Abstract
The importance of preserving both protected areas and their surrounding landscapes as one of the major conservation strategies for tigers has received attention over recent decades. However, the mechanism of how land-use surrounding protected areas affects the dynamics of tiger populations is poorly understood. We developed Panthera Population Persistence (PPP)—an individual-based model—to investigate the potential mechanism of the Sumatran tiger population dynamics in a protected area and under different land-use scenarios surrounding the reserve. We tested three main landscape compositions (single, combined and real land-uses of Tesso-Nilo National Park and its surrounding area) on the probability of and time to extinction of the Sumatran tiger over 20 years in Central Sumatra. The model successfully explains the mechanisms behind the population response of tigers under different habitat landscape compositions. Feeding and mating behaviours of tigers are key factors, which determined population persistence in a heterogeneous landscape. All single land-use scenarios resulted in tiger extinction but had a different probability of extinction within 20 years. If tropical forest was combined with other land-use types, the probability of extinction was smaller. The presence of agroforesty and logging concessions adjacent to protected areas encouraged the survival of tiger populations. However, with the real land-use scenario of Tesso-Nilo National Park, tigers could not survive for more than 10 years. Promoting the practice of agroforestry systems surrounding the park is probably the most reasonable way to steer land-use surrounding the Tesso-Nilo National Park to support tiger conservation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The importance of managing protected areas together with the surrounding landscape is a central issue for the conservation of large endangered mammals such as the Sumatran tiger (Johnson and others 2006; Nyhus and Tilson 2004; Kusters and others 2008; Maddox and others 2007). As a large carnivore, the Sumatran tiger requires large protected areas for habitat, food availability and reproduction. The social, political and economic realities of many landscapes in Asia have turned large suitable habitats into small, fragmented ones (Nyhus and Tilson 2004) and the difficulty of halting forest loss has made the situation even worse (Kinnaird and others 2003). Therefore, the need for integrated and comprehensive management strategies that take into account the complexity of landscapes surrounding protected areas has become more urgent over recent decades.
In order to integrate the surrounding landscape with protected areas, particular attention should be paid to the landscape structure. Landscape structures play an important role in population dynamics (Fahrig 2007) with landscape composition providing different risks and benefits to maintain wildlife populations. Sumatran tiger populations have been facing habitat fragmentation and possible population isolation due to forest conversion over the last three decades (Franklin and others 1999). As a result, the landscape of Sumatra is comprised of many land-use types such as logging concessions, agriculture, settlements, oil-palm plantations as well as mining operations. A challenge for conserving the Sumatran tiger is to understand how tigers respond to those land-uses Seindensticker and others (1999) and which landscape configurations are able to support tiger populations.
The response of tigers to different land-use types should consider their relation to prey species. As a carnivore, tigers depend on prey populations (Miquelle and others 1999; Ramakrishnan and others 1999; Karanth and Stith 1999; Karanth and others 2004). However, the response of predators to the loss and fragmentation of habitat depends on the complex interaction between predator, prey and their habitat (Ryall and Fahrig 2006). Therefore, in order to be able to understand the response of the Sumatran tiger to a heterogeneous landscape, we have to understand the mechanism of individual behaviour and the relation with prey distributions and landscape configurations.
To the best of our knowledge, it is hard to find any information on the mechanisms behind the response of the Sumatran tiger to a heterogeneous landscape. Several studies have shown the distribution pattern of the Sumatran tiger and its prey in different habitat types, such as tropical forest within protected areas (Franklin and others 1999; Kinnaird and others 2003; Linkie and others 2003; O’Brien and others 2003), areas with agroforestry (Nyhus and Tilson 2004), palm oil plantations (Maddox and others 2007) and logging concessions (Linkie and others 2008). However, those studies did not explain the mechanistic process involved in responding to different habitat types. In addition, the cryptic characteristics of the tiger and the complexity of the existing landscape configuration have resulted in very few behavioural studies of this species, presenting us with further limitations to understanding their response to habitat change.
