Abstract
Purpose
To detect the variable relationship between sciatic nerve and piriformis muscle and make surgeons aware of certain anatomical features of each variation that may be useful for the surgical treatment of the piriformis syndrome.
Methods
The gluteal region of 147 Caucasian cadavers (294 limbs) was dissected. The anatomical relationship between the sciatic nerve and the piriformis muscle was recorded and classified according to the Beaton and Anson classification. The literature was reviewed to summarize the incidence of each variation.
Results
The sciatic nerve and piriformis muscle relationship followed the typical anatomical pattern in 275 limbs (93.6 %). In 12 limbs (4.1 %) the common peroneal nerve passed through and the tibial nerve below a double piriformis. In one limb (0.3 %) the common peroneal nerve coursed superior and the tibial nerve below the piriformis. In one limb (0.3 %) both nerves penetrated the piriformis. In one limb (0.3 %) both nerves passed above the piriformis. Four limbs (1.4 %) presented non-classified anatomical variations. When a double piriformis muscle was present, two different arrangements of the two heads were observed.
Conclusions
Anatomical variations of the sciatic nerve around the piriformis muscle were present in 6.4 % of the limbs examined. When dissection of the entire piriformis is necessary for adequate sciatic nerve decompression, the surgeon should explore for the possible existence of a second tendon, which may be found either inferior or deep to the first one. Some rare, unclassified variations of the sciatic nerve should be expected during surgical intervention of the region.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Piriformis syndrome (PS) is an often overlooked cause of buttock and leg pain [15, 17]. It is a poorly understood clinical syndrome, which is thought to be a nerve entrapment syndrome characterized by the entrapment of the sciatic nerve (SN) by piriformis muscle (PM). PS accounts for approximately 6–8 % of sciatica [21]. Proposed mechanisms for PS include overuse and hypertrophy of the PM [6, 32]. It has been found that during flexion adduction and internal rotation of the hip joint the infrapiriformis foramen becomes narrower [19]. Moreover, the PM can be spastic, inflamed or it can be seen as myofascial syndrome [43]. Anatomical variations between SN and PM constitute another cause of the syndrome [12, 25], while a history of direct trauma to the pelvis or buttock is usually elicited in approximately 50 % of cases [31]. Some authors use PS for any sciatica arising from nerve trunk compression, regardless of PM involvement, such as compression by osteophytes, hematomas, myositis ossificans, pseudo-aneurysms, endometriotic cysts in the pelvis and prolonged external pressure [23, 43]. Fractures of the femoral neck and of the ischial tuberosity as well as hip arthroplasty may also cause sciatica [23]. Such cases have been called secondary PS or pseudosciatica [23].
The three commonest symptoms and signs of PS include buttock pain, pain aggravated by sitting and external tenderness near the greater sciatic notch [22]. The management of PS includes conservative measures, local injections and operative neurolysis along with dissection of the PM, or part of it, in recalcitrant cases or when there is a documented PM anatomical variation [9, 38]. Such anatomical variations in the relationship between the PM and the SN have been intraoperatively recognized as a cause of PS [12, 24–26].
The possible anatomical relationships between SN and PM were classified by Beaton and Anson [7] in 1937 into six types. Several authors have described the incidence, topographic anatomy and potential clinical significance of those six morphologic types [9, 20, 34, 39]. However, the literature is scarce regarding certain anatomical features of these variations that could affect the outcome of PM dissection during surgical decompression of the SN. Purpose of the present study was to detect the variable relationship between SN and PM in a large sample of cadavers, review the relevant literature summarizing the reported incidence of the variations and make surgeons aware of those anatomical features of each variation which may be useful for the surgical treatment of PS.
