Abstract
Background
The recurrence rate for incisional hernias following reconstruction depends not only on the size of the initial hernia or abdominal wall defect but also on the number of the concomitant diseases. The surgical approach chosen and the level of tension affecting the sutures both represent essential indicators of long-term success in abdominal wall reconstruction. Several techniques have been advocated to reconstruct the abdominal wall, either with or without use of alloplastic material. A number of authors even recommended separating the lateral components of the abdominal wall to preserve innervation and blood supply of the advanced parts.
Methods
This retrospective study is based on a patient collective consisting of 23 patients. In addition to hernias or abdominal wall defects, all of them suffered several concomitant diseases and were treated at the German university hospitals in Erlangen-Nürnberg and Freiburg. All procedures were performed between the years 2000 and 2006. In all cases, reconstruction of the abdominal wall was achieved by employing the separation of components technique by Ramirez. Some of these procedures entailed the use of alloplastic material. Use of this material was based on intraoperative tensiometry results. Surgical outcome was determined by evaluating postoperative complications and the hernia recurrence rate.
Results
We achieved complete anatomic reconstruction of the abdominal wall in 61% of cases. Alloplastic mesh was used in 39% of the cases. Results of a long-term follow-up (56 months) revealed that only 18% of patients experienced hernia recurrence.
Conclusions
Intraoperative tensiometry provides an additional important parameter for the surgical algorithm. Depending on the results, the appropriate surgical method for each case can be chosen. The decision on whether this procedure entails the use of alloplastic mesh can also be based on intraoperative tensiometry results.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
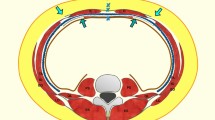
The reconstruction of massive abdominal wall defects, especially recurring midline hernias, represents a surgical challenge [1–4]. Obese patients with a body mass index (BMI) of 30 kg/m2 or higher and patients suffering from much co-morbidity represent an even greater surgical challenge [5–7]. The introduction of the “components of anatomic separation technique” by Ramirez et al. [8–10] is considered a landmark development for autologous tissue reconstruction of difficult to treat problem wounds [11, 12] (Fig. 1). There are two main factors influencing the rate of incisional hernia recurrence: the operative technique itself and the tension applied to the suture lines. The ultimate goal is to improve a patient’s well-being by reconstructing the abdominal wall. Although complete anatomic reconstruction (CAR) may be possible in even highly challenging cases, aiming for CAR may well lead to inferior functional results in comparison to a nonanatomic restoration. In certain cases, nonanatomical restoration may prove to be functionally sound, whereas CAR may not.
Components of anatomic separation technique by Ramirez. a Frontal view modified after "Reoperative Aesthetic and Reconstructive Plastic Surgery" by James C. Grotting, Vol. II, 1995. b Transversal view. E: external oblique muscle; I: internal oblique muscle; T: transversus abdominis muscle; R: rectus abdominis muscle; H: hernia; S: sutures; 1, 3: mobilization of the external oblique muscle; 2: mobilization of the rectus abdominis muscle through the anterior layer of the rectus sheath
Incisional hernias must be closed, and the abdominal wall has to be reconstructed, if possible, in an anatomic fashion [13]. However, sutures on the abdominal wall may not be subjected to excessive tension. Application of arbitrary tension results in a considerable rate of hernia recurrences irrespective of the kind of reconstruction technique used [11, 14–16]. In previous studies it was shown that the operative technique, and the tension applied to the sutured margins could be adjusted based on intraoperative tensiometry results [17–19]. In addition, the decision on whether to use alloplastic mesh can be made on the basis of these results [17–19]. In selected cases, where use of nonabsorbable alloplastic material is not deemed appropriate, an anatomic approach featuring the component separation technique can be the method of choice (Fig. 2). In addition to having an abdominal wall defect, a number of patients also suffer from wound healing problems, unstable scars, and infections following radiation treatment. As a consequence, this particular group of patients requires a strategic surgical approach, if possible, without the use of prosthetic materials, as they entail several risks. To date, there have been no published reports on routine measurement of applied tension during abdominal wall reconstruction.
