Abstract
Evaluation of MRI-findings in patients with involvement of psoriatic arthritis (PsA) in small joints in hands and feet. Twenty-five patients with symptomatic joint involvement were studied by MRI. All patients were found to be positive for one or more imaging criteria. Soft tissue oedema was identified in 22/25 (88%) patients. Joint effusion was observed in 23/25 (92%) patients, whereas bone erosion was seen in 20/25 (80%) patients. Bone marrow oedema was evident in 21/25 (84%) cases. In 12/25 (48%) cases, bone proliferation was noted. Tendon sheath effusion was present in 17/25 (68%) patients. Contrast enhancement of the synovia was detected in all patients (n = 25) (100%), whereas adjacent periost was enhanced in 22/25 (88%) and epiphysial bone marrow in 18/25 (72%) patients. MRI allows the assessment of PsA-alterations of soft tissue, cartilage, bone, bone marrow, and adjacent tendon sheath in patients with hand and foot involvement.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The first description of arthritis in a patient with psoriasis was published in 1818. The notion of a specific form of arthritis associated with psoriasis was first evaluated by Wright in 1956 [1]. There is still debate as to whether psoriatic arthritis (PsA) is a distinct entity [2]. Nowadays, attention focuses on the role of T-cells, cytokines, adhesion molecules, and angiogenetic factors of the skin and synovial membrane [2]. PsA as a spondyloarthropathy may be accompanied by skin and nail alterations [2]. Both, psoriasis and arthritis are frequent clinical conditions, and thus their occurrence is expected in the same patient population [3, 4]. There is a 5–7% incidence of PsA in patients with psoriasis [1]. Other authors found an even higher incidence of nearly 40% [3]. Psoriasis is primarily a skin disease that can affect the musculoskeletal system including synovial tissue, tendons, ligaments and bone [4, 5]. Synovial involvement causes inflammation of tendon sheaths, bursae and adjacent bone and bone marrow of the affected joints [6]. Unlike PsA, is inflammatory in nature, and affecting proximal and distal joints, and differing from rheumatoid arthritis by the lack of gender preference, frequent involvement of DIP-joints, and a tendency to asymmetry [5]. Moreover, lack of rheumatoid factors, presence of spondyloarthropathy, association with HLA-B-27, and extra-articular features are common to the spondyloarthropathies. Characteristic radiographic features of PsA include asymmetric distribution, affection of the DIP-joints, bone erosions with new bone formation, bony ankylosis, and resorption of distal phalanges [9–13]. By Ultrasound and MRI, articular and periarticular changes can be detected earlier than with conventional radiography [7–14]. Preliminary studies suggest that MRI features like synovialitis, bone erosions and bone marrow oedema may predict and indicate future joint damage [15, 16]. Other authors point out that the localisation of inflammatory changes may be useful in differentiating between rheumatoid arthritis and spondyloarhtropathy [12, 14–19].
MRI has a key impact on therapy decisions in patients with PsA because it visualises bone, bone marrow, and soft tissue changes [12, 14, 15, 19]. MRI is thus sensitive in assessing soft tissue and bone involvement in patients with inflammatory arthritis [5, 12, 15, 17]. The purpose of this study was to evaluate the frequency of MRI features in the detection of hand and foot involvement in patients with symptomaticPsA.
Materials and methods
Patients
Between April 2002 and May 2005, 25 consecutive patients (20 males, 5 females, mean age: 41.4 years, range 22–53 years) with PsA and symptomatic involvement of small joints in hand and feet were studied prospectively. The onset of psoriasis varied from 1 month to 5 years of age.
The following inclusion criteria were applied:
-
1.
Clinical diagnosis of PsA made by an experienced rheumatologist (U.W).
-
2.
No systemic therapy or photochemotherapy prior to the MRI-examination.
-
3.
Sero-negative for rheumatoid factors.
The most symptomatic hand (n = 13) or foot (n = 12) was examined.
