Abstract
The aim of this prospective study was to analyze Ming’s classification in correlation with other currently used classification systems of gastric cancer. In addition, we wanted to define the prognostic significance of the Ming classification system. The present study analyzed material of 117 patients with gastric carcinoma who underwent D2-gastrectomy with curative intent. All specimens were catagorized according to International Union Against Cancer (UICC) classification, World Health Organization (WHO) classification, Borrmann classification, Laurén classification, Goseki classification, Ming classification, and tumor differentiation. For analysis of correlation between the classification systems, the correlation coefficient according to Spearman was calculated. The survival curves have been calculated according to the Kaplan-Meier method. According to the Ming classification, 38.5% of the carcinomas exhibited an expanding growth pattern, and 61.5% of specimens showed an infiltrating growth pattern. The subtypes according to the Ming and Laurén classification correlated significantly (P < 0.001). WHO classification (P < 0.001), tumor differentiation (P < 0.001), and Goseki classification (P < 0.001), as well as the macroscopic classification of Borrmann (P < 0.001) and the pT and pN categories of the UICC classification exhibited a highly significant correlation with the Ming classification (P < 0.001 and 0.001, respectively). Median overall survival was 31.3 months. In Kaplan-Meier analysis, the 3-year survival rates were lower in the infiltrative tumor type when compared to the expansive tumor type according to Ming (P = 0.0847). In multivariate analysis, only the UICC system presented as an independent prognostic factor in multivariate analysis (P < 0.001). This study shows that the Ming classification correlates significantly with the currently used classification systems for gastric cancer and with the UICC staging system, especially, the pT and pN category. The 3-year survival rates were lower in the infiltrative tumor type than in the expansive tumor type according to Ming. However, the Ming classification is not an independent prognostic factor.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Gastric carcinomas exhibit various pathological features and show great differences with regard to their histogenetic origin.1,2 A main problem in the histological classification of gastric cancer is the great variability of architectural and cytological features that may be present within the same tumor.3 As a result, many different systems have been proposed for the histological classification of gastric carcinoma. The fact that so many systems are in use simultaneously indicates that none of them are completely satisfactory. For clinical use, a classification system should ideally be easy to use, reliable, reproducible, biologically meaningful, and clinically relevant. In addition, to optimize therapy strategies, it would be very desirable to have a classification system of gastric carcinoma that allows preoperative evaluation of tumor progression and lymph node metastasis. As we know, especially in gastric carcinoma, the exact preoperative staging is of great importance in choosing the best therapy options (concept of targeted surgery). At present, the classification systems most widely used are those of Laurén4 and the World Health Organisation (WHO).5,6 The widely applied Laurén classification divides stomach cancers into intestinal, diffuse, and mixed types. In contrast, the WHO classification categorizes cancers according to features of histopathological differentiation.6 Papillary, tubular, mucinous, and signet ring cell types are defined. In 1992, Goseki et al., 7,8 described a novel grading system based on tubular differentiation and intracellular mucin; it was used to categorize gastric cancers in a series of 200 patients investigated by autopsy. The Goseki system classifies gastric carcinomas into four groups. The Ming classification, based on the growth pattern of the cancer, recognizes two main growth patterns: the expanding growth pattern accounted for 67% of gastric cancers and the infiltrating growth pattern which accounted for 33% of gastric cancers in previous studies1,9,10 The aim of the present prospective study was to analyze the relationship between the Ming classification and other histopathological classification systems, and to investigate the clinical and prognostic relevance of the Ming system.
Methods
Patients
The specimens included in this prospective study were obtained from 117 consecutive patients who underwent gastric surgery with curative intent for primary gastric adenocarcinoma at the Department of Surgery, University of Cologne, Germany, between 1996 and 2000. A total of 68 specimens were obtained from men and 49 were obtained from women. The patients ranged from 33 to 85 years of age (median: 65 years). Of these patients, 105 (89.7%) underwent gastrectomy with D2-lymphadenectomy with curative intent, and 12 (10.3%) underwent subtotal gastric resection with D2-lymphadenectomy, also with curative intent. A mean number of 39 lymph nodes were resected per patient. Fifteen (12.8%) patients with locally advanced tumors had been treated preoperatively with cytotoxic drugs and/or radiation following the concept of neoadjuvant therapy.
Methods
The tumor samples were routinely fixed in 5% phosphate-buffered formalin and embedded in paraffin. Specimens were stained with hematoxylin and eosin and periodic acid–Schiff (PAS) staining according to standard histological techniques. Appropriate tissue samples were categorized according to the classifications of Borrmann, the International Union Against Cancer (UICC; sixth edition, 2002),11 the World Health Organization (WHO), Laurén, Goseki, and Ming. All tumor specimens were independently evaluated by two pathologists. There was a deviation of median values <5%; in these cases, a consensus was obtained between the two pathologists.
