Abstract
Pathology of the hip abductor muscles and their associated tendons is implicated in the aetiology of lateral hip pain (LHP). Muscle atrophy is an important factor to consider in the diagnosis of this condition as it could result in reduced muscle volume and associated decreases in strength.
Purpose
(1) To estimate the volumes of the gluteus medius (GMed), gluteus minimus (GMin) and tensor fascia lata (TFL) muscles, and (2) to examine pathological changes of the soft tissues in the vicinity of the hip joint, in women with and without LHP.
Methods
Twenty female participants (10 with LHP and 10 age-matched controls) underwent magnetic resonance imaging. Two radiologists reviewed the images for signs of pathological changes. Hip abductor muscle volumes were estimated using cross-sectional areas and Cavalieri’s method. Differences in volume between sides, study groups and the three muscles were assessed.
Results
The volume of GMed was the largest (292.5 ± 33.3 cm3), followed by GMin (82.1 ± 12.1 cm3), then TFL (49.7 ± 18.9 cm3). No differences were evident in the volumes of the hip abductor muscles in individuals with LHP when compared to age- and sex-matched controls (GMed, p = 0.30; GMin, p = 0.40; TFL, p = 0.90). Pathology of the soft tissues was not specific to the symptomatic hips.
Conclusions
Novel muscle volume data are presented for GMed, GMin and TFL in the context of LHP. Further research is needed to determine if symptom severity and duration have an impact on the extent of muscle atrophy in this population.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Lateral hip pain (LHP) is a problem most prevalent in females aged between 40 and 70 years [2, 6, 25]. This non-specific condition is characterized by pain and tenderness over the greater trochanter of the femur [4, 25] and individuals report intermittent, chronic pain, which is often recurrent over a number of years [33, 35].
Due to the complexity and variety of symptoms in patients with LHP, conflict exists regarding the exact source(s) of pain. In the past, trochanteric bursitis has been considered the primary cause [2, 33], but pathology of the hip abductor muscles [specifically, gluteus medius (GMed) and gluteus minimus (GMin)] and their associated tendons has recently emerged as an indicator of symptoms [4, 6, 9, 25]. In addition to tendon pathology, individuals with LHP may present with atrophy of these muscles [6, 9, 30, 35]. The presence of atrophy is likely to alter muscle volumes. This is significant as force production depends on physiological cross-sectional area, which is linked to muscle volume [14]. A reduction in hip abductor muscle volume in individuals with LHP could impact on function and pelvic stability. It has been shown that specific strengthening of the hip abductor muscles is effective in the treatment of LHP [16, 32], but the probable associated changes in muscle volume have not been quantified. Currently, there is limited data available on the muscle volumes of GMed, GMin and tensor fascia lata (TFL), calculated from healthy, living individuals [17, 18, 21–23, 31], and no muscle volume data have been reported for persons with LHP. Therefore, the primary aim of this study was to estimate the volumes of GMed, GMin and TFL from magnetic resonance images of individuals with LHP and compare them to age- and sex-matched controls. Second, this research aimed to identify and report associated pathology of the soft tissues surrounding the hip joint.
Methods
Ethical approval was granted by the Lower South Regional Ethics Committee. Twenty female participants were recruited for this study; 10 with clinically diagnosed unilateral LHP (duration ≥3 months), and 10 age-matched (within 6 months) controls, with no history of hip pain in the preceding 12 months. A qualified health professional confirmed the presence of LHP. This was confirmed by the reproduction of pain with palpation over the greater trochanter, and resisted hip abduction [2]. Individuals whose symptoms were thought to be referred from the lumbar spine, who had received a steroid injection within the last 6 months, or had any contraindication to magnetic resonance imaging were excluded from the study.
Participants with LHP filled in a questionnaire regarding symptoms, work load and daily activities. They also marked the site(s) of their pain on a body chart and completed a visual analogue scale to quantify pain intensity. A similar version of the questionnaire (excluding pain site and intensity) was completed by the control participants ("Appendix").
The following definitions have been used for the purpose of comparing hips from each study group: SLHP, the symptomatic hip of an individual with LHP; ALHP, the asymptomatic hip of an individual with LHP; SControl, the hip of a control participant matched to the same side as the symptomatic hip of the age-matched individual with LHP; AControl, the hip of a control participant matched to the same side as the asymptomatic hip of the age-matched individual with LHP.
