Abstract
The purpose of this study was to correlate histopathological with CT findings in patients suffering from hepatocellular carcinoma (HCC) eligible for orthotopic liver transplantation (OLT), with a special focus on the antitumoral effect of transarterial chemoembolization (TACE) therapy. A total of 42 consecutive patients suffering from HCC had been treated prior to OLT by means of TACE. TACE was carried out with a mixture of Lipiodol (10–20 ml) and mitomycin C (max. dosage, 10 mg). TACE was performed at 6- to 8-week intervals. Follow-up investigation included contrast-enhanced multislice CT controls and laboratory control. Liver explants were evaluated macroscopically and microscopically to determine the number and size of the tumor lesions as well as the degree of tumor necrosis. Necrosis was investigated in H&E-stained sections. The degree of necrosis was classified as follows: 0–25%, 26–50%, 51–75%, 75–99%, and complete necrosis. Two hundred thirty-one TACE procedures (5.5 ± 2.9; range, 1–14) were performed. Mean tumor size in CT before and after TACE was 4.1 ± 2.4 (range, 1.0–12.0 cm) and 2.7 ± 1.2 (range, 1.0–6.0 cm; p < 0.001). Mean tumor number before and after TACE in CT was 2.5 ± 1.5 (n = 105; range, 1–8) and 2.4 ± 2.0 (n = 103; range, 1–6; p = 0.99). In the surgical specimen tumor size and tumor number were 2.8 ± 1.6 (range, 1.0–7.0 cm; p = 0.78) and 1.9 ± 1.2 (range, 1–7; p = 0.003). Mean tumor necrosis was 67.8% ± 28.1%. Tumor necrosis was subtotal or complete in 17 of 42 (40.5%) patients. Tumor necrosis correlated significantly with the degree of arterial devascularization in CT (p = 0.001), the amount of Lipiodol washout (p = 0.002), and the number of tumor lesions (i.e., unifocal vs. multifocal). Furthermore, elevated serum levels of bilirubin (p = 0.005) and decreased albumin (p = 0.004) affected the local antitumoral effect. A poor necrosis rate (< 25%) significantly correlated with the number of TACE procedures accomplished (p = 0.023). In conclusion, TACE provided an acceptable local antitumoral effect in patients scheduled for liver transplantation. Tumor necrosis depended significantly on the degree of arterial devascularization and the accumulation of Lipiodol within the HCC lesions. Unifocal tumors and preserved liver function were positive predictors for a more favorable local antitumoral effect. Poor necrosis rates were found in patients with significant Lipiodol washout and who received a limited number of TACE procedures.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Although a variety of therapeutic modalities is available for treating hepatocellular carcinoma (HCC), orthotopic liver transplantation (OLT) represents the only curative option. By means of OLT both the tumor and the underlying cirrhosis can be cured [1–3]. Considering the shortage of donor organs, the time that HCC patients must remain on the waiting list might increase [4].
Thus, “bridging the time to transplant” by means of interventional radiological therapy is of particular interest and importance [5–9]. Local ablative treatment modalities such as transarterial chemoembolization (TACE), percutaneous ethanol injection (PEI), and radiofrequency thermoablation (RFTA) have been introduced as therapeutic options in the pretransplant phase; these treatments aim to prevent tumor progression, downsize tumors, or keep patients in stable condition on the list. Whereas PEI and RFTA are restricted for patients with a more limited disease, TACE is the treatment of choice in patients with multifocal tumor spread [10–12]. Due to conflicting results of TACE the discussion concerning its efficacy is still ongoing [7, 13–17]. Therefore, we reviewed 42 consecutive patients who were treated in a prospective study by TACE prior to OLT. This evaluation presents the correlations between multislice CT imaging, on the one hand, and the macroscopic and histomorphological findings of the explanted specimens, on the other. In this regard, particular focus was directed to determining the local ablative effect of the TACE procedure and the reliability of CT imaging in evaluating the number and size of the tumors. Positive and negative predictors for a more or less favorable effect of the TACE procedure are formulated and discussed.
