Abstract
The etiopathogenesis of rheumatoid arthritis (RA) is partially understood. Genetic, environmental, and hormonal factors and their interactions are considered to play an important role on disease development. The relative contribution of environmental factors to RA development is probably larger than previously thought. The aim of this review is to appraise robust evidence about the role of environmental and hormonal risk factors for RA. We will discuss inhaled pollutants, nutritional habits, infectious, hormonal, and reproductive factors. As some of these factors are potentially modifiable, understanding their impact on RA development opens new opportunities for potential interventions and disease prevention.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Rheumatoid arthritis (RA) has become a model for autoimmune diseases, both to understand the development of autoimmunity and to study novel therapeutic interventions [1]. RA is the most prevalent systemic autoimmune inflammatory disease affecting between 0.5 and 1% of the adult population worldwide. The etiopathogenesis of RA is only partially understood but is believed to result from a multi-step process, whereby environmental factors induce specific post-translational modifications of proteins, which in turn trigger a pathologic activation of the immune system in genetically susceptible individuals, ultimately leading to the clinical onset of the disease. Specific pre-clinical phases of RA development have been proposed (Fig. 1) [2, 3].
Phases of RA development. UA undifferentiated arthritis, RA rheumatoid arthritis. Reproduction with authorisation from [3]
A genetic contribution to the development of RA has long been suspected because of the increased risk in first-degree relatives of patients with RA [4, 5]. A variety of susceptibility genes have been identified, notably different alleles of the HLA-DRB1 gene that share a common sequence in the third hypervariable region, referred to as the “shared epitope.” However, the overall risk associated with established genetic markers remains modest and the relatively low penetrance of the disease suggests that environmental factors play an important role in the etiology of RA [6]. Nevertheless, individual environmental factors, such as smoking, have generally demonstrated only weak associations with RA. Over the last years, striking gene-environment interactions have been discovered, which have changed our concepts of RA etiopathogenesis. The importance of gene-environment interactions [7, 8] is growing, because it opens opportunities for potential interventions and disease prevention.
In the following pages, we will review specific environmental risk factors for RA, without attempting to be comprehensive. Many studies regarding environmental and hormonal factors show controversial results. Some of these conflicting outcomes may be related to heterogeneity in the definition of the exposure, in the study designs and in the study populations. We will consider environmental factors broadly, as potentially modifiable exposures, in contrast to genetic factors. We will discuss inhaled pollutants, nutritional habits, chronic infections, hormonal and reproductive factors, focusing on the most robust findings or on results that have been replicated. We will underscore gene-environment interactions or environment-environment interactions, when these have been established. Other environmental factors, such as acute infections, geographic or ethnic differences, socio-economic status, stress or specific industrial pollutants have also been associated with the development of the disease [9–13]; however, these findings still need confirmation.
Inhaled pollutants
Tobacco smoke has long been known to moderately increase the risk of developing RA. A meta-analysis of 16 studies estimated that ever smoking increases the risk of developing RA by 40% (odds ratio (OR), 1.40; 95% confidence interval (CI), 1.25–1.58) [14]. Smoking primarily increases the risk of rheumatoid factor (RF) or anti-citrullinated protein antibody (ACPA)-positive (seropositive) RA and only in the presence of specific genetic risk factors (the “shared epitope” or some PTPN22 alleles) [8]. For instance, the combination of a smoking history and two copies of the shared epitope increased the risk of seropositive RA by 21-fold (95% CI, 11–40) [2]. The precise etiopathogenic mechanism by which tobacco induces the development of RA is only partially understood. Tobacco increases the risk of RA only when inhaled but not when chewed (OR, 1.0; 95% CI, 0.8–1.2), suggesting that nicotine is not involved in the etiopathogenesis [15]. Klareskog et al.’s seminal work first demonstrated that smoking induces protein citrullination in the lungs [2]. In the SERA cohort, investigators were able to demonstrate inflammatory airway abnormalities in healthy ACPA-positive subjects, before the development of clinically apparent RA, even in nonsmokers, suggesting that the initial inflammation and immune onset of the disease begins in the lungs [16]. The major histocompatibility complex carrying the shared epitope allele has an increased affinity for citrullinated proteins [17], which provides a rationale of how citrullinated proteins could lead to a loss of tolerance and a pathologic activation of the immune system. Other genetic interactions may modify the association of smoking with RA, in particular enzymes involved in tobacco smoke detoxification. For instance, some studies have suggested an increased risk of RA with smoking in individuals with lower copy numbers of the GSTM1 gene [18, 19] or NAT2 polymorphisms [20].
Likewise, other inhaled pollutants have shown associations with RA development, particularly in nonsmokers. In the Nurses’ Health Study, women living within 50 m from the highway, a proxy for exposure to traffic pollution, had a moderately increased risk of RA (hazard ratio (HR), 1.31; 95% CI, 0.98–1.74), particularly in nonsmokers (HR = 1.62; 95% CI, 1.04–2.52) [10]. These results were replicated in British Columbia, Canada with very similar results [21]. A longitudinal, population-based study in Taiwan found an increasing risk of RA with higher nitrogen dioxide (NO2) levels, as well as a clear dose response [22]. While there are still inconsistencies in epidemiological studies on air pollution and RA development [23], the overall pattern suggests that air pollution is an environmental risk factor for RA [24].
