Abstract
The use of the buccal fat pad (BFP) has increased in popularity in recent years because of its reliability, ease of harvest, and low complication rate during oral and maxillofacial procedures. The aim of this study was to evaluate the volumetric variations of the BFP with a CT and MRI, as well as the thickness, weight and volume with conventional methods. We have examined the BFP from 80 formalin fixed adult cadavers (mean age 59) derived from both males (45) and females (35). In addition, we also examined 20 cadaveric BFPs using MR and CT imaging. Digital image analysis software was used to measure the volumetric distribution and to characterize the morphology of BFP. The BFP can be divided into three lobes (anterior, intermediate, and posterior) and has four extensions (buccal, pterygoid, pterygopalatine, and temporal). The BFP is fixed by six ligaments, to the maxilla, posterior zygoma, inner and outer rim of infraorbital fissure, temporalis tendon, and buccinator membrane.
The mean volume in males was 10.2 ml and ranged 7.8–11.2 ml, while in females the mean volume was 8.9 ml and ranged 7.2–10.8 ml. Additionally, the mean thickness was 6 mm, with a mean weight of 9.7 g. These facts may be important when considering the use of the BFP in reconstruction, particularly whether the correct volume has been removed from each side in aesthetic, oral, or maxillofacial procedures.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Although descriptions of the buccal fat pad (BFP) are typically very brief and lacking in detail in anatomical textbooks [1], they have recently received increased attention in the clinical literature. This stems largely from documentation of the use of BFP in oral and maxillofacial reconstruction [2–8]. Bichat [9] first considered the BFP, in 1802, as a well-circumscribed mass of fat (according to Khan, BFP is surrounded by a well-defined capsule giving the appearance of a well-circumscribed mass) without functional importance [10]. In modern medicine, however, clinicians realize that the BFP has several functional and therapeutic significances. In addition to its importance in filling deep tissue spaces and serving as a gliding pad during masticatory and facial muscle contraction, the BFP also acts to cushion important structures from the extrusion of muscle contraction or outer force impulsion [11]. Further exploration also indicates the BFP’s role as an important structure in plastic and reconstructive procedures [12].
Several surgical procedures involve the use of the BFP as a graft or pedicled flap, including correction of a cleft palate, closure of chronic buccal fistulas and softening bone graft contours in infraorbital and maxillary deformities [5, 13, 14].
In addition, BFP herniations are very common, especially in infants and children. These herniations usually arise due to a tear in a mucosal surface or in the buccinator muscle and may proceed to push the BFP into the oral cavity [14]. The BFP can also herniate into the maxillary sinus in which case it can potentially be misdiagnosed as a blood clot [15]. Another common diagnostic error occurs following facelift procedures, fat resections, or reparation of soft tissues directly over the buccal extension of the BFP, any of which may result in iatrogenic herniation. The resulting BFP herniation may then be confused with a lipoma [15]. Due to the similar morphology between a lipoma and the BFP, physicians have termed this instance “traumatic pseudolipoma” [16–18]. The risk of misdiagnosis between BFP herniation and other neoplastic lesions could possibly be minimized with a more accurate knowledge of volumetric variations of the BFP.
Despite the multitude of clinical and aesthetic uses, the significance of BFP volumetric variations and their relevance in use in grafting procedures have yet to be fully elucidated [12, 13, 15]. The aim of our study, therefore, was to provide an examination of the BFP with special emphasis on its volumetric variations amongst age and gender groups.
Materials and methods
We examined 80 adult human cadavers (160 hemifaces) during the “Human Body” course at Harvard Medical School, throughout the academic semesters of 2001, 2002, and 2003. The cadavers derived from female and male subjects (35 females/45 males) with an age range of 55–86 years old and a mean age of 59 years. The cadavers were divided into three groups according to their age: Group A, <60 (n=38); Group B, 60–70 (n=32); Group C, >70 (n=10). The division of the cadavers into three groups was based upon the narrow intra-group age distribution and not due to functional criteria. All the cadavers were fixed in formalin/phenol/alcohol solution. Dissections were performed by M.L and T.K. to elucidate the anatomy of the BFP and its surrounding structures. All the dissections were performed with the aid of surgical loupes (magnification 8.0×, http://www.microsurgeryusa.com/product.htm) in order to precisely identify the examined structures.
In addition, we also examined 20 cadaveric BFPs using MR and CT imaging. The BFPs used for imaging studies were obtained from the Department of Anatomy, American University of the Caribbean, School of Medicine, Netherlands Antilles. The specimens were derived from female and male subjects (8 females/12 males) with an age range of 58–76 years old and a mean age of 64 years. None of the examined cadavers had a previous history of facial surgery, congenital facial deformities, or any known contraindications to MRI.