Imaging
MRI examinations were performed using a 1.5 T Magnetom (Symphony, Siemens, Erlangen, Germany) with a surface coil. The following sequences were applied in axial, coronal, and sagittal planes according to the anatomy and PsA site: fast STIR sequence (TR 4890–5792/TE 60/TI 120), T1-weighted spin echo (SE) (TR 715–838/TE 20), T2-weighted turbo spin echo (TSE) (TR 3474/TE 96).
Slice thickness was 3–5 mm, matrix size 220 × 256, and the field of view (FOV) was 16 cm. Intravenous contrast media was applied in a standard dosage of 0.1 mmol Gd-DTPA (Magnevist, Schering, Berlin, Germany) per kg body weight.
After IV contrast media application, T1-weighted fat-suppressed spin echo sequences were performed in two planes.
Evaluation
Two expierenced musculo-skeletal radiologists (MU, NG) who had been blinded to clinical findings evaluated the MRI images in a consensus fashion. Manifestation of hand and foot involvement of PsA was classified based on a patient analysis and a regional assessment according to the following three joint regions in each patient:
- I:
-
= carpus/tarsus (n = 1)
- II:
-
= metatarsophalangeal (MTP)/metacarpophalangeal (MCP) joints (n = 5)
- II:
-
= proximal distal interphalangeal (PIP/DIP) joints (n = 9)
All in all, 15 different joint regions of the most seriously affected symptomatic hands and/or feet were evaluated. In our patient cohort nearly 375 different small joints and regions had to be considered.
A semiquantitative assessment of bone, bone marrow, and soft tissue findings was performed as reported in literature [8] using the following criteria: Soft tissue oedema, bone marrow oedema, bone erosion and proliferation, and tendon sheath effusion. Furthermore, we evaluated contrast enhancement of the adjacent synovia, periost and bone marrow of the affected joints. Semiquantitative assessment of abnormalities of joints, soft tissue, bone, bone marrow and tendons was based on a four-point grading score (0 = no, 1 = slight, 2 = moderate, 3 = strong abnormality).
Results
All patients were found to be positive for one or more signs of PsA. MRI-features were evaluated in 12 hands and 13 feet.
Based on the patient analysis, a transversal distribution pattern of joint involvement was seen in 11/25 (44%) patients (Figs. 1, 3). A mixed type was observed in 2/25 (8%) patients with hand manifestation of PsA. One ray distribution was described in 12 cases (Figs. 2, 3, 8).
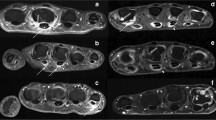
Fifty-three-year-old male with a 2 years history of psoriatic arthritis. A transversal distribution pattern of the symptomatic right hand is demonstrated by MRI. An axial STIR image shows diffuse soft tissue swelling suggesting for oedema in digitus I (strong) and IV (mild), especially in the plantar region consistent with sausage digits presenting dactylitis (arrows). Please note the concomitant bone marrow odema in the first digit and the tendon sheath effusion in the fourth digit indicating tendosynovialitis (arrow)
Table 1 summarised the morphologic and contrast enhancement MRI findings in PsA.
Soft tissue oedema was identified in 22/25 (88%) patients (Figs. 1, 4, 5, 6, 7, 8); and joint effusion in 23/25 (92%) patients (Fig. 2). Bone erosion was seen in 20/25 (80%) patients (Fig. 3), whereas adjoining bone marrow oedema was evident in 21/25 (84%) cases (Figs. 1, 4, 8, 9a). Bone proliferation was found in 12/25 (48%) cases (Fig. 3). Tendon sheath effusion was seen in 17/25 (68%) patients (Figs. 1, 6).
Thirty-eight-year-old male patient with a long history of severe foot involvement. Axial T1-weighted fat-suppressed postcontrast image shows osteopro-liferative changes in the metatarsophalangeal joint. A strong contrast enhancement is visible in the periost and surrounding soft tissue, especially in the plantar fascie and adjacent flexor tendon sheath
Forty-four-year-old patient with a long history of psoriatic arthritis presenting with a symptomatic right hand. The coronal STIR image (a) reveals bone marrow oedema in the os lunatum. At this final stage of disease, no contrast enhancement of the carpus, the adjacent bony, synovial and bone marrow structures is visible (b)
Contrast enhancement of the synovia was detected in all patients (n = 25). The adjacent periost was enhanced in 22/25 (88%) patients, and epiphysial bone marrow in 8/25 (32%) patients (Figs. 4, 5, 8).
