Abstract
Background
National trauma registries have helped improve patient outcomes across the world. Recently, the idea of an International Trauma Data Bank (ITDB) has been suggested to establish global comparative assessments of trauma outcomes. The objective of this study was to determine whether global trauma data could be combined to perform international outcomes benchmarking.
Methods
We used observed/expected (O/E) mortality ratios to compare two trauma centers [European high-income country (HIC) and Asian lower-middle income country (LMIC)] with centers in the North American National Trauma Data Bank (NTDB). Patients (≥16 years) with blunt/penetrating injuries were included. Multivariable logistic regression, adjusting for known predictors of trauma mortality, was performed. Estimates were used to predict the expected deaths at each center and to calculate O/E mortality ratios for benchmarking.
Results
A total of 375,433 patients from 301 centers were included from the NTDB (2002–2010). The LMIC trauma center had 806 patients (2002–2010), whereas the HIC reported 1,003 patients (2002–2004). The most important known predictors of trauma mortality were adequately recorded in all datasets. Mortality benchmarking revealed that the HIC center performed similarly to the NTDB centers [O/E = 1.11 (95 % confidence interval (CI) 0.92–1.35)], whereas the LMIC center showed significantly worse survival [O/E = 1.52 (1.23–1.88)]. Subset analyses of patients with blunt or penetrating injury showed similar results.
Conclusions
Using only a few key covariates, aggregated global trauma data can be used to adequately perform international trauma center benchmarking. The creation of the ITDB is feasible and recommended as it may be a pivotal step towards improving global trauma outcomes.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Trauma has emerged as the preeminent global cause of death and disability accounting for an estimated 10 % of the world’s deaths. According to the World Health Organization (WHO), 5.8 million people die each year as a result of injuries; 32 % more than deaths resulting from malaria, tuberculosis, and HIV/AIDS combined [1]. Millions more suffer the nonfatal consequences of injury caused by lasting mental and physical disabilities. The global cost of road traffic crashes alone is estimated to be U.S. $518 billion; for some countries, this equates to 5 % of their gross national product [1]. Consequently, there exists an urgent need for dedicated research and health policy implementation to mitigate the disastrous societal and economic effects of trauma.
A successful strategy to improve trauma outcomes has been through improvements in quality of care (QoC). Research from multiple areas in healthcare, including trauma, has demonstrated that quality improvement (QI) initiatives reduce morbidity, mortality, complications, and costs [2–7]. In a recent systematic review by the World Health Organization-International Association for Trauma Surgery and Intensive Care (WHO-IATSIC), Juillard et al. [8] concluded that hospital-based and system-wide trauma QI initiatives “have been consistently shown to improve the process of care, decrease mortality, and decrease costs.” While the majority of the publications reviewed were from high-income countries, experiential evidence from other areas of medicine, particularly obstetric care, strongly support the feasibility and efficacy of QI programs in lower-middle income countries (LMIC) [9–13].
The cornerstone of any QI program is standardized collection of relevant healthcare information as databases [14, 15]. These databases typically enable measurement of the three Donabedian components of health quality: structure, process, and outcomes [16]. Analysis of these data helps to establish baselines, identify factors affecting QoC, monitor improvements temporally, and make interprovider comparisons. Injury-specific data recorded in trauma registries (but not in administrative hospital discharge datasets) is considered critical in improving QoC [17]. In the past two decades, numerous national trauma databases have been setup across Europe, North America, Israel, Japan, and Australia [18]. These databases have helped to identify and improve multiple areas of trauma care in their respective regions/countries [3, 19–23].
Recently, the American College of Surgeons Committee on Trauma (ACSCOT) suggested the development of a global trauma registry—the International Trauma Data Bank (ITDB)—with contributions from trauma registries from across the world. First proposed by Raul Coimbra, MD, at the 2011 ACSCOT spring meeting (personal communication), the central idea of the ITDB is to establish a mechanism for global comparative assessments of quality of trauma care to identify potential area for improvements and promote data-driven performance enhancement initiatives on a wider scale. However, a key problem identified remains the feasibility of data aggregation given the lack of standardized data collection practices across the world [24]. The objective of this study is to determine whether trauma data from across the world could be combined to explore the feasibility of performing international benchmarking using the proposed ITDB concept.
