Abstract
Background
In malignant hepatic neoplasm, anatomic resection could improve survival and limit complications from hepatectomy. Our purpose was to develop an intraoperative method for identifying segment and subsegment of the liver with high-sensitivity near-infrared fluorescence imaging.
Methods
The subjects were 35 patients with hepatic malignant liver disease who received hepatectomy in 2006. The segments of liver method of identification that used infrared observation camera system termed Photo Dynamic Eye-2 (PDE-2) with indocianine green (ICG) for the patient with malignant liver tumor (hepatocellular carcinoma: 13 cases; metastatic liver cancer: 18 cases; intrahepatic cholangio carcinoma: 4 cases) were performed before liver resection.
Results
Although greenish stain of the liver surface after the injection of ICG via portal vein is not visible clearly without infrared observation camera system PDE-2, 1 minute after injection of ICG with fluorescent using infrared observation camera system PDE-2, demarcation of liver segment and subsegment was clearly detected. Ten minutes after injection of ICG with fluorescent using infrared observation camera system PDE-2, fluorescence of liver subsegment remained. Stained subsegment and segment of liver were identifiable in 33 (94.3%) of the 35 patients. There were no complications or side-effects related to the injection of patent blue dye.
Conclusion
We demonstrated here that near-infrared fluorescence imaging system is a novel and reliable intraoperative technique to identify hepatic segment and subsegment for anatomical hepatic resection.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
There has been a significant increase in the number of patients undergoing hepatic resections for hepatic malignancies. Improved understanding of the segmental anatomy of the liver has resulted in the evolution of liver resection procedures. Anatomic resection, which was originally introduced as segmentectomy and subsegmentectomy by Makuuchi et al. [1] involves systematic removal of a hepatic segment confined by tumor-bearing portal tributaries. This is necessitated by the high likelihood of the cancer cells, after effective eradication, producing intrahepatic metastases of hepatocellular carcinoma (HCC) [2, 3]. Moreover, segment-oriented resection allows maximal conservation of normal liver parenchyma while eliminating the tumor burden [4].
After injection of dye into the portal vein bordering the tumor-bearing area under intraoperative ultrasound guidance, anatomic segmentectomy and subsegmentectomy were performed. Although there is a technique for identifying the demarcation line for the segment or subsegment of the liver, which involves dividing the liver after occlusion of the portal vein and hepatic artery, conventional indigo carmine injection remains the “gold standard” [1] for identifying the segments and subsegments of the liver. This method stained the surface of the liver clearly; however, it was impossible to follow the intersubsegmental or intersegmental plane during division of the liver parenchyma because the dye disappeared soon after injection [5]. Sakairi and Makuuchi [5] demonstrated the modified Makuuchi’s method with a surgical clip. With their method, staining of the portal unit persists even after resection is complete, and the margin of the portal unit within the parenchyma is easily followed during transection. However, their procedure needed to create a slit approximately 1 cm wide from the surface of liver for insertion of Surgical clip applier.
Recently, illuminated with near-infrared light (NIR) has been used to detect the sentinel lymph node in breast [6, 7], gastric [8, 9], lung [10, 11], and esophageal [12] cancers and to assess intraoperative graft patency [13–18]. Using this novel technology, we showed for the first time, as described in this preliminary report, the use of this novel and highly sensitive imaging technique for intraoperative identification of liver segments, although in this study the comparison with the conventional dye injection method advocated by Makuuchi et al. could not be addressed. The technique is based on capture, by a charge-coupled device videocamera, of the fluorescence of ICG when illuminated with NIR using a PDE -2 imaging system (Hamamatsu Photo Inc., Hamamatsu, Japan).
Patients and methods
The subjects were 35 patients with malignant liver disease who underwent hepatectomy in 2006. Their ages ranged from 46 to 80 (mean age, 66) years. There were 20 men and 15 women. The liver segments method of identification, using the PDE-2 with ICG fluorescence for patients with malignant liver tumor (HCC: 13 cases, metastatic liver cancer: 18 cases, intrahepatic cholangio carcinoma: 4 cases), was performed before liver resection (Table 1). The procedure was conducted in accordance with the ethical standards of our institution’s Committee on Human Experimentation.
PDE imaging system
The PDE-2 imaging system is based on fluorescence of ICG. Fluorescence images were obtained from Hamamatsu Photonics, Hamamatsu, Japan. The light source, a light-emitting diode (LED), emitted light at a wavelength of 760 nm, and the detector was a charge-coupled device (CCD) camera, which subsequently filtered light with a wavelength <820 nm. Thirty-six LEDs were aligned on a board in an area, and the CCD camera was set in the center. The fluorescence signals were sent to a digital video processor for display on a TV monitor. When ICG binds to human plasma, ICG is detectable as fluorescence using an infrared observation camera system PDE-2 (Fig. 1)
Identification of liver segments and subsegments
One milliliter of ICG (Wako Pure Chemical Industries, Osaka, Japan) (5 mg/ml) was injected using a 23-gauge needle into the portal vein branch, with a Pringle maneuver under ultrasonography (Fig. 2). The intermittent Pringle maneuver required periods of inflow clamping that last 15 min for the injection of ICG. The amount and concentration of ICG was not adjusted depending on the volume of the liver subsegment or segment to be stained. An infrared observation camera system termed PDE-2 was linked for observation to the fluorescence excitation of ICG with LED immediately after the operating room was darkened. The observers, at 20–30 cm, took their eyes off the abdominal wall, allowing observation on a liquid crystal display television monitor. At this point, the extent of segment and subsegment in the liver can be determined according to the results of the dye injected through the portal vein.
