Abstract
Background and aims
Pancreatic ductal adenocarcinoma (PDAC) represents the fourth cause of cancer-related death. We aimed to evaluate whether gemcitabine treatment shapes the gut microbiota in a model of PDAC xenografted mice.
Materials and methods
Pancreatic cancer xenograft mice were subjected to gemcitabine injection once per week for 3 weeks to assess the tumor volume as compared to control mice injected with normal saline solution. The composition of fecal microbiota, the activation of NF-kB pathway in cancer tissues and the serum metabolomics were further analyzed.
Results
Gemcitabine considerably decreases the proportion of Gram- positive Firmicutes (from about 39 to 17%) and the Gram- negative Bacteroidetes (from 38 to 17%) which are the two dominant phyla in the gut of tumor-bearing control mice. This downshift was replaced by an increase of Proteobacteria (Escherichia coli and Aeromonas hydrophila) from 15 up to 32% and Verrucomicrobia (Akkermansia muciniphila) from 5 to 33% in the gut of drug-receiving mice. An overall increase in inflammation-associated bacteria was observed upon gemcitabine. Consistently, activation of the NF-kB canonical pathway was found in cancer tissues from gemcitabine-treated mice. Serum metabolomics revealed a significant decrease of the purine compounds inosine and xanthine, and a decreasing trend for their metabolically-related molecule hypoxanthine.
Discussion
Understanding chemotherapy side effects may explain the lack of activity or the chemoresistant processes and it may help to set up strategies to improve the effectiveness of therapy.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Pancreatic ductal adenocarcinoma (PDAC) accounts for 85–95% of pancreatic cancers [1, 2], which represent the fourth lethal malignancy, with only less than 5% of patients reaching 5-year survival [3]. This poor outcome is attributable to lack of early symptoms, to limited biomarkers and screening tools which delay diagnosis. The latter is responsible of the aggressiveness of the disease with early metastatic spreading and chemoresistance processes [4,5,6]. From the point of view of clinical management, PDAC can be classified into (1) localized, resectable cancer, (2) locally advanced, unresectable cancer and (3) metastatic cancer [1]. Only 10–20% of patients are eligible for surgical resection, which remains the only curative option [1, 7, 8]. Chemotherapy represents the first-line approach in the treatment of the advanced and metastatic disease [2, 4]. Among chemotherapeutic agents, gemcitabine represents the standard of care in the treatment of such disease, as a single agent or in combination with other drugs to improve its efficacy [2, 7]. Despite being the gold standard in the treatment of PDAC, gemcitabine-based therapies provide only a modest improvement in prognosis, mainly because of both intrinsic or acquired drug resistance phenomema.
Nowadays, it is evident that inflammation plays a central role both in the onset and in the progression of PDAC [9, 10], not only as a risk factor but also as a consequence of cancer itself [11]. The persistence of an inflammatory state hinders the curability of the disease, that is why different anti-inflammatory drugs are being currently evaluated as supportive strategies in the management of PDAC [12, 13]. Given the poor responsiveness of PDAC to traditional chemotherapeutic agents, and the pivotal role of inflammation in the course of the disease, finding alternative additional approaches which may improve the clinical outcome is an interesting and promising challenge.
A growing and exciting field of research is represented by the study of microbiota, the community of commensal, symbiotic and pathogen microorganisms living within host’s body [14], mostly (99%) in the gut [15], which are known to take part and modulate many physiological and pathological processes [16]. Carcinogenesis, tumor progression and response to anticancer therapies are important processes known to be influenced by gut microbiota [17]. The majority of reports linking gut microbiota to cancer concern colorectal cancer, as the natural seat where microorganisms reside. Intestinal microbiota, however, not only acts locally but also at distant sites, through bacterial translocation or through the release of microorganism-associated molecular patterns (MAMPs) that reach other organs anatomically linked to the gut [15]. In this regard, changes in gut microbiota composition are suggested to influence also pancreatic cancer [11, 14, 18], as it was demonstrated for alterations in oral microbiota [11, 14, 19, 20].
Several pre-clinical and clinical studies were conducted with the aim to evaluate the impact of chemotherapy on the composition of gut microbiota and the subsequent development of gastrointestinal mucositis, which is a frequently observed side effect in patients receiving chemo- or radiotherapy [21].
In this study, we aimed to evaluate whether gemcitabine treatment influences the gut microbiota and serum metabolomic in a model of PDAC xenografted mice. Reducing the side effects of chemotherapy may be useful to set up integrative therapeutic strategies which may help to enhance the effectiveness of therapy.