Methods
The goal of this study was to understand the opportunities and challenges associated with trying to compare trauma outcomes from different parts of the world, with dissimilar data collection practices. We compared mortality outcomes of two trauma centers (TCs) [one European high-income country (HIC) and one Asian lower-middle income country (LMIC)] with centers included in the United States/Canadian National Trauma Data Bank (NTDB). The European HIC center was an academic medical and TC (Lyon South Hospital) located in Lyon, France. This institution is one of two academic TCs in Lyon, serving a population of nearly 1.6 million. Injured patients are brought to the trauma center by prehospital physician providers. We included patients triaged to the center’s trauma resuscitation unit during 2002–2004.
The Asian LMIC center (Aga Khan University Hospital) is an academic medical and TC located in Karachi, Pakistan. This private, primary, and referral TC serves a population of 2.1 million. The hospital functions as part of a decentralized trauma system where patients receive no prehospital care. Over the past decade, as part of an institutional trauma quality improvement initiative, this center has hired dedicated trauma care providers, built new facilities, and implemented trauma care protocols based on adapted ACSCOT guidelines [13]. All patients meeting the institution’s trauma activation criteria during 2002–2010 were included [25, 26].
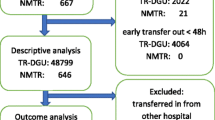
To explore the feasibility of international comparisons, these two centers were compared to one another and to data from North American centers in the NTDB. The NTDB is maintained by the American College of Surgeons and is the largest trauma database in the world, comprising annually submitted data on approximately 700,000 patients from more than 900 centers across the United States and Canada [27]. Since 2007, the quality of data in the NTDB has improved substantially with the institution of the National Trauma Data Standard (NTDS), which has standardized definitions, data collection, and reporting procedures [28]. Submission to the NTDB is voluntary; however, 97 % of level I and 75 % of level II TCs contribute data. Adult trauma patients (≥16 years of age) with blunt and/or penetrating injuries from all three datasets were included in the analysis. Patients who were dead on arrival were excluded. Hospitals in the NTDB missing >20 % data on covariates used to risk adjust were excluded [29]. Given the known association between hospital trauma volume and patient outcomes, the main analysis included NTDB hospitals with annual trauma volumes within 2 standard deviations of annual patient volumes at the HIC and LMIC, i.e., between 25 and 400 patients (Fig. 1) [30]. A sensitivity analysis, including all NTDB centers, also was performed.
Multiple patient demographic and injury severity measures were recorded in each of the three datasets. To ensure robust performance assessment, we included patient-level covariates that were uniformly and consistently reported for all centers and considered to be the most important predictors of in-hospital mortality following injury [31]. These included age, gender, type and mechanism of injury, presence of hypotension on arrival, total Glasgow Coma Scale (GCS), and Injury Severity Score (ISS). Because ISS scores reported in the different registries were derived using multiple various versions of the Abbreviated Injury Scale (AIS) scores, data from the three sources were categorized and standardized as described in Fig. 2, before its synthesis into an aggregate dataset. Age was categorized into deciles (16–25, 26–35, 36–45, 46–55, 56–65, 66–75, 76–85, and >85 years). Type and mechanism of injury were determined using International Classification of Diseases 9th edition Matrix of External-cause-of-injury codes (E-codes), where possible, and classified as blunt or penetrating, and stab, fall, gunshot wound, motor vehicle collision, pedestrian, struck-by/or against, or other mechanism, respectively. Hypotension at admission was defined as systolic blood pressure <90 mmHg on arrival and categorized as a binary variable (yes/no). Total GCS was categorized as 3–5, 6–8, 9–11, and 12–15. ISS was categorized as 0–8, 9–15, 16–24, and 25–75.
