Abstract
Purpose
To determine the role of prophylactic N-acetylcysteine in the prevention of contrast-induced nephrotoxicity.
Methods
One hundred and sixteen patients undergoing noncoronary angiography, with or without pre-existing renal impairment, were randomly assigned to receive prophylactic oral N-acetylcysteine or no treatment. Serum creatinine (sCr) was measured prior to angiography and 48 hr after the procedure. Urine samples were collected before and after the examination for measurement of malondialdehyde (MDA) concentration. Contrast-induced nephrotoxicity (CIN) was defined as a rise in serum creatinine of 0.5 mg/dl (44 mmol/l) at 48 hr.
Results
Complete data were available on 106 patients, 53 of whom had received N-acetylcysteine. There were no significant differences between the two groups in baseline characteristics, type of angiogram, or volume and concentration of contrast used. Three patients (2.8%), all of whom had received N-acetylcysteine, developed CIN. In the N-acetylcysteine group, the mean serum creatinine in patients with renal impairment was 151.0 ± 44.2 μmol/l prior to the procedure and 155.6 ± 48.6 μmol/l (p = 0.49) after the procedure. Respective values for those without renal impairment were 79.6 ± 15.1 μmol/l and 81.2 ± 20.0 μmol/l (p = 0.65). In the group that had not received N-acetylcysteine, the mean serum creatinine levels before and after the procedure were 150.0 ± 58.1 and 141.4 ± 48.0 μmol/l (p = 0.17) in patients with renal impairment and 79.7 ± 14.2 and 81.4 ± 15.4 μmol/l (p = 0.34) in those without renal impairment. In both groups, no significant change in urinary MDA concentration was observed.
Conclusion
There is no benefit to the prophylactic administration of N-acetylcysteine in patients undergoing peripheral angiography using current contrast media.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Contrast-induced nephrotoxicity (CIN) is a significant cause of morbidity and mortality [1]. Although the incidence in the general population is reported to be less than 2% [2], in high-risk patients, namely those with both diabetes and pre-existing renal impairment, the reported incidence ranges from 9% to 50% [2–5].
The mechanism of renal injury is poorly understood and is likely to be multifactorial. Experimental models have demonstrated that following the injection of radiographic contrast media there is transient intrarenal vasodilatation followed by a longer period of vasoconstriction [6–8]. The resulting reduction in glomerular filtration rate (GFR) is compounded by a further response likely to be mediated by oxygen free radicals and resulting in direct cellular damage [9–14]. Previous work by our group demonstrated a significant rise in urinary malondialdehyde (a urinary marker of free radical injury) concentration following infusion of low-osmolar radiographic contrast medium in humans [13].
The administration of antioxidants may therefore prevent CIN and against this background recent randomized controlled studies have compared the prophylactic administration of the antioxidant N-acetylcysteine with placebo in high-risk patients undergoing contrast-enhanced computed tomography [15] and coronary angiography [16–22]. The results of these individual studies are conflicting. Furthermore, three meta-analyses of the published literature have concluded a significant benefit in using prophylactic N-acetylcysteine in patients with chronic renal insufficiency [23–25] whilst others have failed to reach a definite conclusion [26–29].
The aims of this study were twofold: to determine whether the prophylactic use of N-acetylcysteine reduces the incidence of CIN in patients undergoing peripheral angiography, and to test the hypothesis that N-acetylcysteine prevents contrast-induced oxidative damage to the kidney, by measuring levels of urinary malondialdehyde (MDA).
Materials and Methods
Study Patients
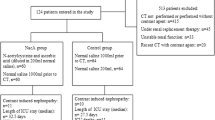
Patients referred for noncoronary diagnostic angiography, i.e., head and neck, extremity and visceral abdominal angiography, with or without renal impairment, were considered eligible for the study. Patients with acute renal failure and patients with renal transplants were excluded from the study. All patients underwent diagnostic angiography using either a nonionic iso-osmolar or low-osmolar radiographic contrast agent.
The study protocol was approved by the Local Research and Ethics Committee and all patients gave written informed consent.