Results
Feasibility
Stained subsegments and segments of the liver were identifiable in 33 (94.3%) of the 35 patients. The procedure was unsuccessful in two patients: one because of blockage of portal flow secondary to portal hypertension, and another because of technical failure with the injection. There were no complications or side-effects related to the injection of patent blue dye.
Mapping of liver segments and subsegments
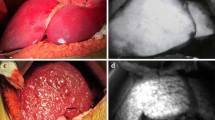
In the present study, mapping of the liver stained with ICG alone was not consistently visualized by all attending surgeons (Fig. 3A), whereas segments and subsegments of the liver were immediately identified distinctly 1 min after ICG injection with the PDE-2 camera system (Fig. 3B). The demarcation line of a liver segment or subsegment liver was distinguished with fluorescence, even 10 min after injection with fluorescence using an infrared observation camera system (Fig. 3C), the PDE-2 showed the demarcation line between S6 and S7 to be clearly indentified (Fig. 3D). Thus, demarcation of liver subsegments was readily achieved using elecrocautery (Fig. 3E). In addition, when a tumor was located near the liver surface, it was depicted as a defect image and could not be removed surgically with a neoplastic locus, and a locus correlation for the corresponding area was clear. Hepatic fluorescence was retained even after resection of the liver (Fig. 4A, B).
(A) Greenish staining of the liver surface after the injection of ICG via the portal vein is not visualized clearly. (B) One minute after injection of fluorescent ICG using the fluorescent using infrared observation camera system PDE -2, the liver subsegment is clearly demonstrated. (C) Ten minutes after injection of fluorescent ICG using the infrared observation camera system PDE -2, fluorescence of liver subsegment persisted. (D) Demarcation between S6 and S7 is clearly identified. (E) Demarcation of this liver subsegment was marked using electrocautery
Discussion
It is standard for HCC patients to undergo anatomic resection, because cancer cells from HCC can easily go through the portal vein and create metastatic lesions in the liver [1, 2]. Moreover, segmental resection is a safe technique that allows complete resection of liver tumors with preservation of normal liver parenchyma [4]. Thus, anatomical liver resection is a crucial method for treating patients with malignant hepatic neoplasms. To perform anatomical resection of liver for HCC patients, Makuuchi et al. demonstrated a novel method to identify the subsegment or segment of liver using dye staining method under intraoperative ultrasonography. This method stained the surface of the liver clearly however, it was impossible to follow the intersubsegmental or intersegmental plane during division of the liver parenchyma because the dye disappeared soon after injection [5]. Sakairi et al. showed their modified Makuuchi’s method with a surgical clip. With their method, staining of the portal unit persists even after resection is complete, and the margin of the portal unit within the parenchyma is easily followed during transection. However, their procedure needed to create a slit approximately 1 cm wide from the surface of liver for insertion of Surgical clip applier [5].
This study essentially demonstrated clinical development of fluorescence navigation with indocyanine green for mapping of liver segments and subsegments for anatomical hepatic resection. To our knowledge, this is the first report that describes the use of this novel technique in the intraoperative setting for mapping of the liver using fluorescence imaging in liver surgery. Our preliminary results demonstrate that this fluorescence navigation system, based on the fluorescence of ICG, is a safe and highly sensitive method for intraoperatively assessing liver demarcation.
ICG has been widely used in a variety of clinical situations, and adverse reactions are uncommon. Moreover, ICG is known to absorb infrared rays. Light, and specifically NIR in the biological window (700–900 nm), can be exploited for intraoperative imaging guidance. Tanaka et al. demonstrated that (1) the appearance of the surgical field is not altered, (2) it is safe, (3) wavelengths in the 800 nm range penetrate relatively deeply into living tissue, and (4) there has been a tremendous recent effort in developing general-purpose NIR fluorophores that can be conjugated to targeting or other molecules, thus creating “contrast agents” matched to any desired surgical application [19]. Therefore, ICG injection combined with the fluorescence navigation system is now widely used to detect sentinel lymph nodes [7–12], because this technique is convenient and safe for assessing lymph node status in the oncological field. We speculate that this novel technique can be readily adapted to detect demarcation lines of the liver in patients with hepatic malignant neoplasm. Herein, although mapping of the liver stained with ICG alone was not consistently visualized by all attending surgeons, the liver segment or subsegment was immediately well visualized and we obtained semiquantitative fluorescence of the liver with real-time imaging with the PDE camera system. A major benefit is that this system provides a clear image of the demarcation lines in the liver and continuous fluorescence imaging during Pringle maneuver.