Materials and methods
Animal study
The animal study was performed in an AAALAC (Association for Assessment and Accreditation of Laboratory Animal Care International) accredited experimental facility. Principles of laboratory animal care were followed for the welfare of animals in experimental neoplasia. Protocols were approved by the Institutional Animal Care and Use Committee. As previously reported [24], BxPC-3-luc cells in log phase of growth were suspended in a PBS/matrigel mixture (1:1) and 5 × 106 cells/mouse were injected subcutaneously into the right flank of 5–6 weeks old female Nu/Nu nude mice. The tumor was allowed to grow until the desired size (~ 100 mm³) was reached, then mice were randomly allocated into two different treatment groups (6 per group): Group 1 (Normal saline, i.p, qw), group 2 (Gemcitabine, 25 mg/kg, i.p, qw). Gemcitabine was dissolved in saline (0.9% NaCl w/v in water) to generate a final concentration of 10 mg/ml. The drug was freshly dissolved before use. The intraperitoneal injection volume was 100 μl/10 g mouse weight. Treatment was administered for up to 3 weeks with one i.p. injection of gemcitabine per week after which the animals were sacrificed once tumor volume reached 2500 mm3 or they showed signs of a deteriorating health. Fresh fecal samples from animals of both experimental groups were collected into sterile eppendorfs and kept at − 80 °C until use.
Microbiota analysis
Analysis of microbiota composition was performed as already described [18]. Briefly, genomic DNA was extracted from feces using MoBio Power Fecal DNA extraction kits (MoBio, Carlsbad, Canada) according to the manufacturer’s instructions. Amplicons (~ 450 bp) obtained with universal primers targeting the V3–V4 hypervariable region of the 16S rRNA gene were sequenced on Illumina MiSeq 2 × 250 v2 platform. Sequence data were quality-controlled and analyzed using the open source program BION-meta, according to author´s instructions.
Immunoblotting
Total protein extraction from snap-frozen pancreatic cancer xenograft specimens was obtained through mechanical and detergent-based lysis, using RIPA buffer supplemented with protease inhibitor cocktail (COMPLETE; Roche Diagnostics, Mannheim, Germany), 1 mM sodium orthovanadate and 1 mM sodium fluoride as phosphatase inhibitors. Equal amounts of protein extract, determined by Bradford method, were separated by SDS–PAGE and blotted on PVDF membranes (Whatman, Dassel, Germany). The membranes were blocked with 5% milk dissolved in TBS containing 0.1% Tween 20 and incubated overnight at 4 °C with primary antibodies as follows: anti-phospho-NF-κB p65 (Ser536) (1:1000, Cell Signaling Technology, Cat. # 3033), anti-NF-κB p65 (1:1000, Cell Signaling Technology, Cat. # 8242), anti-phospho-IκBα (Ser32) (1:1000, Cell Signaling Technology, Cat. # 2859), anti-IκBα (1:1000, Cell Signaling Technology, Cat. # 4814), anti-GAPDH (1:8000, Merck Millipore, Cat. # ABS16). After incubation of the membranes with respective horseradish-peroxidase-conjugated secondary antibodies, protein bands were detected by enhanced chemo-luminescence (Thermo Fisher Scientific).
LC–MS analysis of serum metabolomics
Serum proteins were precipitated using acetonitrile. The supernatant was diluted 1:1 in a acetonitrile/milliQ water mixture. Liquid chromatography/Mass spectrometry was used to analyze serum metabolites. The samples were analyzed using a slightly modified version of the acidic protocol (positive and negative ionization) described by Paglia et al. [22]. For quality control, a mixed pooled sample was created by taking a small aliquot from each sample. Every 4–5 samples this QC sample was analyzed. A targeted approach was used to extract the response of compounds included in a standard list of 97 compounds.
Statistical analysis
Data are expressed as mean ± standard deviation of the mean. Statistical significance was assumed as p ≤ 0.05 (*), p ≤ 0.01 (**) or p ≤ 0.001 (***). To explore the association of gut microbiota taxa with metabolites and both phosphorylated and total IκBα protein levels, Pearson’s correlations were estimated using SAS software.