Study Protocol
The patients were randomly assigned, by selection of a sealed envelope, to receive N-acetylcysteine at a dose of 600 mg twice daily the day before the procedure and on the day of the procedure. The volume, concentration of iodine per milliliter, and type of contrast agent used varied according to the site of interest. Iopamidol, a low-osmolar non-ionic contrast medium (Niopam, Bracco) was used for lower limb angiography at an iodine concentration of 300 mg/ml, and in upper limb and mesenteric angiography at an iodine concentration of 370 mg/ml. Iodixanol, an iso-osmolar, non-ionic contrast medium (Visipaque, Amersham Health) was used for renal angiography at an iodine concentration of 320 mg/ml.
Serum creatinine was measured before the examination and 48 hr after the administration of the contrast medium. Renal impairment was defined as a serum creatinine concentration above 1.2 mg/dl (106 μmol/l). Urine samples were collected before and after the examination for measurement of MDA concentration. These data were correlated with the urine creatinine concentration. CIN was defined as an acute rise in the serum creatinine of at least 0.5 mg/dl (44 μmol/l) 48 hr after the administration of the radiographic contrast medium.
Statistical Analysis
Student’s t-test was used to compare the baseline characteristics and procedural details between the two groups. The t-test and Wilcoxon ranked nonparametric test were applied to the data relating to the serum and urinary variables.
Results
Study Patients
From October 2001 to October 2002, 116 patients were enrolled in the study. Of the 116 patients, 58 were randomized to receive prophylactic N-acetylcysteine and 58 were randomized to receive no treatment. Ten patients with incomplete study data were excluded from the analysis. The demographic data and cardiovascular history were similar between the two groups (Table 1). The proportion of patients with renal impairment was higher in the group receiving N-acetylcysteine: 50% versus 34%. The baseline mean serum creatinine concentration was similar between the two groups as was the estimated creatinine clearance calculated using the Cockcoft-Gault formula. There was no difference between the types of procedure or volume, strength and type of contrast used between the two groups (Table 2). Twenty-one patients (47%) with renal impairment received iso-osmolar contrast compared with 7 (11%) without renal impairment.
The mean time to collection of the urine samples for MDA and creatinine analysis was similar between the two groups.
Effect on Serum Creatinine and Urinary Malondialdehyde Concentrations
The overall incidence of CIN was 2.8% (n = 3) and in patients with pre-existing renal impairment the incidence was 4.4% (n = 2). All 3 patients were in the group receiving N-acetylcysteine. Two of the patients had pre-existing renal impairment and one patient had normal renal function prior to angiography. There was no significant difference in serum creatinine concentration or urinary MDA/creatinine ratio between the two groups before or after angiography (Tables 3, 4).
Discussion
This study demonstrates that the incidence of CIN in patients attending for diagnostic peripheral angiography is low, i.e., 2.8%. Even in patients with pre-existing renal impairment the incidence of CIN was 4.4%.
The reasons for the low incidence of CIN are unclear. CIN is thought to result from complex changes in intrarenal blood flow causing free radical formation and subsequent tubular damage. Many ways of preventing CIN have been tried, of which hydration [30], the use of low-osmolar and iso-osmolar contrast agents [4, 31–35] and prophylactic N-acetylcysteine [16–24] have been shown to be of benefit but the data are inconclusive.
In this study, intravenous hydration was not part of the study protocol in order to represent the reality of peripheral angiographic procedures, which are often performed as an emergency or as a day case.
The type of contrast agent may have influenced the incidence of CIN. The use of low-osmolar contrast agents is associated with a lower incidence of CIN [4, 31–35], although these studies still report an incidence of 12–50%. More promising results have been reported with the use of iso-osmolar contrast agents, which theoretically may not cause volume depletion and subsequent alteration in intrarenal blood flow [35, 36]. Aspelin et al. showed an increase in the post-angiography creatinine concentration of 0.5 mg/dl in 3% of patients receiving the iso-osmolar agent iodixanol, compared with 26% of patients receiving the low-osmolar agent iohexol [35]. Chalmers and Jackson also reported an incidence of CIN of 3.7% in patients receiving iodixanol compared with 10% in the iohexol group [36]. In the current study, 46.6% of patients with pre-existing renal impairment received iodixanol and the incidence of CIN was 4.4%. Furthermore, all 3 patients who developed CIN had received iopamidol. However, on subgroup analysis there was no difference in mean creatinine or MDA/Cr ratio after angiography according to the type of contrast agent used.