Magnetic resonance imaging
A 1.5-T Phillips Achieva (Software Release 2.5.3.4, Philips Healthcare, Massachusetts, USA) was used for all scans. Each hip was imaged separately with the participants positioned supine, using a sensitivity encoding cardiac coil placed anteriorly over the hip. The anatomical area covered for each scan was the highest point of the iliac crest superiorly, to the base of the lesser trochanter inferiorly and from the anterior to posterior skin surfaces of the participant. A whole body reference scan was completed for calibration.
The magnetic resonance images that were obtained for each participant are outlined in Table 1. T1-weighted scans were used for assessing hip abductor muscle volume, and T2-weighted scans for diagnoses of pathology.
Muscle volume estimation
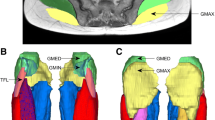
Using OsiriX (Antoine Rosset, 2003–2009, v.2.7.5) the circumferences of GMed, GMin and TFL were traced. Areas of atrophy were excluded, with the adipose-infiltrated tissue appearing as a T1-high-intensity signal in comparison to muscle [15]. On the completion of a trace, the corresponding cross-sectional area was automatically calculated and, using Cavalieri’s method, the total volume of each muscle was estimated. After a random choice of start point, therein and systematically [19], every third image was used for volumetric analysis. Data were then applied to the formula: volume (cm³) = ΣAd, where A is the cross-sectional area of the muscle in each selected magnetic resonance slice (cm2) and d is the distance between images (15 mm) [19].
Statistics
Mean volume and standard deviation for each muscle were calculated for both study groups. Differences between (a) left and right control participant hips, and (b) between study groups were calculated (Student’s t test), as well as differences in volume between the three muscles (one-way analysis of variance followed by Bonferroni’s post hoc). The muscle perimeter tracings from five participants (randomly selected) were measured three times, 1 week apart, to test intra-rater reliability which was determined using a two-way mixed, intra-class correlation coefficient statistical test (Cronbach’s alpha model). Significance was set at p ≤ 0.05.
Radiological reporting
Two fellowship trained musculoskeletal radiologists (GM, experience 10 years; MR, experience 5 years), both blinded to participant information, independently analyzed the images using Digital Imaging and Communication in Medicine imaging software; disparities were resolved by consensus. Trochanteric bursa location was defined as lateral to the greater trochanter, deep to the fascia lata and gluteus maximus, but superficial to GMed [12]. Sub-GMed bursal location was defined as deep to the lateral GMed tendon, and sub-GMin as beneath the GMin tendon, medial to its superior attachment onto the anterior facet of the greater trochanter [29]. Bursitis was characterized using the definitions of Woodley et al. [35]. The bursa was measured as the maximum length and width in the coronal plane. For example, if the length measured 3 cm and width 0.3 cm on one image and the length 2.5 and 0.5 cm on another, then the dimensions would be 3 × 0.5 cm. If a bursa measured more than 3 cm proximo-distally (cranio-caudally), and was greater than 0.3 cm in thickness (mediolateral dimension) adjacent to the greater trochanter, it was considered pathological [10].
Pathology of the gluteal tendons was classified as either a tear or tendinosis. A tear was identified if a tendon displayed hyperintensive signal on T2-weighted scans, accompanied by disruption of fibers [6, 25]. In addition, tendon elongation, a possible indicator of gluteal muscle atrophy or tendon tears [9]), was recorded if its proximo-distal (cranio-caudal) length was greater than 2 cm on any coronal slice and was in general, longer than in the contralateral hip. Tendinosis was defined as an area of hyperintensity in the absence of fiber disruption [25]. Cases where it was not possible to differentiate the type of tendon pathology were noted. The location of GMed and GMin tendon pathology was classified as either the insertion into the greater trochanter, within the musculotendinous junction or in the middle of the free tendon. For GMed, this was applicable for the posterior and the lateral parts of the tendon [13].
