Abstract
Introduction
Rectus sheath hematoma is rare. It can present to physicians in many specialties, but invariably the surgeon is contacted regarding management. It is frequently difficult to diagnose, which can result in delay of treatment or unnecessary surgery. Its incidence is on the rise.
Methods
We present a series of four cases of rectus sheath hematoma that presented to our hospital within a 5-month period. A comprehensive up-to-date review of the literature was undertaken.
Results
Our research highlights the diagnostic dilemmas, the spectrum of severity (all three grades are represented), the complications, and the need for interdisciplinary awareness.
Conclusions
Rectus sheath hematoma is a rare but important entity in the differential diagnosis of abdominal pain. Interdisciplinary awareness of this condition is essential, as it is frequently difficult to diagnose, leading to delay in treatment or unneeded surgery. Grade 3 hematomas can lead to abdominal compartment syndrome and can be fatal.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Rectus sheath hematoma (RSH) is a rare condition. It enters into the differential diagnosis of abdominal pain but is frequently difficult to diagnose. Patients present with varying symptoms, but the most constant features are abdominal pain and a mass in the lower abdomen, most frequently on the right side. Clinical examination, however, is often misleading; and radiologic imaging is required. The condition encompasses a wide spectrum of severity (self-limiting to fatal) depending on its size, etiology, and the development of complications. RSH is graded according to its size and appearance on the CT scan. It has multiple possible etiologies including, frequently, anticoagulation therapy. Therefore it can present to a variety of specialists, and interdisciplinary awareness of its presentation is essential for early diagnosis. Complications include hypovolemic shock and myocardial infarction, muscle necrosis, intraabdominal hypertension, and death.
Here we report four patients who presented to our hospital within a 5-month period. A review of the literature was undertaken, and we discuss the demographics, pathogenesis, clinical features, diagnosis, and treatment of this condition as well as its complications.
METHODS
We present a series of four cases of RSH that presented to our hospital within a 5-month period. The Royal Glamorgan Hospital is an busy acute district general hospital with 500 beds. Data were collected prospectively after presentation of the first case.
The patients were between 50 and 67 years of age, and all were women. Two patients presented as surgical emergencies via their general practitioners, one presented during routine follow-up for a deep vein thrombosis and anticoagulation in the medical day ward, and the other was seen as a gynecologic emergency.
One patient died. After discharge the other three underwent routine follow-up in our surgical outpatient department until all of the symptoms had settled.
CASE 1
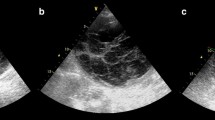
A 50-year-old women presented as a surgical emergency with a 3-day history of right-sided abdominal pain and a mass in the right iliac fossa. Initial investigations included a full blood count (FBC), urea and electrolytes, liver function tests, clotting tests, amylase level, plain abdominal radiograph. and chest radiograph. All of the tests were normal. A provisional diagnosis of cecal or ovarian mass was made, and the patient was admitted for further investigations. A computed tomography (CT) scan was performed, which revealed a lower right RSH (Fig. 1).
The patient was treated conservatively with bed rest and analgesia and was discharged 3 days later. Follow-up outpatient appointments revealed no further complications.
CASE 2
A 64-year-old woman with known chronic obstructive airway disease, asthma, atrial fibrillation, and a past history of deep vein thrombosis treated with warfarin presented acutely with right-sided lower abdominal pain and nausea following a bout of coughing. She was otherwise well. On examination there was an ill-defined fullness (approximately 15 cm) in the right iliac fossa raising suspicion of a cecal or appendicular mass. Investigations on admission were normal, apart from an international normalized ratio (INR) of 6.2. Her warfarin was stopped.
Ultrasonography of the abdomen was inconclusive, so a CT scan was performed (Fig. 2). It showed a large right-sided RSH with a thickness of up to 5.4 cm in places. A repeated FBC showed a drop in hemoglobin (from 12.3 g/dl to 10.2 g/dl), but it did not require transfusion. The patient was treated conservatively with rest and analgesia. The elevated INR returned to normal within 3 days of stopping the warfarin.
After 8 days in hospital with no further complications, the patient was discharged. An outpatient appointment 4 weeks later revealed complete resolution.
CASE 3
A 63-year-old woman was referred to us by the acute medical team. She had a 3-day history of slow onset of diffuse, cramp-like abdominal pain and palpable fullness in the right lower abdomen. She had been diagnosed with a deep vein thrombosis 4 days earlier and treated with Clexane 135 mg and warfarin. There was no other significant past medical history. Initial investigations showed a white blood cell (WBC) count of 11.1 × 109/l, INR 1.6, urea 9.5 mmol/l and creatinine 165 μmol/l.