The recording of important patient level information in each data set and it standardized aggregation as the International Trauma Data Bank (ITDB). *International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM) External cause-of-injury codes. **Abbreviated Injury Scale (AIS). †Facilities reporting data to the National Trauma Data Bank used any one of the following AIS standards (AIS-80 through AIS-05 or AIS-MAP)
Baseline demographic and injury severity characteristics of patients admitted to the HIC and LMIC TC were each compared to NTDB patients using univariate statistics. To profile centers on mortality outcomes, we adapted the American College of Surgeons Trauma Quality Improvement Program (ACS-TQIP) methodology and ranked hospitals on risk-adjusted O/E ratios [32]. A standard multivariable logistic regression analysis was performed adjusting for age, gender, type of injury (blunt versus penetrating), presence of hypotension at admission, total GCS at admission, ISS, year of admission, and annual hospital volume. These covariates were chosen, because (1) they were consistently reported across the three datasets, and (2) included the basic set of covariates deemed necessary when risk-adjusting for trauma mortality [31, 33]. Model discrimination and calibration were assessed using the area under the receiver operating characteristics curve (AUROC) and calibration curves, respectively. Clustering by facility identifier was performed to account for correlated patient outcomes within individual hospitals. Subsequently individual patient probabilities of mortality were estimated and summed to calculate the “expected” number of deaths at each center. The “observed” or actual number of deaths at a center was then divided by expected number of deaths to calculate the O/E ratio along with its 95 % confidence interval. These O/E ratios (95 % CI) were plotted as a “caterpillar” graph and were used to classify individual hospitals as high performing (upper bound 95 % CI <1), average performing (95 % CI overlapping 1) or low performing (lower bound 95 % CI >1). Subset analyses for blunt and penetrating injury also were performed.
All three datasets contained deidentified patient information, and therefore this study was exempted from institutional review board approval. All analyses were performed using Stata12/MP (StataCorp, College Station, TX).
Results
From approximately 4.4 million patients available in the NTDB 2002–2010, a total of 375,433 patients from 301 centers were included in the main analysis (Fig. 1). The LMIC TC contributed 806 patients (2002–2010), whereas the HIC TC reported 1,003 patients (2002–2004). Figure 2 describes the recording of important patient level information in each data set and its standardized aggregation as the ITDB. Most covariates were similarly reported. However, AIS scores (and subsequent ISS calculation) were derived using different AIS versions. Most NTDB centers reporting data for years 2007 onwards and the HIC center used AIS-98 compared with AIS-90 used by LMIC center.
Table 1 compares the hospital level characteristics from the European HIC, Asian LMIC, and the NTDB. Nearly half of the NTDB centers (48 %) were level 1 centers. Both the HIC (>1,000 beds) and the LMIC center (542 beds) provided definitive patient care and hence were analogously classified as level 1 centers. The number of trauma, orthopedic, and neurosurgeons at these two centers were comparable to the NTDB centers, except that the LMIC center had only one core trauma attending.
Table 2 compares baseline patient demographic, type of injury, and crude mortality rates of patients at NTDB centers versus those at HIC and LMIC centers. Both the HIC and LMIC centers had significantly lower proportions of elderly patients (>65 years) than the NTDB (29.4 % at NTDB centers vs. 11.7 % at HIC and 3.8 % at LMIC centers, p < 0.001 for both comparisons). Both non-NTDB centers had a significantly greater burden of penetrating injury (14.5 % for HIC and 36.8 % for LMIC vs. 9.0 % for NTDB, p < 0.001 for both comparisons) and higher crude mortality rates (16.3 and 4.8 % at HIC and LMIC respectively vs. 3.3 % at NTDB, p < 0.001 for both comparisons).
Table 3 describes the mechanism of injury and injury severity characteristics. Motor vehicle collisions were the most frequent mechanism of injury in HIC (43.5 %) and LMIC (58.8 %) compared with falls (43.1 %) in the NTDB. Non-NTDB patients had more severe physiologic (23.4 and 5.1 % patients hypotensive on arrival at HIC and LMIC, respectively, vs. 2.9 % in the NTDB, p < 0.001 for both comparisons) and anatomic derangements (38.2 and 31.9 % patients with ISS ≥25 at HIC and LMIC, respectively, vs. 6.4 % at NTDB, p < 0.001 for both comparisons). The majority of NTDB and LMIC patients had GCS score above 12 (88.4 and 94.5 %, respectively) compared with only 11.2 % of the patients at the HIC center.
