Abstract
Toxic cyanobacteria blooms are a growing concern for public health and safety, due in part to the production of the hepatotoxin microcystin by certain species, including Microcystis aeruginosa. Management strategies for controlling cyanobacteria blooms include algaecide treatments, often with copper sulfate, and more recently oxidizers such as sodium percarbonate that produce hydrogen peroxide. This study assessed the effects of two copper-containing algaecides and one sodium percarbonate-containing algaecide on mitigating cell numbers and toxin content of cultured M. aeruginosa and summer (July) bloom samples of Anabaenopsis sp. in a brackish stormwater detention pond. Monitoring of the bloom revealed that Anabaenopsis sp. was associated with elevated levels of orthophosphate compared to nitrogen (dissolved inorganic nitrogen to phosphorus ratios were 0.19–1.80), and the bloom decline (September–October) was likely due to lower autumn water temperatures combined with potential grazing by the dinoflagellate Protoperidinium quinquecorne. Laboratory-based algaecide experiments included three dose levels, and cyanobacteria cell numbers and microcystin concentrations (particulate and dissolved) were evaluated over 7 d. Following exposure, copper-containing treatments generally had lower cell numbers than either sodium percarbonate-containing or control (no algaecide) treatments. Addition of algaecides did not reduce overall microcystin levels, and a release of toxin from the particulate to dissolved phase was observed in most treatments. These findings indicate that algaecide applications may visibly control cyanobacteria bloom densities, but not necessarily toxin concentrations, and have implications for public health and safety.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Harmful algal blooms (HABs) include representatives from a wide range of phytoplankton taxa and are a focus of marine and coastal research globally because of their negative impacts on human health and aquatic ecosystems. Examples of negative impacts associated with HABs include shellfish poisoning (Anderson 1997), bivalve and marine mammal mortalities (Scholin et al. 2000; Greenfield and Lonsdale 2002), and ecosystem disruption (Edvardsen and Paasche 1998). The annual frequency and intensity of HABs have increased (Hallegraeff 1993; Anderson et al. 2008). Cyanobacteria in particular are responsible for the largest number of HABs in freshwater environments worldwide (Chorus and Bartram 1999; Huisman and Visser 2005), but they are also common in brackish and coastal waterbodies (Paerl 1988; Lewitus et al. 2008). Cyanobacteria HABs are primarily associated with certain genera such as Microcystis, Anabaenopsis, Oscillatoria, Nostoc, and Planktothrix that produce microcystins, a group of hepatotoxins that include ~90 cyclic congeners (Sivonen and Jones 1999). Microcystins represent a serious health concern because they can induce severe gastroenteritis, liver failure, and even death (Pouria et al. 1998; Falconer 2005). Blooms of microcystin-producing cyanobacteria are common in a wide range of aquatic ecosystems, particularly lakes and ponds associated with human residential and recreational activity (Gobler et al. 2007; Lewitus et al. 2008), presenting a considerable public health risk.
Cyanobacteria blooms are caused by a variety of physical, chemical, and biological factors. Warm, stagnant conditions, such as those typical of mid to late summer, are often associated with cyanobacteria bloom formation (Paerl 1988; Huisman et al. 2005; Lewitus et al. 2008; Elliot 2010). Additionally, numerous studies have shown that enrichment of nitrogen and phosphorus associated with runoff from urban, suburban, and agricultural lands, fertilization, and other anthropogenic activities promote the development of HABs, especially those caused by cyanobacteria (reviewed in Anderson et al. 2008; Heisler et al. 2008). Studies have shown that biological control of cyanobacteria blooms includes grazing pressure by microzooplankton (Gobler et al. 2007; Davis et al. 2012), but it has also been suggested that cyanobacteria are not always the preferred prey due to their chemical defenses and reduced nutritional quality (Wilson et al. 2006; Paerl and Paul 2012), so the extent to which top-down processes mitigate a bloom remains uncertain. The physical and chemical factors that cause cyanobacteria blooms are predicted to become enhanced under future climate conditions (Paerl and Huisman 2009; Elliot 2010; Paerl and Paul 2012), so it is reasonable to suggest that incidences of HABs caused by cyanobacteria will also increase. This is compounded by the tendency for cyanobacteria HABs to occur in waters with a high likelihood of human contact. For example, shallow brackish detention ponds are routinely built within residential and recreational (golf course) developments along the southeastern coast of the US to mitigate stormwater (Drescher et al. 2011). Although many detention ponds, such as the site considered here, are either directly or daisy-chain linked to tidal creeks and thus exchange water with receiving estuaries, long residence times make them prone to frequent stagnation during warmer months such that they accumulate fertilizers and other runoff, making them ideal settings for cyanobacteria HABs (Lewitus and Holland 2003; Serrano and DeLorenzo 2008; Lewitus et al. 2008). Given the ecological and public health threats of toxic cyanobacteria, combined with the likelihood of increased bloom occurrences, safe and effective bloom management is of paramount importance.
Management strategies for mitigating cyanobacteria blooms include nutrient reduction, aeration, flocculation, dilution, grazing, and a variety of algaecides (reviewed in Chorus and Bartram 1999; Huisman et al. 2005). Copper sulfate (CuSO4) in particular is a common ingredient of commercial algaecides because it is inexpensive and cyanobacteria are more sensitive to high concentrations of copper than other phytoplankton taxa (Brand et al. 1986; Chorus and Bartram 1999; Le Jeune et al. 2006). Copper (Cu) toxicity acts on cyanobacteria through multiple routes, including substitution for magnesium in chlorophyll (Jančula and Maršálek 2011) and associated thylakoid disruption combined with inhibition of electron transport in photosystem II (Verhoeven and Eloff 1979; Baron et al. 1995; Qian et al. 2010). In addition to cyanobacteria, elevated copper levels pose serious physiological and ecological threats to secondary and tertiary consumers, such as fish and invertebrates (Wilson and Taylor 1993; Grosell et al. 2007; Korosi and Smol 2012; Al-Bairuti et al. 2013), and consequently they can have detrimental long-term consequences for aquatic food webs (Korosi and Smol 2012). Thus, chemical alternatives to copper-based algaecides have been proposed, particularly hydrogen peroxide (H2O2) (Barroin and Feuillade 1986; Drábková et al. 2007; Barrington and Ghadouani 2008). Similar to copper sulfate, hydrogen peroxide inhibits electron transport and photosynthetic activity in cyanobacteria, rendering photosystem II inactive (Samuilov et al. 2004; Barrington and Ghadouani 2008). Research has shown that cyanobacteria tend to be more sensitive to hydrogen peroxide than other phytoplankton taxa (Barroin and Feuillade 1986; Barrington and Ghadouani 2008; Matthijs et al. 2012). As a result, hydrogen peroxide has been applied to control cyanobacteria in several systems, including waste stabilization ponds (Barrington et al. 2011, 2013) and a lake (Matthijs et al. 2012). Safety concerns prevent adding large volumes of concentrated hydrogen peroxide directly to water bodies. However, it can be produced through oxidation, such as from sodium percarbonate, so algaecides containing oxidizers are available commercially.