Several occupational exposures to inhaled pollutants have also been suggested as risk factors for RA development. Over half a century ago, physicians first noticed a striking association between silicosis and RA in miners, entitled Caplan’s syndrome [25, 26]. Since then, the association between silica dust and RA has been confirmed in the mining industry [27], and also in occupations outside of mining, such as construction workers or the pottery industry [28, 29]. Interestingly, several studies have found an interaction between silica dust and smoking [30, 31], with a modest elevation of the risk of ACPA-positive RA in nonsmokers (OR, 1.15; 95% CI, 0.42–3.15) and a strong elevation of the risk in current smokers (OR, 7.36; 95% CI, 3.31–16.38) [30]. How inhaled silica dust induces autoimmunity is poorly understood; however, it has been shown that silica particles can activate the innate immune system, promote pro-inflammatory cytokine production and function as an adjuvant for antibody production [32]. Other professional inhaled exposures have been found to increase the risk of RA. In a Malaysian case-control study, occupational exposure to textile dusts was associated with an increased risk of developing RA in women (OR, 2.8; 95% CI, 1.6–5.2), and a strong gene-environment interaction with the shared epitope in ACPA-positive RA patients (OR, 39.1; 95% CI, 5.1–297.5) [33]. Recent studies have also suggested that certain agricultural pesticides could increase the risk of RA in farmers and their families [34, 35].
Nutritional habits
Patients often attribute the occurrence of an autoimmune disease such as RA to nutritional habits. We will focus on a few nutritional factors that have been convincingly associated with the disease, whether protective or hazardous.
In animal models, it has been known for over 10 years that adding small doses of ethanol to mice’s drinking water defers the onset of collagen-induced arthritis and prevents the development of erosions, suggesting preventive properties of low-dose and persistent alcohol consumption [36]. In humans, a meta-analysis of nine observational studies found a significant protective effect of alcohol on the development of RA with an OR of 0.78 (95% CI, 0.63–0.96), which was more pronounced in ACPA-positive RA (OR, 0.52; 95% CI, 0.36–0.76)) [37]. Consistent with these findings, cohort studies in established RA have confirmed that moderate consumption of alcohol decreases the rate of erosive joint damage progression [38].
Another well-known “cardio-protective food,” omega-3 fatty acids, have been suggested to be protective against the development of autoimmunity associated with RA. In a first-degree cohort of RA relatives, healthy individuals who developed ACPAs were less likely to use omega-3 supplements (OR, 0.14; 95% CI, 0.03–0.68) and had significantly lower concentrations of omega-3 fatty acids in red blood cell membranes [39]. In a large, longitudinal cohort from Sweden, long-lasting intake of omega-3 fatty acids higher than 0.21 g/day decreased the development of subsequent RA by 52% (95% CI: 29% - 67%) [40], as well as a regular consumption of fish at least once per week (risk ratio (RR), 0.71; 95% CI, 0.48–1.04). A meta-analysis examining the association between fish consumption and subsequent development of RA suggested a trend towards a protective effect with one to three portions of fish per week (RR, 0.76; 95% CI, 0.57–1.02) [41]. A randomized controlled trial in early RA, demonstrated that adding fish oil supplements to a standard therapy with conventional anti-rheumatic drugs increased significantly the chance of clinical remission (HR, 2.09; 95% CI, 1.02–4.30), supporting the hypothesis of anti-inflammatory properties of omega-3 fatty acids [42].
High salt consumption has also been suggested as a risk factor for the development of RA, in particular in smokers [43]. In a prospective cohort from northern Sweden, high sodium intake more than doubled the risk of RA among smokers (OR, 2.26; 95% CI, 1.06–4.81) but not in nonsmokers. Furthermore, this interaction between smoking and high sodium intake was stronger for ACPA-positive RA. In the Nurses’ Health Study, regular consumption of sugar-sweetened sodas significantly increased the risk of developing RA. The association was independent of obesity and other socio-economic factors and tended to be stronger for late-onset RA (HR, 2.64; 95% CI, 1.56–4.46). Interestingly, no causal relation was found with diet soda or between sugar-sweetened soda and seronegative RA [44]. Furthermore, akin to high sodium intake, the authors describe an interaction between sugar-sweetened soda consumption and smoking, with a higher risk of developing RA in women who regularly drank sugar-sweetened sodas and had smoked more than 10 pack-years.
Obesity is increasing all over the world, with well-established consequences on a number of chronic diseases. The role of obesity as a risk factor for RA is still debated, but a majority of studies suggest it is a risk factor for RA in women [45]. Obese women (BMI ≥30.0 kg/m2) in the Nurses’ Health Study tended to have an increased risk of RA, in particular in those diagnosed at earlier ages (HR, 1.65; 95% CI, 1.34–2.05) and with adolescent obesity (HR, 1.35; 95% CI, 1.10–1.66) [46]. Similar results were found also in Europe, with obesity increasing the risk for seronegative RA in Swedish women (HR, 1.6; 95% CI, 1.2–2.2) [47]. In men, the effect of obesity is less obvious and some studies have even described a reduced risk of RA in men [48].
Chronic infections
Several infectious agents have been proposed as risk factors for RA; however, findings have not been conclusive. Some viral infections, such as parvovirus B19, chikungunya, and hepatitis C, are known to cause acute and chronic arthralgias and persistent arthritis; however, the association with the development of classifiable rheumatoid arthritis is not solid [49–51]. In a large population-based cohort, chronic hepatitis C was associated with an increased risk of RA, hazard ratio (HR, 2.03; 95% CI, 1.27–3.22) [52]. However, the diagnosis of RA in chronic hepatitis C is difficult to establish because of false-positive auto-antibodies, such as rheumatoid factor and ACPA [53].
Lyme disease caused by the bacterium Borrelia burgdorferi is associated as well with chronic arthritis. RA cases diagnosed after Lyme disease have been reported [54, 55]. However, the distinction between active Lyme infection in joints, post-infectious Lyme arthritis, and another form of inflammatory arthritis, such a RA can be challenging [55].
