Abstract
Background
Residual disease (RD) has been described as one of the most relevant prognostic factors after radical surgical resection for incidental gallbladder cancer (IGC). The purpose of the present study was to analyze patterns of RD and determinant prognostic factors in patients undergoing re-resection for IGC.
Methods
Patients undergoing re-exploration due to IGC between 1990 and 2014 were identified in two referral centers from different South-American countries. Patients submitted to a radical definitive operation were included in the study. Demographics and tumor-treated related variables were analyzed in correlation with RD and survival. The site of RD, local (gallbladder bed) or regional (lymph nodes and bile duct) was correlated with disease-specific survival (DSS).
Results
Of 265 patients with IGC submitted to surgery, 168 underwent a radical re-resection and RD was found in 58 (34.5%). Demographic, clinical and surgical variables were compared between both centers showing differences in type of resection, laparoscopic approach, T stages and disease stage. Location of RD was regional in 34 (20.2%) and local in 24 (14.3%), and no residual disease was found in 110 (65.5%) patients. T stage (T1b = 20%, T2 = 23.8%, T3 = 71.7%, p < 0.001) and disease stage (p < 0.001) were independent predictors of RD. Finding RD at any location reduced the DSS in comparison with non-RD patients (19.6 months vs. 62.7 months p < 0.001). No differences in DSS according to the location of RD were found, and all anatomic sites were equally poor (p = 0.27). RD at any site predicted DSS (p < 0.001), independently of all other IGC variables.
Conclusions
IGC presented similar clinical parameters in two different countries of South America. RD was demonstrated as the most critical prognostic variable in patients with IGC treated by a radical resection. The presence of RD was associated with poor outcome, independently of any anatomic location. Future studies incorporating neoadjuvant chemotherapy in the treatment of patients with prognostic factors for RD are required to improve survival in this entity.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Optimal treatment for incidental gallbladder cancer (IGC) is still a matter of debate. If possible, radical re-resection offers the only potential cure, but there is still controversy about the appropriate selection of patients for the second operation, according to the extent of the disease [1]. The objective of re-resection is the eradication of areas infiltrated by loco regional residual disease (RD) [2]. Disease extent presents a considerable variation, from gross metastatic disease found at the time of re-exploration to unsuspected microscopic disease recognized after complete resection [3, 4]. Previous publications have reported parenchymal involvement in hepatic resections for IGC at a rate of 20–56% [1, 5, 6]. Tumor stage associates with the presence of RD in the liver and the lymph nodes [7]. Prognosis had been associated with nodal status, and lymph nodes metastasis had been individualized as the most frequent location of additional disease [8]. The prognostic benefit from re-resection in patients with RD is still undetermined. In a previous study from our group, all patients with RD died in a period less than 15 month [9]. Other studies have reported poor outcome especially in patients with RD located in the liver parenchyma [10, 11].
The purpose of this study was to analyze the patterns of residual disease and determinant prognostic factors of survival in patients that underwent a radical re-resection for IGC in HPB centers from two different regions of South America.
Methods
A retrospective analysis of data from patients with diagnosis of gallbladder cancer referred to two HPB centers in South America between June 1990 and June 2014 was performed. Identification of patients was from the prospective computed database of both Institutions. Assessment in the preoperative setting included clinical history, routine blood tests, CA 19-9, abdominal ultrasonography and computed tomographic scan (CT) of the chest, abdomen and pelvis with triphasic contrast of the liver.
A total of 260 patients with IGC were identified, being diagnosed by subsequent pathologic examination after cholecystectomy that defined the T stage. Twenty-seven T1a patients were excluded from the analysis. Surgical approach for radical resection consisted of a 4B-5 segmental or wedge liver resection with supra-duodenal lymphadenectomy, selective bile duct and port resection. Operative mortality was defined as death related to the surgical procedure and within 30 days of the resection. The explanted specimens were analyzed by experienced pathologists in each institution. The presence of liver, lymph nodes and port sites involvement, surgical margins, perineural and vascular invasion was reviewed. A complete removal of any clinically evident tumor lesion(s) with negative pathological margins was defined as a curative resection (R0). Any infiltration of the resection margin with tumor cells in the histological specimen was categorized as R1 resection. According to Butte et al. [10], RD was defined in the pathological specimen as local (tumor infiltration in the gallbladder bed, continuous liver involvement) and regional (bile duct invasion, positive lymph nodes). Clinical staging of IGC was performed by the American Joint Committee on Cancer (AJCC) staging system (7th edition) [12]. Adjuvant chemotherapy was given in patients with RD (gemcitabine–platinum-based regimens). The postoperative follow-up was performed by physical examination and CT at 6-month intervals.
