Abstract
Background
Hepatocellular carcinoma (HCC) larger than ten cm belonging to Barcelona Clinic Liver Cancer (BCLC) stage B and C may benefit from hepatic resection (HR), compared to presently recommended management by transarterial chemoembolization and sorafenib, respectively. This study reviews survival outcomes in such patients treated at a tertiary level hospital in Taiwan, and compares survival advantage of surgical resection over embolization therapy using a statistically valid propensity scores matching model.
Methods
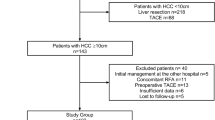
192 patients newly diagnosed with HCC ≥ 10 cm between 2005 and 2010, who had HR (n = 104) and transarterial embolization (TAE) (n = 88), were retrospectively studied. Thirty-two patients in each group were selected by propensity scores matching model for comparison.
Results
Survival rates at 1, 3, and 5 years of patients in BCLC stage B who had HR and TAE were 78.5, 61.4, 54.2 % and 30, 12.9, 12.9 %, (p < 0.001), respectively. For stage C, survival rates were 77.8, 56.4, and 47 % at 1, 3, 5 years in HR group, while it was 12.7 % at 1 year in TAE group, (p < 0.001). Propensity score-based analysis showed estimated 1-, 3-, and 5-year survival rates of patients receiving HR and TAE were 90.2 versus 26.4 %, 64.3 versus 3.3 %, and 51.5 versus 3.3 %, respectively (p < 0.001).
Conclusions
HR had significantly better 5 year survival than TAE for patients with HCC ≥ 10 cm in the propensity score model. Overall survival of BCLC stage B may be improved by considering HR as first treatment option for resectable large HCCs, provided patient is fit for surgery with good liver remnant.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Hepatocellular carcinoma (HCC) is highly lethal, being the third most common cause of cancer deaths worldwide. The prevalence of hepatitis B virus (HBV) and hepatitis C virus (HCV), which are predisposing factors for HCC, is high in Asia. 80 % of all HCC cases occur in Asia, and the number of HCC cases is projected to increase by 80 % over the coming decades [1].
Taiwan has one of the highest incidences of HCC in the world, affecting 52 per 100,000 males and 20 per 100,000 females [2], and a large number of patients present in the intermediate to advanced stage. The optimal management of resection with clear margins is precluded in many cases because of factors including the tumor size, location, number, pathologic characteristics, and the severity of underlying liver disease. Curative treatment modalities like surgical resection, radiofrequency ablation (RFA), and liver transplantation can achieve excellent survival outcomes for tumors within the Milan criteria [3]. However, the best treatment strategy for HCCs larger than ten centimeters (termed as ‘L-HCC’ henceforth) is not clearly defined.
According to the widely recognized Barcelona Clinic Liver Cancer (BCLC) guidelines [4], L-HCCs without extrahepatic spread are considered as intermediate (stage B) or advanced (stage C) cases, depending on liver function and patient’s performance status. American Association for Study of Liver Diseases (AASLD) and European Association for the Study of the Liver (EASL) guidelines [5] recommend transarterial chemoembolization (TACE) and sorafenib as the sole treatment options for stage B and C HCC, respectively, while resection is advised only for Stage A patients. Perioperative complications and mortality after liver resection have reduced significantly over the years, and hence it may have a survival benefit in stage B and C [6, 7]. In this study, we have presented our center’s experience in management of L-HCC by hepatic resection (HR) and transarterial embolization (TAE), and compared the overall survival (OS) using a statistically valid propensity scores matching model.
Methods
Data of all patients presenting at Kaohsiung Chang Gung Memorial Hospital with newly diagnosed L-HCC between 2005 and 2010 and who underwent HR or TAE were retrospectively collected and analyzed. Collated data included patient characteristics such as age, gender, the presence of HBV and HCV infection, and alcohol intake; relevant radiological and biochemical investigations including computed tomography (CT) of liver, complete blood counts, platelets, direct and indirect bilirubin, alanine transaminase (ALT), aspartate aminotranferase (AST), international nationalized ratio (INR), alkaline phosphatase, alpha-fetoprotein (AFP), and indocyanine green (ICG) clearance tests; radiological and pathologic features of the tumor; treatment details including type, date, and duration of therapy; and follow-up. Cases were retrospectively classified according to BCLC criteria to facilitate comparison with other studies.
