Abstract
The aim of our study was to evaluate the clinical efficacy, safety, and tolerability of ornidazole in patients with rheumatoid arthritis (RA). This was 3 months, randomized, double-blind,placebo-controlled study. A total of 160 patients with active RA were randomly assigned to receive 1,000 mg ornidazole (n = 53), 500 mg ornidazole (n = 55), or placebo (n = 52). A significantly greater percentage of patients treated with 1,000 mg ornidazole met the American College of Rheumatology 20% improvement criteria (achieved an ACR20 response) at 3 months compared with patients who received placebo (62.0 vs. 32.4%; P < 0.001). Greater percentages of patients treated with 1,000 mg ornidazole also achieved ACR50 responses (38.3 vs. 10.9%; P < 0.001) and ACR70 responses (19.6 vs. 1.2%; P < 0.001) compared with patients who received placebo. Ornidazole treatment was also associated with significant reductions in pain and duration of morning stiffness, significant improvement in the quality of life and both the physician’s and patient’s global assessments, and significant reductions in disease activity as assessed by objective laboratory measures (erythrocyte sedimentation rate and C-reactive protein level). Ornidazole was well tolerated. There were no dose-limiting toxic effects. In this 3-month-trial ornidazole was safe, well tolerated, and associated with improvement in the inflammatory symptoms of RA.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Rheumatoid Arthritis (RA) is a polyarticular, chronic, inflammatory, and systemic disease [1]. Although many disease modifying antirheumatic drugs (DMARDs) and biological agents are used in the treatment of this disease, new pharmaceutical drugs are required to achieve better efficacy and lower costs.
In many previous studies, this rheumatic disease has been found at frequencies in individuals with periodontitis, and rheumatoid arthritis resembles periodontitis in many pathological aspects [2, 3]. High levels of oral anaerobic bacterial antibodies have been found in the serum and synovial fluid of RA patients [4, 5].
Previously, antibiotic treatment was employed in RA therapy. Sulfasalazine and tetracycline are effective against this disease [6–8]. The chronic use of sulfasalazine decreases gram-negative anaerobic bacteria numbers and Bacteroides in the intestines of patients with rheumatoid arthritis [9].
Ornidazole is a nitroimidazole antiprotozoal drug that also has potent antibacterial activity against anaerobes, including Bacteroides and Clostridium species [10]. Its adverse effects are very mild when compared with those of metronidazole [11]. Adverse effects include nausea, headache, dry mouth, or metallic taste in the mouth. It was demonstrated that it has efficacy surpassing 94% especially against oral anaerobic bacteria in the alveolar canal [12].
Biologic treatment agents and specifically TNF alpha-blockers are effective in rheumatoid arthritis therapy [13, 14]. Periodontopathic bacteria are powerful simulators for TNF alpha and other pro inflammatory cytokines in humans [15–18].
In this study, the purpose was to monitor the efficacy and reliability of ornidazole, an effective antibiotic specifically against Bacterioides genus in rheumatoid arthritis treatment.
Materials and methods
Patients
Patients enrolled in this study met the ACR (formerly, the American Rheumatism Association) criteria for the diagnosis of RA (functional classes I, II, or III) [19]. Patients had active disease defined as ≥ 12 tender joints based on a 68 joint assessment, ≥ 10 swollen joints based on a 66 joint evaluation, and either an erythrocyte sedimentation rate (ESR) ≥ 28 mm/1 h or a serum C reactive protein (CRP) concentration ≥ 2.0 mg/dl.
Previous DMARD therapies such as hydroxychloroquine, gold, sulfasalazine, methotrexate, azathioprine, leflunomide, and cyclosporine were applied and turned out to be insufficient in the treatment of the patients. Disease modifying antirheumatic drugs were discontinued at least 4 weeks before the study began. Stable doses of nonsteroidal antiinflammatory drugs and prednisone (≤ 10 mg daily) were allowed.