As observed on the magnetic resonance images, if a muscle appeared to be less than 25 % of the volume of the same muscle on the contralateral side, it was considered atrophic [9]. If atrophy was observed, the amount of fatty infiltration relative to the amount of muscle tissue was qualified [30]. Pathologies of secondary interest, such as osteoarthritic changes and labral tears were also assessed [35].
Results
The mean age of participants was 55 years (range 43–68 years) for both groups. Mean body mass index (24.8 ± 3.6 kg/m2) for the LHP group was not different from the control group (23.9 ± 4.1 kg/m2, p = 0.15), and no differences were evident for mean height (LHP, 1.63 m; Controls, 1.66 m; p = 0.335). Daily workload ranged between “sedentary” and “moderate” levels of activity for the women, with no obvious difference between groups. The mean visual analogue scale score was 3.8 and a summary of the pain drawings for participants with LHP can be found in Fig. 1. Pain was commonly noted over the lateral aspect of the hip, in the region of the greater trochanter. Some participants also reported pain in the low back and buttock regions, or referring down the lateral thigh to the knee joint.
Muscle volume
The muscle circumference tracing method for volumetric analysis was accurate with excellent intra-class correlation coefficients (range 0.979–0.996 for GMed; (0.646) 0.886–0.995 for GMin; 0.950–0.994 for TFL) [26]. On observation, fatty infiltration was common within the hip abductor muscles in both LHP and control participants. Adipose tissue was mostly feathery and diffuse in appearance (Fig. 2a, c) and was primarily located within GMin. In two participants (one LHP, one control), adipose was “en bloc” in appearance (Fig. 2b, d).
The appearance of two distinct forms of adipose tissue infiltration within GMin of two asymptomatic hips on axial, T1-weighted, fat saturated MR images. a Coronal image showing the diffuse appearance of atrophy (arrows). b Coronal image showing the en bloc appearance of atrophy (arrows). c Axial image showing the diffuse appearance of atrophy (arrows). d Axial image showing the en bloc appearance of atrophy (arrowheads). GMed gluteus medius, GMin gluteus minimus, GMax gluteus maximus, MR magnetic resonance
Control participant muscle volumes
No difference in muscle volume was observed between the left and right sides of the control participants for GMed (p = 0.40), GMin (p = 0.90) or TFL (p = 0.70). Therefore, data from both sides were combined. For the three muscles, GMed was the greatest in volume (292.5 ± 33.3 cm3), followed by GMin (82.1 ± 12.1 cm3), then TFL (49.7 ± 18.9 cm3). Differences in mean volumes between all three muscles were statistically significant (p < 0.001).
Side to side difference in muscle volumes between groups
There were no differences in mean volumes of SControl and AControl hips, for any muscle (Table 2) (GMed, p = 0.6; GMin and TFL, p = 0.90). Similarly, the mean muscle volumes in ALHP hips were not significantly different to those in SLHP hips (GMed, p = 0.60; GMin, p = 0.40; TFL, p = 0.97).
Differences in muscle volumes between groups
The mean muscle volumes from SControl hips were greater than SLHP hips. However, there were no significant differences between study groups for GMed (p = 0.30), GMin (p = 0.40) or TFL (p = 0.90).
Radiological diagnoses
Radiological evidence of pathology was evident in SLHP, ALHP and control hips with no evidence of pathology detected in four hips (SLHP, 1; ALHP, 1; control, 2). For control and SLHP hips, the most common diagnosis was isolated bursitis (25 and 40 %, respectively) and for ALHP hips, isolated bursitis (30 %) and isolated osteoarthritis (30 %) were most frequently observed. Different pathologies often occurred concurrently (Fig. 3). However, the combination of bursitis and tendon pathology was only observed in SLHP hips (20 %, 2). Muscle atrophy was not commonly reported (5 %, 2).
Combinations of radiological diagnoses of all study hips. This figure shows the different combinations of pathologies observed, and the percentage of cases diagnosed in all three study groups (SLHP, n = 10; ALHP, n = 10; control, n = 20). ALHP asymptomatic lateral hip pain hip, OA osteoarthritis, SLHP symptomatic lateral hip pain hip, TP tendon pathology
Bursitis was diagnosed in 67.5 % (27) of the individual hips imaged, nine SLHP, six ALHP and 12 control hips. Isolated bursitis was found in 12 hips and, although most commonly observed in SLHP hips (40 %, 4), it also occurred in ALHP (30 %, 3) and control (25 %, 5) hips. In 77.8 % (21) of LHP participants presenting with trochanteric bursitis, the proximo-distal (cranio-caudal) size of the bursa was larger in the SLHP hips compared to the ALHP hips. In contrast, the mean thickness of the bursae in the SLHP hips was not significantly greater than that of the ALHP hips (Table 3).