Materials and methods
The study included 23 patients (15 males, 8 females). In addition to abdominal wall defects, they all suffered from several concomitant diseases. The mean age of the patients was 57 years. They were treated at the university hospitals in Erlangen-Nürnberg and Freiburg between the years 2000 and 2006 (Table 1). These patients represent a special patient collective. All patients experienced either wound healing problems, unstable scars, or infections. Furthermore, abdominal wall defects (e.g., incisional hernia) and/or preoperative cancer radiation treatment of the abdominal wall added to the complexity of the cases. The average BMI was 30 kg/m2. Prior to the operation, all 23 patients were clinically examined, and ultrasonography was performed. The average preoperative hernia size was 107 cm2 (Table 1).
Intraoperatively, a method of tensiometry first described by Klein et al. [17] was used (Fig. 3). One reason we performed intraoperative tensiometry was to determine if we could avoid the use of alloplastic material. The other reason was to help us decide on the type of suture to be used. Depending on the intraoperative clinical findings and tensiometry results, either direct suture or component separation of the lateral abdominal wall was performed. In cases where tensiometry results were below 1.5 kp, direct suture was performed. If the results exceeded 1.5 kp, component separation of the lateral abdominal wall was carried out. The specific value of 1.5 kp is the result of our clinical findings and 12 years of experience working with the intraoperative tensiometry technique [17–19].
Component separation of the lateral abdominal wall, first described by Ramirez et al., involves bilateral, innervated, bipedicled, rectus abdominis–transversus abdominis–internal oblique muscle flap complexes, which are transposed medially to reconstruct the central abdominal wall. In cases where intraoperative tension exceeded 1.5 kp, we used absorbable alloplastic materials to close and reconstruct the abdominal wall without any tension. In two cases, polypropylene mesh was used, as these patients showed no signs of infection during the preoperative clinical examination or during surgery.
We conducted a follow-up evaluation that including all 23 patients. The follow-up took place an average of 6 months after surgery. In addition, we performed a long-term follow-up study an average of 56 months after the operation. This particular follow-up entailed a questionnaire and telephone call. The three patients who experienced hernia recurrences were seen at the clinic for a physical examination.
Results
In this patient collective, there were no intraoperative indications to perform direct closure of the abdominal wall. Intraoperative tensiometry results were never below 1.5 kp in any of the cases. Following the Ramirez procedure, however, intraoperative tensiometry results were <1.5 kp in 14 patients (61%). As a consequence, we performed reconstruction of the abdominal wall without the use of alloplastic material. In 9 patients (39%), suture tension exceeded 1.5 kp following the Ramirez procedure. In these cases, we decided to use absorbable alloplastic material such as Vicryl (n = 7) or nonabsorbable alloplastic material such as polypropylene (n = 2) using the inlay/onlay technique. We achieved a CAR of the abdominal wall in 14 of 23 cases (61%). We defined all cases as CAR when we did not use alloplastic material.
None of the patients had suffered a recurring incisional hernia when examined during the 6-month postoperative follow-up. Overall, the postoperative complication rate was low. Five patients (22%) were treated for seroma, and two patients (9%) had secondary wound healing. However, one of these two patients had been given high doses of cortisone to treat severe asthma. At the long-term follow-up (56 months), we were able to contact 17 of the original 23 patients. We found that two of the six missing patients had died, and the other four could not be located. The long-term follow-up revealed hernia recurrence in three patients (18%) (patients 9, 17, and 21). However, when compared to the preoperative hernias, these hernias were 50% smaller.
Discussion
There has been much debate regarding the optimal surgical treatment of abdominal wall defects such as incisional hernias [1, 2]. Especially, high-risk patients suffering from concomitant diseases and individuals with a BMI exceeding 30 kg/m2 pose a surgical challenge [5]. Prior to the implementation and regular use of alloplastic meshes, a full-thickness abdominal wall defect >6 cm in diameter represented and, in some difficult cases still represents, a major surgical problem.
In 1990, Ramirez et al. presented an operative technique for closure of full-thickness abdominal wall defects >6 cm in diameter without using prosthetic material [8–10]. Abdominal wall component separation featuring bilateral, innervated, bipedicled rectus abdominis–transverses abdominis–internal oblique muscle flap complexes, which are transposed medially to reconstruct the central abdominal wall, allow the advancement of the medial rectus muscle borders up to 6 cm in the upper part on each side. In the middle part, as well as in the lower part, 5 cm can be gained on each side.