Results
Effect of gemcitabine on pancreatic tumor growth and fecal microbiota
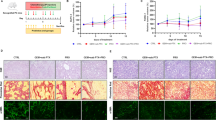
As a first step, we evaluated the effect of gemcitabine treatment on pancreatic tumor growth in our xenograft mouse model. As shown in Fig. 1a, mice subjected to gemcitabine treatment displayed a ~ 35% reduction in tumor volume at the end of the therapy (p = 0.05). Moreover, mice treated with gemcitabine displayed a trend toward decreased body weight (22.7 ± 1.5 gr control vs 20.3 ± 2.7 gr gemcitabine treated) compared with control mice, without reaching statistical significance (Fig. 1b). Furthermore, Fig. 1c shows the relative abundance of the top 15 most represented bacterial taxa in both control and gemcitabine-treated group. In agreement with the literature [23, 24], the Gram- positive Firmicutes (to which belong Lachnospiraceae, Lactobacillusanimalis, Ruminococcaceae, Blautia, Anaerotruncus, Roseburia, Erysipelatoclostridium and Peptoclostridium difficile) and the Gram-negative Bacteroidetes (to which belong Bacteroidales and Alistipes) are the two dominant phyla in the gut of tumor-bearing control mice. Gemcitabine, however, considerably decrease the proportion of both phyla (from about 39 to 17% as regards Firmicutes, from 38 to 17% as regards Bacteroidetes) and concomitantly supported the growth of Proteobacteria (mainly Escherichia coli and Aeromonas hydrophila, 32 vs 17% in control mice) and Verrucomicrobia (mainly Akkermansia muciniphila, 33 vs 5% in control mice) that are the most represented phyla in the gut of drug-receiving mice (Table 1). Looking at lower taxonomic levels, the bacteria belonging to the Bacteroidales order were approximately halved in the gemcitabine group compared to the control, and similarly, a net decrease in the relative abundance of the Lachnospiraceae family (from about 16 to 6%) and of the Ruminococcaceae (from 10 to ~ 2%) was observed in chemotherapy-treated mice. Bacteria belonging to the genus of Erysipelatoclostridium, from about 4% in control group, almost disappeared in gemcitabine-treated mice, and the same drop was observed for the Alistipes and Anaerotruncus (less than 2% in control group versus ~ 0% in gemcitabine treated mice). However, the most striking differences between the two experimental groups were observed at the species level, with A. muciniphila and Escherichia coli significantly increased while B. acidifaciens decreased in gemcitabine- treated mice compared to controls. Less pronounced but still noteworthy, L. animalis, representing about 4% of the bacterial abundance in controls was detectable in minor amounts in treated animals; conversely P. difficile, which was not detected at all in control mice, was found to represent about 4% of gut microbiota in the gemcitabine group. As we will discuss later, the overall alteration observed in microbiota composition is suggestive of a pro-inflammatory bacterial profile in the gut of gemcitabine-treated mice.
Tumor volume evaluation (a). Body weight of xenografted pancreatic cancer mice vs gemcitabine-treated mice (b). Results are expressed as means ± SD. Differences were considered as significant when p < 0.05 (*). Mean composition of bacterial taxa in mice fecal samples (c). The average abundance (average sum of reads in relative scale, %) of the most represented 15 bacterial taxa is shown
Gemcitabine treatment influences NF-κB inflammatory pathway
To confirm whether gemcitabine could be responsible for a worsening of inflammation which notoriously fuels pancreatic cancer [11, 25], we assessed the activation status of the NF-κB pathway, which plays a critical role in inflammatory conditions, in the biopsies of pancreatic cancer mice. The most frequently activated form of NF-kB is a heterodimer composed of p65 and p50, which is sequestered in the cytoplasm by binding with a member of IkB proteins (e.g. IkBα), in basal conditions. In the canonical activation pathway, IkB is phosphorylated and subsequently polyubiquitinylated and degraded by proteasome. This allows NF-kB to move to the nucleus and promote transcription of its target genes [26]. In addition, phosphorylation events involving the NF-κB proteins themselves can also occur, as alternative ways of activation [27].
Figure 2a–c shows significant changes upon gemcitabine treatment in the total amount and the phosphorylated content of IkBα. In detail, IkBα phosphorylation increased (p = 0.03) (Fig. 2b), while the total amount of the protein decreased (p = 0.03) (Fig. 2c), indicating an activation of the inflammatory pathway. We also investigated the phosphorylation level of NF-kB p65 (S536) (Fig. 2d, e). A trend towards an increase in NF-kB p65 phosphorylation, without reaching statistical significance, was observed in the gemcitabine-treated mice.
Immunoblot detection (a) and quantification of relative phospho-IkBα normalized to total IkBα protein expression (b) and of relative total IkBα protein level (c) in tumor tissue of control and gemcitabine-treated mice. Immunoblot detection (d) and quantification (e) of relative phospho-p65NF-kB normalized to total p65NF-kB protein expression in control tumor tissue and gemcitabine-treated mice. Results are expressed as means ± SD. Differences were considered as significant when p < 0.05 (*)
Metabolomic profile in gemcitabine-treated PC mice
We next evaluated the metabolomic profile of serum samples collected from both control and gemcitabine-treated animals. Using a targeted approach, a total of 42 out of 97 compounds were detected in mice sera, namely: (iso)leucine, acetyl-carnitine, a-ketoglutaric acid, arginine, asparagine, aspartic acid, carnitine, citric acid, citrulline, creatine, creatinine, cystine, cytidine, dihydroxyacetone-phosphate, fumaric acid, glucose, glucose-x-phosphate, glutamic acid, glutamine, histidine, hypoxanthine, inosine, itaconic acid, lactic acid, lysine, malic acid, methionine, myoinositol, nicotine amide, ornitine, pantothenic acid, phenylalanine, phosphoenolpyruvic acid, proline, pyridoxamine, serine, succinic acid, taurine, tryptophane, tyrosine, uridine, and xanthine (Fig. 3). Among these compounds, a significant decrease of the purine bases inosine and xanthine, and a decreasing trend for their metabolically-related compound (i.e. hypoxanthine) were observed in the gemcitabine group as compared to the control group.