A population-based case-control study (N = 6401) examined the association of recent infections with the risk of RA and found that gastrointestinal and urogenital infections were associated with a lowered risk of RA, OR of 0.7 (95% CI, 0.6–0.8) and OR of 0.8 (95% CI, 0.7–0.9), respectively. This finding was hypothesized to be related to antibiotic treatment; however, no information about antibiotic use was specified [56].
The most prevalent chronic infection consistently associated with RA is periodontitis. RA patients have a higher prevalence of chronic periodontitis and tooth loss compared with patients without RA [57, 58]. A longitudinal population study based on the First National Health and Examination Survey (NHANES I) suggested that subjects with periodontal disease and missing teeth (p for interaction = 0.05) had an increased risk of developing RA. In severe periodontal disease associated with an edentulous state, the risk of incident RA was almost doubled (OR, 1.92; 95% CI, 1.0–3.7) [59]. Porphyromonas gingivalis, a common periodontal pathogen associated with chronic periodontitis, is known to express an endogenous peptidyl-arginine deiminase (PAD) that is able to citrullinate different human proteins [60]. Citrullinated proteins are present in serum, synovial, and periodontal tissues of RA patients. DNA of P. gingivalis has been found in the synovial tissue [61, 62]. Factors that contribute to the loss of tolerance to the citrullinated proteins are still unclear. Studies have not found an association between the presence of antibodies to periodontal bacteria and RF seropositivity [63]. An additive interaction between ACPA positivity, the shared epitope and antibodies against the P. gingivalis virulence factor, arginine gingipain-type B (RgpB) has been reported (OR, 5.7; 95% CI, 4.2–7.6) [64]; however, other studies have not found an association between anti-RgpB and later development of RA [65]. A recent study reported an association with another periodontitis related bacteria, Agregatibacter actinomycetemcomitans, which induces citrullination and dysregulates activation of citrullinating enzymes in neutrophils. In addition, A. actinomycetemcomitans was associated with ACPA positivity (p = 0.01) in a sample of 196 patients with RA [66]. Nevertheless, in spite of the biological arguments for the role of periodontitis in RA development, the causal relationship is still not clearly established. The association between periodontitis and RA could be confounded by behavioral and environmental exposures common to both conditions [67]. Furthermore, the entity of periodontitis has often been poorly defined in scientific investigations [57, 59].
Microbiota comprises the diverse population of bacteria that live in the human body. Changes in nutrition, immune competence, incidence of disease, and use of medications, such as antibiotics, could alter the composition of the microbiota, generating an imbalance between beneficial and harmful species. A dysbiotic microbiota has been observed in many conditions, including obesity, diabetes, and inflammatory bowel diseases [68, 69]. In RA patients, oral and gut dysbiotic microbiota has been reported as well [70, 71]. Particularly, increased occurrence of Prevotella species have been identified in patients newly diagnosed with RA [72, 73]. Dysbiotic microbiota in RA patients is partially resolved after disease-modifying anti-rrheumatic drug (DMARD) treatment. In addition, fecal, dental, and salivary samples from RA patients who show improvement with treatment contain a greater number of virulence factors than those without improvement [71]. As the reports of dysbiotic microbiota are based on association studies, causality is difficult to establish with the information available so far. Interestingly, in DR4-transgenic mice, a sex-biased mouse model of collagen-induced arthritis, the fecal microbiota differed in males and females. In females, the microbiota was dysbiotic and less dynamic, and males showed significantly higher abundance of Bacteroides [74]. These findings suggest that gut microbiota could contribute to the sexual dimorphism of RA.
Hormonal and reproductive factors
RA is more prevalent in women. The female-to-male sex ratio of the disease varies between 4:1 in patients younger than 50 years to less than 2:1 in the older population [75, 76]. The reason for the increased prevalence in females is still unclear. An increased risk of developing RA has not been reported in X-chromosome dosage disorders, such as Klinefelter syndrome, characterized by an extra chromosome [77, 78]. In addition, the familial aggregation of RA is not preponderant in women, suggesting that the familial risk is not related to chromosome X [79].
There are many controversies regarding the role of hormonal factors in the development of RA. For example, both factors related to high and to low estrogen exposure have been associated with increased risk of RA. In a simplistic manner, it has been proposed that estrogens have a pro-inflammatory and androgens an anti-inflammatory effect. However, estrogens can have both stimulatory and inhibitory effects on the immune system [80]. Overall, a host of studies suggest that factors related with the decline of estrogens are risk factors and those related with high exposure to estrogens are rather protective factors.
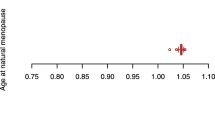
Two examples of factors characterized by a decline of estrogens are menopause and the use of anti-estrogen agents. Post-menopause has been associated with an increased risk of developing seronegative RA in a large cohort study (HR, 2.1; 95% CI, 1.5–3.1), and particularly early age at menopause defined as <44 years old (HR, 2.4; 95% CI, 1.6–3.5) [81]. Other studies have confirmed this finding with even earlier ages at menopause [82, 83].
An interesting study analyzed the incidence of RA in users of anti-estrogen agents in a large US national database for breast cancer. The use of anti-estrogen agents, both selective estrogen receptors modulators and aromatase inhibitors, was associated with RA, with a dose- and duration-dependent effect (for >12 months, OR, 2.4 (95% CI, 1.9–3.0) and OR, 1.9 (95% CI, 1.6–2.1), respectively) [84].