Considering the rapid loss of tropical forests on the Island of Sumatra, the emergence of various habitat types surrounding protected areas and the lack of knowledge on the response of tigers and prey to various habitat types, we require an alternative approach to deal with this situation. The approach should be able to be used to understand individual behaviour as influenced by different landscapes. Modelling is one possible approach for predicting population consequences from landscape structure. Individual-based modelling (IBM) has been used to deal with individual behaviour patterns and their emergence at higher levels such as the population or landscape level. IBMs have the ability to simulate the behaviour of individuals and predict interactions depending on complex environmental conditions (Ahearn and others 2001; Grimm and Railsback 2005). It is almost impossible to set up experiments with different land-use types surrounding a protected area to monitor the response of tiger populations. The use of IBM would therefore appear to be a promising option that would complement existing monitoring programs. For these reasons, IBM was chosen for this study as a tool for understanding the response of tiger populations to very dynamic changes in land-use.
Methods
Study Area and the Development of Land-Use Maps
We selected the Tesso-Nilo National Park and its surrounding landscape with a total size of 3,841 km2. The national park is situated in the lowland area of Riau province in Central Sumatra, Indonesia. The park was established in 2004 and was formerly an area designated for logging concessions. The park is surrounded by different land-use types as shown in Fig. 1. We developed a land-use map from a satellite image of LANDSAT (Path 126 row 60 date 07 August 2005). To adjust it to the current situation of the study area, we compared it with a land-cover map from WWF-Riau and a ground check was conducted in November and December 2009. We classified the study area into six major land-use types: tropical forest, logging concessions, agroforestry, acacia (Acacia mangium) plantations, palm-oil plantations, and settlements.
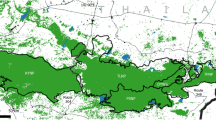
Initial conditions of three different landscape compositions: a single land use b combined land-use c real land use in Tesso Nilo National Park and the surrounding landscape of Central Sumatra. The white line indicates the border of the park. Different grid cell colours represent different land-use types (very light grey settlements, light grey palm oil plantations, medium grey acacia plantations and agroforestry, dark grey logging concessions and very dark grey tropical forest)
Model Description
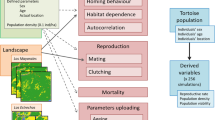
We developed the Panthera Population Persistence (PPP) model, which was derived from the TIGMOD model (Ahearn and others 2001). The TIGMOD model has been successfully used to simulate basic behaviour of the tiger and its potential conflict with humans in Nepal. The model description follows the ODD (overview, design concepts and details) protocol for describing individual- and agent-based models (Grimm and Railsback 2005; Grimm and others 2006).
Purpose
The purpose of the model is to investigate the potential mechanisms of tiger population dynamics in the reserve under different land-uses.
State Variables and Scales
The PPP model comprises three animal species: the Sumatran tiger (Panthera tigris sumatrae) and two of its prey species, the Sambar deer (Rusa unicolor) and the red muntjac (Muntiacus muntjac). The individuals of tigers have different sex and age classes, hunger and starvation levels and reproductive-based state variables. Sambar deer and red muntjac have age and hunger levels. Table 1 shows the description of state variables for each species in the PPP model. The simulated area is Tesso Nilo National Park and its surrounding landscape (Fig. 1).
Process Overview and Scheduling
Ageing. As in TIGMOD (Ahearn and others 2001), tigers and prey increase in age with a time-step of 0.5 days with the age-classes changing accordingly. The age classes and internal state conditions lead to different behaviour patterns such as cubs that only follow the mother or hunger-levels which determine whether an individual will hunt or not. Figure 2 shows the behaviour of tigers in different age classes within the PPP model.
Movement. The PPP simulates two types of movement for tigers; random and directed movement. Random movement is not related to any object. It is part of the behaviour involved in maintaining a home-range, reproduction and parenting. Directed movement includes “following the mother”, hunting, mating, and parenting (described below). Appendix 1 shows further details and parameters. A prey decides to move or to stay based on the habitat quality of the patch.
Hunger and Starvation. The hunger level of a tiger starts at 0 and increases by 10 per time-step and by 12.5 for a female with cubs. If an individual has a hunger-level above 90, starvation will start. In that case, the hunger level is fixed to 90, and the starvation-level increases by 0.5 per time step. If a tiger reaches a starvation level of 30, it will die. When hunger levels are >60, hunting is stimulated. The hunger levels of a tiger will decrease when it has successfully caught and consumed its prey, the starvation level is then set to zero again and the hunger level is reduced by 12.5 per time-step over the entire feeding period. Foraging behaviour of a prey is controlled by its hunger level.
