Summary
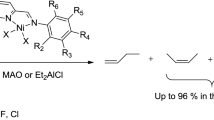
Palladium (II) compounds (palladium dichloride, acetate and acetylacetonate) in conjunction with triphenylphosphine dissolved in 1-butyl-methyl imidazolium tetrafluoroborate, hexafluorophosphate or trifluoromethanesulfonate are able to dimerize butadiene into 1,3,6-octatriene in a typical two-phase catalytic reaction with 100% selectivity. The reaction product is easily separated from the reaction mixture by simple decantation, and the recovered ionic liquid catalyst solution can be reused several times without significant changes in catalytic activity or selectivity.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Author information
Authors and Affiliations
Additional information
Received: 24 November 1997/Revised version: 5 January 1998/Accepted: 7 January 1998
Rights and permissions
About this article
Cite this article
Silva, S., Suarez, P., de Souza, R. et al. Selective linear dimerization of 1,3-butadiene by palladium compounds immobilized into 1-n-butyl-3-methyl imidazolium ionic liquids. Polymer Bulletin 40, 401–405 (1998). https://doi.org/10.1007/s002890050269
Issue Date:
DOI: https://doi.org/10.1007/s002890050269