Both copper sulfate and hydrogen peroxide disrupt cyanobacteria cells, but the extent to which algaecides using these reactive compounds mitigate toxin content is less understood, representing an important gap in our understanding of bloom management. The objective of this study was to investigate the relative abilities of three commercially available algaecides (two containing copper sulfate and one containing sodium percarbonate) for controlling cyanobacteria cell concentrations and toxin content (as microcystin levels). Specifically, we conducted laboratory experiments using cultured and bloom cyanobacteria to evaluate the efficacy of each algaecide for controlling (1) bloom levels of cyanobacteria and (2) the partitioning of microcystin in both the particulate (intracellular) and dissolved (extracellular) fractions. Each experiment was conducted at three treatment levels to assess the effects of dose and chemical nature of algaecides on microcystin levels and cell densities.
Materials and Methods
Phytoplankton Culturing and Enumeration
Microcystis aeruginosa (SC isolate) was grown under a 12:12 light:dark cycle at 25 °C (temperatures typical during mid-summer in coastal SC ponds) and 140 μE m−2 s−1. This irradiance level is within range of those associated with maximum Microcystis growth and cellular microcystin level (Wiedner et al. 2003) but below irradiances that cause severe oxidative stress (Drábková et al. 2007). M. aeruginosa was grown in sterile 0.2-μm filtered f/2 medium (Guillard and Ryther 1962) without silica using water collected from the South Atlantic Bight that was diluted with sterile distilled water to a final salinity of 5 psu. M. aeruginosa was counted by sampling triplicate 1 mL aliquots of late log-phase culture, staining each aliquot with a drop of Lugol’s iodine solution, and enumerating individual cells by light microscopy using a 0.1 mm haemocytometer. Anabaenopsis sp. was sampled from bloom water by removing triplicate 1 mL aliquots, staining each aliquot with a drop of Lugol’s iodine solution, then enumerating as for M. aeruginosa.
Sampling Location
Kiawah Island is a barrier island located ~45 km southwest of Charleston, South Carolina (SC), USA (Fig. 1). It is characterized by extensive landscaping, golf course construction, and networks of detention ponds as catchments for stormwater runoff (Lewitus et al. 2003, 2008; Holland et al. 2004). There are 136 detention ponds on Kiawah Island within ~335 acres (1.356 km2), and the majority of these ponds are shallow (1–3 m depth), brackish to marine systems (Lewitus et al. 2003). Monitoring and research of water quality, nutrient loading, and phytoplankton in Kiawah Island detention ponds has been ongoing since 2001. These and related efforts have shown that due to low flushing, SC detention ponds often accumulate nutrient runoff from fertilizers, and this enrichment makes them susceptible to HABs (Lewitus and Holland 2003; Lewitus et al. 2003, 2008; Drescher et al. 2011). Blooms caused by cyanobacteria, including HAB species, commonly recur in Kiawah Island detention ponds, with Microcystis, Anabaenopsis, Oscillatoria, Cylindrospermopsis, and Aphanizomenon being among the most common genera (Brock 2006; Lewitus et al. 2008; Siegel et al. 2011).
The site used for this study, Kiawah Island pond number 093 (henceforth K093), is a shallow (~1.5 m maximum depth) mesohaline (typical salinity range 15–25 psu) system located on the northeastern end of Kiawah Island that covers ~13 acres (0.053 km2) of surface area (Fig. 1). During summer (June–September), water temperatures are usually 24–32 °C. K093 is surrounded by residential housing, a golf course, salt marsh, and road, and it is connected to a tidal creek by stormwater pipes, such that tidal exchange occurs between the pond and the creek. K093 is used for recreational shrimping, crabbing, and fishing.
Field Sampling and Analyses
Triplicate 1 L surface (0.3 m depth) water samples were collected from the K093 dock (Fig. 1) at mid-ebb tide using opaque Nalgene bottles that were previously acid-washed (immersion in 10 % hydrochloric acid, HCl, for 4 h followed by rinsing three times with distilled water), placed in the dark inside a cooler, then immediately transported to the laboratory for processing. Sample intervals were frequent (2–3 times wk−1) following initial observation of the Anabaenopsis sp. bloom (20 July, 2009), then every 1–2 wks while the bloom persisted (through 1 October, 2009). During each sampling event, standard water quality parameters (temperature, salinity, and dissolved oxygen) were recorded from surface depths using a hand-held YSI 85 unit (YSI Incorporated, Yellow Springs, OH, USA), and pH was measured using a hand-held pH meter.
Upon return to the laboratory, whole water samples (up to 40 mL) from each replicate were filtered through a 0.7 μm glass fiber filter (GF/F) for total chlorophyll a (a common proxy for phytoplankton biomass). Sampling processing, evaluations of pigment standards, and quality assurance/quality control (QA/QC) procedures followed well-established methods (non-acidification version of EPA method 445.0; Arar and Collins 1992). Briefly, filters containing sample were placed into acid-washed (using 10 % HCl as described above) 25 mL scintillation vials, then 1 mL of saturated magnesium carbonate (MgCO3) was added to ensure that cell membranes ruptured completely, and samples were frozen (−20 °C) until analysis. To evaluate pigment levels, 9 mL of high-performance liquid chromatography (HPLC) grade acetone (90 %) were added to each replicate, then chlorophyll a was extracted (−20 °C for 36 h). Following extraction, concentrations (μg L−1) of chlorophyll a were evaluated using a Turner Design 700 fluorometer according to manufacturer specifications (Arar and Collins 1992).