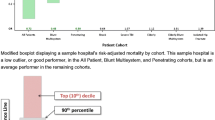
Figure 3 shows the position of the HIC and LMIC risk-adjusted O/E based mortality performance on caterpillar plot relative to NTDB centers. The HIC center’s performance was statistically no different than the average performing NTDB centers [O/E = 1.11(95 % CI 0.92–1.35)]. However, the LMIC TC showed significantly worse survival [O/E = 1.52 (1.23–1.88)]. The multivariable logistic model used to benchmark hospitals demonstrated excellent discrimination between survivors and nonsurvivors (AUROC >0.90) and adequate model fit (as assessed using calibration curves). A sensitivity analysis comparing the LMIC and HIC center to all centers in the NTDB did not significantly alter the results. Subset analyses stratified by injury type (blunt/penetrating) revealed a similar pattern; the LMIC demonstrated significantly higher O/E [blunt 1.55 (1.25–1.92), penetrating 1.63 (1.07–2.50)] compared with HIC [blunt 1.18 (1.00–1.41), penetrating 0.70 (0.44–1.12)] (Fig. 4).
Observed/expected (O/E) mortality ratios (95 % CI) for a hospital in a high-income and a lower-middle income country compared to trauma centers included in the NTDB; adjusted for age, gender, type of injury, presence of hypotension (systolic blood pressure <90), Glasgow Coma Scale, Injury Severity Score, year of admission, and hospital volume. Black line at one indicates that the hospital is performing as expected given its patient case-mix
Subset caterpillar plots by injury type; observed/expected (O/E) mortality ratios (95 % CI) for a hospital in a high-income and a lower-middle income country compared with trauma centers included in the NTDB; adjusted for age, gender, presence of hypotension (systolic blood pressure <90), Glasgow Coma Scale, Injury Severity Score, year of admission, and hospital volume > Black line at one indicates that the hospital is performing as expected given its patient case-mix. Both caterpillar plots truncated at O/E = 7 for clarity. a Blunt injury. b Penetrating injury
Discussion
Using trauma data from three different continents, this study establishes a proof-of-concept that global benchmarking of trauma center performance is feasible using aggregated data from countries across the globe. The most important covariates predicting postinjury outcomes were found to be recorded adequately and reported in all three datasets. As few as seven variables can be used to reliably predict in-hospital mortality with excellent discriminative ability. Hospitals from both a European HIC and an Asian LMIC were successfully benchmarked against NTDB TCs using the well-accepted, observed-to-expected (O/E) mortality ratios. This work shows that comparing outcomes using global trauma data is feasible. Therefore, we strongly support creation of the ITDB as a pivotal step towards improving global trauma outcomes.
Trauma registries help to improve patient outcomes and are considered an integral component of regional, national, and local trauma QI initiatives [3, 13, 19–23]. Initially, registries were simply in-patient administrative hospital records of trauma patients [17]. With the growing understanding of the impact of outcomes data on trauma care, patient safety, and performance improvement processes, these gradually evolved into regional/national repositories and increasingly included trauma-specific clinical information. Concurrently, complex injury severity assessment systems and risk-adjustment methodologies were developed to predict postinjury outcomes accurately [33–49]. Currently, using these large datasets and robust statistical methodologies, observational trauma studies help to guide physicians, researchers, and policy makers to improve quality of trauma care [3, 13, 19–23].
One key barrier to establishing large trauma data repositories is the lack of standardized data. While uniform reporting procedures can be developed, implemented, and enforced locally, international standardization is difficult to achieve given the inherent differences in national health policies and medical practices. Although trauma systems within single countries have successfully established standardized reporting practices, similar endeavors at the international level have yet to occur. A recent study by the European Trauma Audit Research Network (EuroTARN) found that trauma registries across Europe differed sufficiently to rule out meaningful outcomes comparisons [50]. Similar findings were reported by a group exploring the possibility of a Scandinavian Major Trauma Outcome Study [51]. To mitigate these concerns, a consensus panel of European experts has proposed a uniform data reporting standard, the Utstein Trauma Template (UTT), containing 36 core variables [52–54]. Since introduction of the NTDS, the U.S./Canadian-based NTDB already contains the majority of these core elements.