Diagnosis of HCC was based on the typical CT scan or MRI findings; percutaneous needle biopsy was not routinely performed [5]. Liver status was assessed using liver function tests, Child-Turcotte-Pugh score, ICG clearance, and CT volumetry of liver. The management of patients with HCC was discussed in multidisciplinary tumor board meetings.
HR was offered to patients with resectable tumor, provided that functional reserve was sufficient. Criteria for resectability and assessment of liver functional reserve at our center have been previously described [8]. Tumors invading ipsilateral hepatic or portal veins, and tumors with satellite nodules in the same liver lobe were also considered for resection. A reverse L abdominal incision was used in all cases. Abdominal exploration for distant metastases and intraoperative Doppler ultrasound was performed after liver mobilization. The anterior approach and liver hanging maneuver were utilized, often together, in cases where mobilization of the perihepatic attachments was likely to cause increased bleeding or tumor rupture [9]. Parenchymal transection was done using Cavitron Ultrasonic Surgical Aspirator (CUSA; Valleylab Inc., Boulder, Colorado), bipolar diathermy, and crushing with a Kelly clamp, with intermittent inflow occlusion.
TAE was offered to patients of Child-Pugh class A or B having unresectable HCC, or resectable HCC with low-predicted remnant liver volume, and patients unwilling to undergo surgery. Technique of TAE at our institute has been described previously [10]. Patients were followed up by sonography, CT scan, and AFP at interval of 1–2 months within 2 years post-operative period then every 2–3 months after 2 years.
Baseline patient characteristics were presented using descriptive statistics and nominal data were compared using χ 2 test and Student’s t test. Data were labeled as significant if p value was <0.05. OS was computed using Kaplan–Meier analysis, and differences in survival curves were compared using log-rank test. Survival statistics were calculated from the date of HR or TAE. All statistical analyses were done using SPSS version 16.0.0
Propensity score-based model
Since the distribution of patients with L-HCCs into HR and TAE groups was non-randomized, results obtained by direct comparison could be biased. So the propensity score matching model was used for accurate comparison between the two modalities. Variables potentially affecting the outcomes were assigned propensity score after logistic regression analysis. The nearest neighbors in each group were matched one-on-one based on the generated propensity scores, and the survival outcomes were then compared between the two groups using appropriate non-parametric tests as described previously. Two propensity score match model were selected. In addition to propensity score, one model matched sex, Child-Pugh, ascites, tumor number, AJCC staging (model 1), and BCLC staging, and the other matched sex only (model 2).
Results
Study population
108 patients underwent HR, while 88 received TAE (Table 1). 83 per cent of the cases were male, and HBV was a predisposing factor in 64.6 % patients. The patients receiving TAE were older, with higher incidence of ascites and higher CTP scores (p < 0.001). 30 % tumors (n = 37) were larger than 15 cm in diameter. Fifty-six percent of the patients had solitary tumor and 72 % of these underwent HR, while those with more than 3 nodules and with vascular invasion seen on CT scan (36.5 %) were more likely to have TAE, p < 0.05.
According to American Joint Committee on Cancer (AJCC) 7th edition criteria, 61 % cases were in Stage III, and Stage I and II cases were significantly more likely to have undergone HR (Table 1). According to the BCLC classification, 45.3 % cases were in the intermediate stage and were more likely to have HR, while most of the advanced stage cases and all the terminal stage cases received TAE. These observations result from our center’s policy to offer HR as primary option for all resectable L-HCC cases. Patients undergoing HR had better liver reserve as evident by Child-Pugh classification (74 % Child’s A, p < 0.001), albumin (3.8 g/dl p < 0.001), platelet count (247 × 109/L, p = 0.034), INR (1.00, p = 0.001), and bilirubin levels (0.9 mg/dL, p = 0.005) when compared to TAE group.