Makuuchi et al. [1] showed with their novel technique that dye was injected under the clamp of only the hepatic artery with maintaining the portal flow for identification of segment or subsegment of liver. When ICG was injected into the portal vein branch with PDE-2 camera system to identify the subsegment or segment of liver according to their method, ICG was systemically circulated, and then, gradually, whole liver was stained because of remaining portal flow. To avoid this phenomenon, we performed ICG injection under Pringle maneuver. Although in this study the comparison with the conventional dye injection method advocated by Makuuchi et al. [1] could not be addressed, we need to perform a comparative study in near future, which is essential to establish whether our method is superior.
The fluorescence imaging technique for identification of segments and subsegments of the liver described herein has the advantages of safety and reproducibility. This study provides insights into the imaging system for subsegmentectomy or segmentectomy in patients with malignant hepatic neoplasms.
References
Makuuchi M, Hasegawa H, Yamazaki S (1985) Ultrasonically guided subsegmentectomy. Surg Gynecol Obset 161:346–350
Makino I, Chijiiwa K, Kondo K et al (2005) Prognostic benefit of selective portal vein occlusion during hepatic resection for hepatocellular carcinoma. Surgery 137:626–631
Hasegawa K, Kokudo N, Imamura H et al (2005) Prognostic impact of anatomic resection for hepatocellular carcinoma. Ann Surg 242:252–259
Billingsley KG, Jarnagin WR, Fong Y et al (1998) Segment-oriented hepatic resection in the management of malignant neoplasms of the liver. J Am Coll Surg 187:471–481
Sakairi T, Makuuchi M (1991) Identification of the intersegmental or subsegmental plane in the liver with a surgical clip. Surgery 110:903–904
Kitai T, Inamoto T, Miwa M et al (2005) Fluorescence navigation with indocyanine green for detecting sentinel lymph nodes in breast cancer. Breast Cancer 12:211–215
Motomura K, Inaji H, Komoike Y et al (1999) Sentinel node biopsy guided by indocyanine green dye in breast cancer patients. Jpn J Clin Oncol 29:604–607
Nimura H, Narimiya N, Mitsumori N et al (2004) Infrared ray electronic endoscopy combined with indocyanine green injection for detection of sentinel nodes of patients with gastric cancer. Br J Surg 91:575–579
Ishikawa K, Yasuda K, Shiromizu A et al (2007) Laparoscopic sentinel node navigation achieved by infrared ray electronic endoscopy system in patients with gastric cancer. Surg Endosc 21:1131–1134
Ito N, Fukuta M, Tokushima T et al (2004) Sentinel node navigation surgery using indocyanine green in patients with lung cancer. Surg Today 34:581–585
Soltesz EG, Kim S, Laurence RG et al (2005) Intraoperative sentinel lymph node mapping of the lung using near-infrared fluorescent quantum dots. Ann Thorac Surg 79:269–277
Parungo CP, Ohnishi S, Kim SW et al (2005) Intraoperative identification of esophageal sentinel lymph nodes with near-infrared fluorescence imaging. J Thorac Cardiovasc Surg 129:844–850
Taggart DP, Choudhary B, Anastasiadis K et al (2003) Preliminary experience with a novel intraoperative fluorescence imaging technique to evaluate the patency of bypass grafts in total arterial revascularization. Ann Thorac Surg 75:870–873
Detter C, Russ D, Iffland A et al (2002) Near-infrared fluorescence coronary angiography: a new noninvasive technology for intraoperative graft patency control. Heart Surg Forum 5:364–369
Rubens FD, Ruel M, Fremes SE (2002) A new and simplified method for coronary and graft imaging during CABG. Heart Surg Forum 5:141–144
Sekijima M, Tojimbara T, Sato S et al (2004) An intraoperative fluorescent imaging system in organ transplantation. Transplant Proc 36:2188–2190
Raabe A, Beck J, Gerlach R et al (2003) Near-infrared indocyanine green video angiography: a new method for intraoperative assessment of vascular flow. Neurosurgery 52:132–139
Kuroiwa T, Kajimoto Y, Ohta T (2001) Development and clinical application of near-infrared surgical microscope: preliminary report. Minim Invasive Neurosurg 44:240–242
Tanaka E, Choi HS, Fujii H et al (2006) Image-guided oncologic surgery using invisible light: completed pre-clinical development for sentinel lymph node mapping. Ann Surg Oncol 13:1671–1681
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Aoki, T., Yasuda, D., Shimizu, Y. et al. Image-Guided Liver Mapping Using Fluorescence Navigation System with Indocyanine Green for Anatomical Hepatic Resection. World J Surg 32, 1763–1767 (2008). https://doi.org/10.1007/s00268-008-9620-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00268-008-9620-y