We successfully demonstrate that trauma data from three different global regions can be aggregated to perform adequately mortality-based external benchmarking using only a few critical variables. These findings are similar to those reported by Nathens et al., while evaluating patient and injury factors that most affected case-mix across NTDB TCs. They concluded that few variables are needed to risk-adjust adequately for mortality outcomes, obviating the need for extensive data collection [55]. This finding is important given the enormous costs associated with the implementation and maintenance of trauma registries. While standardized trauma data reporting initiatives are crucial and work well in HICs, they may not be feasible in resource-depleted LMICs [24]. Therefore, few important predictors of trauma mortality could be considered for the proposed ITDB to perform international benchmarking.
Most variables used to perform risk-adjustment in this present study were recorded uniformly. However, individual hospitals differed in their use of multiple AIS versions. Several studies have identified important differences between AIS versions and have suggested against the use of ISS derived from these varied sources when comparing outcomes [56–58]. These differences can, at least partially, be resolved by using mapping software to standardize the reported ISSs [59]. However, a more pressing challenge remains in the development of a globally accessible injury severity assessment system. Most injury scoring systems are resource intensive and are difficult to implement and maintain in LMICs. While simple, low-cost alternatives, such as the Kampala Trauma Score, have been specifically developed for LMICs, using different injury scoring systems may undermine the standardization of a global trauma repository [60]. Benchmarking HICs and LMICs separately would again segregate and regionalize global trauma initiatives rather than bring all regions onto a level playing field to compare outcomes appropriately. Additionally, this geographic segregation would not account for the existence of highly variable healthcare settings, resources, and access within each region.
Rather than pursuing comparisons that are global geographically, a more reasonable approach would be to compare similarly resourced centers with one another, because not all HIC hospitals are abundantly resourced and not all LMIC hospitals are ill-resourced. Using this system, low-resourced centers could use simpler injury scoring systems while higher-resourced centers could use more elaborate systems. This would create resource-based global benchmarking tiers, perhaps similar to how trauma centers in the United States are designated, which may offer greater intra-tier homogeneity of trauma data and enable more appropriate comparisons without regionalizing trauma quality improvements. However, without specific data on hospital resource profile, we restricted our comparisons to similar-volume NTDB centers. This volume-based comparison alone enabled us to identify NTDB centers that performed worse than the LMIC center, raising red flags regarding efficiency of resource utilization.
We compared mortality-based hospital performance using the validated techniques currently used by ACS-TQIP [32]. The ACS-TQIP has been modeled to replicate the methodology and success achieved by the ACS National Surgical Quality Improvement Program (NSQIP), a program that has helped reduce morbidity and mortality rates after major surgery across U.S. hospitals [5]. We specifically chose this methodology, because it remains the most well-recognized and widely cited comparative assessment of TC performance. However, other regional/national performance evaluation systems should be considered in the future to determine the optimal methodology to ensure the global trauma quality improvement initiative remains objective, evidence-based, and data-driven.
Our study has several limitations. First, the non-NTDB centers were chosen based on convenience and we included only one center each from a HIC and a LMIC, which may potentially be a source of bias, because they are not necessarily representative of their respective country’s injury profile. However, the goal of the analysis was not to assess performance of TCs across the world but to explore the challenges associated with the future conglomeration of trauma data. Using limited data on a few important patient variables, we demonstrated a proof-of-concept in support of global trauma benchmarking. Second, only mortality was used as a quality endpoint. While several studies recommend other important quality metrics to corroborate mortality-based performance assessments, such as complication rates or failure-to-rescue, these are not uniformly reported even in well-established trauma datasets [61–63]. Hence, we restricted our evaluation to the most commonly used outcome measure: in-hospital mortality.
In conclusion, this study demonstrates the feasibility of aggregating predictors of trauma mortality from existing trauma registries from around the world to undertake comparative performance assessments. This study highlights key areas for future exploration, such as global injury severity scoring systems and resource-based benchmarking. These findings may have important implications as we enter the era of evidence-based global trauma care.