Patients and Methods
A total of 42 consecutive patients suffering from HCC were included in this study. HCC had been histologically confirmed in all cases. All patients were scheduled for liver transplantation at the Department of Liver Transplantation and Hepatobiliary Surgery according to the common recommendations for liver transplantation. Patients with multifocal tumors and tumors larger than 5.0 cm that exceeded the Milan criteria for OLT (≤3 tumor nodules, none larger than 3 cm in diameter, or patients with a solitary nodule ≤5 cm in maximum diameter) were initially not considered as transplant candidates. However, in case of tumor regression and relevant downstaging, those patients might become secondary candidates for OLT and were placed on the transplant list.
To bridge the time to transplantation, the patients had been treated with TACE and all 42 patients were finally transplanted. The decision to perform TACE was made by an interdisciplinary tumor board in each individual case. Approval for TACE as a bridging concept for patients with HCC eligible for liver transplantation was granted by the institutional investigation and ethics committee. The TACE protocol included CT evaluation after each single treatment session. In all cases data from both the initial helical CT investigation and the last CT scans were available for evaluation. The last CT scan was performed prior to liver transplantation within a period of 2 weeks.
Patients were excluded from TACE if they had decompensated liver disease (Child-Pugh class C, Okuda stage III), bacterial infection, extrahepatic tumor spread, or any contraindication for an arterial procedure (impaired clotting test, platelet count <50,000/mm3, or prothrombin activity <50%). Provided that there were no contraindications, TACE was continued until OLT at 6- to 8-week intervals.
Histopathology
The whole hepatectomy specimens were routinely examined at the Department of Pathology to identify features of the tumor disease and determine the impact of TACE. The explanted liver was serially cut into sections of approximately 1.0 cm in the sagittal plane. Standard procedure included macroscopic evaluation, assessing the number, size, and location of the different tumor nodules. Gross vascular invasion and the presence of a tumor capsule were checked additionally. Histological sections were made for all suspicious lesions. Histological specimens were routinely stained with hematoxylin and eosin (H&E) as part of the routine histological assessment. Tumors were graded according to the WHO criteria as well, moderately, and poorly differentiated into classes G1–G3 [18].
The necrosis rate of the tumor was defined as the proportion of necrotic area to the presumed tumor area. Necrosis was termed complete when no viable tumor cells were found. The diagnosis of active neoplasm was based on the presence of viable neoplastic hepatocytes. The amount of viable tumor was classified on a percentage basis during conventional optical microscopy as follows: 0–25% necrosis, 26–50% necrosis, 51–75% necrosis, 76–99% necrosis, and 100% necrosis. The results were documented for each single nodule. Due to the retrospective design of this evaluation by means of the written pathological reports, the nodules were allocated to the CT images and analyzed lesion by lesion. Pathological data and CT results were only compared for those lesions that could be correctly matched.
Imaging Modalities
Prior to TACE, contrast-enhanced helical CT was performed. Dynamic CT images were obtained using a four-row multislice CT scanner (Siemens Volume Zoom; Erlangen, Germany). A native CT scan was obligatory, including arterial and portal venous phase acquisitions. Investigation parameters were as follows: slice thickness, 3 mm; increment, 2.5 mm; collimation, 2 mm; 150 ml contrast medium i.v. (Imeron 300; Bracco Altana Pharma GmbH, Konstanz, Swiss); 50-ml 0.9% NaCl bolus; flow, 5 ml/s; 150 mAs; 120 kV. After TACE, a native CT scan was carried out on the same or the following day to document the Lipiodol distribution and to exclude displacement of the embolizing agent.