Following preliminary examination, images from all dissected specimens were recorded with a Sony digital camera (model: Sony Cyber-Shot DSC-f717) and studied using a computer-assisted image analysis system [all measurements were carried out with the Lucia program (1998 edition for Windows), made by Nikon (Laboratory Imaging Ltd., Precoptic Co., Medical and Optical Instruments, Poland)] as previously described [19]. Digitized images of the BFP, together with their surrounding structures, were stored in the Lucia program, (1152×864 pixels), and converted to intensity gray levels from 0 (darkest) to 32 bit (lightest). After applying a standard 1 mm scale to all pictures within the program, Lucia was able to use this information to calculate pixel differences between two selected points (diameter, length, and width) on a given structure. The purpose of the software was to allow easy and accurate translation of pixel differences into metric measurements.
The BFPs were transected (according to their extensions), photographed, and width, length and volume measured. Similarly, the aforementioned dimensions were calculated with the Lucia software for CT and MR images. All BFPs after exposed and photographed were completely removed from the faces and weighed on an electronic scale, as previously described [20].
The magnetic resonance imager (1994 SIEMENS 1.5T VISION MRI) was operated at 1.5 T. The technique used for scan acquisition using a custom-designed radiofrequency surface coil has been previously described by Gosain et al. [21, 22]. The orientation of the images was such that the slices were parallel to the line from the outer canthus to the commissure of the mouth, as previously described [21, 22]. All cadaveric heads were scanned for a mean of 5 min (±55 s) so that 10–12 slides were obtained of each BFP. All measurements were recorded on a PC and were calculated with the Lucia software. The volume area measurements from each BFP were applied to the following formula to reconstruct a volumetric representation.
, where n is the number of images in the series, A represents the surface area of the BFP and 2.5 represents the sum of slice thickness (=1.5 mm) plus the interslice gap length (=1.0 mm).
Results were analyzed using Student’s t test and ANOVA (Statistica for Windows, version 6.2) and differences between means were considered statistically significant when values were P<0.05.
Results
Anatomically, the BFP consists of three independent lobes: anterior, intermediate, and posterior. Each lobe is encapsulated by an independent membrane and separated by a natural space. The BFP lobes are attached by six ligaments to the maxilla, posterior zygoma, inner and outer rim of infraorbital fissure, temporalis tendon, and buccinator membrane. The maxillary ligament was a fibrous condensation from the anterior lobe to the maxilla. The posterior zygomatic ligament connected the intermediate lobe with the zygomatic process. The medial and lateral infraorbital ligaments connected the medial and lateral side of the intermediate lobe, respectively, to infraorbital rim. The temporalis tendon ligament connected the posterior lobe with the temporalis tendon posteriorly. Finally, the buccinator ligament connected the anterior lobe with the buccinator membrane. These ligaments, which were not present in all specimens, appeared with the following prevalences: 95% (152) maxilla, 100% (160) posterior zygoma, 80% (128) inner and 80% (128) outer rim of infraorbital fissure, 90% (144) temporalis tendon, and 60% (96) buccinator membrane.
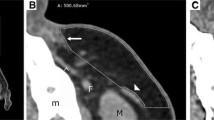
In order to examine, in detail, the volumetric variations of the different segments of BFP, we followed the classification proposed by Khan et al. [9]. All dissected specimens revealed that the main body of the BFP (specifically the posterior lobe) gave rise to four extensions: buccal, pterygoid, temporal (superficial temporal), and pterygopalatine (deep temporal) (Fig. 1). The main body lies on the anterior border of the masseter muscle and extends deeply to lie on the posterior maxilla and forward along the buccal vestibule. The parotid duct and zygomatic and buccal branches of the facial nerve cross the anterior and lateral surfaces of the BFP (Fig. 2). The anterior surface of the BFP was covered by buccal branches of the facial nerve in 75% (120) of specimens, while the lateral border of the BFP was covered by zygomatic branches in 90% (144) of cases.
reveals the relationships of the BFP with important landmarks of the face. The skin, together with a small amount of subcutaneous fat, has been retracted medially. The superficial musculoaponeurotic system (SMAS) has been left intact in the areas above and below an imaginary horizontal line, which connects the parotid gland to the BFP. The parotid duct and the parotid gland are exposed. The branches of the facial nerve are identified and retracted slightly upward to expose the BFP under a thick membrane of SMAS
The buccal extension, which together with the body accounts for about half the total weight, lies superficially within the cheek and is largely responsible for the contour of the cheek. The temporal, pterygoid and pterygopalatine extensions are smaller and situated more deeply (Table 1).
