Abstract
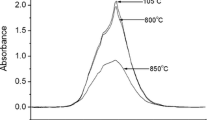
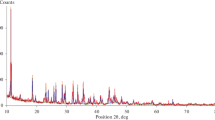
Muscovite-2M1 shows a major phase transition at about 800°C, which is generally attributed in the literature to the structural dehydroxylation process, although a number of structural models have been proposed for the dehydroxylated phase, and different transformation mechanisms have also been put forward. The observed first order transformation involves an increase in the cell volume, and it is not clear to date how the cell expansion is related to the loss of hydroxyl groups. The phase change has been re-investigated here by in situ high temperature powder diffraction, both in non-isothermal and isothermal modes, to combine for the first time the structural and the kinetic interpretation of the transformation. The results unequivocally confirm that the reaction taking place in the temperature range 700–1000°C is truly a dehydroxylation process, involving the nucleation and growth of the high temperature dehydroxylated phase, having Al in 5-fold coordination. Structural simulations of the basal peaks of the powder diffraction patterns indicate that the model originally proposed by Udagawa et al. (1974) for the dehydroxylated phase correctly describes the high temperature phase. The kinetic analysis of the isothermal data using an Avrami-type model yields values for the reaction order compatible with a reaction mechanism limited by a monodimensional diffusion step. Apparent activation energy of the process in vacuum is about 251 kJ/mol. Experiments carried out at temperatures much higher than the onset temperature of the reaction show that the dehydroxylation reaction overlaps with the reaction of formation of mullite, the final product in the reaction pathway.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Author information
Authors and Affiliations
Additional information
Received: 24 April 1998 / Revised, accepted: 12 October 1998
Rights and permissions
About this article
Cite this article
Mazzucato, E., Artioli, G. & Gualtieri, A. High temperature dehydroxylation of muscovite-2M1: a kinetic study by in situ XRPD. Phys Chem Min 26, 375–381 (1999). https://doi.org/10.1007/s002690050197
Issue Date:
DOI: https://doi.org/10.1007/s002690050197