For nutrient analyses, aliquots (25 mL) of whole water from each sample replicate were filtered through pre-combusted (450 °C for 4 h) GF/F filters into acid-washed (using 10 % HCl as described above) scintillation vials. Subsequent sample processing followed well-established methods for orthophosphate (PO4 3−) (Hach method 31-115-01-1-H), nitrite + nitrate combined (NO2 − + NO3 −) (Hach method 31-107-04-1-D), and ammonium (NH4 +) (Hach method 31-107-06-1-B) using a Lachat Quick-Chem 8000 nutrient auto-analyzer with an ASX 500 autosampler (Grasshoff 1983; Johnson and Petty 1983; Zimmerman and Keefe 1991). QA/QC procedures followed manufacturer guidelines. Briefly, a calibration curve was generated at the beginning and end of each analysis batch (up to 30 samples) using reference standards from the manufacturer (Hach Inc.). If R 2 > 0.98, the curve was considered valid. One blank (ultrapure distilled water) and one standard (mid-range concentration) sample was run at the beginning, end, and every 15 samples to assess potential instrument drift and recalibrated as warranted.
To determine qualitative phytoplankton community composition, an aliquot (~2–3 mL) of whole water from one sample replicate (chosen at random) was dispensed into a Lab-Tek II Chamber slide, settled for 5 min, and then viewed with a Nikon Eclipse TS100 inverted microscope. All observed phytoplankton were identified to the lowest taxonomic level possible. Bloom taxa were enumerated by preserving an aliquot (100 mL) from the same replicate with Lugol’s iodine solution (3 % final preservative concentration), then individual cells (within colonies/chains and excluding heterocysts for Anabaenopsis sp.) were counted with a 0.1 mm haemocytometer until either a minimum of 300 cells or the entire chamber was counted, whichever occurred first.
Algaecide Exposure Experiments
Two copper-containing algaecides (Captain* and K-Tea™, trademarks of SePRO Corporation) and one non-copper-containing algaecide (Pak™ 27, formerly by Solvay Chemicals Inc., currently manufactured by SePRO Corporation) were evaluated for controlling the growth and toxin release of cultured M. aeruginosa and Anabaenopsis sp. bloom water. Captain* is a chelated elemental copper compound containing 15.9 % active copper carbonate that targets filamentous and planktonic algae, including cyanobacteria. K-Tea™ is also a chelated elemental copper compound, but it contains 8 % elemental copper derived from a copper–triethanolamine complex and copper hydroxide. K-Tea™ targets a wide range of algae taxa, including cyanobacteria, chlorophytes, diatoms, and dinoflagellates. The active ingredient in Pak™ 27 is sodium percarbonate (85 %), and it presents an alternative to copper-derived compounds, primarily targeting cyanobacteria. While each of the three products comes with a label describing specific instructions for use, application of algaecides to water bodies often varies with user. Therefore, three dose levels were tested: half the manufacturer’s recommended dose level (0.5×), at the manufacturer’s recommended dose (1×), and twice the manufacturer’s recommended dose (2×). Manufacturer’s recommended dosages were as follows: Captain (0.4 ppm), K-Tea™ (0.5 ppm), and Pak™ 27 (6.5 lbs acre-ft−1). We recognize that the molar concentrations of active compounds varied between algaecides, but since the goal of this study was to evaluate algal responses to products as managers typically use them, we did not normalize to account for these differences. Control treatments contained no algaecide.
Two series of experiments were conducted. The first used log-phase cultured M. aeruginosa (culturing conditions described above), and the second used water from K093 during an Anabaenopsis sp. bloom (collected 21 July, 2009). To evaluate the efficacy of each algaecide for mitigating cyanobacteria, initial concentrations typical of those observed during blooms (target concentration ~5.00 × 105 cells mL−1) were used. For each experiment, triplicate algal treatments (1 L each) were incubated using a 12:12 light:dark cycle at 140 μE m−2 s−1 at 25 °C in 0.2-μm filtered sterile seawater at the same salinities used for cultured (5 psu for M. aeruginosa) and field collected (21 psu for Anabaenopsis sp.) algae using sterile glass flasks according to the above three dose levels.
To evaluate the effects of each algaecide on cyanobacteria cell concentrations, flasks were gently mixed, then a 1 mL aliquot was collected, placed in an opaque 1.5 mL microcentrifuge tube, and preserved with 1 drop of Lugol’s iodine solution when algaecides were first added (t0), then at 4, 8, 24, 48, 72 h and 7 d following inoculation. This experimental duration was chosen because algaecide applications to ponds and lakes usually visibly reduce blooms in less than 1 week’s time. All preserved samples were subsequently enumerated using light microscopy and a 0.1 mm haemocytometer as described above.
Microcystin concentrations were evaluated using commercially available enzyme-linked immunosorbent assay (ELISA) kits (Abraxis microcystins/nodularins kit, product number 520011) according to the manufacturer’s procedure and using reference standards included within the kits. When complete, optical density (450 nm) was analyzed using a Synergy HT microplate reader (Biotek). To evaluate particulate (intracellular) microcystin levels, flasks were gently mixed as above, then whole water (1 mL) was removed from each replicate, placed in a 1.5 mL microcentrifuge tube, and immediately frozen for subsequent analyses. To evaluate dissolved (extracellular) microcystin levels, additional aliquots (~5–10 mL) were removed from each replicate then gently syringe-filtered through pre-combusted GF/F filters (0.7 μm), and 1 mL of the filtrate was stored in a 1.5 mL microcentrifuge tube as above. All toxin analyses were performed within 14 d of sampling, and no samples were thawed more than once. Particulate (intracellular) microcystin was calculated as the difference between whole and dissolved (extracellular) fractions. Microcystin was evaluated for the following time points: 0, 4, 24 h and 7 d following addition of algaecide.
