, Lai CH (2010) Nationwide population-based epidemiologic study of systemic lupus erythematosus in Taiwan. Lupus 19:1250–1255
Cervera R, Khamashta MA, Hughes GRV (2009) The Euro-lupus project: epidemiology of systemic lupus erythematosus in Europe. Lupus 18:869–874
Kariuki SN, Moore JG, Kirou KA, Crow MK, Utset TO, Niewold TB (2009) Age- and gender-specific modulation of serum osteopontin and interferon-[alpha] by osteopontin genotype in systemic lupus erythematosus. Genes Immun 10:487–494
Kaslow RA (1982) High rate of death caused by systemic lupus erythematosus among U.S. residents of Asian descent. Arthr Rheum 25:414–418
Johnson SR, Urowitz MB, Ibañez D, Gladman DD (2006) Ethnic variation in disease patterns and health outcomes in systemic lupus erythematosus. J Rheumatol 33:1990–1995
Bernatsky S, Boivin JF, Joseph L et al (2006) Mortality in systemic lupus erythematosus. Arthr Rheum 54:2550–2557
Chen MJ, Tseng HM, Huang YL et al (2008) Long-term outcome and short-term survival of patients with systemic lupus erythematosus after bacteraemia episodes: 6-yr follow-up. Rheumatology 47:1352–1357
Chang DM, Chang CC, Kuo SY, Chu SJ, Chang ML (1998) The clinical features and prognosis of male lupus in Taiwan. Lupus 7:462–468
Prete PE, Majlessi A, Gilman S, Hamideh F (2001) Systemic lupus erythematosus in men: a retrospective analysis in a Veterans administration healthcare system population. J Clin Rheumatol 73:142–150
Manger K, Manger B, Repp R et al (2002) Definition of risk factors for death, end stage renal disease, and thrombo-embolic events in a monocentric cohort of 338 patients with systemic lupus erythematosus. Ann Rheum Dis 61:1065–1070
Author information
Authors and Affiliations
Corresponding authors
Additional information
Kuo-Wei Yeh and Chong-Hua Yu contributed equally to this work.
Rights and permissions
About this article
Cite this article
Yeh, KW., Yu, CH., Chan, PC. et al. Burden of systemic lupus erythematosus in Taiwan: a population-based survey. Rheumatol Int 33, 1805–1811 (2013). https://doi.org/10.1007/s00296-012-2643-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00296-012-2643-6