Technical Procedure
For TACE a suspension consisting of 10 ml Lipiodol (Lipiodol ultra fluid; Andre Guerbet, Aulnay-sous-Bois, France) and 10 mg mitomycin C was employed. The two components had been mixed directly before administration with two syringes connected via a three-way stopcock. First, any atypical liver blood supply was checked by selective catheter exploration of the mesenteric and celiac arteries. Then the common hepatic artery was cannulated, and, according to the individual anatomical disposition, the catheter was placed distal to the origin of the gastroduodenal artery in the proper hepatic artery or in the right or left lobar artery. A superselective approach on the level of the segment arteries using a 2.2-Fr microcatheter (Boston Scientific Corp., Natick, MA, USA) was selected if a stable catheter position could not be achieved. The treatment was performed under continuous fluoroscopy and terminated after reaching the total dose and/or stasis or reflux in the appropriate section. The total dose was distributed under continuous fluoroscopy to both liver lobes in the case of bilateral tumor spread or only to either the right or the left lobar artery in cases of unilateral tumor growth. The patients were usually hospitalized for one night and were dismissed the next day if no complications developed. Nausea, pain, or fever that occurred in association with the TACE treatment was treated symptomatically with antiemetics (e.g., Navoban, 5–15 mg i.v.), analgesics (e.g., Metamizol i.v.), or antipyretics (e.g., Paracetamol, 500–1000 mg p.o.). No antibiotic prophylaxis was given.
CT Evaluation
Tumor was diagnosed in each single nodule by summarizing the findings from native CT scans, contrast-enhanced arterial and portal-venous phase and late scans, and scans made after Lipiodol administration. A nodule was rated as HCC if it showed arterial hypervascularization, a hypo- or isodense pattern in the portal-venous phase, and a positive Lipiodol accumulation after intra-arterial administration of the oily medium.
By CT tumor density (categories: hyperdense, hypodense, and mixed), number of tumor lesions, and maximal transverse and longitudinal diameter of the marker lesion in the disease process were assessed. The occurrence and/or the absence of pathological lymph nodes was noted, as was extrahepatic tumor spread. Tumor response was assessed on the basis of the cross-sectional CT images and according to RECIST criteria [19].
Up to five marker lesions were analyzed and the sum of the nonmeasurable lesions, i.e., <1 cm in diameter in cross-sectional spiral CT, was registered. According to RECIST, unidimensional measurements of the sum of the longest diameters of the marker lesions were carried out. Response was defined as follows: complete response, disappearance of all target lesions; partial response, >30% decrease from baseline; progressive disease, >20% increase from baseline, any new tumor lesion; and stable disease, all other cases. In cases of repeated treatment, the best interim result (best response) was taken.
Lipiodol accumulation was rated according to a visual analogue score as follows: complete–moderate–poor. Lipiodol washout was graded as follows: complete–strong–minor–none.
Tumor vascularization after TACE was evaluated based on the measurement of contrast enhancement expressed as Houndsfield units (HU) in the central and lateral aspect of the tumor nodule in comparison to native and arterial contrast-enhanced images. Mean enhancement of <10 HU was rated as no arterial contrast uptake. Mean and peak enhancement as well as standard deviation was documented. According to the maximum diameter of the tumor nodule the still vascularized part of the tumor was calculated according to a visual analogue score and classified as follows: none (i.e., completely devascularized), <25%, 25–49%, 50–74%, and 75–100%. These results were compared to the histopathological degree of necrosis.
Statistics
The retrospective database was analyzed using the statistical program SPSS (version 12.0 for Windows). Continuous ordinal data are expressed as mean ± SD and as qualitative data by frequency and rate. The statistical significance of quantitative data was determined by means of the Kruskall Wallis test and categorical data were compared using the chi-square test. A two-tailed p-value of <0.05 was considered to be statistically significant. All statistical tests were carried out bilaterally.
Laboratory Parameters, Clinical Findings
In all patients the serum AFP levels, transaminases (GOT, GPT), serum cholinesterases, and blood count and coagulation parameters (PTT, thrombin time, Quick, and/or INR) were checked every 6 weeks. Patients were clinically classified according to the Child Pugh stages (Child stages A–C).
Results
During the investigation period, a total of 42 patients (33 men, 11 women; mean age, 59.4 ± 6.1 years) were included in the study. All patients had undergone liver transplant owing to histologically verified HCC. Alcoholism was the most frequent cause for the underlying liver cirrhosis (42.9%; n = 18), followed by hepatitis C and hepatitis B infection. Prior to OLT, chemoembolization was performed as a bridge to transplant. Median time from the first TACE procedure to OLT was 252 ± 203 days (range, 54–973 days) and median time on the transplant list was 215 ± 145 days (range, 20–705 days). Prior to the first TACE procedure liver function was classified according to the Child-Pugh stages as Child A in 90.5% (n = 38) and as Child B in 9.5% (n = 4). Overall a total number of 231 TACE procedures were accomplished. The mean number of TACE procedures was 5.4 ± 2.9 (range, 1–14). Demographics are listed in Table 1.