All data are presented as means of triplicate measurements (n = 3) ± standard deviations (SD). Statistical comparisons were made using nonparametric analyses by analysis of variance (ANOVA; α = 0.05) (Sokal and Rohlf 1995) followed by a Dunnett’s post hoc test.
Results
Field Sampling and Analyses
During the study period, surface water temperatures of K093 ranged from 23.3 to 32.4 °C, and salinities ranged from 12.7 to 22.9 psu (Table 1). Dissolved oxygen levels exhibited wide fluctuations during the sampling period with a minimum of 1.65 mg L−1 (hypoxia) and a maximum of 16.28 mg L−1 (super-saturation). Surface pH was fairly alkaline, ranging from 7.8 to 9.1. Dissolved inorganic nitrogen primarily consisted of ammonium, with mean levels ranging from 1.36 to 4.73 μM (Table 1). By comparison, nitrite + nitrate was typically <1 μM, with mean concentrations ranging from 0.34 to 1.64 μM. Mean concentrations of dissolved inorganic phosphorus, as orthophosphate, were often 1–2 orders of magnitude greater than total dissolved inorganic nitrogen (ammonium, nitrite, and nitrate combined), with levels of orthophosphate ranging from 2.57 to 23.13 μM.
Mean phytoplankton biomass (chlorophyll a) was greatest between 21 and 30 July, reaching a maximum of 917.9 μg L−1 compared to a minimum of 33.9 μg L−1 on 1 October (Table 1). This exceptionally high biomass was coincident with a dense bloom of the cyanobacteria Anabaenopsis sp. and cell concentrations exceeding 3.49 × 107 cells mL−1. Other phytoplankton observed over the study period included cyanobacteria (Oscillatoria sp.), diatoms (the genera Navicula, Amphiprora, and the species Cylindrotheca closterium), a raphidophyte (Chattonella subsalsa), and dinoflagellates (Prorocentrum minimum, Heterocapsa rotunda, and Amphidinium sp.). The dinoflagellate Protoperidinium quinquecorne was the only species besides Anabaenopsis sp. to reach bloom concentrations at ~200 cells mL−1 on 16 September.
Algaecide Exposure Experiments
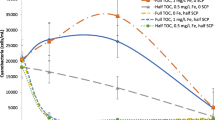
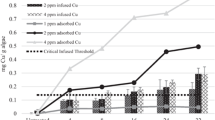
Algaecides tested in this study controlled the cell concentration, but not overall toxin levels, of cyanobacteria. For cultured M. aeruginosa, Pak™ 27 was the least effective algaecide in reducing cell concentrations in all treatments by day 7. Specifically, cell concentrations in Captain* and control treatments were not significantly different (ANOVA, P > 0.05) from each other at 0.5×, but cell concentrations were slightly more than double in the 0.5× Pak™ 27 treatment (Fig. 2a). Cell concentrations in Captain* and K-Tea™ treatments were significantly (ANOVA, P < 0.05) less than both Pak™ 27 and control treatments after 7 d for 1× and 2× (Fig. 2b, c). Experiments using bloom cyanobacteria (Anabaenopsis sp.) produced trends similar to M. aeruginosa, with cell concentrations in both Captain* and K-Tea™ treatments being significantly (ANOVA, P < 0.05) less than cell concentrations in either Pak™ 27 or control treatments for all dose levels by day 7 (Fig. 3).
Mean (n = 3 ± SD) cell concentrations of Microcystis aeruginosa exposed to commercially available algaecide mixtures at a half the manufacturer’s recommended dose strength (0.5×), b at the manufacturer’s recommended dose (1×), and c twice the manufacturer’s recommended dose (2×). Control treatments received no algaecide
Mean (n = 3 ± SD) cell concentrations of Anabaenopsis sp. exposed to commercially available algaecide mixtures at a half the manufacturer’s recommended dose strength (0.5×), b at the manufacturer’s recommended dose (1×), and c twice the manufacturer’s recommended dose (2×). Control treatments received no algaecide
Evaluations of toxin levels focused on the M. aeruginosa culture because microcystin concentrations of bloom samples were <0.1 ppb and therefore not a public health concern. The initial range for total (particulate + dissolved) microcystin concentration at the start of the M. aeruginosa experiments was typically 11–30 ppb, a moderate toxin range. Accordingly, the ratio of particulate:dissolved microcystin was typically >1, indicating that the majority of toxin was intracellular (Table 2). After 7 d, the ratio of particulate:dissolved microcystin shifted such that microcystin levels were comparatively higher in the dissolved phase, particularly for the 0.5× and 2× doses, although particulate microcystin remained comparable to or slightly greater than initial at 1×. Total microcystin levels generally increased over the duration of each experiment such that the final toxin content was often 2–10× greater than initial levels.
Discussion
Cyanobacteria are responsible for more HABs worldwide than any other phytoplankton taxa, and incidences of toxic blooms are expected to continue to increase (Hallegraeff 1993; Chorus and Bartram 1999; Paerl and Huisman 2009; Paerl and Paul 2012). This growing environmental and public health concern makes effective and environmentally safe management a critical need. While others have investigated the individual effects of either copper sulfate or hydrogen peroxide on cyanobacteria (García-Villada et al. 2004; Samuilov et al. 2004; Le Jeune et al. 2006; Barrington and Ghadouani 2008), only a few published studies have compared the two (Qian et al. 2010; Zhou et al. 2013; Fan et al. 2014), and of these none have evaluated cultured and field-collected algae. This study assessed the short-term effects of three treatment concentrations of commercially available copper sulfate and sodium percarbonate (an oxidizing agent for hydrogen peroxide production)-based algaecides on the growth and toxin content of cyanobacteria using cultures and field samples. All three algaecides reduced cell numbers of cyanobacteria relative to control treatments within a 7 d period. However, the two copper sulfate-based products (Captain* and K-Tea™) generally resulted in a greater reduction of cell numbers relative to treatments containing the sodium percarbonate-based compound (Pak™ 27). Despite reductions in cell numbers, none of the treatments mitigated the overall toxin levels.