Materials and methods
The gluteal region of 147 formalin embalmed Caucasian cadavers (294 limbs) was dissected by means of classical anatomical dissection during a 15-year period, both for tutorial and research purposes. In particular, the gluteus maximus and medius were dissected vertically and the muscular flaps were reflected laterally, in order to obtain a clear view of the PM and the SN. The anatomical relationship between the SN and the PM was detected and classified according to the Beaton and Anson classification; type I, undivided SN below the PM; type II, one division of SN through and the other below the PM; type III, one division above and the other below the PM; type IV, undivided SN through the PM; type V, one division through and the other above the PM; type VI, undivided nerve above the PM (Fig. 1) [7]. From the medical history of the cadavers, no data concerning the presence of symptoms indicative of PS during their lives were available. The cadavers utilized for our research were adult males and females and demonstrated no evidence of previous pathologic disorders, traumatic conditions or surgical interventions in the gluteal region. Special attention was paid to distinguish any anatomical features that might be useful for the surgical treatment of the PS. The literature was reviewed to document the reported incidence of the anatomical variations in the relationship between SN and PM.
Results
The SN and PM relationship followed the typical anatomical pattern in 275 limbs (93.6 %). In 12 out of the 294 (4.1 %) studied limbs the SN was divided and the common peroneal nerve (CPN) passed between the two heads of a double PM, while the tibial nerve (TN) passed below the PM. The 12 limbs belonged to four male and five female cadavers. The variation was bilateral in three cadavers. In these 12 limbs, we observed two different arrangements of the two heads of the double PM. Namely, in seven limbs (two right and five left) the second muscle belly was located inferior to the first one (Fig. 2a), while in the remaining five limbs (three right and two left) the second muscle belly was located deep to the other one (Fig. 2b). In the right side of a female cadaver (0.3 %), the SN was divided and the CPN passed superior to the PM and afterwards it was reunited with the TN that coursed below the PM (Fig. 3). In the left limb of a female cadaver (0.3 %), the SN was divided and both nerves passed between the superficial and the deep belly of the PM and distal to the muscle they merged into a single nerve (Fig. 4). In the right side of a male cadaver (0.3 %) the SN was divided and both nerves passed above the PM (Fig. 5).
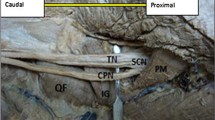
Type II variation per Beaton and Anson: the sciatic nerve was divided and the common peroneal nerve (P) passed between the two heads of a double piriformis muscle, while the tibial nerve (T) passed below the muscle. a Right subgluteal region. The piriformis muscle had two heads, one superior (1) and one inferior (2). b Left subgluteal region. The piriformis muscle had two heads, one superficial (1) and one deep (2)
Right subgluteal region having a type III variation per Beaton and Anson: the sciatic nerve was divided and the common peroneal nerve (P) passed superior to the piriformis muscle (M) and afterwards it was reunited with the tibial nerve (T) that passed below the muscle. The sciatic nerve was painted (asterisk gluteus maximus fibers)
Four limbs (1.4 %) displayed rare anatomical variations, which are not described in the Beaton and Anson classification. In the right side of a male cadaver, the PM had three muscle bellies. The SN was divided and the CPN passed between the superficial and the intermediate muscle belly, whereas the deep muscle belly passed through a branch of the TN (Fig. 6). In the right side of another male cadaver, the SN was divided and the CPN passed between the superficial and the deep muscle belly of a double PM, while the TN passed below the muscle. The inferior gluteal vein was also located below the PM, but it passed through a branch of the TN (Fig. 7). In both sides of a male cadaver, the SN was running below the PM. However, there was a bilateral supernumerary muscle located just superior to the PM, in the suprapiriform foramen. This muscle was originated with a well-formed fibrous tendon from the posterior rim of the ilium bone, inferior to the posterior inferior iliac spine. It ran parallel to the PM, just below the superior gluteal neurovascular structures and appeared a short and broad fibrous insertion into the greater trochanter (Fig. 8). Results are summarized in Table 1.