This is contrary to our previous findings in which infusion of intra-arterial iopamidol did result in a significant rise in urinary MDA levels suggesting that part of the injury in CIN is free-radical-induced [13]. On this basis the current study was designed to test the hypothesis that the antioxidant N-acetylcysteine may prevent CIN. However, no benefit was observed and all 3 patients who developed CIN had received N-acetylcysteine. These results are in keeping with other studies [19–22] and it is interesting that in one of these studies [21] an iso-osmolar contrast agent was used. Furthermore, four recent meta-analyses of all the trials that have evaluated the use of prophylactic N-acetylcysteine have concluded that there is marked heterogeneity in the available data, with some trials reporting a significant benefit and others reporting no renoprotective effect, so that the routine use of N-acetylcysteine cannot be supported [26–29].
However, the current study has its limitations. The power calculations were based on a higher incidence of CIN and therefore the sample size may be too small. Furthermore this study did not evaluate the clinically relevant parameters of outcome such as mortality, morbidity, and length of hospitalization.
Clearly the etiology of CIN is more complex than currently understood. Large studies are needed to clarify the pathophysiology before useful preventive measures can be introduced. Based on the results of our study there is no role for the routine prophylactic administration of N-acetylcysteine in patients undergoing peripheral angiography with either low-osmolar or iso-osmolar contrast agents.
References
Rihal CS, Textor SC, Grill DE, et al (2002) Incidence and prognostic importance of acute renal failure after percutaneous coronary intervention. Circulation 105:2259–2264
Lautin EM, Freeman NJ, Schoenfeld AH, et al (1991) Radiocontrast-associated renal dysfunction: Incidence and risk factors. AJR Am J Roentgenol 157:66–68
Cochran ST, Wong WS, Roe DJ (1983) Predicting angiography-induced acute renal function impairment: Clinical risk model. AJR Am J Roentgenol 141:1027–1033
Manske CL, Sprafka JM, Strony JT, et al (1990) Contrast nephropathy in azotemic diabetic patients undergoing coronary angiography. Am J Med 89:615–620
Parfrey PS, Griffiths SM, Barrett BJ, et al (1989) Contrast material-induced renal failure in patients with diabetes mellitus, renal insufficiency, or both. A prospective controlled study. N Engl J Med 320:143–149
Caldicott WJ, Hollenberg NK, Abrams HL (1970) Characteristics of response of renal vascular bed to contrast media. Evidence for vasoconstriction induced by the renin–angiotensin system. Invest Radiol 5:539–547
Katzberg RW, Schulman G, Meggs LG, et al (1983) Mechanism of the renal response to contrast medium in dogs: Decrease in renal function due to hypertonicity. Invest Radiol 18:74–80
Sherwood T, Lavender JP (1969) Does renal blood flow rise or fall in response to diatrizoate? Invest Radiol 4:327–328
Bakris GL, Lass N, Gaber AO, et al (1990) Radiocontrast medium-induced declines in renal function: A role for oxygen free radicals. Am J Physiol 258:F115–120
Yoshioka T, Fogo A, Beckman JK (1992) Reduced activity of antioxidant enzymes underlies contrast media-induced renal injury in volume depletion. Kidney Int 41:1008–1015
DiMari J, Megyesi J, Udvarhelyi N, et al (1997) N-acetyl cysteine ameliorates ischemic renal failure. Am J Physiol 272:F292–F298
Bakris GL, Gaber AO, Jones JD (1990) Oxygen free radical involvement in urinary Tamm-Horsfall protein excretion after intrarenal injection of contrast medium. Radiology 175:57–60
Sandhu C, Newman DJ, Morgan R, et al (2002) The role of oxygen free radicals in contrast-induced nephrotoxicity. Acad Radiol 9 (Suppl 2):S436–437
Drager LF, Andrade L, Barros de Toledo JF, et al (2004) Renal effects of N-acetylcysteine in patients at risk for contrast nephropathy: Decrease in oxidant stress-mediated renal tubular injury. Nephrol Dial Transplant 19:1803–1807
Tepel M, van der Giet M, Schwarzfeld C, et al (2000) Prevention of radiographic-contrast-agent-induced reductions in renal function by acetylcysteine. N Engl J Med 343:180–184