The criteria for exclusion from the study were as follows: use of antibiotics in the last 30 days prior to the study, some other infection, renal or hepatic functional dysfunction, and pregnancy. Women of reproductive age were required to take appropriate contraceptive measures.
Study protocol
This study was conducted from August 2001 to August 2005 in Salihli and Nazilli State Hospital in Turkey. This was 3 months, randomized, double-blind, placebo-controlled study. The study was conducted in accordance with the principles of Good Clinical Practice, according to the Declaration of Helsinki. Before entering this study, all patients gave written informed consent. Basic clinical measures included the below parameters: complete count of swollen and tender joints (68 joints evaluated; cervical spine and hips evaluated only for tenderness), duration of morning stiffness, disability [disability index of the health-assessment questionnaire (HAQ)] [20], physician’s and patient’s global assessment on scale from 0 (asymptomatic) to 10 (severe symptoms), patient’s assessment of pain on a visual–analogue scale from 0 (no pain) to 10 (severe pain) [21], Westergren erythrocyte sedimentation rate, and CRP [22]. The disease activity measures taken after the first day were repeated once every week in the first month and biweekly in the following period.
The patients were taken in for control first on a biweekly basis following the 3-month study. These controls continued until the number of patient inflamed joints reached pre-study levels or the patient required a previously administered therapy or a new one.
During the study as well as post-study evaluation period, hematological tests, serum biochemistry, and urinary analysis test assays were performed. All patients were evaluated for side effects and laboratory anomalies.
Treatment
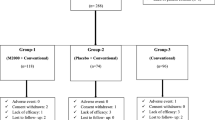
Patients were randomly assigned to one of the three treatment groups: placebo orally, 500 mg ornidazole (250 mg twice a day) orally, or 1,000 mg ornidazole (500 mg twice a day) orally.
Concomitant medications
The use of analgesics such as acetaminophen, codeine, or propoxyphene for pain relief was permitted.
Statistical analysis
The percent change from base line to 3 months in the swollen-joint count, tender-joint count, and total count of swollen or tender joints was the primary measure of efficacy. Secondary end points included pain, quality of life, duration of morning stiffness, erythrocyte sedimentation rate, C-reactive protein level, and physician’s and patient’s global assessments. If a subject withdrew from the study, the last available value was used as the 3 month value. The data were also analyzed to determine the number of patients meeting American College of Rheumatology criteria for 20, 50, and 70% improvement.
An ACR 20 response is defined as a reduction of at least 20% in the number of tender joints and swollen joints plus an improvement of at least 20% in at least three of the following five criteria: patient’s assessment of pain, patient’s assessment of disease activity, physician’s assessment of disease activity, patient’s assessment of physical function, and serum C-reactive protein concentration [23, 24]. ACR50 and ACR70 were defined in the same manner as ACR20 but with ≥ 50 or ≥ 70% degree of improvement, respectively.
The values were compared with the use of analysis of variance. The percentages of patients with ACR 20, ACR 50, and ACR 70 responses were compared with the use of chi-square tests.
Results
The pre-treatment baseline characteristics of patients are given in Table 1. A total of 160 patients between the ages 19 and 69 were included in the study, 35 of them males and the remaining 125 females. There were no significant differences between the groups in neither baseline disease activity nor pre-treatment characteristics. A total of 53 patients completed 1,000 mg ornidazole daily, 55 patients completed 500 mg ornidazole daily, and 52 patients completed placebo therapy. The primary response for withdrawal from the study was the noncontrol of arthritis symptoms. About 5% of the patients in 1,000 mg daily ornidazole group withdrew from the study. The ratios of withdrawals for 500 mg ornidazole and placebo groups were 19 and 42%, respectively.
