Abstract
Background
A nutritional indicator suitable for predicting complications after esophagectomy has not been confirmed. The nutritional screening tool CONUT is a potential candidate.
Methods
We retrospectively analyzed 352 patients who underwent elective esophagectomy with lymphadenectomy for esophageal cancer between April 2005 and December 2014. Patients were divided into three groups according to the malnutrition degree in controlling nutritional status (CONUT): normal, light malnutrition, moderate or severe malnutrition.
Results
The numbers of patients assigned to the normal, light malnutrition, and moderate or severe malnutrition groups were 205, 126, and 21, respectively. One hundred forty-seven (41.8 %) patients were considered malnourished. Patients with moderate or severe malnutrition had a significantly high incidence of any morbidity, severe morbidities, and surgical site infection. Hospital stay in patients with moderate or severe malnutrition was significantly longer. Logistic regression analysis suggested that moderate or severe malnutrition was an independent risk factor for any morbidity [hazard ratio (HR) 2.75, 95 % confidence interval (CI) 1.081–7.020; p = 0.034] and severe morbidities (HR 3.07, 95 % CI 1.002–9.432; p = 0.049).
Conclusions
CONUT was a convenient and useful tool to assess nutritional status before esophagectomy. Patients with moderate or severe malnutrition according to CONUT are at high risk for postoperative complications.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Recently in Japan, operative morbidity and mortality risk models were established for various gastroenterological cancer surgeries based on the National Clinical Database [1–7]. The models were constructed for the following surgeries and published in succession: esophagectomy, distal gastrectomy, total gastrectomy, right hemicolectomy, low anterior resection, hepatectomy, and pancreaticoduodenectomy. In those risk models, factors related to nutrition such as weight loss, low serum albumin, anemia, and low blood urea nitrogen were proved to be independent risk factors in many surgeries.
Esophagectomy is the most invasive of these gastrointestinal surgeries. Despite advances in perioperative management, esophagectomy remains correlated with a high incidence of postoperative morbidity [8–12]. Several studies demonstrated that preoperative malnutrition predisposed to complications after esophagectomy [8, 13–15], and several markers related to nutrition, such as low body mass index (BMI), weight loss, hypoalbuminemia, and sarcopenia could be independent predictors of postoperative complications [1, 15–17]. To reduce postoperative complications and to shorten length of hospital stay, perioperative management techniques such as the enhanced recovery after surgery (ERAS) pathway are being emphasized [18]. Perioperative nutrition is one of the important components of ERAS. Several studies demonstrated that perioperative nutritional intervention might reduce complications following esophagectomy [19, 20].
Determination of an appropriate method to preoperatively assess malnutrition likely to adversely affect surgical outcomes is an important clinical problem. Although serum albumin, transthyretin, transferrin, cholesterol, and total lymphocyte count (TLC) are some of the markers of nutritional status, a comprehensive indicator suitable for the prediction of complications after esophagectomy has not been clarified.
Controlling nutritional status (CONUT) is a screening tool for nutritional status reported by de Ulíbarri et al. in 2005 [21]. CONUT consists of three parameters, serum albumin, cholesterol, and TLC, and can be calculated conveniently. Previous studies suggested that CONUT could estimate liver function and fracture risk in patients with cirrhosis of the liver and could also predict the 3-year survival of patients with chronic heart failure [22, 23]. On the other hand, the correlation between preoperative CONUT and surgical outcomes has seldom been confirmed.
Therefore, the current study aimed to investigate whether CONUT could identify preoperative malnutrition, which has a risk of the incidence of complications after subsequent esophagectomy.