Survival comparison between TAE and HR groups
The OS for all L-HCC cases, including both treatment groups, was 50.5, 35, and 30.9 % at 1, 3, and 5 years. The overall 1, 3, and 5 year survival rates for BCLC stage B cases were 64.8, 47.6 and 42.9 %, respectively. (Fig. 1)
The OS in the HR group at 1, 3, and 5 years was 78.2, 59.3, and 51.6 % which was significantly better than TAE group with 16.9, 6, and 6 %, respectively, p < 0.001.(Table 2). Even when cases were classified according to BCLC criteria, patients undergoing HR had significantly increased OS compared to those undergoing TAE in both the intermediate and advanced group cases. Similarly, when cases were stratified according to the AJCC classification, the 1-, 3-, and 5-year OS rates were significantly better in those with HR than those undergoing TAE for stage I, II, and III.
Comparison of patients selected in the propensity score matching model
In model 1, 32 pairs of patients each were selected from the HR and TAE groups, after matching according to the propensity score model. There were no significant differences between these two groups for the following patient characteristics: gender, body mass index (BMI), history of alcoholism, HBV and HCV coinfection, ascites, and Child-Pugh classification. The two groups were also matched for the following tumor characteristics: number and size of tumor nodules, vascular invasion, serum AFP levels, AJCC stage, and BCLC stage. The comparison of the patient and disease characteristics, and the survival outcomes are shown in Tables 3 and 4. On analysis, the patients in the HR group were significantly younger than those in the TAE group. The 1-, 3-, 5-year OS for the patients in the HR group (90.2, 64.3, and 51.5 %, respectively) was significantly better than for those who underwent TAE (26.4, 3.3, and 3.3 %, respectively, p < 0.001).
In model 2, we included 54 pairs of patients in HR and TAE. The comparison of the patient and disease characteristics, and the survival outcomes of 54 patients in HR and TAE are shown in Tables 5, 6. The survival showed similar results in both matching models.
Discussion
The tumor volume of L-HCCs is greater than tumors belonging to BCLC stage A (maximum 3 tumors with maximum 3 cm diameter). They are usually associated with poor prognostic factors such as increased incidence of vascular invasion, presence of multiple or satellite nodules, and poor degree of differentiation, in addition to tumor size [11]. These patients have a dismal prognosis, even with locoregional control treatments [12]. While the single large capsulated type HCC in a Child A patient is often resectable, surgical resection for most L-HCCs in previous decades was associated with high perioperative morbidity and mortality [13, 14]. The EASL and APASL (Asian Pacific Association for the Study of the Liver) guidelines [1] recommend TACE for treatment of BCLC stage B tumors. TACE undoubtedly improves overall outcomes in BCLC stage B patients, and can achieve good tumor control when average size of HCCs is between 5 and 10 cm. However, this survival benefit does not seem to extend to L-HCCs even though they may be a subset of BCLC stage B; in fact, large size may be considered a relative contraindication to TACE therapy [15]. Response to TACE correlates with the extent of necrosis achieved. The chemotherapeutic dose and number of sessions required to achieve an adequate antitumoral response in L-HCCs significantly increases the risk of liver failure. There is wide variation in the technique, drugs, dosage, number of sessions, and intervals between sessions for TACE described in the literature. The survival benefit of TACE over TAE has not been definitively demonstrated, especially for L-HCCs [16, 17].
