Abstract
This study investigates the differences in proportion and structure of the larynx in both children and adults, as well as changes which the larynx undergoes during puberty and adolescence. Furthermore, we have tried to answer the question on the existence of laryngeal sexual dismorphism in pre-pubertal (or pubertal) children. Larynges of 20 adults (10 men and 10 women) and 34 larynges of children not more than 12 years of age were acquired from the Forensic Medicine Department of the Medical University of Warsaw, Poland. Methods used are based on (1) anatomical preparation, (2) anatomical description and (3) measurements performed with the use of a digital camera and a computer-aided system MULTISCAN. Statistics, the Student’s test and Pearson’s linear correlation coefficient were calculated. Results show that proportions of the larynx depend on the age of the individual. The younger the individual is the more slender the larynx would be. However, proportion is not gender-related and hence is about the same in both males and females of any given age-group. Nevertheless, basic parameters were larger in males.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
In recent years, more and more information about the human larynx has been discovered [6, 7, 8, 9, 17, 26, 27]. Though it is well known that the most pronounced changes in the structure of the larynx happen during the adolescent period, we feel that these changes have not yet been sufficiently investigated. This insufficiency may prove to be an obstacle in medical practice. To perform laryngeal surgery, excellent knowledge of its gross anatomy as well as its varying structural proportions is essential. This is particularly so when small children are concerned. For instance, intubation is usually performed without much difficulty in adults, but in children it might result in complications such as torsion of arytenoids due to the relatively short intra-membranous part of the rima glottidis [20]. Likewise, tracheotomy in children carries a considerable risk of complications [2].
During ontogeny, the larynx undergoes peculiar changes. Not only does its size increase, but its proportion changes as well [9, 8, 10, 11, 23, 24]. Despite all the changes observed during puberty and later on in adolescence, the fact that the masculine larynx is larger in all its dimensions than the feminine larynx remains evident. Moreover, the narrowest part of a child’s larynx is the glottis, whereas in adults it is the subglottis [3, 13, 15, 19, 21, 25, 27]. The most pronounced differences between a child and an adult are found in the glottis [14], as children’s intercartilaginous part builds 60–75% of their glottis length. The anterior intermembranous part of the glottis becomes longer than the intercartilaginous part not earlier than the 3 years of life [10]. Furthermore, the subglottal area grows significantly during the first 2 years of life, and henceforth, growth becomes slow and steady and has a linear character [10, 22, 28]. It is also a known fact that during ontogeny the larynx descends [15, 26], causing the voice to deepen [29], and its shape changes from conoidal to cylindrical [19].
The main aim of this research is to determine differences in structure, proportion and size of the larynx as observed in children and adults; secondly, to find out if sex dimorphism in larynx structure is present during puberty; and thirdly, to obtain detailed measuring data characterizing larynx morphology. This data can enrich current knowlegde on the larynx and can help to create models of its physiology.
Material and methods
Twenty larynges of adults (10 men and 10 women) and 34 larynges of children not more than 12 years of age (Table 1) were acquired from the Forensic Medicine Department of the Medical University of Warsaw, Poland. The adults’ age at death varied from 17 to 48.
After staining in 10% formaldehyde, the samples were dissected with the use of a standard microsurgical kit. The methodology of this research is based on the observation of morphology and measurement of dimensions, angles and surfaces.
The causes of death varied from head injuries to heart rupture, heart attacks, abdominal hemorrhage, gunshot injuries, pneumonia (children), meningitis (children), exotoxins, alcohol abuse and electrical shock. Any form of destruction observed in the larynges was only as a result of the autopsy procedures and was not the cause of death in any of the cases.
During forensic medical sections, the larynx is routinely excised with the trachea and cut on its back wall so that the cartilaginous ring of the cricoid cartilage becomes disconnected. During the preparation, however, in some cases, especially in children, the cricoid cartilage fractured because of its very fragile nature and the difficulty involved in preparation of the cartilage. Removal of the perichondral tissue proved extremely difficult. These cases were excluded from the study and modification of method was introduced. One side of cricoid cartilage was sacrificed to obtain an intact opposite side. Hence, only one-sided measurements of samples were available for disposal.
The choice of the method to measure the larynx was suggested by several publications based upon experimental works on animals dealing with laryngeal movements [5, 12, 16, 27].