Tumors were graded as well, moderately, and poorly differentiated in 4.8%, 47.6%, and 33.3% of patients, respectively. Microscopic angioinvasion was found in 73.8%. According to the TNM classification, pT1–pT4 stages were found in 28.6%, 42.9%, 14.3%, and 9.5%, respectively (Table 2). Histomorphological evaluation showed a total number of 78 (1.9 ± 1.2) tumors. By comparison helical, pretransplant contrast-enhanced CT revealed a total number of lesions larger than 1.0 cm in 82 cases (2.0 ± 1.0; range, 1–5) compared to 85 lesions in the initial CT scan (p = 0.98) (Table 3). Furthermore, 21 lesions smaller then 1.0 cm were found in the CT scans that showed an arterial hypervascularized pattern and Lipiodol accumulation. Those lesions could retrospectively not be matched so that the histology remained unclear. Therefore further analysis is according only to the 78 lesions found in pathologic examination that could be correctly attributed.
The great majority of the tumors investigated here was hyperdense on arterial CT scans (84.1%) and the Lipiodol enhancement after the TACE procedure was rated as strong or complete in 75% (n = 33) of the cases. In two-thirds of the cases (n = 28; 66.7%), no or only minor Lipiodol washout on subsequent CT control studies was recognized. In 14 of 42 cases a complete or remarkable Lipiodol washout in follow-up CT scans occurred (Table 2).
Mean tumor size was 4.1 ± 2.4 cm (1.0–12.0 cm) in the initial CT scan and 2.7 ± 1.2 cm (1.0–6.0 cm) in the last CT scan (p < 0.001). Mean tumor size was rated similarly in CT and histological evaluations in most lesions (p = 0.78). CT overestimated the tumor size in the specimen in five cases and underestimated it by more than 1.0 cm in four cases (Table 3).
Comparison of Tumor Necrosis According to CT and Histology
The mean degree of tumor necrosis was 67.8 ± 28.1%. Tumor necrosis was rated as 0–25%, 25–50%, 51–75%, 75–99%, and 100% in 8 (19.0%), 5 (11.9%), 5 (11.9%), 17 (40.5%), and 7 (16.7%) patients, respectively (Table 4). The last CT images after TACE and the histopathological findings correlated well in terms of degree of necrosis and degree of vascularization or devascularisation (p = 0.001). In 12 of 16 cases in which necrosis was >75% in the histopathological study, no contrast enhancement was seen in the multislice CT image. In contrast, in cases of persistent and extensive hypervascularization, >75% (n = 7), sufficient tumor necrosis was excluded in any case by histopathology. Accordingly the local response (i.e., stable condition–response–progression) depended significantly on the degree of devascularization. Regression and stable condition (n = 38) were observed clearly more often in cases of extensive devascularization (n = 27; Figs. 1–4), whereas viable tumor was attributed to progression and occurred significantly more often in cases of a significant residual perfusion after TACE (p = 0.002). In no case was progression found if devascularization was complete (Figs. 5–8), whereas a regression was observed clearly more often in cases in which perfusion was substantially reduced (n = 12/17).
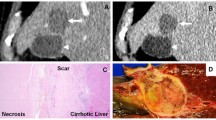
A 40-year-old woman with hepatitis B cirrhosis. Solitary hepatocellular carcinoma lesion in the right liver lobe with hypodense central parts (diameter, 5.0 cm; Fig. 1). Good Lipiodol accumulation after four TACE treatments. Notice the small rim of contrast enhancement (Fig. 3) compared to the native image (Fig. 2) at the medial aspect of the tumor lesion (arrows). H&E-stained section (Fig. 4) demonstrates extensive necrosis (straight arrow) with parts of still viable tumor (curved arrows)
A 52-year-old man with hepatitis B-associated liver cirrhosis. Inhomogeneous, solitary, 3.0-cm hepatocellular carcinoma located in the right liver lobe below the capsule (Fig. 5; arrows). Complete devascularization with lack of contrast enhancement in the arterial phase (Fig. 6) compared to the native scan (Fig. 7). H&E-stained section (Fig. 8) shows complete necrosis of the tumor
For further statistical evaluation two subgroups were defined, as follows: necrosis >75% (n = 23) and necrosis ≤75% (n = 19). Results are listed in Tables 5 and 6. The most significant correlation was found for the degree of Lipiodol washout: the stronger the Lipiodol washout, the smaller the necrotic area (p = 0.003).