Examples of high estrogen exposure are the use of hormonal treatment, such as oral contraceptives (OCs) and hormonal replacement therapy (HRT). The effect of OCs use on RA is controversial [85, 86]. Meta-analyses investigating the association between OCs and development of RA concluded a nonsignificant association. However, the studies were heterogeneous, in particular the dose and the duration of OC exposure varied between studies [87–89]. In several analyses of the duration of treatment, longer use of OCs was protective [90]. OC use for ≥8 years was associated with a decreased risk of ACPA-positive RA (OR, 0.8; 95% CI, 0.7–0.9) and ACPA-negative RA (OR, 0.9; 95% CI, 0.7–1.0) [91]. The fact that older studies tended to show a stronger protective effect for RA suggests a potential dose effect, due to increased estrogen dose in OCs in the past [88]. Overall, the available evidence supports a protective effect of OCs against RA, in particular when used for long periods or at high dose. Regarding hormonal replacement therapy (HRT) a protective association of combined HRT use on ACPA-positive RA has been described in a population-based case-control study (OR, 0.3; 95% CI, 0.1–0.7) but not for estrogen-only HRT (OR, 0.8; 95% CI, 0.5–1.6) [92].
Multiple hormonal changes
Some conditions are characterized by multiple hormonal changes, such as pregnancy and breastfeeding. Pregnancy is associated with high levels of estrogens, but their effect is modified by other hormonal changes, such as high progesterone [80]. In prospective cohorts, pregnancy has been described as protective against developing RA [93, 94]. In a prospective case-control study in women with recently diagnosed RA, parity was significantly associated with a lower risk of RA (RR, 0.6; 95% CI, 0.4–0.8) [95]. Breastfeeding has been consistently associated with decreased risk of RA [13, 96]. In a systematic review, breastfeeding for less than 12 months was protective against RA (OR, 0.8; 95% CI, 0.6–0.9) and a longer breastfeeding period had a stronger protective association (OR, 0.6; 95% CI, 0.4–0.7) [97]. On the contrary, the post-partum period, characterized by a decline of estrogens and other hormonal changes, has been consistently associated with an increased risk of RA [94, 98], particularly during the first year post-partum (OR, 3.4; 95% CI,1.5–9.9) [93].
Androgens
Men with RA have decreased androgen levels, while the androgen levels in women are not different compared to controls [99–101]. Lower androgen to estrogen ratios have been detected in both female and male RA patients [102]. In addition, increased estrogen formation has been described in the synovial tissue of patients with RA [103–105]. In a retrospective cohort study using US administrative health data, 123,460 males aged >19 years with untreated hypogonadism had an increased risk of developing RA (HR = 1.31; 95% CI, 1.3–1.5) compared with men without hypogonadism [106]. In contrast, in women from the Nurses’ Health Study, no significant association was found between total or free testosterone (measured at a single time point prior to RA onset) and risk of RA development [107]. Androgens have been less studied in pre-clinical phases of RA. In a small case-control study, serum androstenedione levels were significantly lower in women before RA diagnosis compared with controls [108].
Nonhormonal reproductive factors
Pregnancies complicated by pre-eclampsia have been associated with an increased risk of RA (RR, 1.4; 95% CI, 1.1–1.8) [109]. The same research group confirmed pre-eclampsia as a risk factor for RA in a larger group of women (HR, 1.9; 95% CI, 1.1–3.6) [110]. One hypothesis for the increased risk of RA during pre-eclampsia as well as during the post-partum period is fetal microchimerism, which is the acquisition of fetal cells and/or DNA persisting in the maternal circulation. It is believed that persisting microchimeric cells carrying one of the SE alleles could confer to the mother additional susceptibility to RA [111]. The rationale is supported by a higher frequency SE microchimerism in RA cases (DRB1*01 microchimerism, 30 vs. 4% (p = 0.001) and DRB1*04 microchimerism, 42 vs. 8% (p < 0.001)) [112, 113].
Conclusions
The understanding of environmental factors and their interactions with other patient-related factors on the development of RA has increased with larger observational studies. The relative contribution of environmental factors to RA development is probably larger the previously thought [6, 114]. As some of the environmental and hormonal factors are potentially modifiable, understanding their impact on RA development opens new opportunities for potential interventions and disease prevention.
References
Furer V, Greenberg JD, Attur M, Abramson SB, Pillinger MH (2010) The role of microRNA in rheumatoid arthritis and other autoimmune diseases. Clin Immunol 136(1):1–15
Klareskog L, Stolt P, Lundberg K et al (2006) A new model for an etiology of rheumatoid arthritis: smoking may trigger HLA-DR (shared epitope)-restricted immune reactions to autoantigens modified by citrullination. Arthritis Rheum 54(1):38–46
Mankia K, Emery P (2016) Preclinical rheumatoid arthritis: progress toward prevention. Arthritis Rheumatol. 68(4):779–788
Silman AJ, Hennessy E, Ollier B (1992) Incidence of rheumatoid arthritis in a genetically predisposed population. Br J Rheumatol 31(6):365–368
