Abstract
Purpose
To identify the molecular mechanisms responsible for tumor cell migration is essential for developing agents that can prevent the relapse or the metastatic spread of hepatocellular carcinoma (HCC).
Methods
In this study, we investigated the effects of the transforming growth factor-β receptor I inhibitor LY2109761 on two different human HCC cell lines, in vitro and in vivo.
Results
LY2109761 inhibits HCC migration in a dose-dependent manner. This inhibition is associated with the decreased phosphorylation of SMAD-2, FAK and β1-integrin, and with increased levels of E-cadherin. By contrast, LY2109761 did not alter the phosphorylation pattern of p38MAPkinase. In a two- and a three-day time-course and in dose-titration experiments, LY2109761 inhibited HCC migration as well as phospho-SMAD-2 and the adhesion proteins. LY2109761 showed the best effect on day 2 at 1 nM and for 3 days at 100 nM concentration. This suggests that maximum effects were sustained for several days and were not dependent on excess concentrations. Finally, in a xenograft model of HCC, LY2109761 strongly inhibits tumor growth, intravasation and metastasis at the aforementioned lower concentrations.
Conclusions
In conclusion, inhibition of transforming growth factor-β (TGF-β) appears to occur at low concentrations of LY2109761 that displays multiple effects on kinases that control HCC cell migration. These findings may help the design of future clinical trials with inhibitors of TGF-β.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Hepatocellular carcinoma (HCC) is the fifth most frequent tumor and the third cause of tumor-related death in the United States and Europe [1]. The so-called “curative” therapies, including liver transplantation, surgical resection and radiofrequency ablation, can only be offered to a minority of patients, and cancer recurrence within 3 and 5 years is a common event [2]. Unfortunately, the majority of patients are not even suitable for such “curative” treatments because of their advanced stage of disease. A significant improvement in terms of overall survival for patients with advanced disease has been achieved with sorafenib, a multi-tyrosine-kinase inhibitor [3]. This finding supports the hypothesis that HCC can potentially be cured 1 day with appropriately targeted drugs. This approach requires, however, that the biological target is well understood, and off target effects are either minimized or re-directed to support the effect on the main signaling pathway.
The transforming growth factor (TGF)-β1 represents an attractive target for a novel drug development strategy for HCC treatment, because of its widely reported role in chronic liver disease and in HCC, where increased levels of TGF-β1 are associated with poor prognosis [4–6]. We have previously described the role of TGF-β1 in triggering the epithelial mesenchymal transition in HCC [7]. As reported previously, in several in vitro and in vivo models, the inhibition of the TGF-β receptor (R) I with the selective inhibitor LY2109761 reduces HCC invasiveness, increases the expression of E-cadherin on cells and blocks neo-vascularization via dephosphorylation of the intracellular tail of the β1-integrin at residues T788-789 [8, 9].
Based on these observations, we here investigated the molecular mechanisms underlying the biological activities of LY2109761. We focused on the kinase activation pattern to characterize the functional aspects of blocking TGF-β signaling in HCC. We correlated this effect of kinase inhibition with HCC spread in vitro and in vivo. This comprehensive assessment led to some novel and unexpected findings, which suggest that TGF-β inhibition has not only a single target hit (i.e., pSMAD2), but also downstream of pSMAD2 multiple kinase inactivation effects. Hence, the global effect of such an inhibition may result in a more comprehensive inhibition of several important pathways involved in the pathogenesis of HCC.
Materials and methods
Cell cultures and reagents
Human HCC cell lines HLE and HLF, periodically purchased and used at low passages, were cultured as previously reported [10]. LY2109761 was kindly provided by Eli Lilly (Indianapolis, IN). Stock solutions were prepared at 10 mM in dimethyl sulfoxide (DMSO) and stored in aliquots at −20°C. Working dilutions were made in culture medium without serum. Fibronectin (Fn) was purchased by Calbiochem (San Diego, CA). The following antibodies were purchased as indicated: against pAkt (T308) Cell Signaling (Danvers, MA) 9275, against MEK1 (Y386) Cell Signaling 9128, against pERK ½ (Y202) Cell Signaling 9101, against mTOR (S2448) Cell Signaling 2971, against p-PTEN (S380) Cell Signaling 9551, against pSmad2 (S465/467) Cell Signaling 3101, against p38-MAPK (Y180/182) Cell Signaling 9211, against p-FAK (Y576/577) Cell Signaling 3281, against E-Cadherin Cell Signaling 4065, and against p-β1-integrin (T788/789) Abcam (Cambridge, MA).