All in all, 60/275 (16%) small joints revealed one or more morphological changes including contrast media uptake. The morphological changes in PsA joints varied between 6 and 14% (Table 1). The leading criteria were synovial contrast enhancement in 55/60 (92%) of small joints (Fig. 6), followed by intraarticular effusion in 51/60 (85%) with a slight distribution pattern predominating in 25/51 cases (42%), as demonstrated in Table 1 and Figs. 2, 6). In nine joints (15%), there was no contrast enhancement as illustrated in Fig. 9b.
No differences were found regarding involvement pattern, frequencies of morphological alterations, and contrast media uptake between the joint regions of hand and feet, with the exception of the tarsus, which only affected in 2/12 (17%) patients, compared to the carpus which was involved in 8/13 (62%). In two of the latter, only the carpus was involved (Fig. 9); in the other six, the adjacent metacarpophalangeal joint were involved as well. Metatarsophalangeal joints were affected in 10/12 patients with foot involvement (83%) (Figs. 2, 7). In those 10 patients 19 MTP-joints were involved to a different extent and in a manner other than that in the above mentioned criteria (Figs. 2, 7). With foot involvement, both a one-ray distribution (Fig. 2) and a transversal-type distribution (Fig. 7) were observed in 6/12 (50%) patients. Mixed distribution was not found with foot involvement.
Metacarpophalangeal-involvement (n = 25) was detected in 11/13 patients with hand involvement (85%) (Figs. 1, 3, 4, 6, 8). Interphalangeal joint involvement was present in 2/13 (15%) patients with foot manifestation, and in 4/12 (33%) of patients with hand manifestation.
Among morphological changes in PsA, the most striking criterium was an intra-articular effusion in 51/60 joints (85%). In 25/51 joints (42%), the intra-articular effusion was classified as slight, whereas a moderate degree was observed in 18 (30%) cases. A large intraarticular effusion was detected in eight cases (13%) as demonstrated in Fig. 2.
Soft tissue oedema adjoining the psoriasis involvement was depicted by MRI in 41/60 cases (68%) (Figs. 1, 4, 8). Ninteen small joints were free of soft tissue oedema. Slight soft tissue oedema was seen in 9 cases (15%), a moderate case in 17 cases (28%) (Figs. 4, 8) and a strong oedema in 15 (25%) (Fig. 1). Bone marrow oedema was observed in 37/60 cases (62%) (Figs. 1, 4, 8, 9a) with the following distribution: slight n = 10 (15%) (Fig. 7), moderate n = 15 (25%) (Figs. 4, 9a), strong n = 12 (20%) (Fig. 1).
We noted tendon sheath effusion respresenting synovialitis in 33/60 small joints (55%) as illustrated in Figs. 1 and 6. The effusion was judged to be slight in 17%, and in 13% to be moderate (Fig. 5).
Bone alterations such as erosion and proliferations were detected in 39/60 (65%), and in only 22/60 (37%) of joints affected with PsA (Figs. 3, 5). Bone erosion were observed in 39/60 cases (65%) with slight erosion in 21/51 cases (35%). A moderate extent of bone erosion was found in 13/60 cases (22%), and in 9/60 of the affected joints (15%), bone erosion as regarded as strong (Fig. 3a). 21/60 joints (35%) showed no bone erosion.
Bone proliferation as detected in 22/60 (37%) with revealing a slight extent in 10/60 (17%). We observed a moderate (Fig. 5) and a slight distribution pattern was seen in six cases (10%), respectively. Thirty-eight out of 60 (63%) joints showed no bone proliferative changes.
In considering the average contrast enhancement for quantitative assessment purposes, the synovia (2.03) were more strongly enhanced than the adjacent periost (1.43) followed by the bone marrow (1.03) (Table 1). The synovia were enhanced in 55 joints (92%), the periost in 41 joints (68%), and the concomitant bone marrow in 34 joints (57%) (Figs. 4, 5, 6).