Shestak et al. described specific limitations and complications of the components repair method performed in 22 patients suffering from severe defects of the abdominal wall [11, 12]. In these cases, the defect area ranged from 84 to 354 cm2. They reported closing defects of 10 cm on each side. However, they did not measure the applied tension. The abdominal walls of two of their patients were examined using a quantitative dynamic measurement system (e.g., Cybex TEF machine). The analysis of the truncal flexion strength was undertaken preoperatively and 6 months following surgery. Their analysis of preoperative and postoperative truncal force generation revealed a 40% increase.
Voigt et al. also published results of nine cases using the Ramirez method of abdominal wall compound separation [20]. They reported that midline defects measuring up to 16 cm at the waistline had been successfully closed without applying any tension; however, they did not measure the tension of the sutures. Ennis et al. used a variation of the original Ramirez technique [21]. For more than 50 years, the use of various local or regional musculocutaneous or musculofascial flaps has been recommended and published [22, 23]. The use of an innervated free flap in this context was discussed by Nincovic et al. [24]. This particular approach should be reserved for special problems, however, because proper reinnervation of this type of flap cannot be guaranteed.
All of these operative procedures involve considerable donor-site morbidity. Additionally, most of these techniques provide poor or even no structural support to the abdominal wall of a patient suffering from many co-morbidities. Denervation of all transferred or transpositioned flaps is unavoidable. The subsequent muscular atrophy and hence tissue laxity are two major problems that lead to the loss of structural support and usually hernia relapse.
In this regard, constant progress and innovation in the field of medical biomaterials also led to an increase in the routine use of alloplastic meshes. There is no doubt that most full-thickness defects of the abdominal wall can be treated well with alloplastic meshes. In recent reviews, De Vries Reilingh et al. [2], Chang et al. [3], and Guzman-Valdivia et al. supported these findings [25]. However, the complication rate accompanying the use of synthetic meshes is significantly higher in obese and irradiated patients [4, 26–32]. Additionally, patients who exhibit signs of infection in the defect area or in general may not receive a nonabsorbable alloplastic mesh, as infection represents a contraindication for the use of such materials. Therefore, having a tissue repair technique that is able reliably to restore the structural integrity of the abdominal wall without the use of synthetic material would be extremely valuable in the treatment of massive abdominal wall defects.
Another important aspect is the fact that there is a relatively high risk of the occurrence of an intraabdominal compartment (IAC) syndrome when sutures are placed with high levels of tension [33–40]. Intraoperatively performed tensiometry may reduce the risk of postoperative IAC syndrome. With suture tension being in the physiological range (<3.5 kp), no undue pressure is transmitted to the intraabdominal compartment [17–19, 37]. Klein et al. showed that primary closure is safest when intraoperative tensiometry pressure measures <1.5 kp [17–19]. To measure it accurately, the patient has to be in a state of complete muscle relaxation.
In principle, midline defect reconstruction employing the component separation technique has been shown to be feasible and was described by several authors [10, 12, 20, 41]. Because this technique relies on preservation of innervated abdominal wall musculature, it seems preferable to other methods, such as lateral relaxing incisions [9, 12, 14, 17, 18, 20, 21, 42]. However, insights gained from midline hernia repair using an alloplastic mesh reveal that long-term success is dependent on suture tension. We recommend using intraoperative tensiometry to decide whether reconstruction with prosthetic materials can be safely performed. Preoperative and intraoperative evaluations of the size and nature of the abdominal defect (e.g., infection and/or the presence of an enterocutaneous fistula) provide the basis for the procedure of choice.
We hypothesize that intraoperative tensiometry is a technique that allows evaluation of an additional important parameter: the tension of the abdominal defect. It can be concluded that knowledge of three parameters—size, nature, and tension of the abdominal defect—can lead to a comprehensive, successful decision on the operative technique to be used in each case. Each abdominal defect has several characteristics that must be taken into account when deciding on the type of procedure to use.