Frisell T, Holmqvist M, Kallberg H et al (2013) Familial risks and heritability of rheumatoid arthritis: role of rheumatoid factor/anti-citrullinated protein antibody status, number and type of affected relatives, sex, and age. Arthritis Rheum 65(11):2773–2782
Sparks JA, Chen CY, Hiraki LT et al (2014) Contributions of familial rheumatoid arthritis or lupus and environmental factors to risk of rheumatoid arthritis in women: a prospective cohort study. Arthritis Care Res (Hoboken). 66(10):1438–1446
Karlson EW, Watts J, Signorovitch J et al (2007) Effect of glutathione S-transferase polymorphisms and proximity to hazardous waste sites on time to systemic lupus erythematosus diagnosis: results from the Roxbury lupus project. Arthritis Rheum 56(1):244–254
Kallberg H, Padyukov L, Plenge RM et al (2007) Gene-gene and gene-environment interactions involving HLA-DRB1, PTPN22, and smoking in two subsets of rheumatoid arthritis. Am J Hum Genet 80(5):867–875
Costenbader KH, Karlson EW (2006) Epstein-Barr virus and rheumatoid arthritis: is there a link? Arthritis Res Ther. 8(1):204
Hart JE, Laden F, Puett RC, Costenbader KH, Karlson EW (2009) Exposure to traffic pollution and increased risk of rheumatoid arthritis. Environ Health Perspect 117(7):1065–1069
Lu B, Solomon DH, Costenbader KH et al (2010) Alcohol consumption and markers of inflammation in women with preclinical rheumatoid arthritis. Arthritis Rheum 62(12):3554–3559
Kallberg H, Jacobsen S, Bengtsson C et al (2009) Alcohol consumption is associated with decreased risk of rheumatoid arthritis: results from two Scandinavian case-control studies. Ann Rheum Dis 68(2):222–227
Karlson EW, Mandl LA, Hankinson SE, Grodstein F (2004) Do breast-feeding and other reproductive factors influence future risk of rheumatoid arthritis? Results from the Nurses’ Health Study. Arthritis Rheum 50(11):3458–3467
Sugiyama D, Nishimura K, Tamaki K et al (2010) Impact of smoking as a risk factor for developing rheumatoid arthritis: a meta-analysis of observational studies. Ann Rheum Dis 69(1):70–81
Jiang X, Alfredsson L, Klareskog L, Bengtsson C (2014) Smokeless tobacco (moist snuff) use and the risk of developing rheumatoid arthritis: results from a case-control study. Arthritis Care Res (Hoboken) 66(10):1582–1586
Demoruelle MK, Weisman M, Simonian P (2011) Airways abnormalities and rheumatoid arthritis-related autoantibodies in subjects without arthritis: early injury or initiating site of autoimmunity? Arthritis Rheum 64:1756–1761
Hill JA, Bell DA, Brintnell W et al (2008) Arthritis induced by posttranslationally modified (citrullinated) fibrinogen in DR4-IE transgenic mice. J Exp Med 205(4):967–979
Lundström E, Hartshorne T, Li K et al (2011) Effects of GSTM1 in rheumatoid arthritis; results from the Swedish EIRA study. PLoS One 6(3):e17880
Criswell LA, Saag KG, Mikuls TR, et al. Smoking interacts with genetic risk factors in the development of rheumatoid arthritis among older Caucasian women. Ann Rheum Dis. 2006
Mikuls T, LeVan T, Gould K et al (2012) Impact of interactions of cigarette smoking with NAT2 polymorphisms on rheumatoid arthritis risk in African Americans. Arthr Rheum 64(3):655–664
De Roos A, Koehoorn M, Tamburic L, Davies H, Brauer M (2014) Proximity to traffic, ambient air pollution, and community noise in relation to incident rheumatoid arthritis. Environ Health Perspect 122(10):1075–1080
Chang K, Hsu C, Muo C et al (2016) Air pollution exposure increases the risk of rheumatoid arthritis: a longitudinal and nationwide study. Environ Int 94:495–499
Hart J, Källberg H, Laden F et al (2013) Ambient air pollution exposures and risk of rheumatoid arthritis: results from the Swedish EIRA case-control study. Ann Rheum Dis 72(6):888–894
Farhata SCL, Silva CA, Orione M et al (2011) Air pollution in autoimmune rheumatic diseases: a review. Autoimmun Rev 11(1):14–21
Caplan A (1953) Certain unusual radiological appearances in the chest of coal-miners suffering from rheumatoid arthritis. Thorax 8:29–35
Sluis-Cremer G, Hessel P, Hnizdo E, Churchill A (1986) Relationship between silicosis and rheumatoid arthritis. Thorax 41:596–601
Klockars M, Koskela R, Jarvinen E, Kolari P, Rossi A (1987) Silica exposure and rheumatoid arthritis: a follow up study of granite workers 1940–81. Br Med J (Clin Res Ed) 294:997–1000
Blanc P, Järvholm B, Toren K (2015) Prospective risk of rheumatologic disease associated with occupational exposure in a cohort of male construction workers. Am J Med 128(10):1094–1101
Turner S, Cherry N (2000) Rheumatoid arthritis in workers exposed to silica in the pottery industry. Occup Environ Med 57:443–447
Stolt P, Yahya A, Bengtsson C et al (2010) Silica exposure among male current smokers is associated with a high risk of developing ACPA-positive rheumatoid arthritis. Ann Rheum Dis 69(6):1072–1076
Yahya A, Bengtsson C, Larsson P, et al (2014) Silica exposure is associated with an increased risk of developing ACPA-positive rheumatoid arthritis in an Asian population: evidence from the Malaysian MyEIRA case-control study. Mod Rheumatol.24(2)
Pollard K (2016) Silica, silicosis, and autoimmunity. Front Immunol 7:97
Too C, Muhamad N, Lar A et al (2016) Occupational exposure to textile dust increases the risk of rheumatoid arthritis: results from a Malaysian population-based case-control study. Ann Rheum Dis 75(6):997–1002