The abdominal radiograph showed features consistent with constipation. Ultrasonography of the abdomen was reported as normal but incomplete owing to bowel gas. She was admitted to a surgical ward, and the warfarin was stopped. Five hours later her oxygen saturation dropped to 77%. Following a medical review, the concern was of a possible pulmonary embolism. Repeat investigations showed a reduction in hemoglobin from 11.1 g/dl to 6.9 g/dl. Blood transfusion and aggressive fluid resuscitation was commenced. A CT scan was performed 13 hours after admission, at 5 AM (Fig. 3). It showed a massive RSH with extraperitoneal extension and active bleeding. The INR had increased to 4.2, for which she was given fresh frozen plasma. Despite our efforts the patient continued to deteriorate and was transferred to the high dependency unit, where she suffered a myocardial infarction. Her poor condition made anesthesia impossible. Furthermore, her renal function and urine output deteriorated despite fluid resuscitation and full inotropic support.
She died 36 hours after admission. Postmortem examination revealed more than 5 liters of intraperitoneal blood and a massive RSH.
CASE 4
A 67-year-old woman was admitted under the care of a gynecologist with a 3-day history of sudden onset of left iliac fossa pain radiating down the left leg. She was on warfarin for a previous pulmonary embolism. Initial investigations were normal, apart from the INR, which was 3.6. Abdominal ultrasonography showed a mass in the left iliac fossa, more likely to be a bowel mass than an ovarian lesion. A surgical opinion was sought and a CT scan requested. The scan revealed a large anterior and centrally placed mass with a small amount of fluid and air-fluid levels, as well as inflammatory changes in the pelvis extending to the anterior abdominal wall. The radiologic findings were suggestive of a perforated sigmoid cancer. Her clotting abnormality was corrected (vitamin K and fresh frozen plasma), and she underwent emergency laparotomy, which revealed a large RSH bulging into the pelvis. The bowel was normal. The hematoma was evacuated, and the abdomen was closed.
Postoperative recovery was uneventful and the patient was discharged home on day 13 after admission. Follow-up 2 weeks later revealed no further problems.
RESULTS
Our four cases of RSH of varying severity presented to us within a 5-month period. All were women 50 to 67 years of age who presented with abdominal pain and an associated lower abdominal mass. One was misdiagnosed as having a sigmoid tumor and underwent unnecessary emergency surgery.
Two of the patients exhibited a fall in hemoglobin, one of whom required a blood transfusion. One case occurred spontaneously, and the other three were associated with anticoagulation therapy. One of the latter three patients died.
Abdominal ultrasonography was employed in three of the four cases but was diagnostic in only one. CT scanning revealed the correct diagnosis in three instances but was misleading in the other.
Three patients were treated conservatively. The fourth underwent emergency laparotomy because of a misdiagnosis. Patient 3 was considered for surgery, but because of uncorrectable coagulopathy and fresh myocardial infarction she was deemed not suitable. Embolization of the bleeding vessel was discussed with the consultant interventional radiologist, but it was technically not possible in this particular patient.
DISCUSSION
Rectus sheath hematoma is an uncommon and frequently misdiagnosed cause of abdominal pain.1–6 First described by Hippocrates and Galen,1 the first case report was published in 1857. The male/female distribution for RSH is 1.0:2.5–3.0,1 and it occurs most frequently in the right lower abdomen.7 The average age was reported to be 47 years,2 but with the increased use of anticoagulation it may now be higher.