Hunting. A tiger searches for prey within a specific hunting radius. When a tiger has sensed its prey, it will change direction accordingly. The hunting success rate of a tiger varies from 5% to 50% (Sunquist 2010). Hunting success is assumed to be 50% on any hunting occasion. Tigers prefer Sambar deer (Reddy and others 2004), and if no Sambar deer is available within their hunting radius they will prey on red muntjac.
Feeding. Tigers stay close to their hunted prey during the feeding time (Sunquist 1981). The larger the prey, the longer the tiger will stay to consume it. If a tiger successfully kills its prey in a tropical forest, the model simulates two consecutive days to consume a red muntjac and 7 days for a Sambar deer without interruption. Tigers spend less time consuming prey when kills are made in human-affected areas while in undisturbed areas tigers are more likely to eat more meat and spend more time doing so (Kerley and others 2002). To incorporate the effect of human-disturbed areas, we reduce the time of feeding on red muntjac to 1.5 days and on Sambar deer to 5 days in these areas.
Reproduction. The PPP model simulates the reproduction of tigers through three processes: fertility scheduling, mating, pregnancy and giving birth. A female reaches sexual maturity at the age of 825 days (Sunquist 1981). In this case fertility scheduling is initiated. The inter-estrous interval of a female tiger is around 25 days during which the female is fertile for about 5 days. A fertile female will call to an adult male for mating. However, mating will only occur when hunger levels of individuals are lower than 60 and there is no starvation. The duration of mating is two days (Sunquist 1981) and the female will have a 50% chance of becoming pregnant. The gestation period for a female is 102–103 days (Ahearn and others 2001; Sunquist 1981; Sunquist and others 1999). A pregnant female has a random probability of giving birth to 1–3 cubs with a ratio of males to females of 1:3. A new-born tiger will usually adopt all characteristics from its mother except for its sex class, age, its hunger-level and starvation level. The age of cubs is 0 at the time step of birth. The hunger and starvation levels are also 0 until the cubs reach sub-adulthood. A female with cubs will not display any mating behaviour until the cubs reach sub-adult classes. We simulated density dependent birth rate for both red muntjac and Sambar deer.
Mortality. A tiger dies due to starvation. If the starvation-level reaches >30, the tiger will die. If the dead tiger is a female with cubs, the cubs will also die. When an adult male tiger dies, the home-range will be occupied by a sub-adult that is searching for a home-range. Prey mortality is due to tiger killing and starvation. The limit of starvation for prey is defined by hunger level >200.
Dispersal. At the sub-adult level, tigers search for a home range. The home range of a male might overlap with that of one or several females but never with the home range of another male. An adult individual without a home range is removed from the model but is not considered as a dead individual. The PPP model calculates this as a dispersed individual.
Design Concept
The dynamics of the tiger population emerge from the interaction between tiger individuals, prey and habitat. The PPP model explicitly simulates four types of interaction. The first is a prey-habitat interaction, which shows the movement and the foraging behaviour of prey in different habitat types. The prey decides whether to move to the next patch or to stay depending on certain habitat indices. Such indices also determine the energy gained by the prey while foraging. The second type of interaction is a tiger-prey interaction, which represents the behaviour of a tiger hunting prey. The third type of interaction is tiger-prey-habitat, which represents the time taken to consume prey that has been killed in different land-use types. The fourth type is a tiger-tiger interaction, which simulates the behaviour of mating and parental care (between a mother and cubs).
A tiger prefers to kill large prey. However, when there is no large prey in the hunting radius, the tiger will automatically search for small prey. Newborn tigers inherit this preference for large prey. Adult tigers are known to compete for resources and mating (Sunquist 1981). When an adult individual cannot establish a home range, the model considers it as transient and takes out from the landscape. Tigers are able to detect prey and mate, and a cub senses the presence of its mother to be followed.
Stochasticity is applied to the probability of a tiger to successfully hunt for prey, the probability of becoming pregnant, the number of new cubs and the proportion of male and female cubs. Collectiveness occurs during mating and parenting behaviour. A male and a female will remain together over the mating period, and a female will stay with its cubs until they reach the sub-adult class.