Right subgluteal region having an unclassified variation: the piriformis muscle had three muscle bellies. The sciatic nerve was divided and the common peroneal nerve (P) passed between the superficial (1) and the intermediate muscle belly (2), while the deep muscle belly (3) passed through a branch (modified) of the tibial nerve (T)
Right subgluteal region having an unclassified variation: the sciatic nerve was divided and the common peroneal nerve (P) passed between the superficial (1) and the deep muscle belly (2) of a double piriformis muscle, while the tibial nerve (T) passed below the muscle. The inferior gluteal vein (asterisk) was also located below the muscle, but it passed through a branch (modified) of the tibial nerve
Discussion
The etiology of PS is multifactorial with a history of direct trauma, which is usually not dramatic and may occur several months before the initial presentation of symptoms, being involved in approximately 50 % of cases [11, 31]. Anatomical variations in the relationship between PM and SN have been intraoperatively recognized as another cause of PS [12, 24–26]. However, an individual having such a variation may also be asymptomatic [11]. These variations were classified by Beaton and Anson [7] into six types (Fig. 1). In the literature, the reported frequency of the variations ranges between 1.5 and 35.8 % of the limbs (Table 2) [1, 2, 5, 7–10, 13, 14, 16, 18, 20, 27–30, 33–37, 40–42], while in our series variations were present in 10.2 % of the cadavers or in 6.4 % of the limbs studied. Several authors have discussed the incidence and described the potential clinical significance of those six morphologic types for PS [9, 20, 34, 39]. Thus, the present study focused on those anatomical features of each variation that could affect the outcome of PM dissection during surgical decompression of the SN, as well is in anatomical variations of this region which are not included in the classification.
The most common variation is the course of the CPN between the two heads of a double PF muscle (type II) which may be present in 13.7 % of the limbs according to the literature [39], while in the present study it was found in 6.1 % of the cadavers or in 4.1 % of the limbs studied. We observed that a double PM presented always two distinct tendons, even when the muscle bellies had a common origin. For surgical treatment of a PS due to a double PM, dissection of the lower head is suggested in order to restitute its normal relationship to the SN. However, if nerve entrapment is still noted intra-operatively with the hip in flexion and internal rotation, dissection of both PM tendons is required [12]. The typical illustration of the type II variation in Beaton and Anson classification, which is reproduced in many studies, demonstrates that there is a superior and an inferior muscle belly of the PM [7]. Furthermore, Beaton and Anson mentioned that in those cases the superior portion of the PM slightly overlapped the inferior one, except from one case of a PM having a superior portion that covered three-fourths of the inferior one. In the present study, we observed that the superior–inferior arrangement of the two bellies of the PM was present only in seven out of the 12 limbs having a type II variation (Fig. 2a). In the remaining five specimens there was a superficial-deep arrangement of the two muscle bellies (Fig. 2b). In such cases, a thick CPN passing below the superficial head of the PM may be considered as normal by the surgeon and lead to incomplete decompression of the SN.
Anatomical variations classified as type III, IV, V or VI may be found in only 3.2 % of the limbs according to the literature. In the present study, 2.4 % of the cadavers or 1 % of the limbs studied had such a rare variation. Although the sample was large, the anatomical pattern of type V was not found in any limb. Absence of type V and VI is common in most literature studies as it is shown in the Table 2. Nevertheless, though rare, surgeons should bear in mind that the SN or part of it may pass above the PM, through the suprapiriform foramen.
Apart from the six anatomical variations described in the Beaton and Anson classification, we noticed four unclassified variations of the SN around the PM. The unclassified variations included a PM having three fascicles with one of them passing through a branch of the TN, a bilateral supernumerary muscle in the suprapiriform foramen and an inferior gluteal vein passing through a branch of the TN. It is very interesting that both the third fascicle of the PM and the inferior gluteal vein were passing through a branch of the TN, whereas the CPN is usually involved in the variations of the subgluteal region. The supernumerary muscle that was found in the left suprapiriform foramen seems to compromise the available space for the SN emersion through the greater sciatic foramen.
In the literature there are also reports of some other anatomical variations along the course of the SN in the subgluteal region, which are not included in the Beaton and Anson classification. Jawish et al. [24] noted intraoperatively a transverse fibrous band which was located inferior to the PM and compressed the SN, as well as a case of SN impingement by the PM and the sacrosciatic ligament. A case of a superior gemellus muscle passing between the CPN and the TN [4] and another case of a double PM associated with a double superior gemellus and a SN dividing high, where the CPN passing through the double PM and the TN between the lower PM and the upper superior gemellus, have been also reported in the literature [3]. Beaton and Anson found a case of a double PM muscle where the superficial belly passed through a branch of the L4–L5 nerve roots. The S1–S3 nerves passed between the two bellies of the PM and reunited with the L4–L5 trunk, distal to the PM, to form the SN. This variation was not included in the classification [7]. All these findings imply that during surgical intervention of SN entrapment by the PM not only the variations described in the Beaton and Anson classification should be expected, but also rare variations of the SN around the PM may appear. In such cases conventional PM dissection may not be enough for SN decompression.