Contrast uptake of the synovia of the affected small joints was moderate in 27/60 cases (45%) and strong in 20/60 cases (33%). Slight contrast enhancement was detected in eight cases (13%). Five affected joints demonstrated no contrast media enhancement. The adjoining periost was enhanced in 41/60 joints (68%), whereas in 19 cases (32%), no contrast enhancement was seen. Moderate and strong contrast media enhancement was observed in 15 cases (25%) each, respectively, and slight contrast media enhancement in 11/60 joints (18%). As opposed to the adjoining synovia and periost of the epiphyseal bone, adjacent bone marrow was affected in 34/60 cases (57%) with a variegated distribution pattern as demonstrated in Table 1.
When we consider the total number and average values of the quantitative assessment of all small joints affected by PsA, the contrast enhancement of the synovia and intra-articular effusion were the predominant features in MRI. Contrast enhancement of the adjoining periost and morphological changes like soft tissue oedema, bone marrow oedema and tendon sheath effusion were identified in the majority of the small psoriatic joints to a moderate-to-strong extent as demonstrated in Table 1. Features such as bone erosion were observed in 39/60 (65%) to, a slight-to-moderate manner (average value of 1.79). However, we noted bone proliferation were in only 22/60 cases (37%) (average value of 1.82).
Discussion
Despite the advances in imaging over the past 20 years and the important role of MRI in routine clinical diagnostics in patients with musculo-skeletal diseases, there are few papers that systematically analyse the value of MRI in the small joints of the hand and feet [12, 17, 20–22].
Most papers dealing with MR-imaging in small joints focussed on the wrist joints with rheumatoid arthritis [20, 21]. Few investigators have analysed imaging modalities for PsA [12, 15, 17–19, 22]. Wassenberg et al. [23] reported a scoring system in radiographic features of PsA, others dealt with ultrasound imaging findings in patients with PsA [18, 23]. Few authors have investigated the MR-imaging features of the hands of patients with psoriatic disease [12–15, 23–27]. Those found MRI useful in identifying subclinical joint involvement in patients with PsA. Offidani et al. [17] mentioned that MRI allows the aedequate evaluation of joints involved by psoriatic disease and that it was superior to X-rays in revealing clinically and radiologically-silent lesions.
Psoriatic disease affecting the musculo-skeletal system may involve synovial tissue as well as tendons, ligaments and fascia, bone and bone marrow [4–6, 9–15] as illustrated in Table 1 and elsewhere in this paper.
Inflammation of the enthesis is a morphological hallmark of psoriatic disease in association with, tendon sheath involvement (Figs. 1, 5, 6). We observed tendon sheath effusion in 17/25 patients (68%) and a moderate to strong degree in 33/60 affected joints (55%).
Investigating 11 cases of fingers with dactylitis involvement and in comparison to their correspond yet unaffected contralateral fingers, Olivieri et al. [26] did not observe entheseseal involvement of the flexor digitorum tendons using a fast T2-weighted SE sequence with fat saturation. Joint effusion is also common and presents with effusion and synovial proliferation, both which are reliably detected on T2-weighted images and fat-sat contrast-enhanced T1-weighted images (Figs. 2, 6, 7). In more advanced psoriatic disease, destructive arthropathy can occur with severe central erosion resulting in arthritis multilans (Fig. 3) [9–11, 13, 14].
Psoriatic arthritis often involves the hands, varius digits and often multiple joints in one-ray and patchy distribution [9–11, 15]. Two patients had patchy distribution in our study.