Association of gut microbiota profile with metabolites levels and NF-κB signaling
Finally we performed, with only an exploratory intent due to the low number of replicates as feces of the mice housed in the same cage were pooled, a correlation analysis between changes in gut microbiota and the observed alterations of the serum metabolites and NF-κB signaling, irrespective of the treatment administered to animals. Table 2 shows the associations of microbiota taxa with: (1) xanthine, inosine and hypoxanthine serum levels; (2) ph-IkBα/ IkBα and total IkBα protein levels in cancer tissues. The observed statistically significant associations are highlighted in bold (Table 2). In detail, a positive correlation between the phylum Firmicutes and both hypoxanthine and IkBα levels was observed. Lachnospiraceae and Peptoclostridium correlated with ph-IkBα/IkBα, in a negative and a positive fashion, respectively. Lactobacillus, Erysipelatoclostridium, Aeromonas and Enterobacter were found positively associated with hypoxanthine and IkBα levels. A positive correlation was observed between Lachnospiraceae_NK4A136_group and both xanthine and inosine, while Ruminococcaceae_NK4A214_group and Anaerotruncus were positively correlated only with inosine levels.
Discussion
In recent times, extensive efforts have been devoted to the study of human gut microbiota, as it has become clear that the composition and function of this complex microbial community considerably influence a large number of physiological and pathological processes, including metabolism, immunity, inflammation and cancer [16, 28]. For this reason, microbiota is frequently termed the “hidden organ” [29] or the “forgotten organ” [30]. Although microbiota is relatively stable over time [28], a number of environmental factors, such as diet and drugs (above all antibiotics), can nevertheless shape its composition [18, 28], thus producing effects impacting on health. Several pre-clinical and clinical studies, reviewed by Touchefeu [21], have demonstrated that anticancer therapies perturb gut microbial profile, which is often linked to the development of detrimental effects such as gastrointestinal mucositis.
The current study shows that gemcitabine treatment causes substantial modifications in the intestinal microbial community of mice bearing PDAC in comparison to their non-treated counterpart. The vast majority of gut microbiota is composed of bacteria, especially belonging to two dominant phyla, Firmicutes and Bacteroidetes [23, 24, 31]. As a first result, we observed that both these phyla turned out to be widely under-represented in the gut of gemcitabine-receiving mice, whose bacterial composition was shifted in favor of two other phyla, Proteobacteria (mainly E. coli) and Verrucomicrobia (mainly A. muciniphila) which are generally minor constituents of gut microbiota [23]. An overgrowth of Proteobacteria is known to be related to intestinal inflammation [32, 33], and a concomitant decrease in Firmicutes is associated with intestinal bowel diseases (IBD) [33,34,35,36]. Similarly, the Bacteroidetes phylum and the Lachnospiraceae family were found to be reduced in human IBD [37]. Moreover, A. muciniphila, a mucin-degrading bacterium, strongly increased in gemcitabine-treated mice, has been found to favor gut inflammation because of its mucolytic activity [38]. A decrease in unclassified Bacteroidales, whose relative abundance is approximately halved in gemcitabine-treated mice compared to controls, has been described in a mouse model of colitis [39]. This shift of microbiota towards an inflammation-related bacterial profile may, on the one hand, be responsible for gastrointestinal mucositis and side effects which are often observed after chemotherapy treatment and, on the other hand, paradoxically exacerbate the inflammatory state which is a critical factor fueling pancreatic cancer [11, 25]. Inflammation increases intestinal permeability thus allowing bacteria to enter the circulation and move to distant organs [11]. Bacterial translocation through the bloodstream, direct reflux through the main pancreatic ducts or the activation of the immune system by soluble MAMPs are all proposed mechanisms to explain how intestinal microbiota may affect pancreatic carcinogenesis [11, 15]. One of the pathways commonly activated in inflammation is the NF-kB signaling, which is also triggered by the binding of MAMPs to Toll- like receptors (TLRs) of the host [26]. Immunoblots performed on pancreatic cancer tissues, clearly revealed in gemcitabine-receiving mice an activation of the canonical NF-kB pathway, that is the one most frequently triggered by TLR signaling [26]. Indeed, IkBα phosphorylation is increased and, accordingly, IkBα total form was decreased as a consequence of its degradation. When we investigated the phosphorylation of S536 of the p65 subunit as an alternative way of activation, a trend towards an increase was observed in the gemcitabine-treated mice, although not reaching statistical significance. This result, supported by correlation analyses, strongly suggests that the shifts observed in gut microbiota after gemcitabine treatment may have reflected at distant sites and may have had consequences on clinical outcome.