The incidence of RSH as reported in a study of 1257 patients who required abdominal ultrasonography for acute abdominal pain or an unclear abdominal disorder is 1.8%.8 This incidence is thought to be on the rise owing to more frequent use of anticoagulation.3 The overall mortality is reported as 4%, but in anticoagulated patients it increases to 25% and during pregnancy to 13% with 50% fetal mortality.4
Many causes of RSH have been described and include trauma,8 coughing,3,8 straining of the abdominal muscles;1 iatrogenic cases from insertion of peritoneal catheters,5 injections,9 and anticoagulation;6,8 pregnancy;10 and medical conditions such as coagulation disorders, connective tissue disorders, leukemia, and renal disease.11 It has also been described to occur spontaneously without discernible cause.8,12,13
Large RSHs tend to occur in the lower abdomen, related to the anatomy of the rectus sheath. Above the arcuate line of Douglas the rectus muscle is covered by the aponeuroses of the external oblique, internal oblique, and transversalis muscles; it then divides to form the anterior and posterior rectus sheath.7 RSHs in this region tend to be small and confined because the anterior and posterior rectus sheath together with the tendinous intersections limit the size of the hematoma by tamponade.4 Below the arcuate line the posterior rectus sheath is missing, as the aponeuroses of the three muscles pass in front of the rectus muscle,7 enabling hematomas to spread extraperitoneally or even intraperitoneally should the peritoneum be ruptured, as in our case 3. Also most of the change in muscle length on contraction occurs below the arcuate line. The vessels that usually bleed are the inferior epigastric artery and vein. These vessels are firmly attached to the muscle where they penetrate it and are therefore subject to shearing forces.1
Patients can present in numerous ways, but the most constant features are abdominal pain and a mass on palpation.14 Frequently there is mild pyrexia and leukocytosis.4
Fotherhill’s sign is useful for distinguishing between an intraabdominal mass and a mass arising in the rectus sheath.15 It is performed by asking the supine patient to lift his or her head from the pillow. As the abdominal muscles tense, patients with an RSH have increased pain and the mass becomes fixed but is still palpable. An intraabdominal mass is more difficult to distinguish through the tensed muscle.
Late signs of RSH include bruising of the abdomen, which may be periumbilical (Cullen’s sign), associated with intraperitoneal rupture of the hematoma. With extraperitoneal extension4 there may be bruising in the flanks (Grey Turner’s sign). The clinical examination is frequently misleading, and RSHs have been misdiagnosed as bowel-related pathology or an ovarian tumor,16 resulting in unnecessary surgery. Imaging is essential for diagnosing the condition.
Abdominal radiography is not helpful. Although ultrasonography is one of the first-line investigations, it is associated with a sensitivity of only 80% to 90%.17,18 The CT scan is the gold standard for diagnosing fresh RSHs, with nearly 100% sensitivity and specificity.4 The grading of RSH is based on CT findings19 (Table 1). MRI is used for long-standing hematomas, when it is difficult to distinguish between a soft tissue tumor and an RSH on the CT scan.20
Complications of RSH include unnecessary surgery3 hypovolemic shock,21 muscle necrosis,22 medical complications such as myocardial infarction, and death.23 One of the more recently recognized complications is the abdominal compartment syndrome, which was first described by O’Mara et al.24 in 2003. We believe that our third patient suffered from abdominal compartment syndrome. Even though we did not measure the intraabdominal pressure at the time, the clinical course and the postmortem findings (5 liters of free blood in the abdomen) make this diagnosis likely.
Mohapatra25 identified hemorrhage as a risk factor for developing intraabdominal hypertension. Of the 11 clinical parameters for abdominal compartment syndrome identified by Mohapatra in the same article, our patient in case 3 displayed 6 (Table 2, the symptoms of case 3 are shown in italic type): distended abdomen, massive intravenous fluid requirements, oliguria or anuria not responding to volume repletion, decreased cardiac output, hypoxemia refractory to an increase in FiO 2 and positive end-expiratory pressure, and acidosis seen in the blood gases. We believe that the possibility of intraabdominal hypertension should be considered with every grade 3 RSH. This can easily be measured via the urinary catheter that should be in place in all of these patients. Surgical decompression is recommended if the pressure exceeds 26 mmHg, corresponding to grade 3 intraabdominal hypertension.26
Treatment of an RSH depends on its size, the underlying conditions, and its potential complications. Most RSHs settle down with conservative treatment—mainly pain relief and rest. Any underlying coagulopathy should be corrected with fresh frozen plasma and vitamin K,14,17 and any other conditions are treated as appropriate. For severe, uncontrollable pain, aspiration of the hematoma may be necessary. For continued bleeding, embolization of the bleeding vessel is an option. The use of thrombin,27 Gelfoam,28 or coil embolization6,29 has been described. Open surgery is the last resort, whereby the hematoma is evacuated and the bleeding vessel ligated.
CONCLUSIONS
Rectus sheath hematoma is a rare but important part of the differential diagnosis of abdominal pain. It can present to clinicians in many specialties, and its incidence is increasing. The clinical course may range from benign to fatal. The CT scan is the gold standard investigation. Grade 3 hematomas can cause intraabdominal hypertension, and clinicians should be aware of abdominal compartment syndrome. Treatment options are variable and include conservative treatment, intravascular embolization, and surgery. Frequently an interdisciplinary team approach is needed.