Details
Initialization. An adult male Sumatran tiger requires 116 km2 to maintain a home-range, and 70 km2 for a adult female (Franklin and others 1999). We set the population to 10 adult tiger individuals with 5 males and 5 females according to the possible number of tigers occupying Tesso Nilo National Park with an area of approx. 1,000 km2. The densities of red muntjac and Sambar deer in tropical forest were set according to the findings of O’Brien and others (2003). Sambar deer has a density range of 0.88 – 1.42 individuals /hectare and red muntjac has a range of 3.96–4.44 individuals/hectare. For this study, we used the lowest value for both Sambar deer and red muntjac in tropical forest. Due to a lack of data for other land-use types, we proportionately adjusted the density according to the habitat conditions as shown in Table 2.
Input. 203 × 149 grid cells represent the Tesso Nilo National Park and surrounding land-use. Each grid cell represents 12.7 ha and is specified by habitat quality, which corresponds to the land-use types. We developed three main scenarios (single, combined and real land-use scenarios) to test the persistence of the Sumatran tiger population under different land-use compositions. Details on the description of each scenario are given in a later section. The spatial composition of those main scenarios follows the composition of a land-cover map of our study area and can be seen in Fig. 1.
Submodels. We included four main types of prey behaviour: movement, foraging, reproduction and mortality. Prey movement is defined by two main factors: direction and distance. The distance refers to data obtained for red deer movement (Fryxell and others 2008) which varies from 0.23 to 7 km/day. This variation covers both the encamped mode (more sedentary behaviour) and exploratory mode (rapid directional movement). The direction of movement is driven by habitat quality indices. The probability of prey movement is calculated as follows:
with α being the movement probability to the next patch, β1 being the habitat index of the next path and β0 being the habitat index of the current patch (Table 2). If α ≤ 0.5 then the prey will stay in the current patch, otherwise it will move to the next patch. We did not differentiate between the distance and direction for Sambar deer and red muntjac.
The presence of human-affected areas can increase the energy consumed by an animal in order to avoid human contact (Kerley and others 2002). Prey will remain in a patch and consume a certain amount of the food resource in that particular patch. Prey will receive different resource values in different land-use types. At the same rate of increased hunger-level, the greater the human intervention, the less the energy that is gained from the patch, and consequently the more easily the prey becomes hungry. Both red muntjac and Sambar deer increase their hunger-level by 10 levels per time step. Since we do not have any data on the rate of consumption of prey species within different habitat types, we used the same rate for all types of habitats.
Red muntjac start to reproduce annually from the age of 2–4 years with probability of number of litters consisting of 3 individuals. Sambar deer annually reproduce with 1 litter from the age of 2–6 years. Both prey die when they reach a maximum age (approx. 10 years for red muntjac and 17 years for Sambar deer), from acute starving (hunger-level is greater than 200), and/or are killed by tigers. Both Sambar and red muntjac have density-dependent birth rates. Both will continue to reproduce until the population reaches the carrying capacity.
Implementation. The model is implemented in NETLOGO v. 4.1. (Wilensky 1999). Each simulation was carried out for a simulation time of 20 years or until one of the sex classes of the tiger became extinct. We ran each scenario with 100 repetitions.
Scenarios and Analysis
Figure 1 shows the landscape composition of single, combined and real land-use scenario. The single land-use scenario consists of either settlements (ST), palm-oil plantations (POP), acacia plantations (ACP), agroforestry (AF), logging concession (LC) or tropical forest (TF). These scenarios represent habitat qualities from low to high, respectively. The landscape composition for these scenarios is a homogenous area. The combined land-use scenarios are a combination of tropical forest with settlements (CST), palm-oil plantations (CPOP), acacia plantations (CACP), agroforestry (CAF) and logging concessions (CLC). The proportion of tropical forest to other land-uses is 2,594 km2 compared to 1,246 km2.