Efficacy
Ornidazole had an effect of pronounced improvement in the assays of disease activities (Table 2). These improvements were observed to be most prominent in the group that was administered 1,000 mg ornidazole. While the total joint numbers showed a decrease of 60%, in placebo this ratio was 20% (P < 0.001). A positive correlation can be observed between the increased dosage of ornidazole and decrease in swollen joint count and tender joint count. The decreases in the number of swollen and tender joints commenced in the first week and continued until the end of treatment.
Ornidazole treatment was also associated with significant reductions in pain and duration of morning stiffness, significant improvement in the quality of life and physician’s and patient’s global assessments, and significant reductions in disease activity as assessed by objective laboratory measures (erythrocyte sedimentation rate and C-reactive protein level) (Table 2).
While patients complying with ACR20 criteria in the 1,000 mg ornidazole treatment group at the end of the 3-month period were 62.0%, this ratio was 32.4% in the placebo group (P < 0.001). While patients complying with ACR50 criteria in the 1,000 mg ornidazole treatment group at the end of the 3-month period were 38.3%, this ratio was 10.9% in the placebo group (P < 0.001). While patients complying with ACR70 criteria in the 1,000 mg ornidazole treatment group at the end of the 3-month period were 19.6%, this ratio was 1.2% in the placebo group (P < 0.001) (Table 3).
Safety and tolerability
Ornidazole was well tolerated and no deaths were reported by ornidazole-treated patients after 6 months of treatment. In general, adverse events were reported at a similar or lower rate in the ornidazole groups than in the placebo group. The most frequently reported adverse events were headache, dry mouth, and nausea (seen in at least 5% of patients, and excluding worsening of RA).
Only one patient withdrew because of an adverse event related to ornidazole (vertigo). Fewer adverse events were reported in the group given 500 mg ornidazole than in the group given 1,000 mg ornidazole.
No major abnormalities in hematologic findings or serum chemical profiles were noted during or after the study; in fact, improvements in anemia and decreases in platelet counts were seen, which reflect a reduction in disease activity.
Discussion
The results of this randomized, double-blind trial show the clinical efficacy of ornidazole in patients with active rheumatoid arthritis. Treatment with ornidazole for 3 months was associated with a reduction in disease activity as assessed by a number of clinical end points, biochemical markers of disease, and quality of life.
Ornidazole produced significant, rapid, and sustained reductions in disease activity. A strong, consistent dose–response relation was seen in most of the variables measured. The few adverse events noted—headache, dry mouth, and nausea—were mild and easily managed.
Cessation of therapy was associated with an increase in disease activity, suggesting that continued administration of ornidazole is necessary for sustained effect.
Ornidazole, a synthetic nitroimidazole derivative, is used in the treatment of infections caused by periodontopathic bacteria [9]. Porphyromonas gingivalis, Prevotella intermedia, Prevotella melaninogenica, and Bacteroides forsythus are Gram-negative anaerobic bacteria and are considered to be directly responsible for the periodontitis (Periodontopathic bacteria) [25]. These bacteria are members of the normal human mouth flora, where they cause endodontitis, odontogenic inflammation, gingivitis, and mainly periodontitis [26].
High levels of IgG antibodies against this bacteria were found in synovial fluid and blood samples from patients with RA [4, 5]. P. gingivalis has arginine and lysine specific proteinase [27]. Citrullination or deamination of arginine residues in autoantigenic proteins (profilaggrin/filaggrin, fibrinogen/fibrin, keratin, and vimentin) creates epitopes that are targeted by rheumatoid autoantibodies [28]. Arginine is the most important of the amino acids associated with autoantigenicity in proteins.
RA patients have significantly fewer galactose residues on their IgG Fc compared with age-matched healthy control subjects. A lack of terminal galactose residues early in disease is associated with a worse prognosis [29]. P. melaninogenica, as a saccharolytic bacteria, disintegrates galactose. Consequently, P. melaninogenica this causes this condition by binding to the Fc region of the IgG molecule and metabolizing galactose with its enzymes.