Field monitoring during the Anabaenopsis sp. bloom at Kiawah Island pond K093 revealed environmental conditions typical of those found in SC detention ponds during mid to late summer. Surface temperatures were very warm, and salinity ranges were mesohaline, similar to what has been reported in other studies of coastal detention ponds (Lewitus et al. 2004, 2008; Siegel et al. 2011). The large variability in dissolved oxygen levels observed during the sampling period was possibly related to initial stagnation of the water column followed by production of oxygen during photosynthesis as the bloom expanded. Stagnation is well-known to facilitate cyanobacteria blooms (Chorus and Bartram 1999; Huisman et al. 2005; Anderson et al. 2008; Paerl and Huisman 2009; Elliot 2010). It is unlikely that sampling time (and therefore oxygen balance attributed to diel cycling of photosynthesis and respiration rates) played a major role as all samples were collected between 10:30 am and noon. Although organic nutrients were not measured during this study, evidence suggests that inorganic nutrient availability likely contributed to bloom development and persistence. Concentrations of orthophosphate were high during much of the sampling period, and ratios of dissolved inorganic nitrogen (nitrate, nitrite, and ammonium combined) to dissolved inorganic phosphorus (as orthophosphate) ranged between 0.19 and 1.80. These values are substantially below the optimal nitrogen:phosphorus ratio of 16:1 for marine phytoplankton (Redfield et al. 1963), suggesting an enrichment of phosphorus. Numerous studies have linked phosphorus loading with cyanobacteria blooms in freshwater systems (Paerl 1988; Chorus and Bartram 1999; Heisler et al. 2008; Davis et al. 2009), but few have reported this association for brackish systems. Moreover, since members of the genus Anabaenopsis can fix nitrogen (convert atmospheric nitrogen to ammonia), they probably had a competitive advantage over non-nitrogen fixers given the low nitrogen to phosphorus ratios and were consequently able to proliferate.
The termination of the Anabaenopsis sp. bloom was likely attributed to several factors. During September, water temperatures began to drop, and they were considerably lower by 1 October compared to the bloom peak (July–August). In addition, a co-occurring bloom of the heterotrophic dinoflagellate P. quinquecorne (~200 cells mL−1) was observed on 16 September, so it is likely that this species exerted top-down grazing control on cyanobacteria. The extent to which P. quinquecorne grazing reduced Anabaenopsis sp. concentrations relative to the effects of lower water temperatures are not known, however, Davis et al. (2012) showed microzooplankton can graze on cyanobacteria populations in freshwater systems, and cyanobacteria blooms have been shown to persist in Kiawah Island detention ponds during January (Brock 2006).
Both copper-containing algaecides (Captain* and K-Tea™) used in this study reduced numbers of Anabaenopsis sp. and M. aeruginosa more effectively than the sodium percarbonate-containing product (Pak™ 27). These observations agree with prior studies showing that high levels of copper reduce cyanobacteria division rates (Brand et al. 1986; Le Jeune et al. 2006). By the end of the 7 d test period, cells of Anabaenopsis sp. were almost entirely disrupted for all dose levels of Captain* and K-Tea™ experiments. By comparison, M. aeruginosa tended to be more resilient to algaecide treatment than Anabaenopsis sp., and dose did exert an effect. For example, in Captain* experiments, M. aeruginosa cell numbers were greater at 7 d relative to cells numbers at t0 (immediately following algaecide treatment) for the 0.5× treatment whereas cell numbers at 7 d were lower relative to t0 levels for both the 1× and 2× treatments. A similar trend was found for K-Tea™ treatments, but cell numbers were generally lower at the end of the 7 d experiment relative to Captain* treatments. It is uncertain why algaecides seemed to be more effective at reducing cell numbers of Anabaenopsis sp. than M. aeruginosa. Organic carbon levels affect the sensitivity of M. aeruginosa to copper (Zeng et al. 2010), but in this study cyanobacteria were in filtered water making organic carbon unlikely to be a major factor. Laboratory studies have shown that copper resistant mutants can emerge for M. aeruginosa (García-Villada et al. 2004), so this may be a possibility. For Pak™ 27, dose level had a minimal effect on cell numbers; although cell concentrations were typically less than the control by 7 d, this difference was not always statistically significant. The exception is M. aeruginosa 0.5×, where cell concentrations were higher in the Pak™ 27 treatments than control treatments by 7 d. Results from this study differ from Barrington and Ghadouani (2008) who reported a dose-dependent response for hydrogen peroxide at reducing cyanobacteria densities, although their experiments lasted only 48 h. Importantly, since this study tested an algaecide product rather than hydrogen peroxide directly, findings reported here do not necessarily mean that hydrogen peroxide is an ineffective management option for reducing cyanobacteria cell numbers. Sodium percarbonate is the active compound used to generate hydrogen peroxide in Pak™ 27, so one possibility is that this oxidizer or its associated catalyst may be less effective at generating sufficient levels of hydrogen peroxide than other options. Another possibility is that since our field experiment used a mixed phytoplankton assemblage, species that co-occurred with Anabaenopsis sp. may have broken down hydrogen peroxide thus rendering Pak™ 27 less effective on cyanobacteria.