Materials and methods
Patients
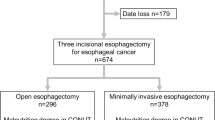
Between April 2005 and December 2014, 501 patients underwent elective esophagectomy for esophageal cancer in the Department of Gastroenterological Surgery, Kumamoto University. Among them, we excluded 15 patients who underwent two-stage esophagectomy, 21 patients who underwent transhiatal esophagectomy, and 105 patients with insufficient data (100 cholesterol, 5 TLC) from the current study. Although eight patients with distant metastasis were excluded, 15 patients with only supraclavicular lymph node metastasis were included in this study. Consequently, 352 patients were eligible and analyzed retrospectively. At the time of admission (2 or 3 days before surgery), the patients were divided into three groups on the basis of preoperative nutritional status according to malnutrition degrees in CONUT (Table 1): normal, light malnutrition, moderate or severe malnutrition. Clinical and surgical data were collected from among the prospectively entered data in the clinical database. Our institutional ethics committee approved the current study (Registry Number 990). Documented comprehensive consent was obtained from all patients.
Treatment strategy
Patients underwent preoperative investigations using esophagoscopy, esophagography, and positron emission tomography–computed tomography (CT) or contrast-enhanced CT. Endoscopic ultrasonography was conducted for T1 tumor which might be curatively treated by endoscopic resection. For patients with node-negative tumors, we performed esophagectomy without neoadjuvant treatment. When lymph node metastases were pathologically confirmed, adjuvant chemotherapy was included. For patients with non-T4, node-positive tumors, either adjuvant (April 2005 through July 2008) or neoadjuvant (August 2008 through December 2014) chemotherapy was administered in addition to esophagectomy. For patients with T4 tumors, chemoradiotherapy (CRT) was indicated. Definitive CRT was considered when patients preferred non-surgical treatment, regardless of the tumor stage. When the use of CRT failed to locally control the carcinoma, we recommended salvage esophagectomy. In the current study, the pretreatment tumor stage was classified according to the Union for International Cancer Control TNM staging, version 7 [24].
Esophagectomy
Esophagectomy was defined in the current study as an esophagectomy with lymphadenectomy requiring chest manipulations. Chest manipulation means esophageal dissection and lymphadenectomy in the thorax. Of 352 patients, 325 underwent subtotal esophagectomy with three incision (neck, chest, and abdomen) and 27 underwent Ivor Lewis esophagectomy. When tumors were located in the upper or middle thoracic esophagus, three-field lymphadenectomy was performed. For lower esophageal tumors, and when the tumor depth was within clinical stage T1, cervical lymph node dissection was omitted. Preventative cervical lymph node dissection was also omitted during salvage esophagectomy. For tumors in the abdominal esophagus, Ivor Lewis esophagectomy was performed. Minimally invasive esophagectomy (MIE) was defined as surgery performed using only a thoracoscopic and laparoscopic technique. MIE for the clinical T1 and T2 cases was adopted from May 2011.
Assessment of malnutrition according to CONUT
The method of assessment of nutritional status according to CONUT is shown in Table 1. Preoperative serum albumin level, TLC, and cholesterol level were classified and scored according to their values. The total score of the three parameters was categorized as normal, light malnutrition, moderate malnutrition, or severe malnutrition and applied to subsequent analyses.
Perioperative management
We preoperatively asked patients to cease smoking at least a month before operation and participate in a breathing training using an adjustable pressure device (Threshold; Philips Respironics Inc, Murrysville, PA, USA), as well as receive oral care by a dentist. Extubation was performed in the operation room subsequent to the surgery. Postoperative enteral nutrition was started from postoperative day 1. Physical rehabilitation was also initiated on postoperative day 1. Other perioperative management such as fluid infusion, antibiotics, timing of oral intake, etc. were conducted according to common clinical care.