Tendon pathology was observed in 22.5 % (9) of all hips (SLHP, 3; ALHP, 1; control, 5); tendinosis was most frequently diagnosed and only one tear was identified (SLHP). GMed tendon pathology was observed in the three SLHP hips, while GMin tendon pathology was observed in one SLHP hip. The distal attachment of the lateral GMed tendon was the primary site for pathology in this muscle; while for GMin, the distal attachments of the tendon and the middle of the tendon were both identified as sites for pathology.
The muscles in two hips (SLHP, 1; control, 1) were considered to be atrophied. GMin was affected in the control hip; both muscles were atrophied in the SLHP hip. Osteoarthritis was observed in just over half of all hips and was most prevalent in controls, but least prevalent in SLHP hips. The severity of osteoarthritis was classified as mild (17) to moderate (control, 4; ALHP, 1; SLHP, 1). Isolated osteoarthritis was not observed in symptomatic hips. Labral abnormalities were observed in 45 % (18), including four SLHP, five ALHP and nine control hips.
Discussion
The primary aim of this study was to estimate the volume of the hip abductor muscles in women with LHP and age- and sex-matched control participants. Second, we aimed to describe pathology of the surrounding soft tissues of the hip joint. We present novel data regarding the volumes of the hip abductor muscles specific to living, female individuals between the ages of 40 and 70 years, demonstrating that the mean muscle volume is 292.5 ± 33.3 cm3 for GMed, 82.1 ± 12.1 cm3 for GMin and 49.7 ± 18.9 cm3 for TFL. These values are comparable to those calculated previously, falling within the mean data range of several studies (GMed, 96.7–351.0 cm3; GMin, 47.5–102.5 cm3; TFL, 43.9–80.6 cm3) [17, 18, 21–23, 31, 34].
We hypothesized that there would be a reduction in hip abductor muscle volume in the symptomatic hips of women with LHP, but no differences were evident between groups. One reason for this outcome may be that in order to detect a reduction in muscle volume, symptoms must be of certain pain intensity and prolonged duration. Certainly, Grimaldi et al. [17, 18] found reduced muscle volumes of the hip abductor muscles in individuals with advanced hip joint osteoarthritis, but did not observe the same in individuals with mild and relatively short-term symptoms. Most participants with LHP in the current study had suffered from the condition for less than a year, and the mean pain score was relatively low. It is possible that individuals with long standing, moderate-to-severe pain may present with hip abductor muscle atrophy. However, unlike osteoarthritis whereby specific criteria are available to define severity [18], no similar standards exist for LHP. Although the sample size was similar to that used by Grimaldi et al. [17, 18], it is possible that the lack of association between muscle volume and LHP is a reflection of the relatively small number of participants. Therefore, a larger, preferably longitudinal, study is required to gain a better understanding of the pathological changes and the development of symptoms over time. It would also be interesting to assess the correlation between clinical changes in muscle strength and alterations in muscle volume, and to reassess these variables following implementation of a specific hip abductor-strengthening program. More details regarding physicality of daily workload and exercise also require further consideration.
The lack of radiological diagnoses of muscle atrophy was also surprising; this was identified in only one individual with LHP and one control participant, despite the appearance of high signal areas within many of the muscles. Previous accounts of atrophy in individuals with LHP have been recorded in up to 40 % of the cohort [9, 35]. It may be that radiological analyses need to incorporate more refined quantitative methods such as measuring the cross-sectional area of muscles and excluding areas of high signal intensity (adiposity) to determine the presence of atrophy, rather than relying on gross examination of images. Interestingly, the two individuals that were diagnosed with muscle atrophy were also two of the eldest participants, showing that aging is also likely to influence atrophy [11], and thus muscle volume, in the hip abductor muscles. A possible association between age and atrophy cannot be determined without conducting a larger study incorporating participants from various age brackets.