The outcomes of TAE for L-HCCs in our study are similar to those reported for TACE for unresectable HCC in previous studies [15], but the criteria for unresectability are different at various centers, making it difficult to accurately compare results. The 5-year survival rate after TAE for BCLC stage B L-HCC in our study is 12.9 %. This is comparable to results after TACE for BCLC stage B HCCs in other studies [5, 18], but significantly less than survival outcomes following HR in this study (54.2 %, p < 0.001). EASL guidelines advise sorafenib monotherapy for BCLC Stage C HCC. However, previous studies have showed survival advantage of other therapeutic options including HR in selected patients in this stage [19]. Several studies recommend HR as better than TACE for L-HCCs, although analysis according to BCLC stages is unavailable in many reports [20–22]. A review in 2011 found a median 5-year survival after HR for L-HCC of 28 % [23]. Our study also indicates that patients in BCLC stages B and C with resectable L-HCC and acceptable liver function may benefit from HR. The importance of proper selection of candidates for surgery cannot be overestimated. Remnant liver volume, liver function, and patient’s physical status have to be carefully assessed before considering tumor resectability. As a large proportion of patients with L-HCC have vascular invasion and multiple nodules, HR in this group requires extended criteria of resectability. Hence, preoperative assessments of patient by imaging studies and investigations of liver function, advanced surgical techniques, expert post-operative management, and experience of large volume of HCC cases are necessary to minimize perioperative complications and achieve good long-term survival outcomes [8].
While surgery is potentially curative, it is technically challenging. En bloc resection of tumor is attempted to obtain the best survival advantage [24]. Feasibility of formal hepatectomy has to be considered in cases with macroscopic invasion of ipsilateral portal or hepatic veins. The anterior approach in combination with the liver hanging maneuver enables right hepatectomy for L-HCCs with significantly less bleeding [9]. Reconstruction of portal vein or hepatic artery has been reported in HR, using interposition grafts of polytetrafluoroethylene (PTFE) or cryopreserved vessels after portal vein resection [25].
The significantly better OS after HR in our study may have resulted from non-randomized selection criteria for surgery or TAE. Hence a propensity score matching analysis was performed to overcome any possible selection bias. Even after matching cases in each group for overcoming confounding variables, the survival advantage was significantly higher for the HR group for intermediate and advanced BCLC stages. The survival benefit of HR over TAE group was also seen when analyzed according to AJCC classification, for Stage I and III. The reasons for better OS with HR in L-HCC could be multifactorial. Significantly decreased perioperative morbidity and mortality as compared to previous decades has improved overall surgical outcomes. The number of TAE sessions required to achieve complete tumor control may be higher, resulting in more time and opportunity for tumor behavior to change. The tumors in the cases undergoing TAE may not have undergone complete tumor necrosis, and the procedure induced ischemia may even have accelerated regeneration of residual tumor [26]. The mean age of the TAE group was higher than that of the HR group, although their performance status was similar. It is notable that the incidence of vascular invasion was equal in both the HR and TAE groups. It is hence possible to improve the OS in patients of BCLC Stage B and C L-HCC by carefully selecting those patients who can safely undergo surgical resection.
Our study is retrospective in nature, and is not a population-based study. Our results may be influenced by the HBV predominant etiology in our patients, unlike the HCV predominant pattern found in other regions [27]. Newer developments may further modify the management of L-HCC. Staged hepatectomies following portal vein ligation or embolization can increase remnant liver volume and hence increase the pool of patients who can undergo surgery. Newer techniques like combination of RFA or sorafenib with TACE, transarterial radioembolization (TARE) with Ytrrium-90 beads, drug eluting beads (DEB) TACE, and brachytherapy should also be investigated extensively for benefit in L-HCCs.
Conclusions
L-HCCs exhibit aggressive tumor behavior and their response to TACE is poor. Surgical resection can significantly improve outcomes; provided patients are carefully screened for remnant liver volume, liver function, and performance status along with assessment of tumor resectability. High volumes, surgeon experience, and advanced surgical techniques can improve outcomes even in advanced BCLC or stage III AJCC cases. The propensity score matching analysis in our study indicates that modern criteria of resection result in significantly higher rates of five-year survival, despite unfavorable tumor pathology. A hospital policy of offering HR to patients when possible, and reserving TAE for unresectable cases can increase OS of patients with L-HCC, provided the surgical mortality and morbidity are within acceptable limits.