The study was performed according to the criteria for prognostic studies.12 The relationship between the Ming classification and the other systems was analyzed using the correlation approach of Spearman. In univariate analysis, overall survival was calculated by the Kaplan-Meier method. The log rank test was used to calculate significance of differences between subgroups, and the Cox regression model was used for multivariate analysis. The following variables were taken into the conditional forward model: UICC stage, sex, Laurén, Goseki, and Ming classification. The median follow-up was calculated according to Bollschweiler.12
All calculations were performed using the SPSS statistical software package (SPSS for Windows release 10.0.7de, SPSS Inc., Chicago, IL); P values lower than 0.05 were considered statistically significant.
Results
R0 resection could be achieved in 106 (90.6%) patients. Lymph node metastases were found in 75 (64.1%) specimens, and distant metastases were seen in 19 (16.2%) patients. The distribution of the different classifications and their association with the Ming classification system are shown in Tables 1 and 2, as are the correlations between the different classifications and the Ming classification.
The bigger the tumor was, the more often the infiltrative tumor type was present (P < 0.001). The higher the incidence of lymph node metastasis, the more frequent the infiltrative tumor type was observed (P < 0.001). Conclusively, the same correlation was found between the Ming classification and the UICC classification; the higher the UICC classification grade, the more frequently the infiltrative tumor type was found (P < 0.001). In our study, poorly differentiated or undifferentiated tumors according to the WHO classification were shown to exhibit exclusively the infiltrative tumor type according to Ming. The lower the Goseki classification, the more often the expansive tumor type was predominant (P < 0.001). The better the grading, the more often the expansive tumor type was found (P < 0.001). And finally, the higher the Borrmann classification, the more often the infiltrative tumor type was observed (P < 0.001).
The median follow-up was 31.3 months. Median overall survival was also 31.3 months (range: 19.8–42.7 months). Briefly, the patients with an expansive tumor type showed a median survival of 46.8 months (range: 17.7–75.9 months), whereas patients with an infiltrative type had a median survival of only 22.7 months (range: 11.8–33.7 months).
In our study, the overall cumulative 3-year survival rate was 45.05% (SE 4.72%). The 3-year survival rate was 56.82% (SE 7.47%) and 35.82% (SE 5.86%), respectively, in patients with the expansive and infiltrative tumor types according to Ming. Thus, the infiltrative type of cancer (according to Ming) had a worse prognosis than the expansive type (P = 0.0847; Fig. 1).
The Ming classification and the Goseki, Laurén, WHO, and Borrmann classifications, as well as tumor differentiation, did not exert a prognostic effect on survival in multivariate analysis. Only the UICC system proved to be an independent prognostic factor in multivariate analysis (P < 0.001).
Discussion
The characteristics of gastric carcinomas can be described according to many of their features: for example anatomic site, clinical and pathological extent of disease, tumor size, histological type, grade, and growth pattern. With regard to their growth pattern, many carcinoma cells maintain a coherent relationship with each other and form relatively circumscribed tumors, like adenocarcinomas, of other organs, for example those of the intestine. However, tumor cells of about one third to one half of all gastric carcinomas show a loss of cohesive growth and invade other organs as individual cells or small groups of cells or glands.13 Some tumors exhibit a significant intratumoral heterogeneity of these morphological features.