RFs have been identified as autoantibodies that react to the IgG molecule in the Fc region, and these antibodies may be of IgM, A, G, or E epitopes. P. gingivalis proteinase is responsible for the epitope development in the RF Fc region. Bonagura et al. identified lysine, histidine, arginine amino acid sequences for the Fc region of the IgG molecule [30]. Because P. gingivalis specifically decomposes lysine and arginine, the IgG3 CH2 and CH3 domains processed by P. gingivalis proteinase become powerful targets for the RF produced by rheumatoid cells [31]. The chronic inflammation associated with periodontitis appears to significantly increase the formation of IgM-RF. The RF detected in the serum of some periodontitis patients may be elicited by certain micro-organisms in the subgingival plaque [32, 33].
High levels of periodontopathic bacteria heat shock proteins were found in the serum of RA patients [34]. P. gingivalis has a 60-kDa heat shock protein (hsp.GroEL). Due to the similarity of the P. gingivalis hsp 60 molecule to the human hsp 60 molecule, it is a key molecule for GroEL homolog autoimmune reactions [35]. P. melaninogenica and P. intermedia heat-shock proteins of approximately 70-kDa have been found in periodontal disease processes [36]. Ueki et al. determined that P gingivalis GroEL and human hsp60 antibodies are found increasingly more often in periodontal patients compared to those without disease [37]. Hsp 70 antibodies have been detected in the synovial tissue of RA patients and when the hsp 70 expression is induced with certain stress stimulating factors, proinflammatory cytokines (tumor necrosis factor-alpha, IL-1, IL-6) develop in the RA synovium [38].
P. gingivalis, P. intermedia, P. melaninogenica, and B. forsythus produce gingival tissue destruction and autoimmune responses in periodontitis patients. T-cell and antibody responses in periodontitis confirm the infiltration of reactive T-cell clones into periodontitis lesions. These lesions demonstrate higher proliferative responses of peripheral blood mononuclear cells, cytokines (gamma-interferon, IL-4), and T-cell clonality. These findings indicate the presence of an active antibody response in synovial tissue and illustrate a potential connection between periodontal and inflammatory joint diseases.
Ornidazole treatment in RA is very economical compared with other treatments. It can be concluded from this study that ornidazole is an effective treatment for RA. The above evidence indicates that antibodies formed against oral anaerobic bacteria could be important in the aetiopathogenesis of RA. However, further studies are needed to confirm this.
References
Albani S, Carson DA (1997) Etiology and pathogenesis of rheumatoid arthritis. In Koopman WJ (ed) Arthritis and allied conditions. Williams and Wilkins, Baltimore, pp 979–992
Mercado FB, Marshall RI, Klestov AC, Bartold PM (2001) Relationship between rheumatoid arthritis and periodontitis. J Periodontol 72:779–787
Greenwald RA, Kirkwood K (1999) Adult periodontitis as a model for rheumatoid arthritis (with emphasis on treatment strategies). J Rheumatol 26:1650–1653
Ogrendik M, Kokino S, Ozdemir F, Bird PS, Hamlet S. Serum antibodies to oral anaerobic bacteria in patients with rheumatoid arthritis. URL: http://www.medscape.com/viewarticle/505458. Accessed 2005 June 16
Moen K, Brun JG, Madland TM, Tynning T, Jonsson R (2003) Immunoglobulin G and A antibody responses to Bacteroides forsythus and Prevotella intermedia in sera and synovial fluids of arthritis patients. Clin Diagn Lab Immunol 10:1043–1050