Demographic and clinical variables, local pathological stages, operative procedures, complications, pathological features and prognostic factors for long-term survival were analyzed. A comparison of variables in both HPB centers was performed.
Statistical analyses were performed using SPSS software v15 with univariate and multivariate logistic regression to assess the relationship between RD at re-operation and pathological variables. Significance was calculated by Chi-square test and correlations using cross-tabs. Survival curves were estimated by Kaplan–Meier method and compared by the long-rank test. p values of <0.05 were considered significant.
Results
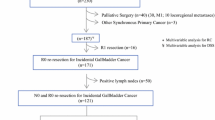
Of the 265 patients with IGC, 168 (63.3%) were candidates for curative treatment and accounts for the population analyzed in the present study. The other 97 patients were contraindicated for radical surgery due to extent of the disease diagnosed in the preoperative or intraoperative setting. Non-curative factors in 97 cases were severe liver invasion in 48 (49.5%), bile duct invasion in 31 (32%) and extrahepatic organ invasion in 18 (18.5%). The study included 137 females (81.5%), and the median age was 56.3 years (30–78). Demographic and tumor-treated related variables are shown in Table 1. One hundred and forty-two patients (53%) were referred from other institutions.
Operative findings and residual disease
Median time from initial cholecystectomy to re-exploration was 3.5 month (range 0.3–8 month). Complete resection was achieved in all patients. Surgical procedures were as listed: segments 4B-5 open resection (34/168, 20.2%), laparoscopic wedge resection (26/168, 15.5%), open wedge resection (108/168, 64.2%). All patients underwent N1 portal lymphadenectomy. Median lymph node count was: 7 (range 0–21). Bile duct excision (12/168, 7.1%), extrahepatic organ resection (10/168, 5.9%) and port site resection (6/168, 3.5%) were performed in selected patients (Table 2).
RD was identified in 58 patients (34.5%) with the following localizations: liver 41.4% (24/58), lymph nodes 60.3% (35/58) and bile duct 21% (12/58). Based in the T stage on the cholecystectomy specimen, RD was found in 20% of T1b, 23.8% of T2 and 71.7% of T3 patients (Table 2). According Butte el al. proposed classification, local RD (only gallbladder fossa) was found in 41% (24/58) and regional RD in 59% (34/58). There was no predominance of regional RD over local RD according to T stage (Table 2). In 18 (31%) patients with regional RD, more than one site was found.
Comparison of variables between both centers demonstrated differences only in type of resection, laparoscopic approach, T stages and disease stage (Table 3).
Statistical analysis
On univariate and multivariate analysis T stage (p < 0.001), TNM stage (p < 0.001) and lympho-vascular invasion (p < 0.001) were associated with RD at re-operation (Table 4). Patients with T3 tumors had a higher incidence of RD in comparison with T2 and T1b patients (p < 0.001). Additionally, patients with stage III had higher incidence of RD than patients with stage II disease (p < 0.001). No statistical difference was found between patients with stages I and II. Poor and moderately differentiated tumors had higher association with RD than well differentiated tumors (p = 0.05 and 0.02).
Overall survival
Median follow-up was 36 months (13.5 month in patients with RD versus 60 months in patients with non-RD). RD at any site was associated with a significant reduction in DSS [19.6 months (RD) vs. 62.7 months (non-RD) p < 0.001] as shown in Fig. 1. There were no differences in DSS according to the site of RD, with all anatomic sites been equally poor (p = 0.27) (Fig. 2). Any site of RD predicted DSS (p < 0.001), independently of all other tumor prognostic variables.