Migration assay
Migration was performed as previously described [11]. Briefly, cells before migration were harvested overnight in serum-free medium. The day after, the medium was changed and cells were cultured for 48 h in serum-free conditions containing LY2109761 at concentrations ranging from 0.01 up to 100 nM added once or twice (every 24 h). Medium was supplemented with 2 mmol/l glutamine dipeptide, glutamax, to avoid toxic catabolites (Invitrogen Carlsbad, CA), penicillin (100 IU/ml) and streptomycin (100 μg/ml)(Gibco Life Technologies, Inc.). Then, cells were washed, trypsinized and seeded (30 × 103), in the absence of the drug, in the upper part of transwell filters (8.0-μm pore size; Corning, New York) coated with Fn. In other experiments, cells were incubated under the same experimental conditions for 24 h with LY2109761 100 nM concentration for a time ranging from 1 up to 5 days. After incubation, cells were washed and challenged in a migration assay in the absence of the drug.
HCC growth, intravasation and metastasis assay
Tumor growth assay was performed as previously described [12]. Briefly, Fertilized white Leghorn chicken eggs (SPAFAS Inc., Norwich, CT) were kept in a forced-air incubator. On day 10, the eggs were candled, the chorioallantoic membrane (CAM) was dropped by applying gentle negative pressure, and a total of 5 × 105 HLE cells were grafted on the dropped CAM. The eggs were then returned to a humidified 37°C incubator. Tumors obtained from 19-day-old embryos were then carefully cut away from normal CAM tissue, and the wet weight of the tumor was determined. In drug experiments, different doses of LY2109761 were administered on days three and five postinoculation of tumor cells. Measurement of intravasation and spontaneous lung metastasis using the chick embryo CAM assay was performed as previously described [12].
ELISA for E-cadherin
E-cadherin was detected in the conditioned media and cell lysates of HLE and HLF cells treated with LY2109761 at concentrations ranging from 1 up to 100 nM for 2 days. In time-course experiments, E-cadherin was detected in the conditioned media and cell lysates of cells treated with LY2109761 at 100 nM for 3 days. All the experiments were performed in serum-free conditions. E-cadherin was detected in the conditioned media using quantikine ELISA kit from R&D Systems, Inc. (Minneapolis MN) and in the cell lysates using duoset IC from R&D following manufacturer’s instructions. Conditioned media were concentrated using the amicon ultra filter device from Millipore (Billerica, MA). In all the samples, protein concentrations were determined by colorimetric Bradford protein assay.
Reverse phase protein microarray construction
The reverse phase protein microarray construction was previously reported [13, 14]. Briefly, samples were lysed, loaded into 384-well plates and printed in duplicate onto nitrocellulose-coated glass slides (FAST Slides Whatman, Florham Park, NJ) using an Aushon 2470 arrayer (Aushon BioSystems, Burlington, MA). Cellular lysate prepared from HeLa ± Pervanadate (Becton–Dickinson, Franklin Lakes, NJ) and Jurkat ± Calyculin (Cell Signaling Technology, Danvers, MA) cell lines were printed on each array for quality control assessments. All antibodies used in this study were validated for specificity by immunoblotting prior to use on the arrays.
Total protein values were assessed by staining the slides with Sypro Ruby Blot Stain (Molecular Probes, Eugene, OR). Stained slides were scanned with Axon GenePix (Molecular Devices, Sunnyvale, CA), and spot intensities were quantitated and normalized to total protein.
Data analysis and statistical procedures
All experiments were done at least twice. Data are presented as mean ± SD of a representative experiment or of normalized data of pooled experiments. To compare means, a repeated measure analysis of variance has been performed.