Necrosis was significantly more extensive in patients with unifocal disease than in those with multifocal disease (p = 0.018). Patients who showed a response of >75% tumor necrosis tended to be younger than patients with less extensive necrosis (p = 0.08). Patients suffering from hepatitis B infection showed larger zones of necrosis than those without hepatitis B infection (p = 0.044). In addition, the degree of necrosis correlated significantly with the preinterventional liver function. Laboratory control of initial albumin and bilirubin levels before TACE indicated that patients with a less favorable response to TACE had significantly decreased serum levels of albumin (p = 0.004) and significantly elevated serum bilirubin levels (p = 0.005).
The degree of necrosis did not correlate with the initial tumor size (p = 0.45) or with the tumor density prior to TACE treatment (p = 0.43).
There was no evident correlation between tumor necrosis and the presence or absence of microscopic angioinvasion or between tumor necrosis and tumor grading (p = 0.35 or p = 0.34, respectively), nor was evidence found for a better or worse response for gender (p = 0.61) or the waiting time to OLT (p = 0.68).
Patients in whom necrosis was insufficient were reviewed separately. The chi-square test showed that Lipiodol washout was the variable with the strongest influence on minor local response (p < 0.0001). Additionally, the number of TACE procedures accomplished was relevant (p = 0.023). In five of seven cases fewer than three TACE procedures were carried out. A mean of 3.5 ± 3.1 TACE procedures was performed, compared to 5.9 ± 2.8 in the other patients. Other factors such as tumor size (p = 0.13), time on the waiting list (p = 0.95), and microscopic patterns such as grading (p = 0.30) and angioinvasion (p = 0.28) had no influence to poorer locoregional response.
Discussion
TACE is the mainstay in the palliative treatment of HCC [10, 11]. Due to the shortage of donor organs and the increasing need for liver transplantation in the last decade, TACE has become more and more important at many centers as a bridge to transplant [5, 8, 9, 17, 20–22, 28].
Besides downstaging patients the most important goal of TACE in transplant candidates is to keep them in a steady state and to avoid dropout from the transplant list [9, 17, 20– 23]. Even if the TACE strategy is well accepted, the local antitumoral efficacy of the TACE protocols has been compared with histological results in only a few reports. We conducted this study to correlate the CT findings with the results of histopathology and to develop predictors for the outcome of our TACE protocol in the future.
In our cohort the mean overall rate of tumor necrosis was calculated to be >65%, ranging from <25% to complete necrosis. In particular, subtotal necrosis of >75% or even total tumor necrosis was found in more than half of the patients treated with the Lipiodol-Mitomycin C combination. In contrast to most of the studies published to date, we abstained from giving temporarily or permanently occluding materials to avoid early deterioration of the target vessels [5, 9, 10, 17]. Nevertheless, our results are comparable to those reported over the last decade [5, 7, 9, 14, 17, 24–29].
One of the first publications dealing with pretransplant TACE was published by Majno et al. in 1997 [17]. They started applying pretransplant TACE in the late 1980s as a substantial part of the pretransplantion treatment. In 54 patients who received TACE prior to OLT they found complete tumor necrosis in 27% and they achieved at least a relevant downstaging in half of their patients [17]. In 2005 Sotiropoulos and colleagues reported comparable results, with tumor regression in more than 50% of the patients [27]. In our cohort, tumor regression—based on RECIST criteria—with a tumor shrinkage of >30% was found in 17 patients (40.4%). In those patients statistical analysis revealed a significant decrease in the maximum tumor diameter between the first and the last CT scan, whereas the number of tumors was stable in most cases.