The BFP was analyzed with coronal and axial CT and MRI. The BFP was clearly visualized by MRI due to its pure adipose nature (Figs. 3, 4) in contrast with CT (Figs. 5, 6) imaging. We were able to differentiate the anatomic border of the BFP in greater detail using T1 sequences in contrast to T2.
The mean volume in males was 10.2 ml with a range of 7.8–11.2 ml, while in females the mean volume was 8.9 ml with a range of 7.2–10.8 ml. Variations between the right and left sides were small and not statistically significantly different (P>0.1). However, a substantial decrease of the BFP volume was observed in older subjects versus younger cadavers (P<0.02) (Table 2 and Fig. 7).
The mean thickness of the BFP was 6 mm with a range 4.8–7.2 mm, with a mean weight of 9.7 g and a range of 7.2–12.3 g. A significant difference was observed between males and females and between older and younger subjects. However, there was no significant difference between left and right sides. The volume distribution of the BFP in groups A, B, and C is shown in Fig. 8. Furthermore, no differences were observed between the specimens received from the two different schools, concerning race, gender, morphology, and percentages.
The volume distribution of the BFP in groups A, B, and C is shown in Scheme 1. With increasing age (group A<B<C), there is a linear decrease in the volume of BFP in both males and females. Furthermore, the volume of the BFP in Group A ** is statistically significantly greater than that of group B (P>0.03) and group C (P>0.02). BFP volume of group B * is statistically significantly greater than that of group C (P>0.03)
Discussion
The focus of the present study was to analyze, in particular, the volumetric and mass specifications of the BFP and to report any differences between genders and amongst different age groups. In keeping with that goal, we have chosen to omit detailed morphological descriptions of each portion of the BFP as they have been sufficiently described by the study of Zhang et al. [9–11, 13]. However, that study, which reported age-related changes of the size of the BFP, did not provide specific volumetric parameters or methods by which these measurements were obtained. For instance, Zhang et al. [11] reported that the anterior lobe of the BFP is smaller in adults than in the aged; a finding which is in contrast with our measurements. We observed that the mean volume for group A specimens was 10.8 ml while the mean volume for Group B was specimens was 9.9 ml and the mean volume of Group C was 8.7 ml.
A study by Amin et al. [3], which described use of the BFP in oral reconstruction, made brief mention of volumetric measurements. In the study, the authors reported a mean weight of the BFP as being 9.3 g and a mean volume of 9.6 ml. However, no references were made as to how these measurements were obtained and whether or not any age or gender differences were noticed with regards to BFP size. The same study did mention the absence of variation between right and left sides: a report which is consistent with our findings. Additionally, Amin et al. [3] reported no significant association between BFP volume and subcutaneous fat stores.
Another study by Matarasso et al. [17, 18, 23] provided similar data to that of Amin et al. [3] reporting a mean weight of 9.3 g and a mean volume of approximately 10 ml [3, 11]. It is interesting to note that neither of these studies reported specific data with regards to variations in BFP volume or mass, either between genders or across different age groups. We are unable to determine whether either of these studies was able to discern such differences, as our research has clearly shown that they exist. It is entirely possible that these data were beyond the scope of both of the aforementioned studies, and therefore not included in their reports. The value of this data is in considering the availability of the BFP for use in plastic and reconstructive surgical procedures. Amin et al. [3] completed a study using the BFP in reconstruction after partial maxillary resection due to neoplastic disease. The technique used in their study, which was the same as that described by others, involved the use of the BFP as a pedicled flap to close bone defects and provide support for dentures [23–26]. It is clear that the availability of the BFP for use in this and similar procedures depends largely on the volume of tissue available for grafting. Taking into account the age associated increases in the incidence of maxillofacial cancer, it is important to note the corresponding decline in BFP volume with age. This fact makes the availability of the BFP for harvest inevitably less likely with advancing patient age.