Meyer A, Sandler D, Beane-Freeman L, Hoffman J, Parks C, editors (2016) Pesticide exposure and risk of rheumatoid arthritis in licensed male pesticide applicators in the Agricultural Health Study [abstract]. ACR annual scientific conference. Washington, DC: Arthritis Rheumatol
Parks C, Hoppin J, De Roos A et al (2016) Rheumatoid arthritis in agricultural health study spouses: associations with pesticides and other farm exposures. Environ Health Perspect 124:1728–1734
Jonsson IM, Verdrengh M, Brisslert M et al (2007) Ethanol prevents development of destructive arthritis. Proc Natl Acad Sci U S A 104(1):258–263
Scott I, Tan R, Stahl D et al (2013) The protective effect of alcohol on developing rheumatoid arthritis: a systematic review and meta-analysis. Rheumatology (Oxford) 52(5):856–867
Nissen MJ, Gabay C, Scherer A, Finckh A (2010) The effect of alcohol on radiographic progression in rheumatoid arthritis. Arthritis Rheum 62(5):1265–1272
Gan R, Young K, Zerbe G et al (2016) Lower omega-3 fatty acids are associated with the presence of anti-cyclic citrullinated peptide autoantibodies in a population at risk for future rheumatoid arthritis: a nested case-control study. Rheumatology (Oxford) 55(2):367–376
Di Giuseppe D, Wallin A, Bottai M, Askling J, Wolk A (2014) Long-term intake of dietary long-chain n-3 polyunsaturated fatty acids and risk of rheumatoid arthritis: a prospective cohort study of women. Ann Rheum Dis 73(11):1949–1953
Di Giuseppe D, Crippa A, Orsini N, Wolk A (2014) Fish consumption and risk of rheumatoid arthritis: a dose-response meta-analysis. Arthritis Res Ther. 16(5):446
Proudman SM, James MJ, Spargo LD, Metcalf RG, Sullivan TR, Rischmueller M, Flabouris K, Wechalekar MD, Lee AT, Cleland LG (2015) Fish oil in recent onset rheumatoid arthritis: a randomised, double-blind controlled trial within algorithm-based drug use. Ann Rheum Dis 74(1):89–95
Sundström B, Johansson I, Rantapää DS (2015) Interaction between dietary sodium and smoking increases the risk for rheumatoid arthritis: results from a nested case-control study. Rheumatology (Oxford) 54(3):487–493
Hu Y, Costenbader KH, Gao X et al (2014) Sugar-sweetened soda consumption and risk of developing rheumatoid arthritis in women. Am J Clin Nutr 100(3):959–967
Finckh A, Turesson C (2014) Impact of obesity in rheumatoid arthritis. Ann Rheum Dis. (in press)
Lu B, Hiraki L, Sparks JA, et al (2014) Being overweight or obese and risk of developing rheumatoid arthritis among women: a prospective cohort study. Ann Rheum Dis
Wesley A, Bengtsson C, Elkan AC et al (2013) Association between body mass index and anti-citrullinated protein antibody-positive and anti-citrullinated protein antibody-negative rheumatoid arthritis: results from a population-based case-control study. Arthritis Care Res (Hoboken) 65(1):107–112
Turesson C, Bergström U, Pikwer M, Nilsson J, Jacobsson L (2016) A high body mass index is associated with reduced risk of rheumatoid arthritis in men, but not in women. Rheumatology (Oxford) 55(2):307–314
Caliskan R, Masatlioglu S, Aslan M et al (2005) The relationship between arthritis and human parvovirus B19 infection. Rheumatol Int 26(1):7–11
Sepulveda-Delgado J, Vera-Lastra OL, Trujillo-Murillo K et al (2016) Inflammatory biomarkers, disease activity index, and self-reported disability may be predictors of chronic arthritis after chikungunya infection: brief report. Clin Rheumatol. doi:10.1007/s10067-016-3419-2
Bouquillard E, Combe B (2009) Rheumatoid arthritis after chikungunya fever: a prospective follow-up study of 21 cases. Ann Rheum Dis 68(9):1505–1506
Su FH, Wu CS, Sung FC et al (2014) Chronic hepatitis C virus infection is associated with the development of rheumatoid arthritis: a nationwide population-based study in Taiwan. PLoS One 9(11):e113579
Liu FC, Chao YC, Hou TY et al (2008) Usefulness of anti-CCP antibodies in patients with hepatitis C virus infection with or without arthritis, rheumatoid factor, or cryoglobulinemia. Clin Rheumatol 27(4):463–467
Hsieh YF, Liu HW, Hsu TC et al (2007) Serum reactivity against Borrelia burgdorferi OspA in patients with rheumatoid arthritis. Clin Vaccine Immunol 14(11):1437–1441
Arvikar SL, Crowley JT, Sulka KB, Steere AC (2017) Autoimmune arthritides, rheumatoid arthritis, psoriatic arthritis, or peripheral spondyloarthritis following Lyme disease. Arthritis Rheumatol. 69(1):194–202
Sandberg ME, Bengtsson C, Klareskog L, Alfredsson L, Saevarsdottir S (2015) Recent infections are associated with decreased risk of rheumatoid arthritis: a population-based case-control study. Ann Rheum Dis 74(5):904–907
de Pablo P, Dietrich T, McAlindon TE (2008) Association of periodontal disease and tooth loss with rheumatoid arthritis in the US population. J Rheumatol 35(1):70–76
de Pablo P, Chapple ILC, Buckley CD, Dietrich T (2009) Periodontitis in the systemic rheumatic diseases. Nat Rev Rheumatol 5:218–224
Demmer RT, Molitor JA, Jacobs DR Jr, Michalowicz BS (2011) Periodontal disease, tooth loss and incident rheumatoid arthritis: results from the First National Health and Nutrition Examination Survey and its epidemiological follow-up study. J Clin Periodontol 38(11):998–1006
McGraw WT, Potempa J, Farley D, Travis J (1999) Purification, characterization, and sequence analysis of a potential virulence factor from Porphyromonas gingivalis, peptidylarginine deiminase. Infect Immun 67(7):3248–3256