As determined radiologically, pathological changes in the vicinity of the hip joint were not specific to the symptomatic hips of individuals with LHP. In this small group of individuals, it is difficult to explain why some individuals, and not others, experienced symptoms of LHP despite similar radiological presentation of pathological changes. This outcome has been described in other studies [5, 9, 10, 35]. De Maeseneer et al. [10] suggest that the identification of a radiological signal change in the region of the trochanteric bursae does not automatically correlate to the clinical presentation of pain. Similarly, Cvitanic et al. [9] found that radiological evidence of trochanteric bursitis is not always confirmed by surgical examination, thereby giving high false-positive diagnoses. Such observations imply that the existing radiological markers of pathology do not necessarily correlate to symptoms of LHP. Furthermore, pathology does not usually occur in isolation, but typically one or more soft tissue changes are evident in the region.
Measurements of proximo-distal (cranio-caudal) dimension and thickness of bursae were used to quantify bursal distention and hence attempt to avoid confusion as to whether increased signal intensity on magnetic resonance images indicated bursitis. According to Blankenbaker et al. [5], a “normal” trochanteric bursa can be defined as having a proximo-distal (cranio-caudal) measurement of less than 3 cm, and a mediolateral thickness of less than 0.3 cm. Significant differences existed between the dimensions of the bursa within SLHP and ALHP hips, and accordingly, the mean measurements calculated for each study group are classified as being “abnormal” and “normal”, respectively. Similarly, although not significantly different, the mean thickness of the bursa in the SLHP hips was considered “abnormal” while that for the ALHP hips was “normal”. By quantifying bursal distension, one may be able to distinguish bursa pathology from normal collections of bursal fluid. Validation of these measurements in a larger cohort of individuals is warranted to determine whether these definitions may be used as radiological diagnostic tools for bursitis.
Little agreement is evident regarding the precise site of gluteal tendon pathology in relation to LHP, and the variability in the literature suggests that more than one site in a given muscle is prone to tendon pathology [4, 6, 9]. We were unable to confirm that the musculotendinous junction is disposed to tearing or tendinosis, but have identified the distal tendon attachments of GMed and GMin, and the middle of the free tendons as sites of pathology. This observation may be significant when considering tendon healing. Tears located at the enthesis or in the midst of the free tendon are of concern with regards to tissue repair, as these areas may be hypovascular (as is seen in the supraspinatus tendon of the rotator cuff muscles) [28], and are thus deprived of nutrients (derived from blood) essential for tissue healing promotion [24]. Therefore, a tendon tear identified at these locations may require intervention, such as surgery, for it to be repaired completely.
Osteoarthritis is often observed in patients with LHP [4, 25, 33, 35] and so, it is important to consider the influence of this pathology on the radiological interpretation. As the prevalence of osteoarthritis increases with age [3], the occurrence of this disease in asymptomatic joints is not surprising. Of more significance may be that in the SLHP hips, osteoarthritis was always accompanied by bursitis (n = 3, 30 %) and/or tendon pathology (n = 1, 10 %), while isolated cases of osteoarthritis were present in only Control or ALHP hips, corroborating the results from previous investigations [25, 35]. Although the association between osteoarthritis and tendon pathology and/or bursitis has not yet been formally investigated, these data suggest that osteoarthritis may be indirectly associated with the pathological changes underlying LHP [20].
Finally, there are a number of other pathological changes that may contribute to symptoms of LHP [2], and the most common in the present study was labral tears. This pathology has been previously identified in patients with LHP [7], but it is still unknown whether the tears are a source of pain, rather than pathology involving the hip abductor muscles. Two extensive reports on the radiological evidence of labral tears in asymptomatic hips suggest that this pathology alone does not lead to symptoms of LHP [8, 27]. Alternatively, the age range of the subjects recruited for the current investigation may explain the asymptomatic presentation of this pathology, as signal intensity in the labrum increases with age [1].