References
World Health Organization (2010) Injuries and violence: the facts [Internet]. http://whqlibdoc.who.int/publications/2010/9789241599375_eng.pdf. Accessed 7 Jul 2013
Marciniak TA, Ellerbeck EF, Radford MJ et al (1998) Improving the quality of care for Medicare patients with acute myocardial infarction: results from the Cooperative Cardiovascular Project. JAMA 79(17):1351–1357
MacKenzie EJ, Rivara FP, Jurkovich GJ et al (2006) A national evaluation of the effect of trauma-center care on mortality. N Engl J Med 354(4):366–378
MacKenzie EJ, Weir S, Rivara FP et al (2010) The value of trauma center care. J Trauma 69(1):1–10
Khuri SF, Daley J, Henderson WG (2002) The comparative assessment and improvement of quality of surgical care in the Department of Veterans Affairs. Arch Surg 137(1):20–27
DiRusso S, Holly C, Kamath R et al (2001) Preparation and achievement of American College of Surgeons level I trauma verification raises hospital performance and improves patient outcome. J Trauma 51(2):294–299 discussion 299–300
Haut ER, Lau BD, Kraenzlin FS et al (2012) Improved prophylaxis and decreased rates of preventable harm with the use of a mandatory computerized clinical decision support tool for prophylaxis for venous thromboembolism in trauma. Arch Surg 147(10):901–907
Juillard CJ, Mock C, Goosen J et al (2009) Establishing the evidence base for trauma quality improvement: a collaborative WHO-IATSIC review. World J Surg 33(5):1075–1086. doi:10.1007/s00268-009-9959-8
Dumont A, Gaye A, de Bernis L et al (2006) Facility-based maternal death reviews: effects on maternal mortality in a district hospital in Senegal. Bull World Health Organ 84(3):218–224
Ifenne D, Essien E, Golji N et al (1997) Improving the quality of obstetric care at the teaching hospital, Zaria, Nigeria. Int J Gynaecol Obstet 59(Suppl 2):S37–S46
Mbaruku G, Bergström S (1995) Reducing maternal mortality in Kigoma, Tanzania. Health Policy Plan 10(1):71–78
Ronsmans C (2001) How can we monitor progress towards improved maternal health goals? Stud HSO&P 17:317–342
Hashmi ZG, Haider AH, Zafar SN et al (2013) Hospital-based trauma quality improvement initiatives: first step toward improving trauma outcomes in the developing world. J Trauma Acute Care Surg 75(1):60–68
U.S. Department of Health & Human Services (2013) Health Resources and Services Administration (HRSA) clinical quality & performance measures supplemental technical assistance: quality improvement [Internet]. Available at: http://www.hrsa.gov/quality/toolbox/methodology/qualityimprovement/index.html. Accessed 17 Jul 2013
Varkey P, Reller MK, Resar RK (2007) Basics of quality improvement in health care. Mayo Clin Proc 82(6):735–739
Donabedian A (2005) Evaluating the quality of medical care. 1966. Milbank Q 83(4):691–729
Moore L, Clark DE (2008) The value of trauma registries. Injury 39(6):686–695
European Trauma Registry Network (2013) Trauma Registries worldwide [Internet]. http://www.eurotrauma.net/site2/index.php?option=com_content&view=article&id=54&Itemid=59. Accessed 17 Jul 2013
Jurkovich GJ, Mock C (1999) Systematic review of trauma system effectiveness based on registry comparisons. J Trauma 47(3 Suppl):S46–S55
Patel HC, Bouamra O, Woodford M et al (2005) Trends in head injury outcome from 1989 to 2003 and the effect of neurosurgical care: an observational study. Lancet 366(9496):1538–1544
Fuller G, Bouamra O, Woodford M et al (2012) Recent massive blood transfusion practice in England and Wales: view from a trauma registry. Emerg Med J 29(2):118–123
Lecky F, Woodford M, Yates DW (2000) Trends in trauma care in England and Wales 1989-97. UK Trauma Audit and Research Network. Lancet 355(9217):1771–1775
Siman-Tov M, Radomislensky I, Peleg K (2012) Reduction in trauma mortality in Israel during the last decade (2000-2010): The impact of changes in the trauma system. Injury 44(11):1448–1452