The majority of patients showed a transversal type of distribution (n = 11) or one ray distribution (n = 12) leading to dactylitis of a finger (n = 6) or toe (n = 6) (Figs. 1, 4, 5, 7, 8). Dactylitis was thought to be due to concomitant flexor tendon synovialitis and arthritis in the MTP/MCP or IP-Joints [1, 18, 24]. On the other hand, Olivieri et al. reported recently that ultrasound and MRI revealed flexor tenosynovialitis as the main cause of finger dactylitis [24]. One observes adjoining joint effusion in Fig. 4, but joint capsula distention was found in only 2/36 toe joints with dactylitis in their study. No joint involvement was reported with the dactylitis [26]. As far as our results are concerned, the sausage-like appearance of fingers and toes indicating dactylitis is caused by a combination of soft tissue oedema, tendon sheath effusion and intra-articular joint effusion in patients with one-ray distribution (Fig. 1–8). On MR imaging, one sees in a sausage digit a diffuse soft tissue oedema and tendosynovitis with increased tendon sheath fluid and enhancement related to synovial proliferation [25], as demonstrated in Fig. 1. However, this can be found in all forms of spondyloarthro-pathies, especially in Reiter disease [12, 27].
The most frequent MR imaging feature was synovial contrast enhancement observed in 55/60 cases (92%) (Fig. 6). This is caused by active inflammation of tendon sheath, bursae and joints, and is clearly depicted by contrast enhanced, fat-suppressed images [6, 12, 15] (Fig. 6). The adjoining periost was enhanced in 41/60 joints (68%), and adjacent bone marrow was affected in 34/60 joints (57%) with a variegated distribution pattern (Table 1; Figs. 5, 6). Fluid and enhancing synovial tissue is observed in joints and adjacent bursae (Figs. 4, 6). In contrast-enhanced sequences, the inflamed synovial tissue is strongly enhanced as reported previously [12, 15, 16].
In comparison to the concomitant periost (68%) and adjoining epiphyseal bone marrow (70%), the synovial enhancement was seen in 92%. In semiquantitative assessment, synovial enhancement was seen in a moderate manner (2.03), whereas values were average for the periost (1.43) and bone marrow (1.03). Bone marrow oedema is an important imaging feature in inflammatory disease. There are reports bone marrow oedema is a predictor of bone erosion [21]. We detected bone marrow oedema in nearly 70% of all affected joints (Figs. 1, 4, 5, 7, 8, 9a).
Totterman [27] stressed that the location and the extent of bone marrow in PsA varies from those seen in rheumatoid arthritis and osteoarhritis.
In conclusion, MRI can visualize inflammatory soft tissue, cartilage, bone, bone marrow and tendon sheath in patients with psoraitic arthritis. STIR-Sequences demonstrate bone marrow, soft tissue oedema, joint and tendon sheath effusion indicating PsA.
Using intravenous contrast media, MRI is capable of demonstrating contrast media uptake of the synovia, adjoining periost, epiphyseal bone marrow.
The assessment of small joints in hands and feet in MRI should assume an integral part in the diagnosis of PsA. Moreover, MRI`s clinical value must be proven in prospective studies monitoring new drug regimens. Treatment of PsA can be modulated according to the activity and severity of psoriatic disease in patients with hand and foot manifestation.
References
Wright V (1956) Psoriasis and arthritis. Ann Rheum Dis 15:348
Gladmann DD (1995) Criteria for psoariatic arthritis. Baillieres Clin Rheumatol 9:319
Cats A (1990) Psoriasis and arthritis. Cutis 46:323
Van Romunde LKJ, Cats A, Hermans J, Valkenberg HA, de Vries E (1984) Psoriasis and arthritis III A cross sectional comparative study of patients with psoriatic arthritis and seronegative and seropositive polyarthritis: radiological and HLA-aspects. Rheumatol Int 4:67