In our study, gemcitabine treatment produced a considerable decrease in the relative abundance of two bacterial families, namely Lachnospiraceae and Ruminococcaceae. Numerous members of these families are known for their ability to generate butyrate as a product of fermentation of non-digestible carbohydrates [40]. Aside from being the main energy source for colonic epithelial cells [41], butyrate has shown anti-proliferative and pro-apoptotic effects in several cancer cell lines [41,42,43,44,45], including pancreatic cancer cells [44, 46] and anti-angiogenic properties [47,48,49]. Moreover, butyrate inhibits in vitro invasiveness of pancreatic cancer cells by altering integrin expression [50]. Even though, to the best of our knowledge, these in vitro findings still need confirmation in in vivo models of PDAC, it is tempting to speculate that strategies aimed at increasing butyrate-producing bacteria may enhance the gemcitabine anticancer effect. Furthermore, butyrate also possesses other beneficial properties, since it has anti-inflammatory effects, activates immunity [51] and stimulates the synthesis of mucus layer counteracting intestinal permeability [41, 51].
The current work revealed that gemcitabine therapy caused the appearance of Peptoclostridium difficile (previously known as Clostridum difficile [52]), which was not detected at all in the microbiota of untreated mice. P. difficile is an infectious microorganism responsible for a range of gastrointestinal manifestations, from mild to severe and lethal, whose growth is favored by factors perturbing the gut microbial eubiosis, such as antibiotic therapy [53]. Previous studies have also documented infections from this bacterium associated with various chemotherapeutic agents [54,55,56]. Together with P. difficile (belonging to the Clostridium cluster XI), overgrowth of Enterobacteriacea, to which E.coli belongs, is a frequent consequence of chemotherapies [57, 58].
Previous studies investigating the effect of chemotherapies on gut microbiota also revealed significant alteration on metabolomics [59, 60], since the interaction between bacteria and the host may impact on the host metabolic phenotype [61, 62]. In this regard, we performed a targeted analysis of mice serum metabolites and found a highly significant decrease of inosine in gemcitabine-receiving mice. Inosine is a naturally occurring metabolite of adenosine, with which it shares anti-inflammatory and immunosuppressive properties [63, 64] and has a protective effect against LPS-induced inflammation [64, 65]. Interestingly, microbiota itself may concur to determine inosine levels, since it has been demonstrated that Lactobacillus reuteri reverses inosine depletion [62]. Together with inosine, its metabolically-related compound (xanthine) was also significantly decreased while a decreasing trend of hypoxanthine was observed. A correlation analysis revealed the association between the abundance of some microbial taxa and the levels of the aforesaid metabolites.
Overall, our study suggests that gemcitabine induces significant modifications of the intestinal microbiota, many of which may be detrimental for its own efficacy. In addition, chemotherapy is associated with the promotion of inflammation at the pancreatic level and with a decreased in serum levels of inosine, xanthine and hypoxanthine, which likely are correlated to the observed dysbiosis. The results of the current study have yielded new insights in understanding the impact of gemcitabine therapy in PDAC on microbiota profile. Despite mouse models are being largely employed in the field of microbiota research [66], it should be taken into account that mouse and human microbiota, although similar at the phylum level, are largely different at lower taxonomic levels [21]. For this reason, further studies are needed to assess the impact of gemcitabine treatment on the human gut microbial communities, which may lead to plan supportive strategies to be used in the clinical setting to improve chemotherapy efficacy, reduce adverse effects and ensure a better prognosis for patients.
References
Cid-Arregui A, Juarez V (2015) Perspectives in the treatment of pancreatic adenocarcinoma. World J Gastroenterol 21(31):9297–9316
Conroy T, Bachet JB, Ayav A, Huguet F, Lambert A, Caramella C, Marechal R, Van Laethem JL, Ducreux M (2016) Current standards and new innovative approaches for treatment of pancreatic cancer. Eur J Cancer 57:10–22
Siegel RL, Miller KD, Jemal A, Cancer statistics (2016) CA Cancer. J Clin 66(1):7–30