Limitations
Although presenting novel data on the muscle volume of the hip abductor muscles, the main limitation of the present study was the sample size, which is likely to have affected the outcomes. This study was also confined to examining the hip abductor muscles in a middle-aged, female population, which means the data are not likely to be applicable to males or younger individuals. However, as LHP is most prevalent in females between the ages of 40 and 70 years [33], it is believed that the choice of sample was appropriate. Due to the nature of this study, we were unable to validate the radiological diagnoses of pathology using surgical confirmation.
Conclusion
Specific to a living, middle-aged, female population, the current study provides accurate preliminary muscle volume data for GMed, GMin and TFL. No difference in muscle volume was found in the hip abductor muscles of females with mild to moderate LHP and age- and sex-matched controls. In addition, radiological diagnoses from magnetic resonance images did not correspond with the clinical presentation of LHP, such that pathologies were commonly found in both symptomatic, asymptomatic and control hips. A longitudinal study may provide further insight into the association of symptom severity, age, tendon and bursal pathology, and muscle atrophy.
Abbreviations
- AControl:
-
The hip of a control participant matched to the same side as the asymptomatic hip of the age-matched individual with LHP
- ALHP:
-
The asymptomatic hip of an individual with LHP
- GMed:
-
Gluteus medius
- GMin:
-
Gluteus minimus
- LHP:
-
Lateral hip pain
- SControl:
-
The hip of a control participant matched to the same side as the symptomatic hip of the age-matched individual with LHP
- SLHP:
-
The symptomatic hip of an individual with LHP
- TFL:
-
Tensor fascia lata
References
Abe I, Harada Y, Oinuma K, Kamikawa K, Kitahara H, Morita F, Moriya H (2000) Acetabular labrum: abnormal findings at MR imaging in symptomatic hips. Radiology 216:576–581
Anderson TP (1958) Trochanteric bursitis: diagnostic criteria and clinical significance. Arch Phys Med Rehabil 39:617–622
Anderson AS, Loeser RF (2010) Why is osteoarthritis an age-related disease? Best Pract Res Clin Rheumatol 24:15–26
Bird PA, Oakley SP, Shnier R, Kirkham BW (2001) Prospective evaluation of magnetic resonance imaging and physical examination findings in patients with greater trochanteric pain syndrome. Arthritis Rheum 44:2138–2145
Blankenbaker DG, Ulrick SR, Davis KW, De Smet AA, Haaland B, Fine JP (2008) Correlation of MRI findings with clinical findings of trochanteric pain syndrome. Skelet Radiol 37:903–909
Chung CB, Robertson JE, Cho GJ, Vaughan LM, Copp SN, Resnick D (1999) Gluteus medius tendon tears and avulsive injuries in elderly women: imaging findings in six patients. Am J Roentgenol 173:351–353
Connell DA, Bass C, Sykes CAJ, Young D, Edwards E (2003) Sonographic evaluation of gluteus medius and minimus tendinopathy. Eur Radiol 13:1339–1347
Cotton A, Boutry N, Demondion X, Paret C, Dewatre F, Liesse A, Chastanet P, Fontaine C (1998) Acetabular labrum: MRI in asymptomatic volunteers. J Comput Assist Tomogr 22:1–7
Cvitanic O, Henzie G, Skezas N, Lyons J, Minter J (2004) MRI diagnosis of tears of the hip abductor tendons (gluteus medius and gluteus minimus). Am J Roentgenol 182:137–143
De Maeseneer M, Gosselin R, De Ridder F, Shahabpour M, Vanderdood K (2008) MR imaging changes in the trochanteric area of asymptomatic individuals: a potential for misdiagnosis of pain in the trochanteric region. Eur J Radiol 72:480–482
Doherty TJ (2003) Invited review: aging and sarcopenia. J Appl Physiol 95:1717–1727
Dunn T, Heller CA, McCarthy SW, Dos Remedios C (2003) Anatomical study of the “trochanteric bursa”. Clin Anat 16:233–240
Duparc F, Thomine JM, Dujardin F, Durand C, Lukaziewicz M, Muller JM, Freger P (1997) Anatomic basis of the transgluteal approach to the hip-joint by anterior hemimyotomy of the gluteus medius. Surg Radiol Anat 19:61–67
Fukunaga T, Miyatani M, Tachi M, Kouzaki M, Kawakami Y, Kanehisa H (2001) Muscle volume is a major determinant of joint torque in humans. Acta Physiol Scand 172:249–255
Foster MA, Hutchison JMS, Mallard JR, Fuller M (1984) Nuclear magnetic resonance pulse sequence and discrimination of high- and low-fat tissues. Magn Reson Imaging 2:187–192