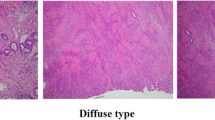
According to the different growth patterns and the varying extent of invasiveness among gastric carcinomas, Ming proposed a classification of gastric carcinomas as “expanding” and “infiltrative” types.1 As the terms suggests, expanding carcinomas grow by enlargement of cohesive tumor nodules or masses, with a well defined periphery; infiltrative carcinoma cells grow independently as dispersed, separate cells and clusters or small glands that show a strong invasive potential with extensive infiltration into the stroma. According to the literature, the expanding and infiltrative growth patterns account for roughly 65% and 30% of all gastric cancers, respectively.1 In the present study, we observed an obviously selected patient series. As a result, 38.3% of our specimens were considered to have an expanding growth pattern and 61.7% an infiltrating growth pattern. The growth patterns are mixed in 5% of tumors, 2% with a predominant expanding pattern and 3% with a prominent infiltrative pattern.13 Some expanding tumors show an infiltrative growth pattern at their advancing margins, a characteristic that reflects increased malignancy of the tumor. The designation of tumor type, however, should be based on the growth pattern of the entire tumor; if the infiltrative pattern is limited to the periphery of the tumor, the basic biology of the expanding carcinoma may have not been substantially altered.13 Expanding carcinomas are usually present in polypoid and fungating carcinomas, whereas diffusely infiltrative carcinomas are made up of non-cohesive infiltrative tumor cells. Grossly ulcerative carcinomas are about equally divided between the expanding and infiltrative growth patterns.13
Many different systems have been proposed for the histologic classification and grading of gastric carcinoma.1,4,6,7,14–16 However, none of them meet the following two requirements of clinical management: prediction of (curative) resectability and determination of prognosis. This lack is illustrated by the fact that many investigators are still trying to find a new classification system with prognostic value. Tumor-nodes-metastases (TNM) staging is a well-known, important prognostic measure of survival.17,18 However, it cannot be assessed preoperatively with accuracy, especially concerning the N status.19 In addition to the commonly accepted systems (UICC classification, WHO classification, Borrmann classification, Laurén classification, Goseki classification), we also analyzed the Ming classification. Compared with the other classifications or grading methods, the Ming system was found to be significantly correlated with the Laurén classification (P < 0.001) and the pT and pN categories (P < 0.001 and 0.001 respectively). Four other systems also showed a significant relationship with the Ming classification: the WHO classification (P < 0.001), tumor grading (P < 0.001), the Goseki classification (P < 0.001), and the macroscopic classification of Borrmann (P < 0.001). A group in the UK20 studied 181 patients with gastric cancers removed by potentially curative resections and compared the Goseki subgrouping with conventional tumor grading as well as the Laurén, Ming, and WHO classifications. In contrast to our results, they did not find any correlation between the Ming classification and the Goseki group.20 Additionally, Piard et al., 21 who reviewed 211 gastric carcinomas, could not correlate the WHO classification with the Ming classification. In accordance with our results, other authors have observed a striking correlation between the Laurén and the Ming classifications.22–24 This may be explained by the definition of the subtypes according Ming’s and Laurén’s classifications. Expanding carcinomas mostly exhibit an intestinal phenotype, and infiltrative carcinomas usually show a diffuse growth pattern. However, the solid carcinoma, unclassified according to Laurén’s system, represents an expanding carcinoma.13
Ming did not consider any prognostic implications for his classification system.1 However, some authors have tried to evaluate the prognostic relevance of Ming’s classification. Up until now, any published findings with regard to the prognostic value of the Ming classification have been controversial. Whereas some authors could demonstrate prognostic value,22,23,25 others21,24 did not observe such an association.
In our study of patients with the expansive and infiltrative tumor types, as defined by Ming, the overall cumulative 3-year survival rate was 45.05% (SE 4.72%). The 3-year survival rate for patients with the expanding type of tumor was 56.82% (SE 7.47%) and for those with the infiltrating type, it was 35.82% (SE 5.86%). Thus the infiltrative type of cancer (according to Ming’s classification) had a statistically nearly significantly worse prognosis than the expansive type (P = 0.0847).
In conclusion, in our study, the Ming classification was found to correlate well with the conventional grading and classification systems of gastric cancer, with findings that are comparable with the results of other groups.22–24 Additionally, the Ming classification was of prognostic relevance in our study, for the infiltrative type of cancer (according to Ming) had a poorer prognosis than the expansive type (P = 0.0847). This result confirmed the findings of other groups.22,23,25 However, the Ming classification does not fulfill the requirements of preoperative prediction of (curative) respectability and preoperative determination of prognosis. This is due to characteristics of tumor growth of gastric carcinomas. Some expanding tumors show an infiltrative growth pattern at their advancing margins, reflecting increased malignancy of the tumor. However, the designation of tumor type should be based on the growth pattern of the entire tumor. If the infiltrative pattern is limited to the periphery of the tumor, the basic biology of the carcinoma may have not been substantially altered.13 As a result, the preoperative classification of small biopsies according to Ming may be misleading because biopsies are generally not representative of the areas at the invasive front of a tumor.26 Thus, the Ming classification should only be applied to the surgically resected specimen.
In accordance with Ming and other authors,21–23,25 our observations confirm and extend the notion that the Ming classification is easy to use; that it is biologically, clinically and prognostically relevant; and that it is easily reproducible. Because of its validity, this classification is suitable for daily practice, but it should not be determined preoperatively from analysis of biopsy specimens.