For the experiment described in Figs. 1 and 2, the repeated measure has been evaluated by profile of concentration. For the experiment described in Fig. 5, the repeated measure by day has been compared with the baseline value (day 1). P values lower than 0.05 have been considered significant. The analysis has been conducted by means of SAS 9.1 for PC.
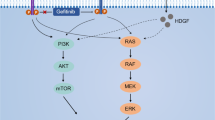
LY2109761 inhibits HCC cell migration via SMAD-2 in a dose-dependent manner and affects intracellular pathways. A HLE and HLF cell migration is inhibited in a dose-dependent manner. However, LY2109761 exerts stronger effects if the drug was added twice to cells (every 24 h) (a). The study of TGF-β-dependent pathways performed using the kinases assay was executed on each condition tested in the migration assay revealed that this effect is achieved through the inhibition of the Smad-2-dependent and nonindependent pathway (b, c). The inhibition of adhesion molecules p-FAK (d) and p-β1 integrin (e), together with the increased expression of E-cadherin (f) is dose dependent, although doubling the drug administration is not more effective than the single treatment. B single or double administration of LY2109761 on HLE and HLF yield a similar inhibition of the phosphorylation of the pathways MEK/ERK and Akt/n-TOR, also including the activation of PTEN
LY2109761 effectiveness in a dose-dependent and time-course experiment. Effects of LY2109761 on HLE and HLF migration at different doses and at different times. Best results were obtained using LY2109761 after 24 h at 1 nM or after 72 h at 100 nM concentration The kinase profile inhibition of the FAK and integrin β1 as well as the E-cadherin expression showed a similar pattern
Results
We investigated the kinase profile to understand the kinase activation profile responsible for pathways involved in TGF-β1-dependent HCC cell migration. HLE and HLF cells were incubated for 48 h in serum-free medium with the TGF-βRI kinase inhibitor LY21098761. A concentration range of 0.01–100 nM was used. We also investigated whether adding the drug twice (every 24 h) led to an enhanced effectiveness of blocking the TGF-b pathway. In both cell lines, LY2109761 strongly inhibited the migration on Fn as indicated by a typical dose–response curve. The maximum effect was observed between 1 and 100 nM, and adding the inhibitor a second time to the media led to a stronger inhibition (P < 0.0001) (Fig. 1A, panel a). Next, we investigated the phosphorylation of pSMAD-2 and p38-MAP-Kinase in the same experiment and compared the phosphorylation pattern with the results of the migration assay. SMAD-2 was dephosphorylated in a dose-dependent manner that perfectly matches the inhibition of cell migration described in panel A (Fig. 1A, panel b). By contrast, p38MAPK was not dephosphorylated even at higher concentrations (Fig. 1A, panel c). Then, we investigated the adhesion molecules commonly involved in cell migration. The kinase profile analyses revealed that FAK and β1-integrin were dephosphorylated, while E-cadherin expression was increased, P < 0.0001 (Fig. 1A, panel d–f). Importantly, while adding compound to the culture increased the inhibition of the migration, doubling the treatment did not consistently increase the dephosphorylation of pSMAD-2 or the adhesion molecules. Therefore, the inhibition of the TGF-β signaling blocks HCC cell migration via selective inhibition of the SMAD-2 pathway and leads to deactivation of the adhesion molecules FAK and β1-integrin while increasing the expression of E-cadherin. In addition to these important kinases, we also evaluated the kinase activation of those kinases generally associated with other cancer-related signal transduction pathways. As shown in Fig. 1B, pAKT/mTOR and MEK/pERK1/2 were dephosphorylated, while the oncosuppressor PTEN was activated, all with a similar dose–response profile.
Based on the above-described results, we restricted the range of concentration (1 up to 100 nM) prior to challenging the cells to migrate after 2 and 3 days of drug administration. In both cell lines, different drug concentrations reached the plateau effect at 1 nM concentration at day two. After 3 days, the stronger effect was gained with progressively increasing doses up to 100 nM concentration P < 0.0001, Fig. 2. The p-SMAD-2 and the adhesion molecules showed a pattern of dephosphorylation or increased expression consistent with the inhibition of cell migration, Fig. 2. As described previously, also pAKT/mTOR and MEK/pERK1/2 were dephosphorylated, while the oncosuppressor PTEN was activated in a time- and dose-dependent manner, P < 0.0001, Fig. 3. Hence, 1 nM after 2 days or 100 nM of LY2109761 after 3 days displayed similar effectiveness on HCC cell migration as well on the kinase profile activation.