Despite the reduction of cyanobacteria cell numbers in algaecide treatments, microcystin was detected throughout all M. aeruginosa experiments. While the actual ratios varied among experiments, the ratio of particulate (intracellular) to dissolved (extracellular) microcystin generally decreased over time. This suggests that as cells lysed, toxin was released into the water, where it persisted. These findings support Barrington et al. (2013), who observed an increase in dissolved microcystin relative to intracellular microcystin within 5 d of hydrogen peroxide application to cyanobacteria assemblages. A tendency for control treatments to also have lower particulate to dissolved ratios after 7 d suggests that leaching of microcystin from intact (non-lysed) cells also occurred. The detection of microcystin for up to 7 d following treatment in a laboratory setting contrasts work by Jones and Orr (1994) who showed that microcystin was detected in a confined area within a lake for less than 24 h following treatment with a copper-containing algaecide. However, their study also reported high levels of microcystin for over 9 d following application in a separate region of the same lake. Mesocosm studies have shown that microcystin can persist in the dissolved and particulate phases for up to 30 and 15 d, respectively, following a bloom (Lahti et al. 1997), but degradation rates follow first order kinetics and may be mediated by light and water temperature (Kenefick et al. 1993; Lahti et al. 1997). Both the current study and Jones and Orr (1994) suggest that algaecide applications do not fully mitigate cyanobacteria bloom toxin levels. Although it was not entirely clear why, the exception to these findings was the 1× treatment, where particulate:dissolved microcystin was generally not statistically different from t0 by 7 d.
From a management perspective, copper-containing algaecides generally controlled cyanobacteria populations more effectively than the sodium percarbonate-based compound, but lingering microcystin in the water itself may still pose a threat to public health and safety for a week or potentially longer following either application. While nutrient reduction strategies are likely the best long-term management solution for diminishing the likelihood of cyanobacteria HABs to develop in the first place, should algaecides be used it is important to consider the advantages and disadvantages of each strategy. Copper-based algaecides, such as Captain* and K-Tea™, have practical advantages over sodium percarbonate-based compounds, such as Pak™ 27, because they are both effective at reducing cyanobacteria cell numbers and cost-efficient. However, numerous studies have shown that copper and copper-based applications are highly toxic to other aquatic taxa and have been linked to food web disruption (Wilson and Taylor 1993; de Oliveira et al. 2004; Grosell et al. 2007; Jančula and Maršálek 2011; Korosi and Smol 2012; Al-Bairuti et al. 2013). Hydrogen peroxide has also been shown to be toxic to certain daphnids (McHuron et al. 2008; Jančula and Maršálek 2011) as well as fish, although the level of sensitivity varies widely among fish species (Gaikowski et al. 1999; Jančula and Maršálek 2011). While hydrogen peroxide poses fewer detrimental consequences to aquatic food webs, it presents a safety hazard if handled improperly. Thus, managers should consider not only cost, but also the ecological consequences of various cyanobacteria bloom prevention and control approaches when determining a strategy for a particular waterbody.
In conclusion, the three algaecides assessed here, Captain*, K-Tea™, and Pak™ 27, all reduced the numbers of cyanobacteria cells, with the two copper-based compounds (Captain* and K-Tea™) being generally more effective. However, none of the algaecides reduced overall microcystin levels, and a greater partitioning of toxin from the particulate to dissolved phase occurred over time. Field studies provided further insight as to the potential role of nutrients, notably inorganic phosphorus and nitrogen, for facilitating cyanobacteria blooms in brackish environments, as well as the potential role of grazing for mitigating blooms. Given the increased frequency of cyanobacteria blooms worldwide, combined with the close association of blooms with human activity, determining environmentally safe management strategies that not only mitigate blooms visibly, but also their toxin content, will be crucial for protecting public health and safety.
References
Al-Bairuti GA, Shaw BJ, Handy RD, Henry TB (2013) Histopathological effects of waterborne copper nanoparticles and copper sulphate on the organs of rainbow trout (Oncorhynchus mykiss). Aquat Toxicol 126:104–115
Anderson DM (1997) Bloom dynamics of toxic Alexandrium species in the northeastern US. Limnol Oceanogr 42:1009–1022
Anderson DM, Burkholder JM, Cochlan WP, Glibert PM, Gobler CJ, Heil CA, Kudela RA, Parsons ML, Rensel JE, Townsend DW, Trainer VL, Vargo GA (2008) Harmful algal blooms and eutrophication: examining linkages from selected coastal regions of the United States. Harmful Algae 8:39–53
Arar AJ and Collins AB (1992) Method 445.0. In vitro determination of chlorophyll a and phaeophytin a in marine and freshwater phytoplankton by fluorescence. Version 1.1. EPA/600/R-92/121
Baron M, Arellano JB, Gorge LJ (1995) Copper and photosystem II—a controversial relationship. Physiol Plant 94:174–180
Barrington DJ, Ghadouani A (2008) Application of hydrogen peroxide for the removal of toxic cyanobacteria and other phytoplankton from wastewater. Environ Sci Technol 42:8916–8921
Barrington DJ, Ghadouani A, Ivey GN (2011) Environmental factors and the application of hydrogen peroxide for the removal of cyanobacteria from waste stabilization ponds. J Environ Eng 137:952
Barrington DJ, Reichwaldt ES, Ghadouani A (2013) The use of hydrogen peroxide to remove cyanobacteria and microcystins from waste stabilization ponds and hypereutrophic systems. Ecol Eng 50:86–94