Furia JP, Rompe JD, Maffulli N (2009) Low-energy extracorporal shock wave therapy as a treatment for greater trochanteric pain syndrome. Am J Sports Med 37:1806–1813
Grimaldi A, Richardson C, Durbridge G, Donnelly W, Darnell R, Hides J (2009) The association between degenerative hip joint pathology and size of the gluteus maximus and tensor fascia lata muscles. Man Ther 14:611–617
Grimaldi A, Richardson C, Stanton W, Durbridge G, Donnelly W, Hides J (2009) The association between degenerative hip joint pathology and size of the gluteus medius, gluteus minimus and piriformis muscles. Man Ther 14:605–610
Gundersen HJG, Bendtsen TF, Korbo L, Marcussen N, Moller A, Nielsen K, Nyengaard JR, Pakkenberg B, Sorensen FB, Vesterby A, West MJ (1988) Some new, simple and efficient stereological methods and their use in pathological research and diagnosis. Act Pathologica Microbiologica Immunologica Scandinavica 96:379–394
Heller A (2003) Anatomy of the trochanteric bursae. Radiology 226:921–922
Inan M, Alkan A, Harma A, Ertem K (2005) Evaluation of the gluteus medius muscle after a pelvic support osteotomy to treat congenital dislocation of the hip. J Bone Joint Surg Am Vol 87:2246–2252
Jaegers S, Dantuma R, de Jongh HJ (1992) Three-dimensional reconstruction of the hip muscles on the basis of magnetic resonance images. Surg Radiol Anat 14:241–249
Jaegers AM, Arendzen JH, de Jongh HJ (1995) Changes in hip muscles after above-knee amputation. Clin Orthop Relat Res 319:276–284
Kajikawa Y, Morihara T, Sakamoto H, Matsuda K, Oshima Y, Yoshida A, Nagae M, Arai Y, Kawata M, Kubo T (2008) Platelet-rich plasma enhances the initial mobilization of circulation-derived cells for tendon healing. J Cell Physiol 215:837–845
Kingzett-Taylor A, Tirman PF, Feller J, McGann W, Prieto V, Wischer T, Cameron JA, Cvitanic O, Genant HK (1999) Tendinosis and tears of gluteus medius and minimus muscles as a cause of hip pain: MR imaging findings. Am J Roentgenol 173:1123–1126
Landis JR, Koch GG (1977) Measurement of observer agreement for categorical data. Biometrics 33:159–174
Levouvet FE, Vande Berg BC, Malghem J, Lebon CJ, Moysan P, Jamart J, Maldague BE (1996) MR imaging of the acetabular labrum: variations in 200 asymptomatic hips. Am J Roentgenol 167:1025–1028
Lohr JF, Uhthoff HK (1990) The microvascular pattern of the supraspinatus tendon. Clin Orthop Relat Res 254:35–38
Pfirrmann CW, Chung CB, Theumann NH, Trudell DJ, Resnick D (2001) Greater trochanter of the hip: attachment of the abductor mechanism and a complex of three bursae—MR imaging and MR bursography in cadavers and MR imaging in asymptomatic volunteers. Radiology 221:469–477
Pfirrmann CWA, Notzli HP, Dora C, Hodler J, Zanetti M (2005) Abductor tendons and muscles assessed at MR imaging after total hip arthroplasty in asymptomatic and symptomatic patients. Radiology 235:969–976
Preininger B, Schmorl K, von Roth P, Winkler T, Schlattmann P, Matziolis G, Perka C, Tohtz S (2011) A formula to predict patients’ gluteus medius muscle volume from hip joint geometry. Man Ther 16:447–451
Rompe JD, Segal NA, Cacchio A, Furia JP, Morral A, Maffulli N (2009) Home training, local corticosteroid injection, or radial shock wave therapy for greater trochanter pain syndrome. Am J Sports Med 37:1981–1990
Shbeeb MI, Matteson EL (1996) Trochanteric bursitis (greater trochanter pain syndrome). Mayo Clin Proc 71:565–569
Sudhoff I, de Guise JA, Nordez A, Jolivet E, Bonneau D, Khoury V, Skalli W (2009) 3D-patient-specific geometry of the muscles involved in knee motion from selected MRI images. Med Biol Eng Comput 47:579–587
Woodley SJ, Nicholson HD, Livingstone V, Doyle TC, Meikle GR, Macintosh JE, Mercer SR (2008) Lateral hip pain: findings from magnetic resonance imaging and clinical examination. J Orthop Sports Phys Ther 38:313–328
Acknowledgments
We thank Karen Rowe and Phillipa Nelson at Otago Radiology in Marinoto Hospital for their technical assistance. This study was supported by the New Zealand Society of Physiotherapists Scholarship Trust Fund and a University of Otago Dean’s Bequest Research Grant.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Appendix
Appendix
List of questions asked of participants with LHP
-
1.