Abbreviations
- AFP:
-
Alpha-fetoprotein
- AJCC staging:
-
American Joint Committee on Cancer staging
- ALT:
-
Alanine aminotransferase
- Anti-HCV+:
-
Hepatitis C virus antibody positive
- AST:
-
Aspartate aminotransferase
- BCLC staging:
-
Barcelona Clinic Liver Cancer staging
- CT:
-
Computed tomography scanning
- EASL:
-
European Association for the Study of the Liver
- HBsAg:
-
Hepatitis B surface antigen
- HCC:
-
Hepatocellular carcinoma
- HR:
-
Hepatic resection
- ICG:
-
Indocyanine green
- INR:
-
International normalized ratio
- L-HCC:
-
Hepatocellular carcinoma larger than 10 cm in widest diameter
- OS:
-
Overall survival
- TACE:
-
Transarterial chemoembolization
- TAE:
-
Transarterial embolization
- γ-GT:
-
Gamma glutamyl transpeptidase
References
Han KH, Kudo M, Ye SL, Choi JY, Poon RT, Seong J et al (2011) Asian consensus workshop report: expert consensus guideline for the management of intermediate and advanced hepatocellular carcinoma in Asia. Oncology 81(Suppl 1):158–164
Ferlay J, Shin HR, Bray F, Forman D, Mathers C, Parkin DM (2010) Estimates of worldwide burden of cancer in 2008: GLOBOCAN 2008. Int J Cancer 127(12):2893–2917
Guglielmi A, Ruzzenente A, Conci S, Valdegamberi A, Vitali M, Bertuzzo F et al (2014) Hepatocellular carcinoma: surgical perspectives beyond the barcelona clinic liver cancer recommendations. World J Gastroenterol 20(24):7525–7533
Llovet JM, Brú C, Bruix J (1999) Prognosis of hepatocellular carcinoma: the BCLC staging classification. Semin Liver Dis 19(03):329–338
European Association for Study of L, European Organisation for R, Treatment of C (2012) EASL-EORTC clinical practice guidelines: management of hepatocellular carcinoma. Eur J Cancer 48(5):599–641
Chang WT, Kao WY, Chau GY, Su CW, Lei HJ, Wu JC et al (2012) Hepatic resection can provide long-term survival of patients with non-early-stage hepatocellular carcinoma: extending the indication for resection? Surgery 152(5):809–820
Choi GH, Han DH, Kim DH, Choi SB, Kang CM, Kim KS et al (2009) Outcome after curative resection for a huge (> or = 10 cm) hepatocellular carcinoma and prognostic significance of gross tumor classification. Am J Surg 198(5):693–701
Wang CC, Iyer SG, Low JK, Lin CY, Wang SH, Lu SN et al (2009) Perioperative factors affecting long-term outcomes of 473 consecutive patients undergoing hepatectomy for hepatocellular carcinoma. Ann Surg Oncol 16(7):1832–1842
Wang CC, Jawade K, Yap AQ, Concejero AM, Lin CY, Chen CL (2010) Resection of large hepatocellular carcinoma using the combination of liver hanging maneuver and anterior approach. World J Surg 34(8):1874–1878. doi:10.1007/s00268-010-0546-9
Cheng YF, Huang TL, Chen TY, Chen YS, Wang CC, Hsu SL et al (2005) Impact of pre-operative transarterial embolization on the treatment of hepatocellular carcinoma with liver transplantation. World J Gastroenterol 11(10):1433–1438
Zhou L, Rui JA, Wang SB, Chen SG, Qu Q (2011) Prognostic factors of solitary large hepatocellular carcinoma: the importance of differentiation grade. Eur J Surg Oncol 37(6):521–525
Forner A, Llovet JM, Bruix J (2012) Chemoembolization for intermediate HCC: is there proof of survival benefit? J Hepatol 56(4):984–986
Abdel-Wahab M, Sultan A, el-Ghawalby A, Fathy O, el-Ebidy G, Abo-Zeid M et al (2001) Is resection for large hepatocellular carcinoma in cirrhotic patients beneficial? Study of 38 cases. Hepatogastroenterology 48(39):757–761
Régimbeau J-M, Farges O, Shen BY, Sauvanet A, Belghiti J (1999) Is surgery for large hepatocellular carcinoma justified? J Hepatol 31(6):1062–1068