Hannonen P, Mottonen T, Hakola M, Oka M (1993) Sulfasalazine in early rheumatoid arthritis. A 48-week double-blind, prospective, placebo-controlled study. Arthritis Rheum 36:1501–1509
Pinals RS, Kaplan SB, Lawson JG, Hepburn B (1986) Sulfasalazine in rheumatoid arthritis. A double-blind, placebo-controlled trial. Arthritis Rheum 29:1427–1434
O’Dell JR, Blakely KW, Mallek JA, Eckhoff PJ, Leff RD, Wees SJ et al (2001) Treatment of early seropositive rheumatoid arthritis: a two-year, double-blind comparison of minocycline and hydroxychloroquine. Arthritis Rheum 44:2235–2241
Kanerud L, Scheynius A, Nord CE, Hafstrom I (1994) Effect of sulphasalazine on gastrointestinal microflora and on mucosal heat shock protein expression in patients with rheumatoid arthritis. Br J Rheumatol 33:1039–1048
Bartlett JG (1982) Anti-anaerobic antibacterial agents. Lancet 2:478–481
Jokipii AM, Jokipii L (1981) Metronidazole, tinidazole, ornidazole and anaerobic infections of the middle ear, maxillary sinus and central nervous system. Scand J Infect Dis Suppl 26:123–129
Kargul B, Kadir T (2001) The antibacterial effects of ornidazole on primary molars with infected pulps. Chemotherapy 47:203–207
Moreland LW, Baumgartner SW, Schiff MH, Tindall EA, Fleischmann RM, Weaver AL et al (1997) Treatment of rheumatoid arthritis with a recombinant human tumor necrosis factor receptor (pg75)-Fc fusion protein. N Engl J Med 337:141–147
Weinblatt ME, Kremer JM, Bankhurst AD, Bulpitt KJ, Fleischmann RM, Fox RI et al (1999) A trial of etanercept, a recombinant tumour necrosis factor receptor:Fc fusion protein, in patients with rheumatoid arthritis receiving methotrexate. N Engl J Med 340:253–259
Seymour GJ, Gemmell E (2001) Cytokines in periodontal disease: where to from here? Acta Odontol Scand 59:167–173
Yoshimura A, Hara Y, Kaneko T, Kato I (1997) Secretion of IL-1 beta, TNF-alpha, IL-8 and IL-1ra by human polymorphonuclear leukocytes in response to lipopolysaccharides from periodontopathic bacteria. J Periodontal Res 32:279–286
Kjeldsen M, Holmstrup P, Lindemann RA, Bendzen K (1995) Bacterial-stimulated cytokine production of peripheral mononuclear cells from patients of various periodontitis categories. J Periodontol 66:139–144
Rossano F, Rizzo A, Sanges MR, Cipollaro de L’Ero G, Tufano MA (1993) Human monocytes and gingival fibroblasts release tumor necrosis factor-alpha, interleukin-1 alpha and interleukin-6 in response to particulate and soluble fractions of Prevotella melaninogenica and Fusobacterium nucleatum. Int J Clin Lab Res 23:165–168
Arnett FC, Edworthy SM, Bloch DA, McShane DJ, Fries JF, Cooper NS et al (1988) The American rheumatism association 1987 revised criteria for the classification of rheumatoid arthritis. Arthritis Rheum 31:315–324
Pincus T, Summey JA, Soraci SA Jr, Wallston KA, Hummon NP (1983) Assessment of patient satisfaction in activities of daily using a modified Stanford health assessment questionnaire. Arthritis Rheum 26:1346–1353
Fries JF, Spitz PW, Young DY (1982) The dimensions of health outcomes: the health assessment questionnaire, disability and pain scale. J Rheumatol 9:789–793
Felson DT, Anderson JJ, Boers M, Bombardier C, Chernoff M, Fried B et al (1993) The American College of Rheumatology preliminary core set of disease activity measures for rheumatoid arthritis clinical trials. The committee on outcome measures in rheumatoid arthritis clinical trials. Arthritis Rheum 36:729–740
Felson DT, Anderson JJ, Boers M, Bombardier C, Furst D, Goldsmith C et al (1995) American collage of rheumatology. Preliminary definition of improvement in rheumatoid arthritis. Arthritis Rheum 38:727–735