The measurements were made with the use of a computer programme Multiscan, which performs image analysis to an accuracy of 0.1 mm. For symmetric dimensions (dexter/sinister), only left-sided measurements were taken because of the frequent destruction of one side of the larynges obtained from the children. The basic measurements are given below:
-
1.
Thyroid cartilage
-
(a)
Vertical dimension in the median line
-
(b)
Distance between the superior cornua
-
(c)
Distance between the inferior cornua
-
(d)
Maximal height of the lamina
-
(e)
Length of the cornu superior
-
(f)
Length of the cornu inferior
-
(g)
Maximal vertical dimension
-
(h)
Angle between the right and left lamina
-
(a)
-
2.
Cricoid cartilage
-
(a)
Width of cricoid cartilage at the level of central point of articular facies of cricothyroid joint
-
(b)
Vertical dimension of the arc in the median line
-
(c)
Vertical dimension of the lamina in the median line
-
(d)
External anteroposterior (AP) dimension measured at the lower edge of the cartilage
-
(e)
Surface of the internal perimeter of the cartilage
-
(f)
Distance between central point of the socket of the cricothyroid joint and anterior pole of cricoid cartilage in a median line
-
(g)
Angle between (f) and AP plane
-
(h)
Distance between the central point of the cricothyroid joint’s socket and ventral pole of cricoid cartilage
-
(i)
Angle between (h) and AP plane
-
(a)
-
3.
Arytenoid cartilage
-
(a)
Maximum height from the base to the apex
-
(b)
Distance between the apex of vocal process and the middle of the socket of the joint of the cricoid cartilage
-
(c)
Distance between the apex of the muscular process and the middle of the socket of the joint of the cricoid cartilage
-
(a)
-
4.
Epiglottic cartilage
-
(a)
Maximum height
-
(b)
Maximum width
-
(a)
-
5.
Whole larynx
-
(a)
Vertical dimension
-
(b)
Length of intramembranous part of rima glottidis
-
(c)
Length of intracartilaginous part of rima glottis
-
(a)
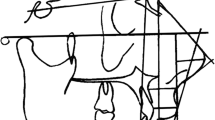
The parameters listed above can be found in Figs. 1–4. After measurement, the basic parameters characterizing larynx’s proportions were calculated. For statistical analysis of sex and body side differences, the Student’s t test was performed. Correlations between measured parameters were investigated using Pearson’s linear correlation test.
Results
The results of measurements of basic larynx parameters (Figs. 1, 2, 3, 4) are presented in Table 2. Statistical analysis showed that the sex factor was not important amongst children, but was very important in the adult group (see parameters 1i, 1d and 5d). This means that in women the average thyroid cartilage angle (1i) was relevantly larger than that in men, while average width of thyroid cartilage (1d) and average length of the membranous part of the rima glottidis (5d) were significantly smaller. Considering all the children used for the experiment, the age factor was important for all parameters except in 2g and 2i. All the parameters of the group of adults were remarkably larger than that in children, with exception of angles of joints’ surfaces of the cricoid cartilages (parameters 2g and 2i). Majority of the parameters were relevantly correlated, which means that larger parameters of individual elements reflected larger parameters of the whole larynx. Moreover, we found a relevant positive correlation between most parameters and age, with exception of the angle of thyroid cartilage or all angles of cricoid cartilage. While analyzing children group, it was proven that important statistical differences exist between children of ages 8–12 years and younger ones. There was no difference between the first two age groups: 0–2 years and 3–7 years.
Results of calculation of chosen proportion parameters are shown in Table 3. Analysis of data from table suggests that the individual’s sex does not significantly influence the ratios that characterize the larynx’s proportions either in adults or in children. On the contrary, the age difference between adults and all the children was found to substantially influence parameters 1, 4, 6, 10, 11, 13 and 17. The relevance level was under 0.01 or even under 0.0001 (for parameters 1 and 17). Moreover, the following parameters were significantly larger in children of both sexes than in adults: the relative height of superior cornua of thyroid cartilage (parameter 1), the height of thyroid cartilage in relation to the height of whole larynx (parameter 4), the height of lamina of cricoid cartilage in relation to its AP dimension (parameter 6), the height of lamina in relation to larynx’s height (parameter 10).