Ringdal KG, Lossius HM, Jones JM et al (2011) Collecting core data in severely injured patients using a consensus trauma template: an international multicentre study. Crit Care 15(5):R237
Zafar H, Rehmani R, Raja AJ et al (2002) Registry-based trauma outcome: perspective of a developing country. Emerg Med J 19(5):391–394
Noordin S, Allana S, Ahmad T et al (2011) Evolution of trauma management at a tertiary care hospital: a cohort study. Int J Surg 9(1):75–78
American College of Surgeons (2010) National Trauma Data Bank: NTDB Research Data Set Admission Year 2010, Annual Report. American College of Surgeons, Chicago
American College of Surgeons. National Trauma Data Standard [Internet]. Available at: http://www.ntdsdictionary.org/index.html. Accessed 30 Aug 2013
Kardooni S, Haut ER, Chang DC et al (2008) Hazards of benchmarking complications with the National Trauma Data Bank: numerators in search of denominators. J Trauma 64(2):273–277 discussion 277–279
Nathens AB, Jurkovich GJ, Maier RV et al (2001) Relationship between trauma center volume and outcomes. JAMA 285(9):1164–1171
Haider A, Hashmi Z, Zafar S et al (2014) Developing best practices to study trauma outcomes in large data bases: an evidence based approach to determine the best mortality risk adjustment model. J Trauma Acute Care Surg 76(4):1061–1069
Nathens AB, Cryer HG, Fildes J (2012) The American college of surgeons Trauma quality improvement program. Surg Clin North Am 92(2):441–454 x–xi
Haider AH, Saleem T, Leow JJ et al (2012) Influence of the National Trauma Data Bank on the study of trauma outcomes: is it time to set research best practices to further enhance its impact? J Am Coll Surg 214(5):756–768
Baker SP, O’Neill B, Haddon W et al (1974) The injury severity score: a method for describing patients with multiple injuries and evaluating emergency care. J Trauma 14(3):187–196
Osler T, Baker SP, Long W (1997) A modification of the injury severity score that both improves accuracy and simplifies scoring. J Trauma 43(6):922–925 discussion 925–926
Kilgo PD, Meredith JW, Osler TM (2006) Incorporating recent advances to make the TRISS approach universally available. J Trauma 60(5):1002–1008 discussion 1008–1009
Osler T, Glance L, Buzas JS et al (2008) A trauma mortality prediction model based on the anatomic injury scale. Ann Surg 247(6):1041–1048
Glance LG, Osler TM, Mukamel DB et al (2009) TMPM-ICD9: a trauma mortality prediction model based on ICD-9-CM codes. Ann Surg 249(6):1032–1039
Haider AH, Villegas CV, Saleem T et al (2012) Should the IDC-9 Trauma Mortality Prediction Model become the new paradigm for benchmarking trauma outcomes? J Trauma Acute Care Surg 72(6):1695–1701
Osler T, Rutledge R, Deis J et al (1996) ICISS: an international classification of disease-9 based injury severity score. J Trauma 41(3):380–386 discussion 386–388
Clarke JR, Ragone AV, Greenwald L (2008) 5) Comparisons of survival predictions using survival risk ratios based on International Classification of Diseases, Ninth Revision and Abbreviated Injury Scale trauma diagnosis codes. J Trauma 59(3):563–567 discussion 567–569
Lavoie A, Moore L, LeSage N et al (2004) The New Injury Severity Score: a more accurate predictor of in-hospital mortality than the Injury Severity Score. J Trauma 56(6):1312–1320
Meredith JW, Evans G, Kilgo PD et al (2002) A comparison of the abilities of nine scoring algorithms in predicting mortality. J Trauma 53(4):621–628 discussion 628–629
Stephenson SCR, Langley JD, Civil ID (2002) Comparing measures of injury severity for use with large databases. J Trauma 53(2):326–332
Sullivan T, Haider A, DiRusso SM et al (2003) Prediction of mortality in pediatric trauma patients: new injury severity score outperforms injury severity score in the severely injured. J Trauma 55(6):1083–1087 discussion 1087–1088
Newgard CD, Fildes JJ, Wu L et al (2013) Methodology and analytic rationale for the American College of Surgeons Trauma Quality Improvement Program. J Am Coll Surg 216(1):147–157