We further studied E-cadherin because of its functional role in cell migration. Untreated HLE and HLF cells display low levels of E-cadherin in the cell lysates, while higher levels are detected in the conditioned media. However, this situation is completely reversed after LY2109761 treatment: cellular expression of E-cadherin progressively increases, while the released or soluble E-cadherin levels decrease in the conditioned media. Similarly, E-cadherin levels after treatment with LY2109761 at 100 nM concentrations increased in the cell lysates and decreased in the conditioned media during the time-course experiments, Fig. 4.
LY2109761 modulates the expression and the release of E-cadherin. E-cadherin levels were measured by ELISA in the cell lysate and conditioned media of HLE and HLF cells. In a, the levels of E-cadherin decrease in the conditioned medium and increase in the cell lysate in a dose-dependent manner, reaching the maximum effect at 100 nM, consistently with the inhibition of cell migration described in Fig. 1. In b, the levels of E-cadherin were investigated in a time-course experiment, in the conditioned medium and cell lysate of HLE and HLF treated with LY2109761 at 100 nM. The best results were obtained after 2 days and remained similar 3 days after treatment
Because of the interesting time-course pattern of the kinase activation profile, we chose to treat the cells only once with 100 nM for the migration assay. HLE and HLF cells were incubated in serum-free condition with 100 nM of LY2109761, from 1 to 5 days. After 24 h, the inhibition of HLE migration was already evident (around 30%), but reached the strongest effect after 48 h (P < 0.0001) of incubation (around 60%), with a progressive declining effect after 3, 4 and 5 days, Fig. 6. We investigated the concomitant intracellular kinase activation profile. SMAD-2 was dephosphorylated by LY2109761 with the maximum effect being evident after 48 h, P < 0.0002, corresponding to the strongest inhibition of cell migration. Consistently, with the biological effect, the adhesion molecules, FAK and β1-integrin, were dephosphorylated, while E-cadherin expression was increased in a time-dependent manner (P < 0.05) corresponding with the dephosphorylation of p-SMAD-2. Finally, MEK/ERK1/2 and Akt/mTOR were dephosphorylated, and PTEN was activated in a time-dependent manner Fig. 5.
LY2109761 effectiveness in a time-course experiment. Effectiveness of LY2109761 at 100 nM on HLE and HLF migration in 5 days time-course experiment (a). Kinase profile of the molecular pathways as well as receptors effective of cell motility performed in the time-course experiment at each condition (b). The inhibition of the molecular pathways matches the inhibition of cell migration
Based on the profile kinase inhibition, we investigated in a chick embryo xenograft model, the effect of LY2109761 added every 3 days in a concentration ranging from 10 up to 50 mg/Kg [12]. As reported in Fig. 6, LY2109761 inhibits HCC metastasis in a dose-dependent manner and furthermore, also intravasation and tumor growth.