Yao et al. presented a prospective study in 2005 that addressed the value of pretransplant locoregional treatment as well [26]. Relevant downstaging was achieved in this cohort in 70% of the patients, and complete tumor necrosis was achieved in 44% of the patients, which is superior to the results published by Majno et al. or Decaens et al. and to our results as well [9, 17]. Whereas Yao et al. presented a single-center study, Decaens et al. reported on a recent multicenter case-control study in 2005. They evaluated two groups separately, the TACE group and the untreated control group. Both consisted of 100 patients. They achieved a total or at least subtotal necrosis in 30% of the patients, which is similar to our data [9].
Which parameters substantially affect the degree of necrosis? Pompili and colleagues evaluated factors that might influence tumor necrosis [13]. A significant correlation was found between the preinterventional size of the HCC lesion (i.e., 3.0 cm) and the extent of necrosis after treatment. Other factors such as serum AFP, number of nodules, and time from treatment to OLT had no influence on the outcome [13].
In our cohort statistical analysis, tumor vascularization after TACE treatment, and Lipiodol washout were ruled out as the most important prognostic factors for tumor necrosis. Patients that demonstrated no relevant contrast enhancement on the postinterventional control CT images had significantly more extensive necrosis, and in contrast, six of the seven patients showing tumor progression in the follow-up examinations demonstrated a >25% vascularized tumor area. As we reported in two former published studies tumor progression during TACE was the most powerful predictor of posttransplant outcome [21, 22]. Whereas stable disease and tumor regression were associated with recurrence-free survival after OLT, a tumor progression according to RECIST criteria was correlated significantly more often with tumor relapse [21, 22].
Before this background the impact of partial necrosis with regard to posttransplant outcome is presently under discussion. Comparable to other series we also found insufficient tumor necrosis, i.e., <25% of necrotic tumor tissue, in 17% of the cases [9, 13, 25]. Insufficient necrosis was associated significantly more often with less frequent TACE treatments. This is in line with results of sequential TACE, suggesting that it is superior to single or only sporadic treatments [22, 30]. Incomplete tumor necrosis might raise concerns that the TACE procedure is not able to control tumor growth, and furthermore there might be worries about tumor recurrence after OLT. Correspondingly, Ravaioli and colleagues reported in 2004 that partial necrosis facilitates the recurrence of HCC in transplanted livers [14]. Of 54 patients who received transplants due to HCC, tumor recurred in 5, whereas only partial necrosis was found in the liver explants from 4 patients. Therefore, they concluded that partial necrosis might be an important risk factor for tumor recurrence after OLT. However, if our data did not support this result in detail, the insufficient response to the treatment must be scrutinized carefully and patients with tumor progression should be discussed critically for transplantation.
For the extent of necrosis induced by repeated TACE treatment, the initial liver function was of significant value. Patients who had elevated serum bilirubin levels of >2.0 mg/dl showed a significantly worse response to TACE treatment. Correspondingly a decreased albumin level of <35 g/L was associated with a smaller degree of tumor necrosis. Although there are no data published in the literature, there is obviously a relationship between the ability of the liver parenchyma to synthesize and to metabolize, on the one hand, and the efficacy of the local treatment, on the other.
The local effect of each TACE procedure is attributed to storage of the administered therapeutics and the prolonged exposure to the drug due to microvascular occlusion and deterioration of the reticuloendothelial system. Tumor necrosis was clearly more extensive in patients with a permanent Lipiodol accumulation in the HCC nodules than in those who presented with a relevant washout in follow-up investigations. In addition to the degree of devascularization and Lipiodol storage, the number of tumor nodules was important. Patients with unifocal tumor disease and unilobar involvement showed a better response to TACE, with more extended tumor necrosis than in patients with multifocal disease. Therefore, the less extensive necrosis found in bilateral and multifocal tumors might be related to the dosage of the administered therapeutics. In the past we administered the antitumoral drug via the segmental or even lobar arteries whenever possible, with the aim of targeting even small nodules by the TACE treatment. This procedure was attributed to the frequently multifocal nature of HCC and the fact that tumors are sometimes discovered only after LT in the pathological investigation. A recently published study by Sotiropoulos and colleagues ostensibly supports this notion [31]. They reported that even if all the radiological techniques are implemented, including MRI and CT, small lesions, <1 cm in diameter, were missed completely and tumor extent was underestimated in more than 50% of cases. However, the authors neglected to specify which imaging modalities they employed or to present the imaging protocols.