Definition of morbidities
We applied the definitions of risk-adjusted morbidity and mortality for esophagectomy for cancer in accordance with the Society of Thoracic Surgeons General Thoracic Surgery Database guidelines [25]. Pulmonary morbidity was defined as the presence of one or more of the following postoperative conditions: initial ventilatory support for >48 h or reintubation for respiratory failure, need for tracheostomy, and pneumonia. Pneumonia was defined as the presence of new infiltrates on chest radiography and a positive culture from bronchoalveolar lavage. Furthermore, any pulmonary morbidity requiring intervention or surgical treatment was also included. Surgical site infection (SSI) was defined as an infection that occurred within 30 days after surgery in the area of the body affected by surgery and included superficial incisional, deep incisional, and organ/space SSI. Anastomotic leak was defined as the various signs of clinical leakage such as rubefaction, skin edema, emission of digestive fluid or pus from the wound or drain, and/or a radiographically apparent leak confirmed by esophagography or CT. Cardiovascular morbidity was defined as the presence of any cardiac disease such as arrhythmia, ischemic heart disease, and pericardial fluid collection requiring pharmacological, electrical, or interventional treatment and the presence of any thrombosis in accordance with the Common Terminology Criteria for Adverse Events (CTCAE) version 4.03 [26]. Morbidity was defined as a morbidity with a Clavien–Dindo classification (CDc) ≥II [27]. Severe morbidity was defined as morbidity with a CDc ≥IIIb, which indicated the need for surgical, endoscopic, or radiological intervention under general anesthesia.
Statistical analysis
The statistical analysis was performed using the software package StatView™ version 5.0 (Abacus Concepts, Inc., Berkeley, CA, USA). Statistical comparisons between the groups were performed using the Chi-square test. The Mann–Whitney U test was used for unpaired samples. When the matrix contained fewer than five patients, the Fisher’s exact test was used. Analysis for postoperative morbidity associated with moderate or severe malnutrition in CONUT was performed. Morbidities with a probability level ≤0.1 (any morbidity, severe morbidity of CDc ≥IIIb, any pulmonary morbidity, SSI, and reoperation) were considered possibly associated with moderate or severe malnutrition in CONUT. Subsequent multivariate analysis was performed in regard to those morbidities. The following clinical factors were adopted for the analyses of independent risk factors for the incidence of postoperative morbidities: age, sex, tumor location, clinical T, clinical N, clinical stage, type of preoperative treatment (NAC, CRT), presence of preoperative CRT, Eastern Cooperative Oncology Group (ECOG) performance status, American Society of Anesthesiologists Physical Status (ASAPS), BMI, Brinkman index (number of cigarettes/day × smoking duration (year)), performance of MIE, number of dissection fields, type of conduit, presence of diabetes mellitus, respiratory comorbidity, cardiovascular comorbidity, operative time, blood loss, and preoperative malnutrition degree according to CONUT. With a probability level ≤0.1, the factor was adopted for subsequent multivariate analysis. Independent risk factors were considered appropriate at a probability level <0.05 in a logistic regression analysis and not adjusted for multiplicity. We thought it was clinically important to clarify the boundary of malnutrition degree related to postoperative complications. When we performed multivariate analysis, we attempted to discriminate two types of boundary lines of the malnutrition degree in CONUT: normal versus any malnutrition (light, moderate, and severe) or normal plus light malnutrition versus moderate plus severe malnutrition. Consequently, the latter grouping was meaningful to predict the postoperative complications. Finally, we examined whether the influence of moderate or severe malnutrition in CONUT on the incidence of postoperative complication was modified by other parameters. (p for interaction >0.05 in all tests) (Supplemental Fig. 1) Only this analysis was conducted using the JMP program (Version 10; SAS Institute, Cary, NC, USA).
Results
The characteristics of the patients are listed in Table 2. The patient cohort included 314 (89.2 %) male patients and the mean age was 66.0 years (range 41–89 years). Neoadjuvant chemotherapy (NAC) and preoperative CRT were administered to 49 (13.9 %) and 39 (11.1 %) patients, respectively. Preoperative CRT was administered to 14 patients as induction CRT and to 25 patients as definitive CRT. MIE was performed in 76 (21.6 %) patients. According to the malnutrition degree in CONUT, the numbers of patients assigned to the normal, light malnutrition, moderate malnutrition, and severe malnutrition groups were 205, 126, 20, and 1, respectively. Consequently, 147 (41.8 %) patients were considered to be malnourished. The relationship between malnutrition degree in CONUT and clinicopathological factors are shown in Supplemental Table 1.