Cheema AR, O’Reilly EM (2016) Management of Metastatic Pancreatic Adenocarcinoma. Surg Clin North Am 96(6):1391–1414
D’Aronzo M, Vinciguerra M, Mazza T, Panebianco C, Saracino C, Pereira SP, Graziano P, Pazienza V (2015) Fasting cycles potentiate the efficacy of gemcitabine treatment in in vitro and in vivo pancreatic cancer models. Oncotarget 6(21):18545–18557
Gharibi A, Adamian Y, Kelber JA (2016) Cellular and molecular aspects of pancreatic cancer. Acta Histochem 118(3):305–316
Korkeila EA (2015) Advanced pancreatic cancer—how to choose an adequate treatment option. World J Gastroenterol 21(38):10709–10713
Stathis A, Moore MJ (2010) Advanced pancreatic carcinoma: current treatment and future challenges. Nat Rev Clin Oncol 7(3):163–172
Greer JB, Whitcomb DC (2009) Inflammation and pancreatic cancer: an evidence-based review. Curr Opin Pharmacol 9(4):411–418
Steele CW, Kaur Gill NA, Jamieson NB, Carter CR (2016) Targeting inflammation in pancreatic cancer: clinical translation. World J Gastrointest Oncol 8(4):380–388
Zambirinis CP, Pushalkar S, Saxena D, Miller G (2014) Pancreatic cancer, inflammation, and microbiome. Cancer J 20(3):195–202
Momi N, Kaur S, Krishn SR, Batra SK (2012) Discovering the route from inflammation to pancreatic cancer. Minerva Gastroenterol Dietol 58(4):283–297
Uomo I, Miraglia S, Pastorello M (2010) Inflammation and pancreatic ductal adenocarcinoma: a potential scenario for novel drug targets. JOP 11(3):199–202
Leal-Lopes C, Velloso FJ, Campopiano JC, Sogayar MC, Correa RG (2015) Roles of Commensal Microbiota in Pancreas Homeostasis and Pancreatic Pathologies. J Diabetes Res. https://doi.org/10.1155/2015/284680
Schwabe RF, Jobin C (2013) The microbiome and cancer. Nat Rev Cancer 13(11):800–812
Blaser MJ (2014) The microbiome revolution. J Clin Invest 124(10):4162–4165
Zitvogel L, Galluzzi L, Viaud S, Vetizou M, Daillere R, Merad M, Kroemer G (2015) Cancer and the gut microbiota: an unexpected link. Sci Transl Med 7(271):271ps271
Panebianco C, Adamberg K, Adamberg S, Saracino C, Jaagura M, Kolk K, Di Chio AG, Graziano P, Vilu R, Pazienza V (2017) Engineered resistant-starch (ERS) diet shapes colon microbiota profile in parallel with the retardation of tumor growth in in vitro and in vivo pancreatic cancer models. Nutrients 9(4). https://doi.org/10.3390/nu9040331
Farrell JJ, Zhang L, Zhou H, Chia D, Elashoff D, Akin D, Paster BJ, Joshipura K, Wong DT (2012) Variations of oral microbiota are associated with pancreatic diseases including pancreatic cancer. Gut 61(4):582–588
Michaud DS, Joshipura K, Giovannucci E, Fuchs CS (2007) A prospective study of periodontal disease and pancreatic cancer in US male health professionals. J Natl Cancer Inst 99(2):171–175
Touchefeu Y, Montassier E, Nieman K, Gastinne T, Potel G, Bruley des Varannes S, Le Vacon F, de La Cochetiere MF (2014) Systematic review: the role of the gut microbiota in chemotherapy- or radiation-induced gastrointestinal mucositis - current evidence and potential clinical applications. Aliment Pharmacol Ther 40(5):409–421
Paglia G, Williams JP, Menikarachchi L, Thompson JW, Tyldesley-Worster R, Halldorsson S, Rolfsson O, Moseley A, Grant D, Langridge J, Palsson BO, Astarita G (2014) Ion mobility derived collision cross sections to support metabolomics applications. Anal Chem 86(8):3985–3993
Eckburg PB, Bik EM, Bernstein CN, Purdom E, Dethlefsen L, Sargent M, Gill SR, Nelson KE, Relman DA (2005) Diversity of the human intestinal microbial flora. Science 308(5728):1635–1638
Ley RE, Backhed F, Turnbaugh P, Lozupone CA, Knight RD, Gordon JI (2005) Obesity alters gut microbial ecology. Proc Natl Acad Sci USA 102(31):11070–11075
Hausmann S, Kong B, Michalski C, Erkan M, Friess H (2014) The role of inflammation in pancreatic cancer. Adv Exp Med Biol 816:129–151
Kawai T, Akira S (2007) Signaling to NF-kappaB by Toll-like receptors. Trends Mol Med 13(11):460–469
Christian F, Smith EL, Carmody RJ (2016) The regulation of NF-kappaB subunits by phosphorylation. Cells 5(1):12
Clemente JC, Ursell LK, Parfrey LW, Knight R (2012) The impact of the gut microbiota on human health: an integrative view. Cell 148(6):1258–1270
O’Hara AM, Shanahan F (2006) The gut flora as a forgotten organ. EMBO Rep 7(7):688–693
Guinane CM, Cotter PD (2013) Role of the gut microbiota in health and chronic gastrointestinal disease: understanding a hidden metabolic organ. Ther Adv Gastroenterol 6(4):295–308