Kurabayashi et al. [27] provided a study describing imaging of buccal space lesions with MRI. Their study was aimed at exploring the capabilities of MR in determining the malignancy of a buccal space lesion. They reported that with a few exceptions they were unable to determine the composition of the lesions purely based on MRI. This is significant in that it demonstrates a common source of diagnostic error. Upon radiological examination of the head, it is difficult, if not impossible, to differentiate between a pseudoherniation of the BFP and other more ominous lesions such as adenoid cystic carcinoma or salivary carcinoma [17, 27]. It is with this diagnostic conundrum that specific knowledge of BFP morphology and measurements would prove particularly useful. For instance, if the diagnostic radiologist is aware of these parameters, it may aid in differentiating between a traumatic lesion and one of proliferative or neoplastic origin. A pseudoherniation of the BFP would involve the displacement of tissue from one compartment to another with no overall increase in tissue mass or volume. On the other hand, a neoplastic lesion would clearly have increased volume, even in the early stages. Careful use of this knowledge could allow for earlier detection of neoplastic masses, while at the same time sparing unnecessarily invasive procedures for those patients in whom the lesion in clearly of traumatic origin.
Several cases exist in the literature, which demonstrate various BFP herniations and the resulting difficulties in their differential diagnosis [28–31]. One report by Marano et al. [32] describes a patient with facial trauma in whom the BFP had herniated into the maxillary sinus. In that case, the radiopacity of the inferior maxillary sinus was incorrectly diagnosed as a fluid level of blood. After surgical exploration, it was discovered that a large portion of the BFP had herniated into the maxillary sinus and virtually obliterated the space there. In this case, where trauma was clearly known, the distinction is not of great clinical significance. It does, however, illustrate the potential for diagnostic errors of a more serious nature, which might involve failure to identify a malignant lesion. Similarly, Ide et al. [33] presented a study which demonstrates this diagnostic paradox by reporting a case of a spindle cell nodule which was misdiagnosed as pseudoherniation of the BFP.
Conclusions
The availability of the BFP for using plastic and reconstructive procedures depends largely on the volume of tissue available for grafting. Taking into account the age associated increases in incidence of maxillofacial cancer, it is important to note the corresponding decline in BFP volume with age. This fact makes the availability of the BFP for harvest inevitably less likely with advancing patient age. It is our hope that these data will prove useful to both anatomists and clinicians alike in both increasing their knowledge of the specific anatomy of the BFP and its relevance in oral and maxillofacial reconstruction.
Limitations of the study: We were unable to measure the degree of subcutaneous fat accumulation in the cadavers or obtain a height weight index. This information could provide data concerning the influence of the degree of subcutaneous fat accumulation on the BFP. We believe that further attention needs to be given to the volumetric investigation of the BFP (related to the age) with MRI and we hope that future studies will follow this avenue of research.
References
Clemente CD (ed) (1985) Gray’s anatomy. 30th Am Ed Williams and Wilkins Baltimore p 447
Adeyemo WL, Ladeinde AL, Ogunlewe MO, Bamgbose BO (2004) The use of buccal fat pad in oral reconstruction—a review. Niger Postgrad Med J 11(3):207–211
Amin MA, Bailey BM, Swinson B, Witherow H (2005) Use of the buccal fat pad in the reconstruction and prosthetic rehabilitation of oncological maxillary defects. Br J Oral Maxillofac Surg 43(2):148–254
Chen GF, Zhong LP (2005) Functional reconstruction of maxilla with titanium mesh and pedicled buccal fat pad flap. Plast Reconstr Surg 115(1):334–336
Dean A, Alamillos F, Garcia-Lopez A, Sanchez J, Penalba M (2001) The buccal fat pad flap in oral reconstruction. Head Neck 23(5):383–388
Holton LH III, Rodriguez ED, Silverman RP, Singh N, Tufaro AP, Grant MP (2004) The buccal fat pad flap for periorbital reconstruction: a cadaver dissection and report of two cases. Plast Reconstr Surg 114(6):1529–1533