Barroin G, Feuillade M (1986) Hydrogen peroxide as a potential algicide for Oscillatoria rubescens DC. Water Res 20:619–623
Brand LE, Sunda WG, Guillard RRL (1986) Reduction of marine phytoplankton reproduction rates by copper and cadmium. J Exp Mar Biol Ecol 96:225–250
Brock LM (2006) Water quality, nutrient dynamics, phytoplankton ecology, and land uses within defined watersheds surrounding six detention ponds on Kiawah Island, South Carolina. Master’s Thesis, College of Charleston
Chorus J, Bartram J (eds) (1999) Toxic cyanobacteria in the water: a guide to their public health consequences, monitoring, and management. E&F Spon, London, 416 pp
Davis TW, Berry DL, Boyer GL, Gobler DJ (2009) The effects of temperature and nutrients on the growth and dynamics of toxic and non-toxic strains of Microcystis during cyanobacteria blooms. Harmful Algae 8:715–725
Davis TW, Koch F, Marcoval MA, Wilhelm SW, Gobler CJ (2012) Mesozooplankton and microzooplankton grazing during cyanobacterial blooms in the western basin of Lake Erie. Harmful Algae 15:26–36
de Oliveira EC, Lopez RM, Paumgartten FJR (2004) Comparative study on the susceptibility of freshwater species to copper-based pesticides. Chemosphere 56:369–374
Drábková M, Admirall W, Maršálek B (2007) Combined exposure to hydrogen peroxide and light—selective effects on cyanobacteria, green algae, and diatoms. Environ Sci Technol 41:309–314
Drescher SR, Law NL, Caraco DS, Cappiella KM, Schneider JA, Hirschman DJ (2011) Research and policy implications for watershed management in the Atlantic Coastal Plain. Coast Manag 39:242–258
Edvardsen B, Paasche E (1998) Bloom dynamics and physiology of Prymnesium and Chrysochromulina. In: Anderson DM, Cembella AD, Hallegraeff GM (eds) Physiological ecology of harmful algal blooms, NATO ASI Series 41. Springer, New York, pp 193–208
Elliot JA (2010) The seasonal sensitivity of cyanobacteria and other phytoplankton to changes in flushing rate and temperature. Glob Change Biol 16:864–876
Falconer IR (2005) Is there a human health hazard from microcystins in the drinking water supply? Acta Hydrochem Hydrobiol 33:64–71
Fan J, Hobson P, Ho L, Daly R, Brookes J (2014) The effects of various control and water treatment processes on the membrane integrity and toxin fate of cyanobacteria. J Hazard Mater 264:313–322
Gaikowski MP, Rach JJ, Ramsay RT (1999) Acute toxicity of hydrogen peroxide treatments to selected lifestages of cold-, cool- and warmwater fish. Aquaculture 178:191–207
García-Villada L, Rico M, Altamirano M, Sánchez-Marin L, López-Rhodas L, Costas E (2004) Occurrence of copper resistant mutants in the toxic cyanobacterium Microcystis aeruginosa: characterization and future implications in the use of copper sulphate as algaecide. Water Res 38:2207–2213
Gobler CJ, Davis TW, Coyne KJ, Boyer GL (2007) Interactive influences of nutrient loading, zooplankton grazing, and microcystin synthetase gene expression on cyanobacterial bloom dynamics in a eutrophic New York lake. Harmful Algae 6:119–133
Grasshoff K (1983) Methods of seawater analysis, 2nd edn. Verlang Chemie GmbH, Weinheim
Greenfield DI, Lonsdale DJ (2002) Mortality and growth of juvenile hard clams Mercenaria mercenaria during brown tide. Mar Biol 141:1045–1050
Grosell M, Blanchard J, Brix JV, Gerder R (2007) Physiology is pivotal for interactions between salinity and acute toxicity of copper to fish and invertebrates. Aquat Toxicol 84:162–172
Guillard RL, Ryther JH (1962) Studies of marine planktonic diatoms. 1. Cyclotella nana Hustedt and Detonula confervacea (Cleve) Gran. Can J Microbiol 8:229–239
Hallegraeff GM (1993) A review of harmful algal blooms and their apparent global increase. Phycologia 32:79–99
Heisler JP, Glibert PM, Burkholder JM, Anderson DM, Cocklan B, Dennison WC, Dortch Q, Gobler CJ, Heil CA, Humphries E, Lewitus A, Magnien R, Marshall HG, Sellner K, Stockwell DA, Stoecker DK, Suddleson M (2008) Eutrophication and harmful algal blooms: a scientific consensus. Harmful Algae 8:3–13
Holland FA, Sanger DM, Gawle CP, Lerberg SB, Santiago MS, Riekerk GH, Zimmerman LE, Scott GI (2004) Linkages between tidal creek ecosystems and the landscape and demographic attributes of their watersheds. J Exp Mar Biol Ecol 298:151–178
Huisman JM, Visser PM (2005) Harmful cyanobacteria. Aquatic ecology series 3. Progr Water Technol 8:359–372
Huisman JM, Matthijs HCP, Visser PM (2005) Harmful cyanobacteria. Springer aquatic ecology series 3. Springer, Dordrecht, p 243
Jančula D, Maršálek B (2011) Critical review of actually available chemical compounds for prevention and management of cyanobacterial blooms. Chemosphere 85:1415–1422
Johnson KS, Petty RL (1983) Determination of nitrite and nitrate in seawater by flow injection analysis. Limnol Oceanogr 28:1260–1266
Jones GT, Orr PT (1994) Release and degradation of microcystin following algaecide treatment of a Microcystis aeruginosa bloom in a recreational lake, as determined by HPLC and protein phosphatase inhibition assay. Water Res 28:871–876
Kenefick SL, Hrudey SE, Peterson HG, Prepas EE (1993) Toxin release from Microcystis aeruginosa after chemical treatment. Water Sci Technol 27:433–440
Korosi JB, Smol JP (2012) Examining the effects of climate change, acid deposition, and copper sulphate poisoning on long-term changes in cladoceron assemblages. Aquat Sci 74:781–792
Lahti K, Rapala J, Färdig M, Niemelä M, Sivonen K (1997) Persistence of cyanobacterial hepatotoxin, microcystin-LR in particulate material and dissolved in lake water. Water Res 5:1005–1012
Le Jeune AH, Charpina M, Deluchat V, Briand JF, Lenian JF, Bandu M, Amblarda C (2006) Effect of copper sulphate treatment on natural phytoplankton communities. Aquat Toxicol 80:267–280
Lewitus AJ, Holland AF (2003) Initial results from a multi-institutional collaboration to monitor harmful algal blooms in South Carolina. Environ Monit Assess 81:361–371