These combined land-use scenarios scenarios were mainly tropical forest in combination with either settlements (CST), palm-oil plantations (CPOP), acacia plantations (CACP), agroforestry (CAF) or logging concessions (CLC). The proportion of tropical forest in the combined land-use is 2,594 km2 compared to 1,246 km2 of other land-use. The forest area follows the distribution of pristine forest and secondary forest in the Tesso Nilo National Park and its surrounding landscape (Fig. 1). In addition, a real land-use scenario also is used to evaluate the current land-use compositions in the study area. The proportion of the size of each land-use type in the real land-use scenario is given in Table 3.
We measured the number of individuals of tigers and prey when extinction or quasi-extinction occurs. Extinction occurs when all tiger individuals (both male and female) become absent in the model. Quasi-extinction is the loss of one of the sex classes from the model due to mortality or dispersal. We refer both extinction and quasi extinction as extinction for further use. We recorded the time-step when extinction occurred. We also calculated the probability of extinction P 0(t20) which represents a ratio between the numbers of simulations resulting in extinction and the total number of simulations in each scenario during 20 years of simulation. If extinction did not occur during a simulation, we calculated number of tigers, prey and dispersed tigers at the end of the simulation.
We calculated the time to extinction, the arithmetic mean time to extinction (Tavg) and the intrinsic mean time to extinction (Tm). The Tavg is the average time-step from all simulations. Compared to Tavg, Tm is a more robust measure that does not depend on the shape of the distribution of extinction time or the initial conditions (Grimm and Wissel 2004). Tm is the slope of the linear regression line based on the ln (1 − P 0(t)) plot. P 0(t) is the cumulative probability of extinction calculated on the basis of the histogram of extinction times. We also calculated the relaxation time (Trel), which specifies the period until the dynamics of the tiger population are no longer affected by the initial conditions of the model. The Trel is derived from the intercept of the regression line of −ln (1 − P 0(t)) with the x-axis. The probability distributions of the state variables are then quasi-stationary (Grimm and Wissel 2004). We did not calculated both of Tm and Trel if the P 0(t ≤ 20) is very low.
For conservation purposes, the intrinsic mean time to extinction is very important to provide a careful prediction. Unlike the arithmetic time to extinction where the initial conditions have an effect on the established phase, the initial conditions of the model do not have any consequences for the relaxation time (Grimm and Wissel 2004). In addition, to understand how landscape structures affect the tiger population, we also recorded the number of dispersed tigers. This value corresponds to the number of tiger individuals leaving the study area because they are unable to establish a home-range.
Results
Tiger and Prey Densities Under Different Land-Use Scenarios
Figure 3 compares densities of tigers, dispersed tigers and prey population at the end of the simulation in each scenario. In general, the densities of tigers at the end of simulations showed a significant difference for all land-use scenarios (Kruskal Wallis test, H = 772.18, df = 11, P < 0.01). Similar patterns could also be seen for the density of dispersed tigers (Kruskal Wallis test, H = 1052.32, df = 11, P < 0.01), and for the remaining prey population (Kruskal Wallis test, H = 969.03, df = 11, P < 0.01).
We separated the analysis for single and combined scenarios. Under single scenario, a strong significant difference among scenarios was also found for remaining tiger densities (Kruskal Wallis test, H = 318.05, df = 5, P < 0.01), dispersed tigers densities (Kruskal Wallis test, H = 556.21, df = 5, P < 0.01) and remaining prey densities (Kruskal Wallis test, H = 505.31, df = 5, P < 0.01). Among all scenarios, the tropical forest scenario showed the highest values for tiger, dispersed tigers as well as prey densities. The combined land-use scenarios showed that the tiger density differed among scenarios (Kruskal Wallis test, H = 397.50, df = 5, P < 0.01), as did the density of dispersed tigers (Kruskal Wallis test, H = 474.53, df = 5, P < 0.01) and the remaining prey density (Kruskal Wallis test, H = 322.89, df = 5, P < 0.01). No significant different on the densities of tigers, dispersed tigers and prey for combination of tropical forest with acacia plantation, agroforestry and logging-concession.
With the real land-use scenario, the density of tiger was different from all other scenarios except for agroforestry. The remained dispersed tiger densities also showed a significant difference with other scenarios except for acacia plantation. The remaining prey densities showed a significant difference to all other scenarios.