Dose-dependent effect of LY2109761 treatment on tumor growth, intravasation and metastasis of human HCC in xenograft model. Analysis of metastatic (a), intravasation (b) and growth (c) of HCC tumors treated with different doses of LY2109761. Intravasation of tumor cells was evaluated by real-time Alu PCR of genomic DNA extracted at day 7 after injection of cells. HCC dissemination to the lung was instead evaluated by fluorescent microscopy of GFP expressing HLE cells during experimental colonization of the chick lung at 7 days postinjection. Dissemination to the lung was measured as the number of metastatic nodules/colonies per lung. Data are expressed as the mean ± SEM from three independent experiments. A total of 5 × 105 HLE cells were grafted on the dropped CAM of 10-day-old chick embryos. Different doses of LY2109761 were administered on days three and five postinoculation of tumor cells. Tumors were then carefully excised from the individual 19-day-old embryos and weighed. *P < 0.05, **P < 0.005, ***P < 0.0005 versus control
Discussion
This study describes, for the first time, the molecular mechanisms by which the inhibitor of the TGF-βRI LY2109761 blocks HCC migration. Taking advantage of a wide profile of kinase examination, we identified that LY2109761 selectively acts via a SMAD-dependent mechanism at different concentrations. This is also consistent with our previous study and with a study in pancreatic cancer cells [8, 15]. In contrast to the pancreatic cancer cells, HCC cells seem to be more sensitive to LY2109761 treatment and show a 500-fold higher sensitivity. In addition, in vivo we found that LY2109761 decreased tumor growth, intravasation and metastatization. We used a low dosage, namely 50 mg/Kg once a day for twice a week, while Melisi et al. [15] used the same drug concentration twice a day for 5 days a week. Unlike the mouse xenograft model, we used a chick embryo model and thus the difference in the animal model may explain our efficacy data on the kinetic of LY2109761. Our in vivo results can be explained by the effects of LY2109761 also on other kinases commonly involved in cancer such as MEK/ERK1-2, Akt/m-TOR and PTEN. We do not know whether these pathways are activated downstream of SMAD-2 or whether this activation is a result of unspecific inhibition by LY2109761. Hence, we cannot rule out the possibility that LY2109761 displays some indirect effect also on Akt and m-TOR downstream pathways of PI3 K, as reported in the case of cell hypertrophy induced by increased glucose concentration [16]. However, given the time-course activation (Fig. 4), it appears more likely that LY2109761 has a strong inhibition on pSMAD2 followed then by the inhibition of downstream kinases. If this is the case, LY2109761 targeting only one receptor will trigger a number of multiple cascades, which can represent a new treatment paradigm for HCC.
Independently of these unsolved specificity issues, LY2109761 demonstrates a strong association between biological effectiveness and the inhibition of kinase profile. In our study, doubling the drug concentration administration had only a small additive inhibitory effect on the HCC migration, while the inhibition of the kinases associated with the migration pathways was unchanged. Furthermore, we also demonstrate that our maximum concentration, 100 nM, is still effective up to 3 days. Therefore, repeated treatment in a short time did not add to the inhibition, and an every 3-day schedule was sufficient for achieving the intended inhibition of the HCC migration. Perhaps this longer-lasting inhibitory effect is consistent with the mechanism of action of LY2109761. Since this small molecule is a competitive inhibitor of adenosine-triphosphate (ATP), it may not be degraded as quickly in the HCC cells [17]. Thus, repeated drug administration may lead to drug accumulation without additional therapeutic effect and perhaps increase the potential of undesired or even toxic effects [12]. Furthermore, we demonstrate that LY2109761 modulates the functional role of downstream molecules involved in cell motility. For instance, we show that E-cadherin is poorly expressed in the HCC invasive cells but highly detected in the conditioned medium. LY2109761 increases the cellular expression of E-cadherin and decreases its concentration in the conditioned medium. The effect of LY2109761 on the increased expression of E-cadherin is evident up to 3 days, further confirming the utility of this therapeutic regimen. This finding is also of interest because E-cadherin can be reliably detected by ELISA in biological specimens including tissue and serum and therefore could be used as a biomarker to assess drug effectiveness.
In conclusion, our results indicate the molecular mechanisms whereby targeting TGF-βRI inhibits HCC migration in vitro and tumor progression in vivo. We also demonstrate the optimal drug concentration and schedule administration in an in vitro and in vivo model. Then, we demonstrate drug effectiveness on both HCC cells and molecular pathways and that E-cadherin may represent a biomarker to assess therapeutic efficacy. All these data provide fundamental indications for the use of LY2109761 in future clinical trials.