How heavy is the work at your normal occupation? (sedentary, light, moderate, heavy)
-
2.
What is your current work situation? (paid employment/self employment, unemployed/jobless, at home parenting/housewife/retired, other—please state)
-
3.
How long have you had this episode of hip pain? (1 day–4 weeks, 1–3 months, 4–6 months, 7–12 months, longer than 1 year, longer than 5 years)
-
4.
Can you identify a specific incident that caused your current hip pain? (yes, no)
-
a.
If yes, please explain:
-
a.
-
5.
Compared to when your pain first started, is it now: (all gone, better, the same, worse)
-
6.
Is your current hip pain: (constant—24 h a day, 7 days a week, intermittent—some periods without pain)
-
7.
How would you describe your pain? (ache, sharp, other—please describe)
-
8.
Does your pain feel: (deep, superficial—close to the surface)
-
9.
Has the pain that you feel in your hip ever spread down the outside of your leg? (yes, no)
-
10.
What types of activities cause your current pain to increase? (standing for longer than a few minutes, sitting for longer than a few minutes, walking for longer than a few minutes, walking downstairs, walking upstairs, lying on affected side, other—please describe)
-
11.
Can you do anything to help reduce your current pain when it’s present? (yes, no)
-
12.
Have you had similar hip pain before? (yes, no)
-
a.
If yes: did it affect the same hip? (yes, no)
-
b.
How long did you have your last episode of hip pain for? (1 day–4 weeks, 1–3 months, 4–6 months, 7–12 months, longer than 1 year, longer than 5 years)
-
a.
-
13.
How much exercise do you regularly do currently, or until very recently? Exercise includes walking, sports, gardening, or other activity that raises your heart-rate or body temperature, for 30 min or more. (I exercise 3 or more times per week, I exercise 1 or 2 times per week, I do no exercise)
-
14.
What sort of exercise and/or sports do you normally do?
-
15.
Have you ever suffered an injury to either of your legs? For example your hip, thigh, knee, calf or ankle (yes, no)
-
a.
If yes, which leg did you injure? (left, right, both)
-
b.
What body part did the injury/injuries involve?
-
c.
What type of injury was it? (e.g. sprain, rupture, fracture)
-
d.
In what year did the injury/injuries occur?
-
a.
-
16.
Do you use a walking aid? (yes, no)
-
a.
If yes, what do you use?
-
a.
-
17.
Have you suffered an episode of lower back pain in the last two years? (yes, no)
-
18.
Do you have lower back pain at the moment? (yes, no)
-
a.
If yes, how long have you had this episode of back pain for?
-
a.
-
19.
Menopausal status: (pre-menopausal, menopausal, post-menopausal)
-
20.
Have you ever had a steroid injection in your painful hip? (yes, no)
-
a.
If yes, how many?
-
b.
How long ago was the last injection?
-
a.
NB: questions also asked of control participants are highlighted in bold.
Rights and permissions
About this article
Cite this article
Flack, N.A.M.S., Meikle, G.R., Reddy, M. et al. Hip abductor muscle volume in women with lateral hip pain: a case-controlled study. Surg Radiol Anat 34, 847–855 (2012). https://doi.org/10.1007/s00276-012-0970-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00276-012-0970-7