References
Ming SC. Gastric carcinoma: a pathobiological classification. Cancer 1977;39:2475–2485
Fenoglio-Preiser CM, Noffsinger AE, Belli J, et al. Pathologic and phenotypic features of gastric cancer. Semin Oncol 1996;23:292–306
Flucke U, Monig SP, Baldus SE, et al. Differences between biopsy- or specimen-related Lauren and World Health Organization classification in gastric cancer. World J Surg 2002;26:137–140
Lauren PA, Nevalainen TJ. Epidemiology of intestinal and diffuse types of gastric carcinoma. A time-trend study in Finland with comparison between studies from high- and low-risk areas Cancer 1993;71:2926–2933
Watanabe A, Oshiro T, Oiwa H, et al. Prognosis of patients with gastric cancer and pyloric stenosis: histological differentiation. Semin Surg Oncol 1994;10:121–124
Watanabe H, Nishimaki T. Factors affecting the growth and extension of gastric cancer—analysis of retrospective observation. Gan To Kagaku Ryoho 1983;10:482–488
Goseki N, Takizawa T, Koike M. Differences in the mode of the extension of gastric cancer classified by histological type: new histological classification of gastric carcinoma. Gut 1992;33:606–612
Monig S, Baldus SE, Collet PH, et al. Histological grading in gastric cancer by Goseki classification: correlation with histopathological subtypes and prognosis. Anticancer Res 2001;21:617–620
You WC, Blot WJ, Chang YS, et al. Diet and high risk of stomach cancer in Shandong, China. Cancer Res 1988;48:3518–3523
Brinton LA, Gridley G, Hrubec Z, et al. Cancer risk following pernicious anaemia. Br J Cancer 1989;59:810–813
Mullaney PJ, Wadley MS, Hyde C, et al. Appraisal of compliance with the UICC/AJCC staging system in the staging of gastric cancer. Union International Contra la Cancrum/American Joint Committee on Cancer. Br J Surg 2002;89:1405–1408
Bollschweiler E. Benefits and limitations of Kaplan-Meier calculations of survival chance in cancer surgery. Langenbecks Arch Surg 2003;388:239–244
Ming SC. Cellular and molecular pathology of gastric carcinoma and precursor lesions: a critical review. Gastric Cancer 1998;1:31–50
Jass JR, Sobin LH, Watanabe H. The World Health Organization’s histologic classification of gastrointestinal tumors. A commentary on the second edition, Cancer 1990;66:2162–2167
Mulligan RM. Histogenesis and biologic behavior of gastric carcinoma. Pathol Annu 1972;7:349–415
Filipe MI, Jankowski J. Growth factors and oncogenes in Barrett’s oesophagus and gastric metaplasia. Endoscopy 1993;25:637–641
Hermanek P. Tumors of the gastrointestinal tract and the pancreas: histopathology, staging and prognosis Anticancer Res 1999;19:2393–2396
Songun I, van de Velde CJ, Arends JW, et al. Classification of gastric carcinoma using the Goseki system provides prognostic information additional to TNM staging. Cancer 1999;85:2114–2118
Monig SP, Zirbes TK, Schroder W, et al. Staging of gastric cancer: correlation of lymph node size and metastatic infiltration. A.J.R. Am J Roentgenol 1999;173:365–367
Dixon MF, Martin IG, Sue-Ling HM, et al. Goseki grading in gastric cancer: comparison with existing systems of grading and its reproducibility. Histopathology 1994;25:309–316
Piard F, Hillon P, Levillain P, et al. Does Ming’s classification of gastric carcinomas have epidemiologic or prognostic value? Ann Pathol 1986;6:329–334
Davessar K, Pezzullo JC, Kessimian N, et al. Gastric adenocarcinoma: prognostic significance of several pathologic parameters and histologic classifications. Hum Pathol 1990;21:325–332
Ribeiro MM, Sarmento JA, Sobrinho Simoes MA, et al. Prognostic significance of Lauren and Ming classifications and other pathologic parameters in gastric carcinoma. Cancer 1981;47:780–784
Garnier P, Vielh P, Asselain B, et al. Prognostic value of the Lauren and Ming classifications in gastric adenocarcinoma. Multidimensional analysis. Gastroenterol. Clin Biol 1988;12:553–558
Cimerman M, Repse S, Jelenc F, et al. Comparison of Lauren’s, Ming’s and WHO histological classifications of gastric cancer as a prognostic factor for operated patients. Int Surg 1994;79:27–32
Dirschmid K, Scheiden R, Zimmermann G, et al. Tubular differentiated stomach carcinoma of the (Ming) infiltrating type. Pathologe 1996;17:122–126
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Luebke, T., Baldus, S.E., Grass, G. et al. Histological Grading in Gastric Cancer by Ming Classification: Correlation with Histopathological Subtypes, Metastasis, and Prognosis. World J. Surg. 29, 1422–1427 (2005). https://doi.org/10.1007/s00268-005-7795-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00268-005-7795-z