The course of SN or part of it through the PM has been also implicated in SN palsy after total hip arthroplasty. Specifically, tenotomy and retraction of the external rotators during posterior approach of the hip joint may cause compression of the SN or part of it, as the PM retracts medially [36].
Conclusions
Anatomical variations of the SN around the PM are not rare since they were present in 6.4 % of the limbs examined in the present study. They constitute a recognized cause of PS and dissection of PM or part of it releases the SN in such cases. When dissection of whole PM is necessary, the surgeon should explore for the possible existence of a second tendon, which may be located either inferior or deep to the first one. Apart from the six anatomical variations described in the Beaton and Anson classification, also some rare variations of the SN around the PM should be expected during surgical intervention of the region.
References
Agur AMR, Dalley AF II (2005) Grant’s Atlas of Anatomy, 10th edn. Lippincott Williams & Wilkins, Baltimore
Anson BJ, McVay CB (1984) Surgical Anatomy, 6th edn. W.B. Saunders Company, Philadelphia
Arifoglu Y, Sargon MF, Tanyeli E, Yazar F (1997) Double superior gemellus together with double piriformis and high division of the sciatic nerve. Surg Radiol Anat 19:407–408
Babinski MA, Machado FA, Costa WS (2003) A rare variation in the high division of the sciatic nerve surrounding the superior gemellus muscle. Eur J Morphol 41:41–42
Bardeen KV (1901) A statistical study of the variations in the formation and position of the lumbosacral plexus in man. Anat Anz 9:209–238
Barton PM (1991) Piriformis syndrome: a rational approach to management. Pain 47:345–352
Beaton LE, Anson BJ (1937) The relation of the sciatic nerve and of its subdivisions to the piriformis muscle. Anat Rec 70:1–5
Beaton LE, Anson BJ (1938) The sciatic nerve and the piriformis muscle: their interrelation a possible cause of coccygodynia. J Bone Jt Surg 20:686–688
Benzon HT, Katz JA, Benzon HA, Iqbal MS (2003) Piriformis syndrome: anatomic considerations, a new injection technique, and a review of the literature. Anesthesiology 98:1442–1448
Brooks JBB, Silva CAC, Soares SA et al (2011) Anatomical variations of the sciatic nerve in a group of Brazilian cadavers. Rev Dor Sao Paulo 12:332–336
Cassidy L, Walters A, Bubb K et al (2012) Piriformis syndrome: implications of anatomical variations, diagnostic techniques, and treatment options. Surg Radiol Anat 34:479–486
Chen WS, Wan YL (1992) Sciatica caused by piriformis muscle syndrome: report of two cases. J Formos Med Assoc 91:647–650
Chiba S, Ishibashi Y, Kasai T (1994) Perforation of dorsal branches of the sacral nerve plexus through the piriformis muscle and its relation to changes of segmental arrangements of the vertebral column and others. Kaibogaku Zasshi 69:281–305
Chiba S (1992) Multiple positional relationships of nerves arising from the sacral plexus to the piriformis muscle in humans. Kaibogaku Zasshi 67:691–724
Filler AG, Haynes J, Jordan SE et al (2005) Sciatica of nondisc origin and piriformis syndrome: diagnosis by magnetic resonance neurography and interventional magnetic resonance imaging with outcome study of resulting treatment. J Neurosurg 2:99–115
Fishman LM, Dombi GW, Michaelsen C et al (2002) Piriformis syndrome: diagnosis, treatment, and outcome: a 10-year study. Arch Phys Med Rehabil 83:295–301
Fishman LM, Schaefer MP (2003) The PS is underdiagnosed. Muscle Nerve 28:626–629
Fukumoto N (1935) Uber den M. piriformis und N. ischiadicus von Japanern. Fukuoka Acta Medica 28:39
Guvencer M, Akyer P, Iyem C et al (2008) Anatomic considerations and the relationship between the piriformis muscle and the sciatic nerve. Surg Radiol Anat 30:467–474