Raoul JL, Sangro B, Forner A, Mazzaferro V, Piscaglia F, Bolondi L et al (2011) Evolving strategies for the management of intermediate-stage hepatocellular carcinoma: available evidence and expert opinion on the use of transarterial chemoembolization. Cancer Treat Rev 37(3):212–220
Pleguezuelo M, Marelli L, Misseri M, Germani G, Calvaruso V, Xiruochakis E et al (2008) TACE versus TAE as therapy for hepatocellular carcinoma. Expert Rev Anticancer Ther 8(10):1623–1641
Marelli L, Stigliano R, Triantos C, Senzolo M, Cholongitas E, Davies N et al (2007) Transarterial therapy for hepatocellular carcinoma: which technique is more effective? A systematic review of cohort and randomized studies. Cardiovasc Intervent Radiol 30(1):6–25
Huang YH, Wu JC, Chen SC, Chen CH, Chiang JH, Huo TI et al (2006) Survival benefit of transcatheter arterial chemoembolization in patients with hepatocellular carcinoma larger than 10 cm in diameter. Aliment Pharmacol Ther 23(1):129–135
Wang JH, Kuo YH, Wang CC, Chen CL, Cheng YF, Hsu HC et al (2013) Surgical resection improves the survival of selected hepatocellular carcinoma patients in Barcelona clinic liver cancer stage C. Diges Liver Dis 45(6):510–515
Hsu CY, Hsia CY, Huang YH, Su CW, Lin HC, Pai JT et al (2012) Comparison of surgical resection and transarterial chemoembolization for hepatocellular carcinoma beyond the Milan criteria: a propensity score analysis. Ann Surg Oncol 19(3):842–849
Pandey D, Lee KH, Wai CT, Wagholikar G, Tan KC (2007) Long term outcome and prognostic factors for large hepatocellular carcinoma (10 cm or more) after surgical resection. Ann Surg Oncol 14(10):2817–2823
Poon RT-P, Fan ST, Wong J (2002) Selection criteria for hepatic resection in patients with large hepatocellular carcinoma larger than 10 cm in diameter. J Am Coll Surg 194(5):592–602
Zhou Y-M, Li B, Xu D-H, Yang J-M (2011) Safety and efficacy of partial hepatectomy for huge (≥ 10 cm) hepatocellular carcinoma: a systematic review. Med Sci Monit 17(3):RA76–RA83
Yap AQ, Millan CA, Wang J-H, Wang C-C, Lu S-N, Wang S-H et al (2014) How to Improve the Outcome in Patients with AJCC Stage I Hepatocellular Carcinoma. Anticancer Res 34(6):3093–3103
Liao G, Hsieh H, Hsieh C, Chen T, Chen C, Yu J et al (2005) Vessel reconstruction for great vessel invasion by hepatobiliary malignancy. J Med Sci 25(6):309
Guan YS, He Q, Wang MQ (2012) Transcatheter arterial chemoembolization: history for more than 30 years. ISRN Gastroenterol 2012:480650
Kim BK, Kim SU, Park JY, Kim DY, Ahn SH, Park MS et al (2012) Applicability of BCLC stage for prognostic stratification in comparison with other staging systems: single centre experience from long-term clinical outcomes of 1717 treatment-naïve patients with hepatocellular carcinoma. Liver Int 32(7):1120–1127
Acknowledgments
The authors would like to express their utmost gratitude to Grace Lin of Division of Hepato-Gastroenterology, Department of Internal Medicine, Kaohsiung Chang Gung Memorial Hospital for all the statistical analyses in this manuscript.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chan, YC., Kabiling, C.S., Pillai, V.G. et al. Survival Outcome Between Hepatic Resection and Transarterial Embolization for Hepatocellular Carcinoma More Than 10 cm: A Propensity Score Model. World J Surg 39, 1510–1518 (2015). https://doi.org/10.1007/s00268-015-2975-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00268-015-2975-y