Table 3 shows short-term outcomes after esophagectomy according to malnutrition degree in CONUT. There were no significant differences in the incidence of any postoperative morbidity between patients with normal nutrition and those with light malnutrition. Patients with moderate or severe malnutrition suffered a higher incidence of any morbidity, severe morbidities, any pulmonary morbidity, SSI, and reoperation. Consequently, the duration of hospital stay in patients with moderate or severe malnutrition was significantly longer than that of patients with normal nutrition or light malnutrition.
Table 4 and Supplemental Table 2 shows the results of univariate and multivariate analyses on the risk factors of complications after esophagectomy. Logistic regression analysis suggested that moderate or severe malnutrition was an independent risk factor for any morbidity [hazard ratio (HR) 2.75, 95 % confidence interval (CI) 1.081–7.020; p = 0.034) and severe morbidities (HR 3.07, 95 % CI 1.002–9.432; p = 0.049)]. Although reoperation was required more frequently in patients with moderate or severe malnutrition, malnutrition degree was not an independent risk factor by a small margin (p = 0.062) (Supplemental Table 2).
Figure 1 shows preoperative total scores of CONUT according to preoperative treatment. Nutritional status in patients after preoperative CRT was significantly worse than that in patients after no treatment or after NAC.
Correlation between type of preoperative treatment and preoperative total score on CONUT. Nutritional status in patients after preoperative CRT was significantly worse than that in patients after no treatment or NAC. NAC neoadjuvant chemotherapy, NACRT neoadjuvant chemoradiotherapy, dCRT definitive chemoradiotherapy
Discussion
In the 1980s, a significant amount of research was performed on the association between preoperative nutritional status and surgical outcomes [28–31]. From those results, poor preoperative nutritional status was considered to correlate with the incidence of postoperative complications. Dickhaut et al. reported that a serum albumin level of less than 3.5 g/dL and a TLC of less than 1500 were risk factors for failure of wound healing in diabetic patients undergoing a Syme’s amputation [28]. Mequid et al. also showed that a serum albumin level of less than 3.5 g/dL and recent weight loss greater than 10 %, or weight/height, mid-arm circumference, and triceps skinfold thickness lower than 10 % were risk factors for a high incidence of postoperative morbidity and mortality after colorectal cancer surgery [29]. Several meta-analyses or systematic reviews suggested that nutritional intervention could reduce a number of postoperative complications [32, 33].
A method to assess preoperative nutrition status that can predict surgical adverse outcomes has been sought for esophageal surgery [33]. Filip et al. reported that the preoperative prognostic nutritional index (PNI) advocated by Onodera et al. [34], which is calculated as 10 × albumin (g/dL) + 0.005 × TLC (/μL), was an independent predictor for major complications after esophagectomy [13]. Nozoe et al. also showed that preoperative PNI was an independent predictor of complications following esophagectomy, although their cohort did not contain any patients who underwent thoracoscopic esophagectomy or neoadjuvant treatment [14]. On the other hand, Han-Geurts et al. showed that PNI and the nutritional risk index (NRI), calculated as 1.519 × serum albumin (g/L) + 41.7 × present weight/usual weight, were not associated with infectious complications after esophagectomy [35]. Other useful formulas related to nutrition to predict postoperative complications after esophagectomy have not been confirmed.
In the current study, preoperative moderate or severe malnutrition as assessed by CONUT was an independent risk factor for the incidence of postoperative complications and severe complications. CONUT consists of three nutritional parameters. According to the original article on CONUT, serum albumin is used as an indicator of protein reserves. Cholesterol is used as a caloric depletion parameter and TLC is used as an indicator of loss of immune defenses caused by malnutrition [21]. Serum albumin was the representative nutrition marker and used frequently to assess nutritional status for the prediction of postoperative outcomes in many studies [1, 36]. TLC is also an important marker of nutrition and immunity. A meta-analysis demonstrated that intervention with immune-enhancing nutrition could increase TLC and reduce postoperative complications [32]. Compared to PNI and NRI, CONUT contained an additional nutritional parameter, cholesterol. Several studies suggested that low serum cholesterol correlated with morbidity or mortality after gastroenterological surgery including esophagectomy [37, 38]. However, most of previous studies with a large cohort aiming to establish the risk factors of postoperative morbidity seldom included serum cholesterol as a variable [1–7]. This might be due to the fact that surgeons do not routinely check the value of preoperative total cholesterol. The possible usefulness of cholesterol level as a predictive marker should be confirmed by the future study.