However, in routine pathological examination the liver specimen is cut into approximately 1.0-cm slices, whereas CT provides overlapping slices with a thickness of 3.0 mm. This involves the danger of missing very small lesions in the pathological evaluation. Therefore, future studies should take this into consideration, and ideally the CT reading and pathological evaluation should be adjusted to each other to obtain maximum information.
Conclusion
The authors are well aware of some substantial disadvantages of the present study. First and most importantly, it is a retrospective evaluation that comprises all the problems associated with retrospective data. In particular, retrospective analysis of the histological sections and the correct attribution to the CT images is burdened with some uncertainties. Nevertheless, we showed that even a simple treatment concept that disclaims from particulate embolization provides degrees of tumor necrosis comparable to those provided by more complex treatment regimens. Moreover, we could show a reliable correlation between the patterns of CT devascularization and Lipiodol accumulation or washout and the degree of tumor necrosis in pathomorphology.
References
El-Serag HB, Mason AC (1999) Rising incidence of hepatocellular carcinoma in the United States. N Engl J Med 340(10):745–750
Llovet JM, Burroughs A, Bruix J (2003) Hepatocelluar carcinoma. Lancet 6:1907–1917
Geschwind JF, Ramsey DE, Choti MA, Thuluvath PJ, Huncharek MS (2003) Chemoembolization of hepatocellular carcinoma: results of a metaanalysis. Am J Clin Oncol 26(4):344–349
Stippel DL, Kasper HU, Schleimer K, et al. (2003) Underestimation of nodules while staging hepatocellular carcinoma prior to neoadjuvant treatmtent on waiting list for transplantation. Transplant Proc 35(4):1423–1424
Graziadei IW, Sandmueller H, Waldenberger P, et al. (2003) Chemoembolization followed by liver transplantation for hepatocellular carcinoma impedes tumor progression while on the waiting list and leads to excellent outcome. Liver Transpl 9(6):557–563
Yao FY, Hirose R, LaBerge JM, et al. (2005) A prospective study on downstaging of hepatocellular carcinoma prior to liver transplantation. Liver Transpl 11(12):1505–1514
Porrett PM, Peterman H, Rosen M, et al. (2006) Lack of benefit of pre-transplant locoregional hepatic therapy for hepatocellular cancer in the current MELD era. Liver Transpl 12(4):665–673
Lencioni R, Della Pina C, Bartolozzi C (2005) Percutaneous image-guided radiofrequency ablation in the therapeutic management of hepatocellular carcinoma. Abdom Imaging 30(4):401–408
Decaens T, Roudot-Thoraval F, et al. (2005) Impact of pretransplantation transarterial chemoembolization on survival and recurrence after liver transplantation for hepatocellular carcinoma. Liver Transpl 11(7):767–775
Llovet JM, Real MI, Montana X, et al. (2002) Arterial embolisation or chemoembolisation versus symptomatic treatment in patients with unresectable hepatocellular carcinoma: a randomised controlled trial. Lancet 359(9319):1734–1739
Lo CM, Ngan H, Tso WK, Liu CL, Lam CM, Poon RT, Fan ST, Wong J (2002) Randomized controlled trial of transarterial lipiodol chemoembolization for unresectable hepatocellular carcinoma. Hepatology 35(5):1164–1171
Marelli L, Stigliano R, Triantos C, et al. Transarterial therapy for hepatocellular carcinoma: which technique is more effective? A systematic review of cohort and randomized studies. CardioVasc Interv Radiol 30(1):6–25
Pompili M, Mirante VG, Rondinara G, et al. (2005) Percutaneous ablation procedures in cirrhotic patients with hepatocellular carcinoma submitted to liver transplantation: assessment of efficacy at explant analysis and of safety for tumor recurrence. Liver Transpl 11(9):1117–1126