Janssen KM, Vissink A, de Smit MJ, Westra J, Brouwer E (2013) Lessons to be learned from periodontitis. Curr Opin Rheumatol 25(2):241–247
Nesse W, Westra J, van der Wal JE et al (2012) The periodontium of periodontitis patients contains citrullinated proteins which may play a role in ACPA (anti-citrullinated protein antibody) formation. J Clin Periodontol 39(7):599–607
Goh CE, Kopp J, Papapanou PN, Molitor JA, Demmer RT (2016) Association between serum antibodies to periodontal bacteria and rheumatoid factor in the Third National Health and Nutrition Examination Survey. Arthritis Rheumatol. 68(10):2384–2393
Kharlamova N, Jiang X, Sherina N et al (2016) Antibodies to Porphyromonas gingivalis indicate interaction between oral infection, smoking, and risk genes in rheumatoid arthritis etiology. Arthritis Rheumatol 68(3):604–613
Fisher BA, Cartwright AJ, Quirke AM et al (2015) Smoking, Porphyromonas gingivalis and the immune response to citrullinated autoantigens before the clinical onset of rheumatoid arthritis in a Southern European nested case-control study. BMC Musculoskelet Disord 16:331
Konig MF, Abusleme L, Reinholdt J et al (2016) Aggregatibacter actinomycetemcomitans-induced hypercitrullination links periodontal infection to autoimmunity in rheumatoid arthritis. Sci Transl Med 8(369):369ra176
da Silva AP, Bissada NF (2016) Arthritis and periodontitis: an association debated for over two centuries. Curr Rheumatol Rev 12(3):202–207
McLean MH, Dieguez D Jr, Miller LM, Young HA (2015) Does the microbiota play a role in the pathogenesis of autoimmune diseases? Gut 64(2):332–341
Erickson AR, Cantarel BL, Lamendella R et al (2012) Integrated metagenomics/metaproteomics reveals human host-microbiota signatures of Crohn’s disease. PLoS One 7(11):e49138
Vaahtovuo J, Munukka E, Korkeamaki M, Luukkainen R, Toivanen P (2008) Fecal microbiota in early rheumatoid arthritis. J Rheumatol 35(8):1500–1505
Zhang X, Zhang D, Jia H et al (2015) The oral and gut microbiomes are perturbed in rheumatoid arthritis and partly normalized after treatment. Nat Med 21(8):895–905
Scher JU, Sczesnak A, Longman RS et al (2013) Expansion of intestinal Prevotella copri correlates with enhanced susceptibility to arthritis. Elife 2:e01202
Pianta A, Arvikar S, Strle K et al (2016) Evidence for immune relevance of Prevotella copri, a gut microbe, in patients with rheumatoid arthritis. Arthritis Rheumatol. doi:10.1002/art.40003
Gomez A, Luckey D, Yeoman CJ et al (2012) Loss of sex and age driven differences in the gut microbiome characterize arthritis-susceptible 0401 mice but not arthritis-resistant 0402 mice. PLoS One 7(4):e36095
de Hair MJ, Lehmann KA, van de Sande MG et al (2012) The clinical picture of rheumatoid arthritis according to the 2010 American College of Rheumatology/European League Against Rheumatism criteria: is this still the same disease? Arthritis Rheum 64(2):389–393
Kvien TK, Uhlig T, Odegard S, Heiberg MS (2006) Epidemiological aspects of rheumatoid arthritis: the sex ratio. Ann N Y Acad Sci 1069:212–222
Kovacs WJ, Olsen NJ (2011) Sexual dimorphism of RA manifestations: genes, hormones and behavior. Nat Rev Rheumatol 7(5):307–310
Hughes GC, Choubey D (2014) Modulation of autoimmune rheumatic diseases by oestrogen and progesterone. Nat Rev Rheumatol 10(12):740–751
Frisell T, Saevarsdottir S, Askling J (2016) Family history of rheumatoid arthritis: an old concept with new developments. Nat Rev Rheumatol 12(6):335–343
Straub RH (2007) The complex role of estrogens in inflammation. Endocr Rev 28(5):521–574
Bengtsson CMS, Sparks JA, Costenbader KH, Karlson EW (2014) Post-menopausal factors and the risk of seropositive and seronegative rheumatoid arthritis phenotypes: results from the Nurses’ Health Study. Arthritis and Rheumatology 66(11 (supplement)):S1261
Beydoun HA, el-Amin R, McNeal M, Perry C, Archer DF (2013) Reproductive history and postmenopausal rheumatoid arthritis among women 60 years or older: Third National Health and Nutrition Examination Survey. Menopause 20(9):930–935
Pikwer M, Bergstrom U, Nilsson JA, Jacobsson L, Turesson C (2012) Early menopause is an independent predictor of rheumatoid arthritis. Ann Rheum Dis 71(3):378–381
Chen JY, Ballou SP (2015) The effect of antiestrogen agents on risk of autoimmune disorders in patients with breast cancer. J Rheumatol 42(1):55–59
Pedersen M, Jacobsen S, Klarlund M et al (2006) Environmental risk factors differ between rheumatoid arthritis with and without auto-antibodies against cyclic citrullinated peptides. Arthritis Res Ther. 8(4):R133
Pedersen M, Jacobsen S, Garred P et al (2007) Strong combined gene-environment effects in anti-cyclic citrullinated peptide-positive rheumatoid arthritis: a nationwide case-control study in Denmark. Arthritis Rheum 56(5):1446–1453
Chen Q, Jin Z, Xiang C et al (2014) Absence of protective effect of oral contraceptive use on the development of rheumatoid arthritis: a meta-analysis of observational studies. Int J Rheum Dis 17(7):725–737
Pladevall-Vila M, Delclos GL, Varas C et al (1996) Controversy of oral contraceptives and risk of rheumatoid arthritis: meta-analysis of conflicting studies and review of conflicting meta-analyses with special emphasis on analysis of heterogeneity. Am J Epidemiol 144(1):1–14