Recently, neoadjuvant CRT has often been administrated to patients with resectable advanced esophageal cancers [39]. Salvage esophagectomy is sometimes included for residual or relapsed cancers after definitive CRT. In the current study, total scores of CONUT according to the preoperative treatment showed that patients were more malnourished after preoperative CRT compared to those who did not receive preoperative CRT. During CRT for esophageal cancer, appetite loss, nausea, vomiting, and odynophagia sometimes occur for a long time. In addition, decrease of TLC due to myelosuppression is also seen. To prevent deterioration of nutrition during CRT, nutritional intervention should be performed. A multicenter randomized controlled trial on the usefulness of disease-specific enteral nutrition clarified the usefulness of nutritional intervention during CRT for head and neck and esophageal cancer [40].
To maintain or improve preoperative nutrition, several novel approaches are being investigated. Ghrelin administration is one of the potential candidates that might help to maintain body weight and minimize the deterioration of nutritional status [41]. Esophageal stenting during preoperative chemotherapy for patients with stenosis has been considered effective to maintain preoperative nutrition [42, 43]. However, most of previous studies with a large cohort aiming to establish the risk factors of postoperative morbidity seldom included serum cholesterol as a variable [1–7]. The possible usefulness of cholesterol level as a predictive marker should be confirmed by the future study.
This study has several limitations. First, it was a retrospective study conducted at a single institute. A number of patients had to be excluded due to insufficient data. Although serum albumin, cholesterol, and TLC were examined in all patients at the first visit, relevant data were often lacking in patients after preoperative treatment. This could have led to selection bias. In addition, there might exist a historical bias, as the period of treatment extended over a period of approximately 10 years. Regarding surgery, patients who underwent two-stage esophagectomy and transhiatal esophagectomy were excluded. In this study, most of patients underwent subtotal esophagectomy with three incision (neck, chest, and abdomen). Two-stage esophagectomy and transhiatal esophagectomy were performed for patients with severe comorbidity, as these surgeries were considered less invasive compared to subtotal esophagectomy. We excluded these types of surgery, since we thought they might affect the general outcome. Finally, the number of patients included was not as large as desirable. We consider further multi-institutional research a prerequisite to undoubtedly establish the usefulness and availability of CONUT.
In conclusion, CONUT is a convenient and useful tool to assess nutritional status before esophagectomy. Patients with moderate or severe malnutrition according to CONUT are at a high risk for postoperative complications. Those patients might be supported by nutritional intervention before surgery and carefully monitored after surgery.
References
Takeuchi H, Miyata H, Gotoh M et al (2014) A risk model for esophagectomy using data of 5354 patients included in a Japanese nationwide web-based database. Ann Surg 260:259–266
Kurita N, Miyata H, Gotoh M et al (2015) Risk model for distal gastrectomy when treating gastric cancer on the basis of data from 33,917 Japanese patients collected using a nationwide web-based data entry system. Ann Surg 262:295–303
Watanabe M, Miyata H, Gotoh M et al (2014) Total gastrectomy risk model: data from 20,011 Japanese patients in a nationwide internet-based database. Ann Surg 260:1034–1039
Kobayashi H, Miyata H, Gotoh M et al (2014) Risk model for right hemicolectomy based on 19,070 Japanese patients in the National Clinical Database. J Gastroenterol 49:1047–1055