Even though this study included 23 patients whose abdominal defects were difficult to treat, we were able to perform a CAR of the abdominal wall in 14 (61%) of them. This was achieved following the algorithm outlined above. Finally, only 3 of 17 patients were found to have a hernia recurrence. Compared with other previously published studies, these data suggest that the described concept yields good long-term results. Using intraoperative tensiometry as a decision tool regarding use of the component separation by Ramirez provides the surgeon with an additional, important parameter when appling the individual algorithm to autologous abdominal wall reconstruction.
References
Borud LJ, Grunwaldt L, Janz B et al (2007) Components separation combined with abdominal wall plication for repair of large abdominal wall hernias following bariatric surgery. Plast Reconstr Surg 119:1792–1798
Chang EI, Foster RD, Hansen SL, et al (2007) Autologous tissue reconstruction of ventral hernias in morbidly obese patients. Arch Surg 142:746–749; discussion 749–751
De Vries Reilingh TS, Bodegom ME, van Goor H et al (2007) Autologous tissue repair of large abdominal wall defects. Br J Surg 94:791–803
De Vries Reilingh TS, van Geldere D, Langenhorst B et al (2004) Repair of large midline incisional hernias with polypropylene mesh: comparison of three operative techniques. Hernia 8:56–59
Iannelli A, Bafghi A, Negri C et al (2007) Abdominal lipectomy and mesh repair of midline periumbilical hernia after bariatric surgery: how to spare the umbilicus. Obes Surg 17:1189–1192
Iljin A, Szymanski D, Kruk-Jeromin J et al (2008) The repair of incisional hernia following roux-en-Y gastric bypass: with or without concomitant abdominoplasty? Obes Surg 18:1387–1391
Rodriguez-Hermosa JI, Codina-Cazador A, Ruiz-Feliu B et al (2008) Incarcerated umbilical hernia in a super-super-obese patient. Obes Surg 18:893–895
Ramirez OM (1994) Abdominal herniorrhaphy. Plast Reconstr Surg 93:660–661
Ramirez OM (2000) Abdominoplasty and abdominal wall rehabilitation: a comprehensive approach. Plast Reconstr Surg 105:425–435
Ramirez OM, Ruas E, Dellon AL (1990) “Components separation” method for closure of abdominal-wall defects: an anatomic and clinical study. Plast Reconstr Surg 86:519–526
Shestak KC (2000) Progressive tension sutures: a technique to reduce local complications in abdominoplasty. Plast Reconstr Surg 105:2587–2588
Shestak KC, Edington HJ, Johnson RR (2000) The separation of anatomic components technique for the reconstruction of massive midline abdominal wall defects: anatomy, surgical technique, applications, and limitations revisited. Plast Reconstr Surg 105:731–738; quiz 739
Milburn ML, Shah PK, Friedman EB et al (2007) Laparoscopically assisted components separation technique for ventral incisional hernia repair. Hernia 11:157–161
Farthmann EH, Mappes HJ (1997) [Tension-free suture of incisional hernia]. Chirurg 68:310–316
Korenkov M, Sauerland S, Arndt M et al (2002) Randomized clinical trial of suture repair, polypropylene mesh or autodermal hernioplasty for incisional hernia. Br J Surg 89:50–56
Ladurner R, Trupka A, Schmidbauer S et al (2001) [The use of an underlay polypropylene mesh in complicated incisional hernias: successful French surgical technique]. Minerva Chir 56:111–117
Klein P, Konzen G, Schmidt O et al (1996) [Reconstruction of scar hernias: intraoperative tensiometry for objective determination of procedure of choice]. Chirurg 67:1020–1027
Klein P, Schmidt O, Reingruber B et al (1998) [Reconstruction of incisional hernias with defined tensiometrically controlled tension: a possibility for anatomically correct reconstruction of the abdominal wall]. Langenbecks Arch Chir Suppl Kongressbd 115:1027–1030
Reingruber B, Kastl S, Stremmel C et al (2001) Incisional hernia repair: tensiometry for the selection of the appropriate procedure. Eur J Surg 167:903–908
Voigt M, Andree C, Galla TJ et al (2001) [Reconstruction of abdominal-wall midline defects: the abdominal-wall components separation]. Zentralbl Chir 126:1000–1004