Guvencer M, Iyem C, Akyer P et al (2009) Variations in the high division of the sciatic nerve and relationship between the sciatic nerve and the piriformis. Turk Neurosurg 19:139–144
Hallin RP (1983) Sciatic pain and the piriformis muscle. Postgrad Med 74:69–72
Hopayian K (2012) The clinical features of the piriformis syndrome. Surg Radiol Anat 34:671
Hopayian K, Song F, Riera R, Sambandan S (2010) The clinical features of the piriformis syndrome: a systematic review. Eur Spine J 19:2095–2109
Jawish RM, Assoum HA, Khamis CF (2010) Anatomical, clinical and electrical observations in piriformis syndrome. J Orthop Surg Res 5:3
Kosukegawa I, Yoshimoto M, Isogai S et al (2006) Piriformis syndrome resulting from a rare anatomic variation. Spine (Phila Pa 1976) 31:664–666
Kouvalchouk J, Bonnet JM, de Mondenard JP (1996) Piriformis syndrome. Apropos of 4 cases treated by surgery and review of the literature. Rev Chir Orthop Reparatrice Appar Mot 82:647–657
Lee CS, Tsai TL (1974) The relation of the sciatic nerve to the piriformis muscle. Taiwan Yi Xue Hui Za Zhi 73:75–80
Ming-Tzu P (1941) The relation of the sciatic nerve to the piriformis muscle in the Chinese. Am J Phys Anthropol 28:375
Misra BD (1954) The relations of the sciatic nerve to the piriformis in Indian cadavers. J Anat Soc India 3:28–33
Nizankowski C, Slociak J, Szybejko J (1972) Variations in the anatomy of the sciatic nerve in man. Folia Morphol (Warsz) 31:507–513
Pace JB, Nagle D (1976) Piriformis syndrome. West J Med 124:435–439
Papadopoulos EC, Khan SN (2004) Piriformis syndrome and low back pain: a new classification and review of the literature. Orthop Clin N Am 35:65–71
Parsons FG, Keith A (1896) Sixth annual report of the committee of collective investigation of the anatomical society of Great Britain and Ireland, 1895–96. J Anat Physiol 31:31–44
Patel S, Shah M, Vora R et al (2011) A variation in the high division of the sciatic nerve and its relation with piriformis muscle. Natl J Res 1:27–30
Pecina M (1979) Contribution to the etiological explanation of the piriformis syndrome. Acta Anat (Basel) 105:181–187
Pokorny D, Jahoda D, Veigl D et al (2006) Topographic variations of the relationship of the sciatic nerve and the piriformis muscle and its relevance to palsy after total hip arthroplasty. Surg Radiol Anat 28:88–91
Pokorny D, Sosna A, Veigl D, Jahoda D (1998) Anatomical variability of the relation of pelvitrochanteric muscles and sciatic nerve. Acta Chir Orthop Traumatol Cech 65:336–339
Skandalakis JE (2004) Surgical anatomy: the embryologic and anatomic basis of modern surgery. Paschalidis Medical Publications, Athens, p 1605
Smoll NR (2010) Variations of the piriformis and sciatic nerve with clinical consequence: a review. Clin Anat 23:8–17
Trotter M (1932) The relation of the sciatic nerve to the piriformis muscle in American Whites and Negroes. Anat Rec 52:321–323
Ugrenovic S, Jovanovic I, Krstic V et al (2005) The level of the sciatic nerve division and its relations to the piriform muscle. Vojnosanit Pregl 62:45–49
Uluutku MH, Kurtoglu Z (1999) Variations of nerves located in deep gluteal region. Okajimas Folia Anat Jpn 76:273–276
Windisch G, Braun EM, Anderhuber F (2007) Piriformis muscle: clinical anatomy and consideration of the piriformis syndrome. Surg Radiol Anat 29:37–45
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Natsis, K., Totlis, T., Konstantinidis, G.A. et al. Anatomical variations between the sciatic nerve and the piriformis muscle: a contribution to surgical anatomy in piriformis syndrome. Surg Radiol Anat 36, 273–280 (2014). https://doi.org/10.1007/s00276-013-1180-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00276-013-1180-7