Santiago BM, Damasceno LM, Primo LG (2005) Bilateral protrusion of the buccal fat pad into the mouth of an infant: report of a case. J Clin Pediatr Dent 29(2):181–184
Scott P., Fabbroni G., Mitchell D.A (2004) The buccal fat pad in the closure of oro-antral communications: an illustrated guide. Dent Update 31(6):363–366
Kahn JL, Wolfram-Gabel R, Bourjat P (2000) Anatomy and imaging of the deep fat of the face. Clin Anat 13:373–382
Racz L, Maros TN, Seres-Sturm L (1989) Structural characteristics and functional significance of the buccal fat pad (Corpus Adiposum Buccae). Biomorphol, Histol, Embryol XXXV(2):73–77
Zhang HM, Yan YP, Qi KM, Wang JQ, Liu ZF (2002) Anatomical structure of the buccal fat pad and its clinical adaptations. Plast Reconstr Surg 109(7):2509–2518
Yousif NJ, Gosain A, Sanger JR, Larson DL, Matloub HS (1994) The nasolabial fold: a photogrammetric analysis. Plast Reconstr Surg 93(1):70–77
Jackson IT (1999) Anatomy of the buccal fat pad and its clinical significance—cosmetic follow-up. Plast Reconstr Surg 103(7):2059–2060 (discussion 2061–2063)
Neder A (1983) Use of buccal fat pad of grafts. Oral Surg, Oral Med, Oral Path 55, 349
Baumann A, Ewers R (2000) Application of the buccal fat pad in oral reconstruction. J Oral Maxillofac Surg 58(4):389–393
Brooke RI (1978) Traumatic herniation of buccal pad of fat (traumatic pseudolipoma). A review. Oral Surg Oral Med, Oral Pathol 45(5):689–691
Matarasso A (2003) Pseudoherniation of the buccal fat pad: a new clinical syndrome. Plast Reconstr Surg 112(6):1716–1720
Matarasso A (1997) Pseudoherniation of the buccal fat pad: a new clinical syndrome. Plast Reconstr Surg 100(3):723–736
Loukas M, Hullett J, Wagner T (2005) The clinical anatomy of the inferior phrenic artery. Clin Anat 18(5):357–365
Martin AD, Ross WD, Drinkwater DT, Clarys JP (1985) Prediction of body fat by skinfold caliper: assumptions and cadaver evidence. Int J Obes 9(1):31–39
Gosain AK, Klein MH, Sudhakar PV, Prost RW (2005) A volumetric analysis of soft-tissue changes in the aging midface using high-resolution MRI: implications for facial rejuvenation. Plast Reconstr Surg 115(4):1143–1152
Gosain AK, Amarante MT, Hyde JS, Yousif NJ (1996) A dynamic analysis of changes in the nasolabial fold using magnetic resonance imaging: implications for facial rejuvenation and facial animation surgery. Plast Reconstr Surg 98(4):622–636
Matarasso A (1991) Buccal fat pad excision: aesthetic improvement of the midface. Ann Plast Surg 26(5):413–418
Alkan A, Dolanmaz D, Uzun E, Erdem E (2003) The reconstruction of oral defects with buccal fat pad. Swiss Med Wkly 23(133):465–470
Arias-Gallo J, Maremonti P, Gonzalez-Otero T, Gomez-Garcia E, Burgueno-Garcia M, Chamorro Pons M, Martorell-Martinez V (2004) Long-term results of reconstruction plates in lateral mandibular defects. Revision of nine cases. Auris Nasus Larynx 31(1):57–63
Shibahara T, Watanabe Y, Yamaguchi S, Noma H, Yamane GY, Abe S, Ide Y (1996) Use of the buccal fat pad as a pedicle graft. Bull Tokyo Dent Coll 37(4):161–165
Kurabayashi T, Ida M, Tetsumura A, Ohbayashi N, Yasumoto M, Sasaki T (2002) MR imaging of benign and malignant lesions in the buccal space. Dentomaxillofac Radiol 31(6):344–349
Haria S, Kidner G, Shepherd JP (1991) Traumatic herniation of the buccal fat pad into the oral cavity. Int J Paediatr Dent 1(3):159–162
Horie N, Shimoyama T, Kaneko T, Ide F (2001) Traumatic herniation of the buccal fat pad. Pediatr Dent 23(3):249–252
Patil R, Singh S, Subba Reddy VV (2003) Herniation of the buccal fat pad into the oral cavity: a case report. J Indian Soc Pedod Prev Dent 21(4):152–154
Zipfel TE, Street DF, Gibson WS, Wood WE (1996) Traumatic herniation of the buccal fat pad: a report of two cases and a review of the literature. Int J Pediatr Otorhinolaryngol 38(2):175–179
Marano PD, Smart EA, Kolodny SC (1970) Traumatic herniation of buccal fat pad into maxillary sinus: report of case. J Oral Surg 28(7):531–532
Ide F, Shimoyama T, Horie N (2000) Post-traumatic spindle cell nodule misdiagnosed as a herniation of the buccal fat pad. Oral Oncol 36(1):121–124
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Loukas, M., Kapos, T., Louis , R.G. et al. Gross anatomical, CT and MRI analyses of the buccal fat pad with special emphasis on volumetric variations. Surg Radiol Anat 28, 254–260 (2006). https://doi.org/10.1007/s00276-006-0092-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00276-006-0092-1