Hashmi ZG, Dimick JB, Efron DT (2013) Reliability adjustment: a necessity for trauma center ranking and benchmarking. J Trauma Acute Care Surg 75(1):166–172
Glance LG, Osler TM, Mukamel DB et al (2009) Impact of statistical approaches for handling missing data on trauma center quality. Ann Surg 249(1):143–148
Oyetunji TA, Crompton JG, Ehanire ID et al (2011) Multiple imputation in trauma disparity research. J Surg Res 165(1):e37–e41
Edwards A, Di Bartolomeo S, Chieregato A et al (2007) A comparison of European Trauma Registries. The first report from the EuroTARN Group. Resuscitation 75(2):286–297
Ringdal KG, Lossius HM (2007) Feasibility of comparing core data from existing trauma registries in Scandinavia. Reaching for a Scandinavian major trauma outcome study (MTOS). Scand J Surg 96(4):325–331
Dick WF, Baskett PJ (1999) Recommendations for uniform reporting of data following major trauma–the Utstein style. A report of a working party of the International Trauma Anaesthesia and Critical Care Society (ITACCS). Resuscitation 42(2):81–100
Ringdal KG, Coats TJ, Lefering R et al (2008) The Utstein template for uniform reporting of data following major trauma: a joint revision by SCANTEM, TARN, DGU-TR and RITG. Scand J Trauma Resusc Emerg Med 16:7
Coats TJ (2010) A revised Utstein Template for trauma. Emerg Med J 27(5):339
Nathens AB, Xiong W, Shafi S (2008) Ranking of trauma center performance: the bare essentials. J Trauma 65(3):628–635
Salottolo K, Settell A, Uribe P et al (2009) The impact of the AIS 2005 revision on injury severity scores and clinical outcome measures. Injury 40(9):999–1003
Palmer CS, Niggemeyer LE, Charman D (2010) Double coding and mapping using Abbreviated Injury Scale 1998 and 2005: identifying issues for trauma data. Injury 41(9):948–954
Palmer CS, Franklyn M (2011) Assessment of the effects and limitations of the 1998 to 2008 Abbreviated Injury Scale map using a large population-based dataset. Scand J Trauma Resusc Emerg Med 19(1):1
Palmer CS, Franklyn M, Read-Allsopp C et al (2011) Development and validation of a complementary map to enhance the existing 1998 to 2008 Abbreviated Injury Scale map. Scand J Trauma Resusc Emerg Med 19:29
MacLeod JBA, Kobusingye O, Frost C (2003) A comparison of the Kampala Trauma score (KTS) with the revised Trauma score (RTS), Injury Severity Score (ISS) and the TRISS method in a Ugandan Trauma registry. Eur J Trauma 29(6):392–398
Moore L, Stelfox HT, Boutin A et al (2013) Trauma center performance indicators for nonfatal outcomes: a scoping review of the literature. J Trauma Acute Care Surg 74(5):1331–1343
Santana MJ, Stelfox HT (2013) Development and evaluation of evidence-informed quality indicators for adult injury care. Ann Surg 259(1):186–192
Glance LG, Dick AW, Meredith JW et al (2011) Variation in hospital complication rates and failure-to-rescue for trauma patients. Ann Surg 253(4):811–816
Acknowledgment
The authors thank Mary Lodenkemper for her editorial assistance and coordination. National Institutes of Health/NIGMS K23GM093112-01 and American College of Surgeons C. James Carrico Fellowship for the study of Trauma and Critical Care (Dr. Haider); Agency for Healthcare Research & Quality K081K08HS017952-01 (Dr. Haut).
Conflict of interest
None of the authors report any potential or real conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
This work was declared as the best oral presentation at the International Association for Trauma Surgery and Intensive Care/the American Association for the Surgery of Trauma (IATSIC/AAST) Scientific Paper Session at the International Surgical Week (ISW) 2013, Helsinki, Finland.
Rights and permissions
About this article
Cite this article
Haider, A.H., Hashmi, Z.G., Gupta, S. et al. Benchmarking of Trauma Care Worldwide: The Potential Value of an International Trauma Data Bank (ITDB). World J Surg 38, 1882–1891 (2014). https://doi.org/10.1007/s00268-014-2629-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00268-014-2629-5