References
El Serag HB (2004) Hepatocellular carcinoma: recent trends in the United States. Gastroenterology 127:S27–S34
Llovet JM, Burroughs A, Bruix J (2003) Hepatocellular carcinoma. Lancet 362:1907–1917
Llovet JM, Ricci S, Mazzaferro V, Hilgard P, Gane E, Blanc JF, de Oliveira AC et al (2008) Sorafenib in advanced hepatocellular carcinoma. N Engl J Med 359:378–390
Bedossa P, Peltier E, Terris B, Franco D, Poynard T (1995) Transforming growth factor-beta 1 (TGF-beta 1) and TGF-beta 1 receptors in normal, cirrhotic, and neoplastic human livers. Hepatology 21:760–766
Tsai JF, Chuang LY, Jeng JE, Yang ML, Chang WY, Hsieh MY, Lin ZY, Tsai JH (1997) Clinical relevance of transforming growth factor-beta 1 in the urine of patients with hepatocellular carcinoma. Medicine (Baltimore) 76:213–226
Abou-Shady M, Baer HU, Friess H, Berberat P, Zimmermann A, Graber H, Gold LI et al (1999) Transforming growth factor betas and their signaling receptors in human hepatocellular carcinoma. Am J Surg 177:209–215
Giannelli G, Bergamini C, Fransvea E, Sgarra C, Antonaci S (2005) Laminin-5 with transforming growth factor-beta1 induces epithelial to mesenchymal transition in hepatocellular carcinoma. Gastroenterology 129:1375–1383
Fransvea E, Angelotti U, Antonaci S, Giannelli G (2008) Blocking transforming growth factor-beta up-regulates E-cadherin and reduces migration and invasion of hepatocellular carcinoma cells. Hepatology 47:1557–1566
Fransvea E, Mazzocca A, Antonaci S, Giannelli G (2009) Targeting transforming growth factor (TGF)-betaRI inhibits activation of beta1 integrin and blocks vascular invasion in hepatocellular carcinoma. Hepatology 49:839–850
Bergamini C, Sgarra C, Trerotoli P, Lupo L, Azzariti A, Antonaci S, Giannelli G (2007) Laminin-5 stimulates hepatocellular carcinoma growth through a different function of alpha6beta4 and alpha3beta1 integrins. Hepatology 46:1801–1809
Giannelli G, Bergamini C, Fransvea E, Marinosci F, Quaranta V, Antonaci S (2001) Human Hepatocellular Carcinoma (HCC) cells require both alpha3 beta1 integrin and matrix metalloproteinases activity for migration and invasion. Lab Invest 81:613–627
Mazzocca A, Fransvea E, Lavezzari G, Antonaci S, Giannelli G (2009) Inhibition of transforming growth factor beta receptor I kinase blocks hepatocellular carcinoma growth through neo-angiogenesis regulation. Hepatology 50:1140–1151
Liotta LA, Espina V, Mehta AI, Calvert V, Rosenblatt K, Geho D, Munson PJ et al (2003) Protein microarrays: meeting analytical challenges for clinical applications. Cancer Cell 3:317–325
Wulfkuhle JD, Aquino JA, Calvert VS, Fishman DA, Coukos G, Liotta LA, Petricoin EF, II I (2003) Signal pathway profiling of ovarian cancer from human tissue specimens using reverse-phase protein microarrays. Proteomics 3:2085–2090
Melisi D, Ishiyama S, Sclabas GM, Fleming JB, Xia Q, Tortora G, Abbruzzese JL, Chiao PJ (2008) LY2109761, a novel transforming growth factor beta receptor type I and type II dual inhibitor, as a therapeutic approach to suppressing pancreatic cancer metastasis. Mol Cancer Ther 7:829–840
Wu L, Derynck R (2009) Essential role of TGF-beta signaling in glucose-induced cell hypertrophy. Dev Cell 17:35–48
Li HY, McMillen WT, Heap CR, McCann DJ, Yan L, Campbell RM, Mundla SR et al (2008) Optimization of a dihydropyrrolopyrazole series of transforming growth factor-beta type I receptor kinase domain inhibitors: discovery of an orally bioavailable transforming growth factor-beta receptor type I inhibitor as antitumor agent. J Med Chem 51:2302–2306
Acknowledgments
We are grateful to Dr. Michael Lahn (Eli Lilly and Company) for critical review of the manuscript. This work was supported by the Italian Association Cancer Research (AIRC) grant to GG number 202240GNN.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fransvea, E., Mazzocca, A., Santamato, A. et al. Kinase activation profile associated with TGF-β-dependent migration of HCC cells: a preclinical study. Cancer Chemother Pharmacol 68, 79–86 (2011). https://doi.org/10.1007/s00280-010-1459-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-010-1459-x