In addition, the width of the cricoid cartilage in relation to its AP dimension turned (parameter 11) out to be significantly smaller in children of both sexes. This indicates that cricoid cartilage increases in width with age, in relation to AP dimension.
Furthermore, the relative length of the muscular process of the arytenoid cartilage (parameter 13) was significantly larger in adults. Also, the length of the intramembranous part in relation to the length of the intracartilaginous part of rima glottis (parameter 17) was significantly larger in adults, which shows that adults have a more elongated intramembranous part in comparison to children.
On top of that, parameters 1 and 17 showed significant negative correlation with age, whereas for parameters 11 and 17 the correlation was positive. Consequently, it means that during puberty, the relative height of the superior cornua of thyroid cartilage and of the cricoid’s arch diminish, whereas the width of the cricoid cartilage and the length of intramembranous part of rima glottis enlarge.
As far as the differences between the subgroups of children are concerned, the statistically significant differences were not so obvious and have been observed only between the group of 8- and 12-year-olds as well as in younger ones, and only for parameters 3, 6, 9 and 12. So, in the 8th to 12th year of life, some important changes in the larynx proportions probably start.
Discussion
To begin with, the given parameters of human larynx are difficult to compare with the results of other authors; nevertheless, an attempt will be made. For simplifying and shortening this chapter, the comparison of our results with results of other authors as far as basic parameters are concerned will be displayed in a table (Table 4). However, for example, the width of the cricoid cartilage (parameter 2a) in men and in women could be only partially compared to the internal dimension of the laryngeal cavity, which was measured by other researchers [18, 30]. The sagittal dimension of the cricoid cartilage (parameter 2d) might be compared with the internal dimension of the laryngeal cavity at this level, which was measured by other researchers [18, 21, 25, 30].
An analysis of the significance of sex- and age-related differences provided some interesting facts. For instance, the thyroid cartilage angle was significantly larger in women and girls than in men and boys. This rule is widely known to be one of the dimorphitic features in humans [1, 26, 27]. It is however strange, though, that it is also found in small children, which is not mentioned in available literature. So, this feature of sexual dimorphism appears before maturation. Moreover, the difference between children and adults is significant, which means that the thyroid cartilage angle lessens substantially with age in both sexes. The children larynges are far smaller in all their parameters than those of adults; this is obvious, in the face of great growth as is experienced during puberty.
However, not all parameters are dependent on sex or age. The exceptions are parameters 2g and 2i, which point to a constancy of proportions between the internal parameters of the cricoid cartilage. It is puzzling that no significant differences between sexes were proven in any parameters, apart from the thyroid cartilage angle, width of thyroid cartilage or length of membranous part of the rima glottidis. In literature, these particular parameters are always shown as presenting significant statistical differences [1, 3, 13, 18, 26, 27, 30]. Additionally, the dependence of size on sex was also proven in the case of the length of the intermembranous part of the rima glottidis (parameter 5b). In women and girls, this parameter is significantly smaller than in men and boys. For adults, it is consistent with results of other researchers. In addition, this parameter significantly differs between sexes as is seen in the children, even though this information was not found in available literature.
Furthermore, interesting observations can be made while analyzing ratios, which were calculated on the ground of basal parameters. In case of the human larynx, it was proven that sex does not significantly influence the laryngeal parameters, whereas when it comes to age, there is proof that parameters 1, 4, 6, 10, 11, 13 and 17 substantially differ in adults and children. The relevance level was under 0.01 or even under 0.0001 (for parameter 1 and 17).
Proportions of the larynx depend mainly on age, not on sex, and they are almost identical in both sexes. Therefore, differences between children and adults were substantial, especially in certain points. First of all, the relative height of the superior cornu of the thyroid cartilage is significantly larger in children of both sexes than it is in adults. Secondly, the height of the lamina of the thyroid cartilage in relation to the height of the whole larynx and the height of the lamina of the cricoid cartilage in relation to its sagittal dimension are also relevantly larger in children than in adults. For this reason, children have more slender thyroid cartilages with a larger predominance of the superior cornu over the inferior one than is found in adults. This fact is consistent with results of other researchers. According to them, the changes in larynx, which develop with age, lead to elongation of all its dimensions, especially the vertical dimension measured at the anterior edge of the thyroid cartilage [15].