Lewitus AJ, Schmidt LB, Mason LB, Kempton JW, Wilde SB, Wolny JL, Williams BJ, Hayes KC, Hymel SN, Keppler CJ, Ringwood AH (2003) Harmful algal blooms in South Carolina residential and golf course ponds. Popul Environ 24:387–413
Lewitus AJ, Hayes K, Kempton J, Mason L, Wilde S, Williams B, Wolny J (2004) Prevalence of raphidophyte blooms in South Carolina brackish ponds associated with housing and golf courses. In: Steidinger KA, Landsburg JA, Tomas CR, Vargo GA (eds) Harmful Algae. Florida Fish and Wildlife Conservation Commission, Florida Institute of Oceanography and the Intergovernmental Oceanographic Commission of UNESCO, St Petersburg, pp 350–352
Lewitus AJ, Brock LM, Burke MK, DeMattio KA, Wilde SB (2008) Lagoonal stormwater detention ponds as promoters of harmful algal blooms and eutrophication along the South Carolina coast. Harmful Algae 8:60–65
Matthijs HCP, Visser PM, Reeze B, Meeuse J, Slot PC, Wijn G, Talens R, Huisman J (2012) Selective suppression of harmful cyanobacteria in an entire lake with hydrogen peroxide. Water Res 46(5):1460–1472
McHuron EA, Greig DJ, Colegrove KM, Fleetwood M, Spraker TR, Gulland FMD, Harvey TD, Meinertz JR, Greseth SL, Gaikowski MP, Schmidt LJ (2008) Chronic toxicity of hydrogen peroxide to Daphnia magna in a continuous exposure, flow-through test system. Sci Total Environ 392:225–232
Paerl HW (1988) Nuisance phytoplankton blooms in coastal, estuarine and inland waters. Limnol Oceanogr 33:823–847
Paerl HW, Huisman J (2009) Climate change: a catalyst for global expansion of harmful cyanobacterial blooms. Environ Microbiol Rep 1:27–37
Paerl HW, Paul VJ (2012) Climate change: links to global expansion of harmful cyanobacteria. Water Res 46:1349–1363
Pouria S, DeAndrade A, Barbosa J, Cavalcanti RL, Barreto VTS, Ward CJ, Presier W, Poon GK, Neild GH, Codd GA (1998) Fatal microcystin intoxication in haemodialysis unit in Caruau, Brazil. Lancet 352:21–26
Qian H, Yu S, Sun Z, Xie X, Liu W, Fu Z (2010) Effects of copper sulfate, hydrogen peroxide, and N-phenyl-2-napthylamine on oxidative stress and the expression of genes involved photosynthesis and microcystin disposition in Microcystis aeruginosa. Aquat Toxicol 99:405–412
Redfield A, Ketchum B, Richards F (1963) The influence of organisms on the composition of sea-water. In: Hill M (ed) The Sea. Interscience, New York, pp 26–77
Samuilov VD, Timofeev KN, Sinitsyn SV, Bezryadnov DV (2004) H2O2-induced inhibition of phytosynthetic O2 evolution by Anabaena variabilis cells. Biochemistry (Moscow) 69:926–933
Scholin CA, Gulland F, Doucette GJ, Benson S, Busman M, Chavez FP, Cordaro J, DeLong R, De Vogelaere A, Harvey J, Haulena M, Lefebvre K, Lipscomb T, Van Dolah FM (2000) Mortality of sea lions along the California coast linked to a toxic diatom bloom. Nature 403:80–84
Serrano L, DeLorenzo ME (2008) Water quality and restoration in a coastal subdivision stormwater pond. J Environ Manag 88:43–52
Siegel A, Cotti-Rausch B, Greenfield DI, Pinckney J (2011) Nutrient controls of planktonic cyanobacteria abundance in coastal stormwater detention ponds. Mar Ecol Prog Ser 434:15–27
Sivonen K, Jones G (1999) Cyanobacterial toxins. In: Chorus I, Bartram J (eds) Toxic cyanobacteria in the water: a guide to their public health consequences, monitoring, and management. E&F Spon, New York, pp 41–111
Sokal RR, Rohlf FJ (1995) Biometry: the principles and practice of statistics in biological research, 3rd edn. Freeman, New York
Verhoeven RL, Eloff JN (1979) Effect of lethal concentrations of copper on the ultrastructure and growth of Microcystis. Proc Electron Microsc Soc S Afr 9:161–162
Wiedner C, Visser PM, Fastner J, Metcalf JS, Codd GA, Mur LR (2003) Effects of light on the microcystin content of Microcystis strain PCC 7806. Appl Environ Microbiol 69:1471–1475
Wilson RW, Taylor EW (1993) The physiological responses of freshwater rainbow trout, Oncorhynchus mykiss, during acutely lethal copper exposure. J Comp Physiol 163:38–47
Wilson AE, Sarnelle O, Tillmanns AR (2006) Effects of cyanobacterial toxicity and morphology on the population growth of freshwater zooplankton: meta-analyses of laboratory experiments. Limnol Oceanogr 52:1467–1479
Zeng J, Yang L, Wang W-X (2010) High sensitivity of the cyanobacterium Microcystis aeruginosa to copper and the prediction of copper toxicity. Environ Toxicol Chem 29:2260–2268
Zhou S, Shao Y, Gao N, Deng Y, Qiao J, Ou H, Deng J (2013) Effects of different algaecides on the photosynthetic capacity, cell integrity and microcystin-LR release of Microcystis aeruginosa. Sci Total Environ 463–464:111–119
Zimmerman CF, Keefe CW (1991) EPA Method 353.4, determination of nitrate + nitrite in estuarine and coastal waters by automated colorimetric analysis. In: An interm manual of methods for the determination of nutrients in estuarine and coastal waters. Revision 1.1, June 1991
Acknowledgments
We thank Norm Shea and the Kiawah Island Community Association for permission to conduct the field work. We also thank SePRO Corporation and Solvay Chemicals Inc. for providing test samples of their products. We would like to thank the valuable input from anonymous reviewers. This project supported summer student research (Duquette and Goodson), and it was funded by the South Carolina Sea Grant Consortium award R-378 to S. Wilde, D. Greenfield, and D. White. This is contribution number 728 from the Marine Resources Research Institute and 1700 from the Belle W. Baruch Institute for Marine and Coastal Sciences, University of South Carolina.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Greenfield, D.I., Duquette, A., Goodson, A. et al. The Effects of Three Chemical Algaecides on Cell Numbers and Toxin Content of the Cyanobacteria Microcystis aeruginosa and Anabaenopsis sp.. Environmental Management 54, 1110–1120 (2014). https://doi.org/10.1007/s00267-014-0339-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00267-014-0339-2