Gladmann DD (1997) Psoraitic arthritis. In: Kelly WN, Ruddy S, Harris ED, Sledge C (eds) Textbook of rheumatology 5th edn. WB-Saunders, Philadelphia, pp 999–1005
Scarpa R, Mathieu A (2000) Psoriatic arthritis: evolving concepts. Curr Opin Rheumatol 12:274–280
Wright V (1987) Skin and arthritis. Clin Exp Rheumatol 5(Suppl 1):75–79
Block SR (1979) Psoriatic arthritis. J Maine Med Assoc 70:326–329
Dihlmann W (1987) Topographische Röntgendiagnostik des Gleitgewebes-Gelenke der Hand In: Gelenk-Wirbelverbindungen Dihlmann W 3. Auflage Thieme Stuttgart, pp 95–190
Freyschmidt J (1993) Erkrankungen der Gelenke: Seronegative Spondarthriden. In: Freyschmidt. J Skeletterkrankungen. Springer, Berlin Heidelberg New York, pp 590–618
Freyschmidt J, Freyschmidt G (1996) Rheumatische Erkrankungen: Psoriasis-pondarthritis und Psoriasisarthritis In: Freyschmidt J, Freyschmidt G Haut-, Schleimhaut-und Skeletterkrankungen SKIBO-diseases Springer, Berlin Heidelberg New York, pp 93–100
McGonagle D (2005) Imaging the joint and enthesis: insights into pathogenes of psoriatic arthritis. Ann Rheum Dis 64:58–60
Resnick D, YU JS, Sartoris D (1997) Imaging. In: Kelly WN, Ruddy S, Harris ED, Sledge C (eds) Textbook of rheumatology 5th edn WB-Saunders, Philadelphia, pp 626–686
Resnick D, Niwayama G (1981) Psoriatic arthritis. In: Resnick D, Niwayama G (eds) Diagnosis of bone and joint disorders. WB-Saunders, Philadelphia pp 1103–1117
Ory RA, Gladman DD, Mease PJ (2005) Psoriatic arthritis and imaging. Ann Rheum Dis 64:55–57
Weishaupt D, Schweitzer ME, Alam F, Karasick D, Wapner K (1999) MR imaging of inflammatory joint diseases of the foot and ankle. Skeletal Radiol 28:663–669
Offidani A, Cellini A, Valeri G, Giovagnoni A (1998) Subclinical joint involvement in psoriasis: magnetic resonance imaging and X-ray findings. Acta Derm Venereol 78:463–465
Coari G, Iagnocco A, Mastrantuono M, De Cata A, Persia S, Passariello R et al (1994) Sonographic and NMR imaging study of sausage digit. Acta Derm Venereol 186:33–34
Giovagnomi A, Grassi W, Terilli F, Blasetti P, Paci E, Ercolani P et al (1995) MRI of the hand in psoriatic and rheumatical arhritis. Eur Radiol 5:590–595
Ostergard M, Hansen M, Stoltenberg M, Gideon P, Klarlund M, Jensen KE, Lorenzen I (1999) Magnetic resonance imaging-determined synovial marker of disease activity and a predictor of progressive joint destruction in the wrists of patients with rheumatoid arthritis. Arthritis Rheum 42:918–929
Mc Queen FM, Stewart N, Crabbe J, Robinson E, Yeoman S, Tan PL, McLean (1999) Magnetic resonance imaging of the wrist in early rheumatoid arthritis reveals progression of erosions despite clinical improvement. Ann Rheum Dis 58:156–163
Yalcin B, Yalcin S (1999) Psoriatic arthritis and magnetic resonance imaging. Acta Derm Venereol 79:407
Wassenberg S, Fischer-Kahle V, Herborn G, Rau R (2001) A method to score radiographic change in psoriatic arthritis. Z Rheumatol 60:156–166
Olivieri I, Padula A, Favaro L, Pierro A, Oranges GS, Ferri S (1995) Dactylitis with pitting oedema of the hand in longstanding ankylosing spondylitis. Clin Rheumatol 14:701–704
Olivieri I, Barozzi L, Pierro A, De Matteis M, Padula A, Pavlica P (1997) Toe dactylitis in patients with spondylarthropathy: assessment by magnetic resonance imaging. J Rheumatol 24:926–930
Olivieri I, Salvarani C, Cantini F et al (2002) Fast spin echo-T2-weighted se-quences with fat saturation in dactylitis of spondyloarthritis. No evidence of en-theseal involvement of the flexor digitorum tendons. Arthritis Rheum 46:2964–2967
Totterman SM (2004) Magnetic resonance imaging of psoriatic arthritis: insight from traditional and three-dimensional analysis. Curr Rheumatol Rep 6:317–321
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ghanem, N., Uhl, M., Pache, G. et al. MRI in psoriatic arthritis with hand and foot involvement. Rheumatol Int 27, 387–393 (2007). https://doi.org/10.1007/s00296-006-0220-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00296-006-0220-6