Moreover, the height of cricoid cartilage in relation to the height of whole larynx (ratio 10) was significantly larger in both sexes. On the contrary, the height of cricoid cartilage in relation to its sagittal dimension was substantially lower in children of both sexes (ratio 11). Therefore, the cricoid cartilage gets wider and relatively loses in its sagittal dimension. That contradicts, at least partially, the conclusions of other authors, which were based on NMR. NMR in their research showed that proportions between the transversal and sagittal dimensions do not change with age [16]. In our research, ratio 11 calculated for adults amounts to 0.71 in men and 0.68 in women. In another research [27], it amounted accordingly to 1.09 and 0.67.
In addition to this, the relative length of the muscular process of the arytenoid cartilage (ratio 13) is significantly larger in the group of adults, which gives it a longer lever for muscles. Consequently, the ability of active adduction/abduction of the arytenoid cartilage is larger in adults.
Finally, the ratio of the intramembranous part to the intracartilaginous part of the rima glottis (ratio 17) is substantially larger in the adult group, and thus adults have a significantly longer intramembranous part than children do. It is worth noting the dramatic changes in this parameter with age: in children before puberty (age group: 8–12) ratio 17 averages to 1.69 in boys and 1.79 in girls, whereas in adults it is 2.04 in men and 2.02 in women. Our results are only partially consistent with the results of other researchers, but their results also vary between one another. According another study [14], in children under 2 years of age, the intracartilaginous part of the rima glottis amounts to 60–75% of the length of the vocal fold. Hence, the ratio of the intramembranous part to the intracartilaginous part amounts from 1.3:1 to 1.6:1. This ratio is close to what was calculated in our research for children under 3 years of age, which oscillated around 1.5:1 (Table 3). From 1 to 12 months of life, the length of the glottis averages 7 mm (5.2–9.5 mm), with an intercartilaginous part of 4.1 mm (2.9–9.5) and an intermembranous part of 2.9 mm (1.7–5.0 mm) [10]. These results are completely consistent with our measurements of the lengths of the glottis in children. Also worth noting is the fact that, in the Eckels et al.’s [10] research, the length of the rima glottis in a 5-year-old child amounted to 10.67 mm (10.2–11.8 mm), with the intracartilaginous part averaging 4.8 mm (4.2–5.2 mm) and the intramembranous part amounting to 5.9 mm (5.3–6.7 mm). In our research, a 5-year-old boy’s glottis was similar amounting to 5.41/3.76 mm.
Dimensions of the larynx in children aged from 8 to 12 years were more or less two times larger than in younger children and more or less two times smaller than in adults.
Conclusions
-
1.
The proportions of human larynx do not depend upon sex but upon age.
-
2.
However, considerable changes in larynx dimensions occur during whole development period; the proportions change dramatically after the 8–12th year of life.
-
3.
The larynges of children before puberty are, in comparison with those of adults, relevantly more slender. This is caused by pronounced predominance of the length above the width.