Turnbaugh PJ, Ley RE, Mahowald MA, Magrini V, Mardis ER, Gordon JI (2006) An obesity-associated gut microbiome with increased capacity for energy harvest. Nature 444(7122):1027–1031
Arthur JC, Perez-Chanona E, Muhlbauer M, Tomkovich S, Uronis JM, Fan TJ, Campbell BJ, Abujamel T, Dogan B, Rogers AB, Rhodes JM, Stintzi A, Simpson KW, Hansen JJ, Keku TO, Fodor AA et al (2012) Intestinal inflammation targets cancer-inducing activity of the microbiota. Science 338(6103):120–123
Shin NR, Whon TW, Bae JW (2015) Proteobacteria: microbial signature of dysbiosis in gut microbiota. Trends Biotechnol 33(9):496–503
Lavelle A, Lennon G, O’Sullivan O, Docherty N, Balfe A, Maguire A, Mulcahy HE, Doherty G, O’Donoghue D, Hyland J, Ross RP, Coffey JC, Sheahan K, Cotter PD, Shanahan F, Winter DC et al (2015) Spatial variation of the colonic microbiota in patients with ulcerative colitis and control volunteers. Gut 64(10):1553–1561
Matsuoka K, Kanai T (2015) The gut microbiota and inflammatory bowel disease. Semin Immunopathol 37(1):47–55
Morgan XC, Tickle TL, Sokol H, Gevers D, Devaney KL, Ward DV, Reyes JA, Shah SA, LeLeiko N, Snapper SB, Bousvaros A, Korzenik J, Sands BE, Xavier RJ, Huttenhower C (2012) Dysfunction of the intestinal microbiome in inflammatory bowel disease and treatment. Genome Biol 13(9):R79
Frank DN, St Amand AL, Feldman RA, Boedeker EC, Harpaz N, Pace NR (2007) Molecular-phylogenetic characterization of microbial community imbalances in human inflammatory bowel diseases. Proc Natl Acad Sci USA 104(34):13780–13785
Ganesh BP, Klopfleisch R, Loh G, Blaut M (2013) Commensal Akkermansia muciniphila exacerbates gut inflammation in Salmonella Typhimurium-infected gnotobiotic mice. PLoS One 8(9):e74963
Berry D, Schwab C, Milinovich G, Reichert J, Ben Mahfoudh K, Decker T, Engel M, Hai B, Hainzl E, Heider S, Kenner L, Muller M, Rauch I, Strobl B, Wagner M, Schleper C et al (2012) Phylotype-level 16S rRNA analysis reveals new bacterial indicators of health state in acute murine colitis. ISME J 6(11):2091–2106
Vital M, Howe AC, Tiedje JM (2014) Revealing the bacterial butyrate synthesis pathways by analyzing (meta)genomic data. MBio 5(2):e00889
Hamer HM, Jonkers D, Venema K, Vanhoutvin S, Troost FJ, Brummer RJ (2008) Review article: the role of butyrate on colonic function. Aliment Pharmacol Ther 27(2):104–119
Canani RB, Costanzo MD, Leone L, Pedata M, Meli R, Calignano A (2011) Potential beneficial effects of butyrate in intestinal and extraintestinal diseases. World J Gastroenterol 17(12):1519–1528
Chopin V, Toillon RA, Jouy N, Le Bourhis X (2002) Sodium butyrate induces P53-independent, Fas-mediated apoptosis in MCF-7 human breast cancer cells. Br J Pharmacol 135(1):79–86
Gaschott T, Maassen CU, Stein J (2001) Tributyrin, a butyrate precursor, impairs growth and induces apoptosis and differentiation in pancreatic cancer cells. Anticancer Res 21(4A):2815–2819
Scheppach W, Weiler F (2004) The butyrate story: old wine in new bottles? Curr Opin Clin Nutr Metab Care 7(5):563–567
Natoni F, Diolordi L, Santoni C, Gilardini Montani MS (2005) Sodium butyrate sensitises human pancreatic cancer cells to both the intrinsic and the extrinsic apoptotic pathways. Biochim Biophys Acta 1745(3):318–329
Blank-Porat D, Gruss-Fischer T, Tarasenko N, Malik Z, Nudelman A, Rephaeli A (2007) The anticancer prodrugs of butyric acid AN-7 and AN-9, possess antiangiogenic properties. Cancer Lett 256(1):39–48
Ogawa H, Rafiee P, Fisher PJ, Johnson NA, Otterson MF, Binion DG (2003) Sodium butyrate inhibits angiogenesis of human intestinal microvascular endothelial cells through COX-2 inhibition. FEBS Lett 554(1–2):88–94
Zgouras D, Wachtershauser A, Frings D, Stein J (2003) Butyrate impairs intestinal tumor cell-induced angiogenesis by inhibiting HIF-1alpha nuclear translocation. Biochem Biophys Res Commun 300(4):832–838
Farrow B, Rychahou P, O’Connor KL, Evers BM (2003) Butyrate inhibits pancreatic cancer invasion. J Gastrointest Surg 7(7):864–870
van Vliet MJ, Harmsen HJ, de Bont ES, Tissing WJ (2010) The role of intestinal microbiota in the development and severity of chemotherapy-induced mucositis. PLoS Pathog 6(5):e1000879
Yutin N, Galperin MY (2013) A genomic update on clostridial phylogeny: Gram-negative spore formers and other misplaced clostridia. Environ Microbiol 15(10):2631–2641
Bien J, Palagani V, Bozko P (2013) The intestinal microbiota dysbiosis and Clostridium difficile infection: is there a relationship with inflammatory bowel disease? Therap Adv Gastroenterol 6(1):53–68
Anand A, Glatt AE (1993) Clostridium difficile infection associated with antineoplastic chemotherapy: a review. Clin Infect Dis 17(1):109–113