Extinction Probability
The probability of extinction-within 20 years of time simulation P 0(t ≤ 20) under a single land-use scenario indicated that the increment of habitat quality reduces the probability of extinction (table 4). Tropical forest was the best scenario with the lowest probability of extinction P 0(t ≤ 20) = 4% followed by logging concessions P 0(t ≤ 20) = 66%, agroforestry P 0(t ≤ 20) = 99%. Acacia plantation, settlements and palm oil plantations resulted in P 0(t ≤ 20) = 100% extinction over the simulation period.
All scenarios with combined land-use showed a lower probability of extinction than a single land-use. However, combined palm oil plantations and settlement only showed a slight difference from a single scenario. The combined scenario of tropical forest with acacia plantations and agroforestry showed a remarkable improvement in the probability of extinction from that of the single acacia plantation and single agroforestry scenarios. In addition, combined logging concession and combined agroforestry was similar as single tropical forest P 0(t ≤ 20) = 4%, while the real land-use scenario showed P 0(t ≤ 20) = 100% of all simulations resulting in an extinction within 20 years of the simulation.
Time to Extinction
Arithmetic Time to Extinction
Among all single land-use scenarios, tropical forest displayed the highest Tavg values whereas settlements showed the lowest. From the combined scenarios, the combination of tropical forest with acacia plantation, agroforestry and logging concessions improved the time to extinction from their single scenarios. The real land-use scenarios were able to support a tiger population with a Tavg of 7.31 years (Table 4).
Relaxation Time
The relaxation time was measured only on the scenarios with ≥30 simulations which resulted tiger extinction/quasi extinction. The relaxation time for single settlements required more than one year to relieve from the effect of initial conditions during simulation. The logging concessions scenarios achieved the longest relaxation compared to the other scenarios, followed by agroforestry and acacia plantations respectively (Table 4). The calculation of Trel were not applicable due to very low extinction probability of tropical forest within 20 years and very clumped data on palm-oil plantation. The relaxation times for combined scenarios were only calculated for palm-oil plantation (CPOP) and settlement (CST). The relaxation time for the real land-use scenarios was the lowest among all other scenarios (Fig. 4).
Intrinsic Time to Extinction
In single land-use scenarios, the settlement displayed the shortest Tm, whereas the logging-concession showed the longest. Agroforestry and acacia plantation delayed the extinction of tiger populations for more than 5 years. In settlements tigers could only survive for less than 2 years (Fig. 4). The combination of tropical forest with acacia plantation, agroforestry and logging concessions were not calculated due to low probability of extinction (see Table 4). Settlements and palm-oil plantation could support tiger populations for more than 5 years when combined with tropical forest. The real land-use scenario could maintain tiger population for not more than 10 years (Fig. 4).
Discussion
The PPP model is the first spatially-explicit individual-based model that has been developed for understanding the consequence of different land-use configurations on the persistence of the Sumatran tiger population. The model has plausibly simulated important key behaviours of tigers to respond to different land-use types in Central Sumatra. The landscape compositions affected the persistence of Sumatran tiger populations due to three types of mechanisms. The first of these mechanisms are habitat conditions, which affect the numbers of prey available for tigers. The second-prey mortality is affected by a variation in the food consumption of prey under different land-use types. The third mechanism of human-affected areas reduces the time to consume prey that has been killed by the tiger.
Tigers respond to the presence of humans by reducing the time they have to consume prey and by exhibiting a less efficient consumption than in undisturbed areas (Kenney and others 1995). The PPP model simulated different times for feeding (t-feeding) for tigers in various land-use types. In their natural habitat, tigers use a maximum time for feeding, but in human-affected land-use areas, tigers consume prey with less time for feeding. Consequently, tigers gain less energy in habitats with anthropogenic land-use than in habitats with tropical forest land-use. The direct consequences of an individual gaining less energy results in a higher probability of starvation, eventually leading to mortality. Since the PPP model only simulates mating behaviour when individuals are not hungry and starving, the probability of mating is reduced when less energy is gained by an individual. In turn, this will reduce the probability of reproduction. These two factors determined the probability of extinction as well as the time to extinction in each scenario.