Discussion
Gallbladder cancer is a common disease in South America with predominant areas that demonstrated a geographic pattern in relation with the Andes mountains [13]. In this region, gallbladder carcinoma is found incidentally in 50–70% of the patients, following an elective cholecystectomy for presumed benign disease [11]. In the present study, no differences were found in preoperative variables among patients treated in both centers. However, a higher predominance of females and a younger age at diagnosis was demonstrated in a previous study based on the American College of Surgeons-National Surgical Quality Improvement Program (ACS-NSQIP) [14]. Time to referral of a gallbladder cancer is still a problem in Latin-American countries, as we see in our population, with a 3.5-month period. It may be explained by delays in the diagnosis and appropriate counseling, geographic location and access to an HPB center. However, the time to re-exploration is still under debate, with some authors proposing a minimum delay of 3 months, while others indicate an optimal time between 4 and 8 weeks after cholecystectomy [11, 15, 16]. Although radical surgery is the best curative option, there is an underutilization of the procedure, not only in the USA but in other countries [17,18,19,20]. However, appropriate treatment of incidental GC is still a matter of controversy [17]. Radical re-resection offers the only potential for cure, but there is still a debate over the appropriate selection and the extent of the resection in the second operation according to each T stage [18]. As depicted in Table 1, the present series demonstrated a lower incidence of T3 and less poorly differentiated tumors in comparison with others [10, 19, 20]. Surgical approach and type of resection were different in each center with a predominance of laparoscopic and wedge resections in the Chilean Institution. The laparoscopic approach is expanding in the field as shown in the present study with 15% (n = 26) of the procedures performed laparoscopically [21]. Other differences between both centers were the T stage and TNM stage with a predominance of T1b and T2 in the Chilean center. Prevalence of the disease in Chile over Argentina and unified criteria for proper pathological evaluation can explain the differences found between both countries [21, 22]. The presence of RD on re-resection has been shown to increase with T stage progression defined in the pathological study of the cholecystectomy specimen [8]. Evidence has shown that is the presence of RD in the explant tissue of the second operation and not the T stage of IGC, the variable that ultimately dictates outcome [6, 8,9,10]. Patients with RD will have a worst prognosis independent of the T stage, but as shown in the present study, patients with a T3 IGC will have a triple chance of having RD at re-operation [10]. RD had been associated with a poor overall and recurrence free survival [6, 9, 18, 23]. In the present analysis, local and/or regional RD was found in 34% of the patients, a figure in concordance with previous studies, but less than the 49% shown in a recent US multicenter study [10, 19, 24]. As shown by Gil et al. [9], RD predominates in the liver (15–36%) or in the lymph nodes (11–35%) and there is no uniform pattern of distribution. A median of 7 dissected nodes in the present series correlates with previous studies [25]. The number of lymph nodes evaluated is critical for stratifying recurrence risk and cancer-specific survival as demonstrated by Ito et al. [7]. Radical surgery of GC with positive lymph nodes can lead to long-term survival in only a subset of patients [26]. Ethun et al. [19] recently published the Gallbladder Risk Score, based on 4 parameters: T stage, grade of differentiation, lympho-vascular invasion and perineural invasion. The model can delineate groups of patients with RD on the radical re-operation and stratify them for neoadjuvant chemotherapeutic regimens [19]. As we see in our study, except for perineural invasion, the other three parameters demonstrated significance in the UV and MV analysis (Table 4). It can be explained by the lower rate of perineural invasion (7%) found in the present study in contrast with other series that range between 39 and 63% [19, 24]. No differences were found according to the location of RD in concordance with previous reports [11, 24]. A neoadjuvant approach, selecting patients with elevated risk of RD that most likely will benefit from re-operation and definitive resection, might be favorable. However, no significant advantages with neoadjuvant chemotherapy have been demonstrated yet in IGC. Some studies have shown positive results with systemic chemotherapy followed by surgery in advanced tumors [27,28,29]. A randomized trial of neoadjuvant chemotherapy according to each T stage in IGC will help to define which are better options for patients with RD.
In conclusion, IGC presented similar clinical parameters in two different countries of South America. RD was a critical prognostic factor in patients after a standardized radical resection in both HPB centers. Long-term survival was poor in the presence of RD, independent of the anatomic location. Future studies with neoadjuvant chemotherapy in patients with prognostic factors for RD will be needed to improve survival in this entity.
References