Felson DT, Anderson JJ, Lange ML, Wells G, LaValley MP (1998) Should improvement in rheumatoid arthritis clinical trials be defined as fifty percent or seventy percent improvement in core set measures, rather than twenty percent? Arthritis Rheum 41:1564–1570
Jousimies-Somer HR, Finegold SM (1991). Anaerobic gram negative bacilli and cocci. In: Balows A, Hausler WJ, Herrmann KL, Isenberg HD, Shadomy HJ (eds) Manual of clinical microbiology. American Society for Microbiology, Washington DC, pp 538–553
Lorber B (2000) Bacteroides, Prevotella, Porphyromonas and Fusobacterium species (and other medically important anaerobic gram-negative bacilli). In Mandell GL, Bennett JE, Dolin R (eds) Mandell, Douglas and Bennett’s principles and practice of infectious Diseases. Churchill Livingstone, Philadelphia, pp 2561–2570
Marsh P, Martin MV (eds) (2001) Oral microbiology. MPG Books, Bodmin
Zhou Z, Menard HA (2002) Autoantigenic posttranslational modifications of proteins: does it apply to rheumatoid arthritis? Curr Opin Rheumatol 14:250–253
Tighe H, Carson DA (2001) Rheumatoid factor. In: Ruddy S, Harris ED Jr, Sledge CB (eds) Kelley’s textbook of rheumatology. W.B Saunders, Philadelphia, pp 151–160
Bonagura VR, Artandi SE, Davidson A, Randen I, Agostino N, Thompson K et al (1993) Mapping studies reveal unique epitopes on IgG recognized by rheumatoid arthritis- derived monoclonal rheumatoid factors. J Immunol. 151:3840–3852
Martin T, Crouzier R, Weber JC, Kipps TJ, Pasquali JL (1994) Structure-function studies on a polyreactive (natural) autoantibody. Polyreactivity is dependent on somatically generated sequences in the third complementarity-determining region of the antibody heavy chain. J Immunol. 152:5988–5996
The J, Ebersole JL (1991). Rheumatoid factor (RF) distribution in periodontal disease. J Clin Immunol 11:132–142
The J, Ebersole JL (1996). Rheumatoid factor from periodontitis patients cross-reacts with epitopes on oral bacteria. Oral Dis 2:253–262
Yoshida A, Nakano Y, Yamashita Y, Oho T, Ito H, Kondo M et al (2001) Immunodominant region of Actinobacillus actinomycetemcomitans 40-kilodalton heat shock protein in patients with rheumatoid arthritis. J Dent Res 80:346–350
Maeda H, Miyamoto M, Kokeguchi S, Kono T, Nishimura F, Takashiba S et al (2000) Epitope mapping of heat shock protein 60 (GroEL) from Porphyromonas gingivalis. FEMS Immunol Med Microbiol 28:219–224
Ando T, Kato T, Ishihara K, Ogiuchi H, Okuda K (1995) Heat shock proteins in the human periodontal disease process. Microbiol Immunol. 39:321–327
Ueki K, Tabeta K, Yoshie H, Yamazaki K (2002) Self-heat shock protein 60 induces tumour necrosis factor-alpha in monocyte-derived macrophage: possible role in chronic inflammatory periodontal disease. Clin Exp Immunol 127:72–77
Schett G, Redlich K, Xu Q, Bizan P, Groger M, Tohidast-Akrad M et al (1998) Enhanced expression of heat shock protein 70 (hsp70) and heat shock factor 1 (HSF1) activation in rheumatoid arthritis synovial tissue. Differential regulation of hsp70 expression and hsf1 activation in synovial fibroblasts by proinflammatory cytokines, shear stress, and antiinflammatory drugs. J Clin Invest 102:302–311
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ogrendik, M. Treatment of rheumatoid arthritis with ornidazole: a randomized, double-blind, placebo-controlled study. Rheumatol Int 26, 1132–1137 (2006). https://doi.org/10.1007/s00296-006-0145-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00296-006-0145-0