References
Casey RG, Mahmoud M, Hurley M, et al. Rectus sheath hematoma: an unusual diagnosis. Ir Med J 2000;93:90–92
Teske JM. Hematoma of the rectus abdominis muscle: report of a case and analysis of 100 cases from the literature. Am J Surg 1946;71:689–695
Decker R, Buhr H, Gahbauer H. Acute rectus sheath hematoma (differential diagnosis of acute abdomen). Chirurg 1986;57:401–405
Werdehoff SG, Blomkalns AL. Rectus sheath hematoma. On eMedicine January 2005
Jayawardene S, Goldsmith D. Clinical picture: rectus sheath hematoma. Lancet 2001;358:1409
Holmes SJ. Rectus sheath hematoma as a cause of acute abdominal pain. Am Fam Physician 2001;64:1681–1682
Cavagna E, Carubia G, Schiavon F. Anatomo-radiologic correlations in spontaneous hematoma of the rectus abdominis muscles. Radiol Med (Torino) 2000;99:432–437
Klingler PJ, Wetscher G, Glaser K, et al. The use of ultrasound to differentiate rectus sheath hematoma from other acute abdominal disorders. Surg Endosc 1999;13:1129–1134
Monsein LH, Davies M. Radionuclide imaging of a rectus sheath hematoma caused by insulin injections. Clin Nucl Med 1990;15:539–541
Humphrey R, Carlan SJ, Greenbaum L. Rectus sheath hematoma in pregnancy. J Clin Ultrasound 2001;29:306–311
Jayavardene SA, Goldsmith DJA. Rectus sheath hematomata in patients with renal disease. Nephrol Dial Transplant 2002;17:1832–1835
Ozaras R, Yilmaz MH, Tahan V, et al. Spontaneous hematoma of the rectus abdominis muscle: a rare cause of acute abdominal pain in the elderly. Acta Chir Belg 2003;103:332–333
Linhares MM, Lopes Filho GJ, Bruna PC, et al. Spontaneous hematoma of the rectus abdominis sheath: a review of 177 cases with report of 7 personal cases. Int Surg 1999;64:251–257
Ergün N, Cevik AA, Holliman CJ, et al. Conservative management of giant abdominal wall hematoma. Ulus Trauma Derg 2004;10:141–144
Fothergill WE. Hematoma in the abdominal wall simulating pelvic new growth. BMJ 1926;1:941–942
Thia EWH, Low JJH, Wee HY. Rectus sheath hematoma mimicking an ovarian mass. Internet J Gynaecol Obstet 2003;2(1)
Moreno Gallego A, Aguayo JL, Flores B, et al. Ultrasonography and computed tomography reduce unnecessary surgery in abdominal rectus sheath hematomas. Br J Surg 1997;84:1295–1297
Fukuda T, Sakamoto I, Kohzaki S, et al. Spontaneous rectus sheath hematomas: clinical and radiological features. Abdom Imaging 1996;21:58–61
Cervantes J, Sanchez-Cortazar J, Ponte RJ, et al. Ultrasound diagnosis of rectus sheath hematoma. Am Surg 1983;49:542–545
Berna JD, Garcia-Medina V, Guirao J, et al. Rectus sheath hematoma: diagnostic classification by CT. Abdom Imaging 1996;1:62–4
Park SH, Oh DR, Kim HK, et al. Spontaneous rectus sheath hematoma with hypovolaemic shock. J Korean Soc Emerg Med 2000;11:586–591
Askew AR. Rectus sheath hematoma causing muscle necrosis. Aust N Z J Surg 1980;50:531–532
Ducatman BS, Ludwig J, Hurt RD. Fatal rectus sheath hematoma. JAMA 1983;249:924–925
O’Mara MS, Semins H, Hathaway D, et al. Abdominal compartment syndrome as a consequence of rectus sheath hematoma. Am Surg 2003;69:975–977
Mohapatra B. Abdominal compartment syndrome. Indian J Crit Care Med 2004;8:26–32
Meldrum DR, Moore FA, Moore EE, et al. Prospective characterization and selective management of the abdominal compartment syndrome. Am J Surg 1997;174:667–672
Shabani AGS, Baxter GM. Inferior epigastric artery pseudo aneurysm: ultrasound diagnosis and treatment with percutaneous thrombin. Br J Radiol 2002;75:689–691
Levy JM, Gordon HW, Pitha NR, et al. Gelfoam embolisation for control of bleeding from rectus sheath hematoma. AJR Am J Roentgenol 1980;135:1283–1284
Zack JR, Ferral H, Postoak D, et al. Coil embolization of rectus sheath hemorrhage. J Trauma 2001;51:793–795
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Luhmann, A., Williams, E. Rectus Sheath Hematoma: A Series of Unfortunate Events. World J. Surg. 30, 2050–2055 (2006). https://doi.org/10.1007/s00268-005-0702-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00268-005-0702-9