Abstract.
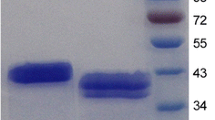
A β-mannanase gene (manA) was isolated from the extremely thermophilic bacterium Dictyoglomus thermophilum Rt46B.1. ManA is a single-domain enzyme related to one group of β-mannanases (glycosyl hydrolase family 26). The manA gene was expressed in the heat-inducible vector pJLA602 and the expression product, ManA, purified to homogeneity. The recombinant ManA is a monomeric enzyme with a molecular mass of 40 kDa and an optimal temperature and pH for activity of 80°C and 5.0. In the absence of substrate, the enzyme showed no loss of activity at 80°C over 16 h, while at 90°C the enzyme had a half-life of 5.4 min. Hydrolysis of the galactomannan locust bean gum (LBG) by purified ManA released mainly mannose, mannobiose, and mannotriose, confirming that ManA is an endo-acting β-mannanase. Sequence comparisons with related β-mannanases has allowed the design of consensus PCR primers for the identification and isolation of related genes.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Author information
Authors and Affiliations
Additional information
Received: 7 June 1999 / Accepted: 6 July 1999
Rights and permissions
About this article
Cite this article
Gibbs, M., Reeves, R., Sunna, A. et al. Sequencing and Expression of a β-Mannanase Gene from the Extreme Thermophile Dictyoglomus thermophilum Rt46B.1, and Characteristics of the Recombinant Enzyme. Curr Microbiol 39, 351–357 (1999). https://doi.org/10.1007/s002849900471
Issue Date:
DOI: https://doi.org/10.1007/s002849900471