Matsubara N, Miyata H, Gotoh M et al (2014) Mortality after common rectal surgery in Japan: a study on low anterior resection from a newly established nationwide large-scale clinical database. Dis Colon Rectum 57:1075–1081
Kenjo A, Miyata H, Gotoh M et al (2014) Risk stratification of 7,732 hepatectomy cases in 2011 from the National Clinical Database for Japan. J Am Coll Surg 218:412–422
Kimura W, Miyata H, Gotoh M et al (2014) A pancreaticoduodenectomy risk model derived from 8575 cases from a national single-race population (Japanese) using a web-based data entry system: the 30-day and in-hospital mortality rates for pancreaticoduodenectomy. Ann Surg 259:773–780
Mantziari S, Hübner M, Demartines N et al (2014) Impact of preoperative risk factors on morbidity after esophagectomy: is there room for improvement? World J Surg 38:2882–2890. doi:10.1007/s00268-014-2686-9
Ferguson MK, Celauro AD, Prachand V (2011) Assessment of a scoring system for predicting complications after esophagectomy. Dis Esophagus 24:510–515
Dutta S, Al-Mrabt NM, Fullarton GM et al (2011) A comparison of POSSUM and GPS models in the prediction of post-operative outcome in patients undergoing oesophago-gastric cancer resection. Ann Surg Oncol 18:2808–2817
Grotenhuis BA, van Hagen P, Reitsma JB et al (2010) Validation of a nomogram predicting complications after esophagectomy for cancer. Ann Thorac Surg 90:920–925
Dhungel B, Diggs BS, Hunter JG et al (2010) Patient and peri-operative predictors of morbidity and mortality after esophagectomy: American College of Surgeons National Surgical Quality Improvement Program (ACS-NSQIP), 2005-2008. J Gastrointest Surg 14:1492–1501
Filip B, Scarpa M, Cavallin F et al (2015) Postoperative outcome after oesophagectomy for cancer: nutritional status is the missing ring in the current prognostic scores. Eur J Surg Oncol 41:787–794
Nozoe T, Kimura Y, Ishida M et al (2002) Correlation of pre-operative nutritional condition with post-operative complications in surgical treatment for oesophageal carcinoma. Eur J Surg Oncol 28:396–400
Ida S, Watanabe M, Yoshida N et al (2015) Sarcopenia is a predictor of postoperative respiratory complications in patients with esophageal cancer. Ann Surg Oncol 22:4432–4437
Yoshida N, Watanabe M, Baba Y et al (2014) Risk factors for pulmonary complications after esophagectomy for esophageal cancer. Surg Today 44:526–532
Sunpaweravong S, Ruangsin S, Laohawiriyakamol S et al (2012) Prediction of major postoperative complications and survival for locally advanced esophageal carcinoma patients. Asian J Surg 35:104–109
Findlay JM, Gillies RS, Millo J et al (2014) Enhanced recovery for esophagectomy: a systematic review and evidence-based guidelines. Ann Surg 259:413–431
Fukuda T, Seto Y, Yamada K et al (2008) Can immune-enhancing nutrients reduce postoperative complications in patients undergoing esophageal surgery? Dis Esophagus 21:708–711
Takeuchi H, Ikeuchi S, Kawaguchi Y et al (2007) Clinical significance of perioperative immunonutrition for patients with esophageal cancer. World J Surg 31:2160–2167. doi:10.1007/s00268-007-9219-8
Ignacio de Ulíbarri J, González-Madroño A, de Villar NG et al (2005) CONUT: a tool for controlling nutritional status. First validation in a hospital population. Nutr Hosp 20:38–45
López-Larramona G, Lucendo AJ, Tenías JM (2015) Association between nutritional screening via the Controlling Nutritional Status index and bone mineral density in chronic liver disease of various etiologies. Hepatol Res 45:618–628
Nochioka K, Sakata Y, Takahashi J, CHART-2 Investigators et al (2013) Prognostic impact of nutritional status in asymptomatic patients with cardiac diseases: a report from the CHART-2 Study. Circ J 77:2318–2326
Edge SB, Byrd DR, Compton CC et al (2009) AJCC cancer staging manual, 7th edn. Springer, New York