Masciullo V, Mainenti S, Lorusso D, Margariti PA, Scambia G (2010) Lethal clostridium difficile colitis associated with paclitaxel and carboplatin chemotherapy in ovarian carcinoma: case report and review of the literature. Obstet Gynecol Int 2010:749789
Raza S, Baig MA, Russell H, Gourdet Y, Berger BJ (2010) Clostridium difficile infection following chemotherapy. Recent Pat Antiinfect Drug Discov 5(1):1–9
Lin XB, Dieleman LA, Ketabi A, Bibova I, Sawyer MB, Xue H, Field CJ, Baracos VE, Ganzle MG (2012) Irinotecan (CPT-11) chemotherapy alters intestinal microbiota in tumour bearing rats. PLoS One 7(7):e39764
Stringer AM, Gibson RJ, Logan RM, Bowen JM, Yeoh AS, Hamilton J, Keefe DM (2009) Gastrointestinal microflora and mucins may play a critical role in the development of 5-fluorouracil-induced gastrointestinal mucositis. Exp Biol Med (Maywood) 234(4):430–441
Montrose DC, Zhou XK, McNally EM, Sue E, Yantiss RK, Gross SS, Leve ND, Karoly ED, Suen CS, Ling L, Benezra R, Pamer EG, Dannenberg AJ (2016) Celecoxib alters the intestinal microbiota and metabolome in association with reducing polyp burden. Cancer Prev Res (Phila) 9(9):721–731
Forsgard RA, Marrachelli VG, Korpela K, Frias R, Collado MC, Korpela R, Monleon D, Spillmann T, Osterlund P (2017) Chemotherapy-induced gastrointestinal toxicity is associated with changes in serum and urine metabolome and fecal microbiota in male Sprague–Dawley rats. Cancer Chemother Pharmacol 80(2):317–332
Daliri EB, Wei S, Oh DH, Lee BH (2017) The human microbiome and metabolomics: current concepts and applications. Crit Rev Food Sci Nutr 57(16):3565–3576
He B, Hoang TK, Wang T, Ferris M, Taylor CM, Tian X, Luo M, Tran DQ, Zhou J, Tatevian N, Luo F, Molina JG, Blackburn MR, Gomez TH, Roos S, Rhoads JM et al (2016) Resetting microbiota by Lactobacillus reuteri inhibits T reg deficiency-induced autoimmunity via adenosine A2A receptors. J Exp Med 214(1):107–123
da Rocha Lapa F, da Silva MD, de Almeida Cabrini D, Santos AR (2012) Anti-inflammatory effects of purine nucleosides, adenosine and inosine, in a mouse model of pleurisy: evidence for the role of adenosine A2 receptors. Purinergic Signal 8(4):693–704
Hasko G, Kuhel DG, Nemeth ZH, Mabley JG, Stachlewitz RF, Virag L, Lohinai Z, Southan GJ, Salzman AL, Szabo C (2000) Inosine inhibits inflammatory cytokine production by a posttranscriptional mechanism and protects against endotoxin-induced shock. J Immunol 164(2):1013–1019
Gomez G, Sitkovsky MV (2003) Differential requirement for A2a and A3 adenosine receptors for the protective effect of inosine in vivo. Blood 102(13):4472–4478
Nguyen TL, Vieira-Silva S, Liston A, Raes J (2015) How informative is the mouse for human gut microbiota research? Dis Model Mech 8(1):1–16
Acknowledgements
We thank Morten Danielsen and the MS-Omics for the excellent technical help. No competing interest is declared.
Funding
This research was supported by the ‘‘Ricerca Corrente RC1703GA31’’ funding granted by the Italian Ministry of Health and by the “5 × 1000” voluntary contributions to our Hospital and also by European Regional Development Fund to Competence Center of Food and Fermentation Technologies (EU48667) and Institutional Research Funding to Tallinn University of Technology (IUT 19-27) of the Estonian Ministry of Education and Research.
Author information
Authors and Affiliations
Contributions
Conceived the study: VP. Designed the experiments: CP, KA, RV and VP. Performed the experiments: CP, MJ, SA and KK. Biostatistics Analysis: MC and AF. Analyzed the data: CP, KA and VP. Contributed reagents/materials/analysis tools: KA, RV and VP. Wrote the paper: CP, KA, AA and VP.
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflicts of interest.
Ethical approval
The animal study was performed in an AAALAC (Association for Assessment and Accreditation of Laboratory Animal Care International) accredited experimental facility. Principles of laboratory animal care were followed for the welfare of animals in experimental neoplasia and protocols were approved by the Institutional Animal Care and Use Committee, with the approval number ANM14-002.
Rights and permissions
About this article
Cite this article
Panebianco, C., Adamberg, K., Jaagura, M. et al. Influence of gemcitabine chemotherapy on the microbiota of pancreatic cancer xenografted mice. Cancer Chemother Pharmacol 81, 773–782 (2018). https://doi.org/10.1007/s00280-018-3549-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-018-3549-0