Alpizar-Rodriguez D, Pluchino N, Canny G, Gabay C, Finckh A (2016) The role of female hormonal factors in the development of rheumatoid arthritis. Rheumatology (Oxford). doi:10.1093/rheumatology/kew318
Berglin E, Kokkonen H, Einarsdottir E, Agren A, Rantapaa DS (2010) Influence of female hormonal factors, in relation to autoantibodies and genetic markers, on the development of rheumatoid arthritis in northern Sweden: a case-control study. Scand J Rheumatol 39(6):454–460
Orellana CKL, Alfredsson, L Bengtsson, C (2015) Breastfeeding, oral contraceptive and the risk of rheumatoid arthritis: results from the Swedish Epidemiological Investigation of RA Study [abstract]. Arthritis Rheumatol 67(supp 10)
Orellana C, Saevarsdottir S, Klareskog L et al (2015) Postmenopausal hormone therapy and the risk of rheumatoid arthritis: results from the Swedish EIRA population-based case-control study. Eur J Epidemiol. doi:10.1007/s10654-015-0004-y
Peschken CA, Robinson DB, Hitchon CA et al (2012) Pregnancy and the risk of rheumatoid arthritis in a highly predisposed north American native population. J Rheumatol 39(12):2253–2260
Silman A, Kay A, Brennan P (1992) Timing of pregnancy in relation to the onset of rheumatoid arthritis. Arthritis Rheum 35(2):152–155
Guthrie KA, Dugowson CE, Voigt LF, Koepsell TD, Nelson JL (2010) Does pregnancy provide vaccine-like protection against rheumatoid arthritis? Arthritis Rheum 62(7):1842–1848
Adab P, Jiang CQ, Rankin E et al (2014) Breastfeeding practice, oral contraceptive use and risk of rheumatoid arthritis among Chinese women: the Guangzhou biobank cohort study. Rheumatology (Oxford) 53(5):860–866
Chen H, Wang J, Zhou W, Yin H, Wang M (2015) Breastfeeding and risk of rheumatoid arthritis: a systematic review and metaanalysis. J Rheumatol. doi:10.3899/jrheum.150195
Wallenius M, Skomsvoll JF, Irgens LM et al (2010) Postpartum onset of rheumatoid arthritis and other chronic arthritides: results from a patient register linked to a medical birth registry. Ann Rheum Dis 69(2):332–336
Heikkila R, Aho K, Heliovaara M et al (1998) Serum androgen-anabolic hormones and the risk of rheumatoid arthritis. Ann Rheum Dis 57(5):281–285
Tengstrand B, Carlstrom K, Hafstrom I (2009) Gonadal hormones in men with rheumatoid arthritis—from onset through 2 years. J Rheumatol 36(5):887–892
Pikwer M, Giwercman A, Bergstrom U et al (2014) Association between testosterone levels and risk of future rheumatoid arthritis in men: a population-based case-control study. Ann Rheum Dis 73(3):573–579
Cutolo M, Seriolo B, Villaggio B et al (2002) Androgens and estrogens modulate the immune and inflammatory responses in rheumatoid arthritis. Ann N Y Acad Sci 966:131–142
Schmidt M, Weidler C, Naumann H et al (2005) Androgen conversion in osteoarthritis and rheumatoid arthritis synoviocytes—androstenedione and testosterone inhibit estrogen formation and favor production of more potent 5alpha-reduced androgens. Arthritis Res Ther. 7(5):R938–R948
Castagnetta LA, Carruba G, Granata OM et al (2003) Increased estrogen formation and estrogen to androgen ratio in the synovial fluid of patients with rheumatoid arthritis. J Rheumatol 30(12):2597–2605
Capellino S, Straub RH, Cutolo M (2014) Aromatase and regulation of the estrogen-to-androgen ratio in synovial tissue inflammation: common pathway in both sexes. Ann N Y Acad Sci 1317:24–31
Baillargeon J, Al Snih S, Raji MA et al (2016) Hypogonadism and the risk of rheumatic autoimmune disease. Clin Rheumatol 35(12):2983–2987
Karlson EW, Chibnik LB, McGrath M et al (2009) A prospective study of androgen levels, hormone-related genes and risk of rheumatoid arthritis. Arthritis Res Ther 11(3):R97
Masi AT, Elmore KB, Rehman AA et al (2013) Lower serum androstenedione levels in pre-rheumatoid arthritis versus normal control women: correlations with lower serum cortisol levels. Autoimmune Dis 2013:593493
Jorgensen KT, Pedersen BV, Jacobsen S, Biggar RJ, Frisch M (2010) National cohort study of reproductive risk factors for rheumatoid arthritis in Denmark: a role for hyperemesis, gestational hypertension and pre-eclampsia? Ann Rheum Dis 69(2):358–363
Jorgensen KT, Harpsoe MC, Jacobsen S, Jess T, Frisch M (2014) Increased risk of rheumatoid arthritis in women with pregnancy complications and poor self-rated health: a study within the Danish National Birth Cohort. Rheumatology (Oxford) 53(8):1513–1519
Nelson JL (2009) Naturally acquired microchimerism: for better or for worse. Arthritis Rheum 60(1):5–7
Wolfberg AJ, Lee-Parritz A, Peller AJ, Lieberman ES (2004) Association of rheumatologic disease with preeclampsia. Obstet Gynecol 103(6):1190–1193
Rak JM, Maestroni L, Balandraud N et al (2009) Transfer of the shared epitope through microchimerism in women with rheumatoid arthritis. Arthritis Rheum 60(1):73–80
Svendsen AJ, Kyvik KO, Houen G et al (2013) On the origin of rheumatoid arthritis: the impact of environment and genes—a population based twin study. PLoS One 8(2):e57304
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is a contribution to the special issue on Immunopathology of Rheumatoid Arthritis - Guest Editors: Cem Gabay and Paul Hasler
Rights and permissions
About this article
Cite this article
Alpízar-Rodríguez, D., Finckh, A. Environmental factors and hormones in the development of rheumatoid arthritis. Semin Immunopathol 39, 461–468 (2017). https://doi.org/10.1007/s00281-017-0624-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00281-017-0624-2