Ennis LS, Young JS, Gampper TJ, et al (2003) The “open-book” variation of component separation for repair of massive midline abdominal wall hernia. Am Surg 69:733–742; discussion 742–733
Ger R, Duboys E (1983) The prevention and repair of large abdominal-wall defects by muscle transposition: a preliminary communication. Plast Reconstr Surg 72:170–178
Wangensteen OH (1946) Repair of large abdominal defects by pedicled to fascial flaps. Surg Gynecol Obstet 82:144
Ninkovic M, Kronberger P, Harpf C et al (1998) Free innervated latissimus dorsi muscle flap for reconstruction of full-thickness abdominal wall defects. Plast Reconstr Surg 101:971–978
Guzman-Valdivia G, Guerrero TS, Laurrabaquio HV (2008) Parastomal hernia-repair using mesh and an open technique. World J Surg 32:465–470
Leber GE, Garb JL, Alexander AI et al (1998) Long-term complications associated with prosthetic repair of incisional hernias. Arch Surg 133:378–382
Voyles CR, Richardson JD, Bland KI et al (1981) Emergency abdominal wall reconstruction with polypropylene mesh: short-term benefits versus long-term complications. Ann Surg 194:219–223
Gesslein M, Horch RE (2006) [Interdisciplinary management of complex chronic ulcers using vacuum assisted closure therapy and “buried chip skin grafts”]. Zentralbl Chir 131(Suppl 1):S170–S173
Grimm A, Dimmler A, Stange S et al (2007) Expression of HIF-1 alpha in irradiated tissue is altered by topical negative-pressure therapy. Strahlenther Onkol 183:144–149
Loos B, Kopp J, Hohenberger W et al (2007) Post-malignancy irradiation ulcers with exposed alloplastic materials can be salvaged with topical negative pressure therapy (TNP). Eur J Surg Oncol 33:920–925
Hunter JE, Teot L, Horch R et al (2007) Evidence-based medicine: vacuum-assisted closure in wound care management. Int Wound J 4:256–269
Dragu A, Kneser U, Horch RE (2008) Post-mastectomy breast reconstruction: pectoralis major myomammary flap versus DIEP and MS-2 TRAM. World J Surg 32:502
Barker DE, Green JM, Maxwell RA, et al (2007) Experience with vacuum-pack temporary abdominal wound closure in 258 trauma and general and vascular surgical patients. J Am Coll Surg 204:784–792; discussion 792–783
Castellanos G, Pinero A, Fernandez JA (2007) [Intra-abdominal hypertension and abdominal compartment syndrome: what should surgeons know and how should they manage these entities?. Cir Esp 81:4–11
De Laet IE, Malbrain M (2007) Current insights in intra-abdominal hypertension and abdominal compartment syndrome. Med Intensiva 31:88–99
De Waele JJ, De laet I, Malbrain ML (2007) Intraabdominal hypertension and abdominal compartment syndrome: we have paid attention, now it is time to understand! Acta Clin Belg Suppl pp 6–8
Larsson A (2007) Clinical significance of elevated intraabdominal pressure during common conditions and procedures. Acta Clin Belg Suppl pp 74–77
Sonne ME, Hillingso JG, Hestad S et al (2007) [Measurement of intraabdominal pressure and abdominal compartment syndrome in surgical and intensive care units]. Ugeskr Laeger 169:705–710
DuBay DA, Wang X, Adamson B et al (2006) Mesh incisional herniorrhaphy increases abdominal wall elastic properties: a mechanism for decreased hernia recurrences in comparison with suture repair. Surgery 140:14–24
Orlando R 3rd, Eddy VA, Jacobs LM Jr et al (2004) The abdominal compartment syndrome. Arch Surg 139:415–422
Iqbal CW, Pham TH, Joseph A et al (2007) Long-term outcome of 254 complex incisional hernia repairs using the modified Rives-Stoppa technique. World J Surg 31:2398–2404
Kingsnorth AN, Shahid MK, Valliattu AJ et al (2008) Open onlay mesh repair for major abdominal wall hernias with selective use of components separation and fibrin sealant. World J Surg 32:26–30
Acknowledgment
We thank Mrs. Maya Wolf for language and grammar correction.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dragu, A., Klein, P., Unglaub, F. et al. Tensiometry as a Decision Tool for Abdominal Wall Reconstruction with Component Separation. World J Surg 33, 1174–1180 (2009). https://doi.org/10.1007/s00268-009-9991-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00268-009-9991-8