Lendoire JC, Gil L, Duek F et al (2012) Relevance of residual disease after liver resection for incidental gallbladder cancer. HPB (Oxford) 14(8):548–553
Butte JM, Waugh E, Meneses M et al (2010) Incidental gallbladder cancer: analysis of surgical findings and survival. J Surg Oncol 102(6):620–625
Maker AV, Butte JM, Oxenberg J et al (2012) Is port site resection necessary in the surgical management of gallbladder cancer? Ann Surg Oncol 19(2):409–417
Goetze TO, Paolucci V (2010) Adequate extent in radical re-resection of incidental gallbladder carcinoma: analysis of the German Registry. Surg Endosc 24(9):2156–2164
Smith GCS, Parks RW, Madhavan KK et al (2003) A 10-year experience in the management of gallbladder cancer. HPB (Oxford) 5(3):159–166
Fuks D, Regimbeau JM, Le Treut Y-P et al (2011) Incidental gallbladder cancer by the AFC-GBC-2009 study group. World J Surg 35(8):1887–1897. https://doi.org/10.1007/s00268-011-1134-3
Ito H, Ito K, D’Angelica M et al (2011) Accurate staging for gallbladder cancer: implications for surgical therapy and pathological assessment. Ann Surg 254(2):320–325
Pawlik TM, Gleisner AL, Vigano L et al (2007) Incidence of finding residual disease for incidental gallbladder carcinoma: implications for re-resection. J Gastrointest Surg 11(11):1478–1487
Gil L, Lendoire J, Duek F et al (2014) Cirugía radical en el cáncer de vesícula incidental: valor del hallazgo de enfermedad residual en el estudio histopatológico diferido. Cir Esp 92(3):168–174
Butte JM, Kingham TP, Gönen M et al (2014) Residual disease predicts outcomes after definitive resection for incidental gallbladder cancer. J Am Coll Surg 219(3):416–429
Ethun CG, Postlewait LM, Le N et al (2017) Association of optimal time interval to re-resection for incidental gallbladder cancer with overall survival. JAMA Surg 152(2):143
Edge SB, Byrd DR, Compton CC et al (eds) (2010) AJCC cancer staging manual, 7th edn. Springer, New York
Arroyo GF, Gentile A, Parada LA (2016) Gallbladder cancer: South American experience. Chin Clin Oncol 5(5):67
Pitt SC, Jin LX, Hall BL et al (2014) Incidental gallbladder cancer at cholecystectomy. Ann Surg 260(1):128–133
Yip VS, Gomez D, Brown S et al (2014) Management of incidental and suspicious gallbladder cancer: focus on early referral to a tertiary centre. HPB 16(7):641–647
Tsirlis T, Ausania F, White SA et al (2015) Implications of the index cholecystectomy and timing of referral for radical resection of advanced incidental gallbladder cancer. Ann R Coll Surg Engl 97(2):131–136
Sternby Eilard M, Lundgren L, Cahlin C et al (2017) Surgical treatment for gallbladder cancer—a systematic literature review. Scand J Gastroenterol 52(5):505–514
Aloia TA, Járufe N, Javle M et al (2015) Gallbladder cancer: expert consensus statement. HPB 17(8):681–690
Ethun CG, Postlewait LM, Le N et al (2017) A novel pathology-based preoperative risk score to predict locoregional residual and distant disease and survival for incidental gallbladder cancer: a 10-institution study from the U.S. extrahepatic biliary malignancy consortium. Ann Surg Oncol 24(5):1343–1350
Shindoh J, de Aretxabala X, Aloia TA et al (2015) Tumor location is a strong predictor of tumor progression and survival in T2 gallbladder cancer. Ann Surg 261(4):733–739
Adsay NV, Bagci P, Tajiri T et al (2012) Pathologic staging of pancreatic, ampullary, biliary, and gallbladder cancers: pitfalls and practical limitations of the current AJCC/UICC TNM staging system and opportunities for improvement. Semin Diagn Pathol 29(3):127–141
Roa I, Ibacache G, Muñoz S et al (2014) Gallbladder cancer in Chile: pathologic characteristics of survival and prognostic factors: analysis of 1366 cases. Am J Clin Pathol 141(5):675–682
Butte JM, Gönen M, Allen PJ et al (2011) The role of laparoscopic staging in patients with incidental gallbladder cancer. HPB (Oxford) 13(7):463–472
Creasy JM, Goldman DA, Gonen M et al (2017) Predicting residual disease in incidental gallbladder cancer: risk stratification for modified treatment strategies. J Gastrointest Surg 21(8):1254–1261
Birnbaum DJ, Viganò L, Russolillo N et al (2015) Lymph node metastases in patients undergoing surgery for a gallbladder cancer. Extension of the lymph node dissection and prognostic value of the lymph node ratio. Ann Surg Oncol 22(3):811–818
Shibata K, Uchida H, Iwaki K et al (2009) Lymphatic invasion: an important prognostic factor for stages T1b–T3 gallbladder cancer and an indication for additional radical resection of incidental gallbladder cancer. World J Surg 33(5):1035–1041. https://doi.org/10.1007/s00268-009-9950-4
Sirohi B, Mitra A, Jagannath P et al (2015) Neoadjuvant chemotherapy in patients with locally advanced gallbladder cancer. Future Oncol 11(10):1501–1509
Engineer R, Goel M, Chopra S et al (2016) Neoadjuvant chemoradiation followed by surgery for locally advanced gallbladder cancers: a new paradigm. Ann Surg Oncol 23:3009–3015
Matsuda T, Matsuda A, Birnbaum DJ et al (2015) Clinical characteristics of incidental or unsuspected gallbladder cancers diagnosed during or after cholecystectomy: a systematic review and meta-analysis. Ann Surg Oncol 22(3):299–305
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gil, L., de Aretxabala, X., Lendoire, J. et al. Incidental Gallbladder Cancer: How Residual Disease Affects Outcome in Two Referral HPB Centers from South America. World J Surg 43, 214–220 (2019). https://doi.org/10.1007/s00268-018-4762-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00268-018-4762-z