Shyu KG, Cheng JJ, Kuan P (2002) Acetylcysteine protects against acute renal damage in patients with abnormal renal function undergoing a coronary procedure. J Am Coll Cardiol 40:1383–1388
Diaz-Sandoval LJ, Kosowsky BD, Losordo DW (2002) Acetylcysteine to prevent angiography-related renal tissue injury (the APART trial). Am J Cardiol 89:356–358
Kay J, Chow WH, Chan TM, et al (2003) Acetylcysteine for prevention of acute deterioration of renal function following elective coronary angiography and intervention: A randomized controlled trial. JAMA 289:553–558
Durham JD, Caputo C, Dokko J, et al (2002) A randomized controlled trial of N-acetylcysteine to prevent contrast nephropathy in cardiac angiography. Kidney Int 62:2202–2207
Briguori C, Manganelli F, Scarpato P, et al (2002) Acetylcysteine and contrast agent-associated nephrotoxicity. J Am Coll Cardiol 40:298–303
Allaqaband S, Tumuluri R, Malik AM, et al (2002) Prospective randomized study of N-acetylcysteine, fenoldopam, and saline for prevention of radiocontrast-induced nephropathy. Catheter Cardiovasc Interv 57:279–283
Fung JW, Szeto CC, Chan WW, et al (2004) Effect of N-acetylcysteine for prevention of contrast nephropathy in patient with moderate to severe renal insufficiency: A randomized trial. Am J Kidney Dis 43:801–808
Birck R, Krzossok S, Markowetz F, et al (2003) Acetylcysteine for prevention of contrast nephropathy: Meta-analysis. Lancet 23:598–603
Alonso A, Lau JB, Jaber BL, et al (2004) Prevention of radiocontrast nephropathy with N-acetylcysteine in patients with chronic kidney disease: A meta-analysis of randomized controlled trials. Am J Kidney Dis 43:1–9
Misra D, Leibowitz K, Gowda RM, et al (2004) Role of N-acetylcysteine in prevention of contrast-induced nephrotoxicity after cardiovascular procedures: A meta-analysis. Clin Cardiol 27:607–610
Nallamothu BK, Shojania KG, Saint S, et al (2004) Is acetylcysteine effective in preventing contrast-related nephropathy? A meta-analysis. Am J Med 117:938–947
Bagshaw SM, Ghali WA (2004) Acetylcysteine for prevention of contrast-induced nephropathy after intravascular angiography: A systematic review and meta-analysis. BMC Med 22:38
Pannu N, Manns B, Lee H, et al (2004) Systematic review of the impact of N-acetylcysteine on contrast nephropathy. Kidney Int 65:1366–1374
Kshirsagar AV, Poole C, Mottl A, et al (2004) N-acetylcysteine for the prevention of radiocontrast-induced nephropathy: A meta-analysis of prospective controlled trials. J Am Soc Nephrol 15:761–769
Asif A, Epstein M (2004) Prevention of radiocontrast-induced nephropathy. Am J Kidney Dis 44:12–24
Rudnick MR, Goldfarb S, Wexler L, et al (1995) Nephrotoxicity of ionic and nonionic contrast media in 1196 patients: A randomized trial. The Iohexol Cooperative Study. Kidney Int 47:254–261
Lautin EM, Freeman NJ, Schoenfeld AH, et al (1991) Radiocontrast-associated renal dysfunction: A comparison of lower-osmolality and conventional high-osmolality contrast media. AJR Am J Roentgenol 157:59–65
Taliercio CP, Vlietstra RE, Ilstrup DM, et al (1991) A randomized comparison of the nephrotoxicity of iopamidol and diatrizoate in high risk patients undergoing cardiac angiography. J Am Coll Cardiol 17:384–390
Barrett BJ, Parfrey PS, Vavasour HM, et al (1992) Contrast nephropathy in patients with impaired renal function: High versus low osmolar media. Kidney Int 41:1274–1279
Aspelin P, Aubry P, Fransson SG, et al (2003) Nephrotoxic effects in high-risk patients undergoing angiography. N Engl J Med 348:491–499
Chalmers N, Jackson RW (1999) Comparison of iodixanol and iohexol in renal impairment. Br J Radiol 72:701–703
Acknowledgments
The authors would like to acknowledge the help of the staff of the surgical vascular unit, blood pressure unit and angiography suite at St. George’s Hospital. Special thanks also go to the late Dr. D.J. Newman for performing all the laboratory analyses.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sandhu, C., Belli, AM. & Oliveira, D.B. The Role of N-Acetylcysteine in the Prevention of Contrast-Induced Nephrotoxicity. Cardiovasc Intervent Radiol 29, 344–347 (2006). https://doi.org/10.1007/s00270-005-0127-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00270-005-0127-8