Society of Thoracic Surgeons (2013) Risk-adjusted morbidity and mortality for esophagectomy for cancer. http://www.sts.org/quality-research-patient-safety/quality/quality-performance-measures. Accessed 10 Oct 2015
National Cancer Institute (2010) Common Terminology Criteria for Adverse Events (CTCAE). United States Department of Health and Human Services. http://evs.nci.nih.gov/ftp1/CTCAE/CTCAE_4.03_2010-06-14_QuickReference_5x7.pdf#search=‘http%3A%2F%2Fevs.nci.nih.gov%2Fftp1%2FCTCAE%2F+CTCAE_4.03_20100614_QuickReference_5x7.pdf. Accessed 10 Oct 2015
Dindo D, Demartines N, Clavien PA (2004) Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg 240:205–213
Dickhaut SC, DeLee JC, Page CP (1984) Nutritional status: importance in predicting wound-healing after amputation. J Bone Joint Surg Am 66:71–75
Meguid MM, Mughal MM, Debonis D et al (1986) Influence of nutritional status on the resumption of adequate food intake in patients recovering from colorectal cancer operations. Surg Clin N Am 66:1167–1176
Detsky AS, Baker JP, O’Rourke K et al (1987) Predicting nutrition-associated complications for patients undergoing gastrointestinal surgery. J Parenter Enteral Nutr 11:440–446
Reilly JJ Jr, Gerhardt AL (1985) Modern surgical nutrition. Curr Probl Surg 22:1–81
Mazaki T, Ishii Y, Murai I (2015) Immunoenhancing enteral and parenteral nutrition for gastrointestinal surgery: a multiple-treatments meta-analysis. Ann Surg 261:662–669
Lawrence VA, Cornell JE, Smetana GW (2006) Strategies to reduce postoperative pulmonary complications after noncardiothoracic surgery: systematic review for the American College of Physicians. Ann Intern Med 144:596–608
Onodera T, Goseki N, Kosaki G (1984) Prognostic nutritional index in gastrointestinal surgery of malnourished cancer patients. Nihon Geka Gakkai Zasshi 85:1001–1005
Han-Geurts IJ, Hop WC, Tran TC (2006) Nutritional status as a risk factor in esophageal surgery. Dig Surg 23:159–163
Noble F, Curtis N, Harris S et al (2012) Risk assessment using a novel score to predict anastomotic leak and major complications after oesophageal resection. J Gastrointest Surg 16:1083–1095
Sanz L, Ovejero VJ, González JJ et al (2006) Mortality risk scales in esophagectomy for cancer: their usefulness in preoperative patient selection. Hepatogastroenterology 53:869–873
Wang Q, Lau WY, Zhang B et al (2014) Preoperative total cholesterol predicts postoperative outcomes after partial hepatectomy in patients with chronic hepatitis B- or C-related hepatocellular carcinoma. Surgery 155:263–270
van Hagen P, Hulshof MC, van Lanschot JJ et al (2012) Preoperative chemoradiotherapy for esophageal or junctional cancer. N Engl J Med 366:2074–2084
Fietkau R, Lewitzki V, Kuhnt T et al (2013) A disease-specific enteral nutrition formula improves nutritional status and functional performance in patients with head and neck and esophageal cancer undergoing chemoradiotherapy: results of a randomized, controlled, multicenter trial. Cancer 119:3343–3353
Hiura Y, Takiguchi S, Yamamoto K et al (2012) Effects of ghrelin administration during chemotherapy with advanced esophageal cancer patients: a prospective, randomized, placebo-controlled phase 2 study. Cancer 118:4785–4794
Brown RE, Abbas AE, Ellis S et al (2011) A prospective phase II evaluation of esophageal stenting for neoadjuvant therapy for esophageal cancer: optimal performance and surgical safety. J Am Coll Surg 212:582–588
Bower M, Jones W, Vessels B et al (2009) Nutritional support with endoluminal stenting during neoadjuvant therapy for esophageal malignancy. Ann Surg Oncol 16:3161–3168
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yoshida, N., Baba, Y., Shigaki, H. et al. Preoperative Nutritional Assessment by Controlling Nutritional Status (CONUT) is Useful to estimate Postoperative Morbidity After Esophagectomy for Esophageal Cancer. World J Surg 40, 1910–1917 (2016). https://doi.org/10.1007/s00268-016-3549-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00268-016-3549-3