Ravaioli M, Grazi GL, Ercolani G, et al. (2004) Partial necrosis on hepatocellular carcinoma nodules facilitates tumor recurrence after liver transplantation. Transplantation 78(12):1780–1786
Sotiropoulos GC, Malago M, Molmenti EP, et al. (2005) Disease course after liver transplantation for hepatocellular carcinoma in patients with complete tumor necrosis in liver explants after performance of bridging treatments. Eur J Med Res 10(12):539–542
Veltri A, Grosso M, Martina MC, et al. (1998) Effect of preoperative radiological treatment of hepatocellular carcinoma before liver transplantation: a retrospective study. CardioVasc Interv Radiol 21(5):393–398
Majno PE, Adam R, Bismuth H, et al. (1997) Influence of preoperative transarterial lipiodol chemoembolization on resection and transplantation for hepatocellular carcinoma in patients with cirrhosis. Ann Surg 226(6):688–701
Edmonson H, Steiner P (1954) Primary carcinoma of the liver: a study of 100 cases among 48,900 necropsies. Cancer 7:462–503
Therasse P (2002) Measuring the clinical response. What does it mean? Eur J Cancer 38(14):1817–1823
Obed A, Beham A, Pullmann K, et al. (2007) Patients without hepatocellular carcinoma progression after transarterial chemoembolization benefit from liver transplantation. World J Gastroenterol 13(5):761–767
Otto G, Heise M, Moench C, et al. (2007) Transarterial chemoembolization before liver transplantation in 60 patients with hepatocellular carcinoma. Transplant Proc 39(2):537–539
Herber S, Schneider J, Brecher B, et al. (2005) [TACE: therapy of the HCC before liver transplantation—experiences]. Rofo 177(5):681–690
Mazzaferro V, Regalia E, Doci R, et al. (1996) Liver transplantation for the treatment of small hepatocellular carcinomas in patients with cirrhosis. N Engl J Med 334(11):693–699
Kim YS, Rhim H, Lim HK, et al. (2006) Completeness of treatment in hepatocellular carcinomas treated with image-guided tumor therapies: evaluation of positive predictive value of contrast-enhanced CT with histopathologic correlation in the explanted liver specimen. J Comput Assist Tomogr 30(4):578–582
Wong LL, Tanaka K, Lau L, et al. (2004) Pre-transplant treatment of hepatocellular carcinoma: assessment of tumor necrosis in explanted livers. Clin Transplant 18(3):227–234
Yao FY, Hirose R, LaBerge JM, et al. (2005) A prospective study on downstaging of hepatocellular carcinoma prior to liver transplantation. Liver Transpl 11(12):1505–1514
Sotiropoulos GC, Malago M, Molmenti E, Paul A, Nadalin S, Brokalaki EI, Verhagen R, Dirsch O, Gerken G, Lang H, Broelsch CE (2005) Efficacy of transarterial chemoembolization prior to liver transplantation for hepatocellular carcinoma as found in pathology. Hepatogastroenterology 52(62):329–332
Bharat A, Brown DB, Crippin JS, et al. (2006) Pre-liver transplantation locoregional adjuvant therapy for hepatocellular carcinoma as a strategy to improve longterm survival. J Am Coll Surg 203(4):411–420
Cheng YF, Huang TL, Chen TY, et al. (2005) Impact of pre-operative transarterial embolization on the treatment of hepatocellular carcinoma with liver transplantation. World J Gastroenterol 11(10):1433–1438
Jaeger HJ, Mehring UM, Castaneda F, Hasse F, Blumhardt G, Loehlein D, Mathias KD (1996) Sequential transarterial chemoembolization for unresectable advanced hepatocellular carcinoma. CardioVasc Interv Radiol 19(6):388–396
Sotiropoulos GC, Malago M, Molmenti E, et al. (2005) Liver transplantation for hepatocellular carcinoma in cirrhosis: Is clinical tumor classification before transplantation realistic? Transplantation 79(4):483–487
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Herber, S., Biesterfeld, S., Franz, U. et al. Correlation of Multislice CT and Histomorphology in HCC Following TACE: Predictors of Outcome. Cardiovasc Intervent Radiol 31, 768–777 (2008). https://doi.org/10.1007/s00270-007-9270-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00270-007-9270-8