References
Ajmani ML, Jain SP, Saxena SK (1980) A metrical study of laryngeal cartilagines and their ossification. Anat Anz 148:42–48
Carr MM, Poje CP, Kingston L, Kielma D, Heard C (2001) Complications in pediatric tracheostomies. Laryngoscope 111:1925–1928
Carter DR, Meyers AD (1979) Anatomy of the subglottic larynx. Otolaryngol Head Neck Surg 87:203–206
Cinar U, Yigit O, Vural C, Alkan S, Kayaoglu S, Dadas B (2003) Level of vocal folds as projected on the exterior thyroid cartilage. Laryngoscope 113:1813–1816
Choi HS, Berke GS, Ye M, Kreiman J (1993) Function of the posterior cricoarytenoid muscle in phonation: in vivo laryngeal model. Otolaryngol Head Neck Surg 109:1043–1051
Dover K, Howdieshell TR, Colborn GL (1996) The dimensions and vascular anatomy of the cricothyroid membrane: relevance to emergent surgical airway access. Clin Anat 9:291–295
Eckel HE, Sittel C, Zorowka P, Jerke A (1994) Dimensions of the laryngeal framework in adults. Surg Radiol Anat 16:31–36
Eckel HE, Sittel C (1995) Morphometry of the larynx in horizontal sections. Am J Otolaryngol 16:40–48
Eckel HE, Koebke J, Sittel C, Sprinzl GM, Pototschnig C, Stennert E (1999) Morphology of the human larynx during the first five years of life studied on whole organ serial sections. Ann Otol Rhinol Laryngol 108:232–238
Eckel HE, Sprinzl GM, Sittel C, Koebke J, Damm M, Stennert E (2000) Anatomy of the glottis and subglottis in the pediatric larynx. HNO 48:501–507
Fishman RA, Pashley NR (1981) A study of the premature neonatal airway. Otolaryngol Head Neck Surg 89:604–607
Garrett CG, Coleman JR, Reinisch L (2000) Comparative histology and vibration of the vocal folds: implications for experimental studies in microlaryngeal surgery. Laryngoscope 110:814–824
Goding GS, Bierbaum RW (1999) Relationship of the posterior cricoarytenoid muscle to the posterior cricoid lamina. Otolaryngol Head Neck Surg 120:493–498
Hirano M, Kurita S, Yukizane K, Hibi S (1989) Asymmetry of the laryngeal framework: a morphologic study of cadaver larynges. Ann Otol Rhinol Laryngol 98:135–140
Kahane J (1978) A morphological study of the human prepubertal and pubertal larynx. Am J Anat 151:11–20
Kasperbauer JL (1998) A biomechanical study of the human cricoarytenoid joint. Laryngoscope 108:1704–1711
Koufman JA, Fortson JK, Strong MS (1983) Predictive factors of cricoid ring size in adults in relation to acquired subglottic stenosis. Otolaryngol Head Neck Surg 91:177–182
Lieberman DE, McCarthy RC, Hiiemae KM, Palmer JB (2001) Ontogeny of postnatal hyoid and larynx descent in humans. Arch Oral Biol 46:117–128
Litman RS, Weissend EE, Shibata D, Westesson PL (2003) Developmental changes of laryngeal dimensions in unparalyzed, sedated children. Anesthesiology 98:41–45
Navsa N, Nossel G, Boon JM (2005) Dimensions of the neonatal cricothyroid membrane—how feasible is a surgical cricothyroidotomy? Paediatr Anaesth 15:402–406
Randestad A, Lindholm CE, Fabian P (2000) Dimensions of the cricoid cartilage and the trachea. Laryngoscope 110:1957–1961
Reidenbach MM (1996) The attachments of the conus elasticus to the laryngeal skeleton: physiologic and clinical implications. Clin Anat 9:363–370
Sasaki CT, Levine PA, Laitman JT, Crelin ES (1977) Postnatal descent of the epiglottis in man. A preliminary report. Arch Otolaryngol 103:169–171
Sellars SL, Keen EN (1990) Laryngeal growth in infancy. J Laryngol Otol 104:622–625
Seymour AH (2003) The relationship between the diameters of the adult cricoid ring and main tracheobronchial tree: a cadaver study to investigate the basis for double-lumen tube selection. J Cardiothorac Vasc Anesth 17:299–301
Sprinzl GM, Eckel HE, Sittel C, Pototschnig C, Koebke J (1999) Morphometric measurements of the cartilaginous larynx: an anatomic correlate of laryngeal surgery. Head Neck 21:743–750
Tayama N, Chan RW, Kaga K, Titze IR (2001) Geometric characterization of the laryngeal cartilage framework for the purpose of biomechanical modeling. Ann Otol Rhinol Laryngol 110:1154–1161
Too Chung MA, Green JR (1974) The rate of growth of the cricoid cartilage. J Laryngol Otol 88:65–70
Woisard V, Percodani J, Serrano E, Pessey JJ (1996) The voice of the child, morphological evolution of the larynx and its acoustic consequences. Rev Laryngol Otol Rhinol 117:313–317
Zieliński R (2001) Morphometrical study of the senile larynx. Folia Morphol 60:73–78
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wysocki, J., Kielska, E., Orszulak, P. et al. Measurements of pre- and postpubertal human larynx: a cadaver study. Surg Radiol Anat 30, 191–199 (2008). https://doi.org/10.1007/s00276-008-0307-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00276-008-0307-8