The results from the single scenarios clearly indicate that different habitat qualities are important for population persistence analysis. The PPP model plausibly showed the effect of habitat conditions on the foraging and movement behaviour of prey and the movement and feeding behaviour of tigers. Even though we used an optimistic value for the hunting radius, the results of our simulation still showed that the survival chance was relatively low except under the tropical forest scenario. The tropical forest scenario is the best scenario among all of the scenarios. The model showed that large protected natural landscapes are preferable for tigers to survive and reproduce, in line with the empirical study by Linkie and others (2003) that tigers are best conserved in large protected areas.
The combined scenarios highlighted the importance of the land-uses surrounding protected areas for tiger conservation (Baeza and Estades 2010). Although logging-concessions, agroforestry and acacia plantation as single land-use scenarios were not the best habitat for Sumatran tigers, the combination of tropical forest with them resulted in an improved situation. However, since we did not test the simulation for more than 20 years, further analysis beyond this time horizon will be more meaningful.
Among all anthropogenic land-use types, selective logging concessions provided the best alternative to maintain forest cover and ensure the availability of alternative food for prey (Meijaard and Sheil 2008). In addition, the gaps created from logging provide tigers with the opportunity to have a better access for dispersal and hunting (Linkie and others 2008). Selective logging concessions close to a protected area provide a good habitat for tigers (Linkie and others 2008). The fact that Tesso-Nilo National Park used to be logging concessions but is still able to support a tiger population suggests that it is reasonable to combine this type of land-use when it is close to a protected area as part of an overall tiger conservation strategy.
Agroforestry and forest plantations have become increasingly more apparent in the Sumatran landscape. The probability of these two scenarios for the persistence of tiger populations was more than 90% over 20 years if combined with tropical forest. Our findings can be used to explain why agroforestry has the potential to conserve tiger populations in Nepal (Gurung and others 2008; Dinerstein and others 1999) and Sumatra (Nyhus and Tilson 2004). Agroforestry surrounding strict protected areas can support the conservation of the Sumatran tiger through a greater availability of prey (Nyhus and Tilson 2004). The Agroforestry system on the island of Sumatra varies in its vegetation compositions such as rubber (Beukema and others 2007), multi-storied tree gardens (Michon and others 1986), Damar/resin production agroforestry (Kusters and others 2008) as well as coffee agroecosystems (Philpott and others 2008). These different vegetation compositions might bring about different consequences for both the Sumatran tiger and its prey. A large-scale forest plantation might provide protection from human pressure. However, most forest plantations are managed in a monoculture system, which might not support many species of prey, but could lead to a population explosion for some species, such as wild boar.
The settlement and oil-palm plantation scenarios clearly showed the detrimental effect of these land-use types on tiger persistence. Both single and combined scenarios resulted in extinction within a relatively short period of time. This finding is consistent with the absence or non-detection of tigers in settlements and palm-oil plantations (Maddox and others 2007). Settlements and oil-palm plantations do not provide good habitat for prey, provide poor habitat and experience high human pressure, leading to the absence of tigers.
The real land-use scenario resulted in a relatively short time to tiger extinction. A small proportion of tropical forest (6.75%) and a large proportion of settlements and palm-oil plantations (32.2%) might explain the disappearance of tigers due to a low density of prey (see Table 3). However, since the PPP model integrated a land-cover map which is not directly represented by a habitat-matrix map for prey species, the incorporation of a habitat suitability index map might improve the prediction. In addition, the pressure from poaching under anthropogenic land-use scenarios is crucial for the persistence of both tiger and prey (Nyhus and Tilson 2004). Therefore, to improve our understanding of the effect of these land-uses, poaching should also be included.
The PPP model simulated the response of both tiger and prey on the presence of anthropogenic land-use. However, we assume that the movement decision of Sambar deer and red muntjac are similar. The Sumatran tiger is known to predate wild-boar which behaves differently toward the presence of human. Studies have shown that the movement decisions of individuals are important factors for populations of the lynx (Kramer-Schadt and others 2004) and the Florida panther (Cramer and Portier 2001). Therefore considering species-dependent different movement decision of prey would be worth in the next model development.
Conservation Implications
The PPP model successfully simulated the response of tigers and their prey to various land-use types. The model demonstrates the mechanisms of how tigers and their prey are affected by different land-use scenarios. The PPP model proves that tropical forest is the best scenario. However, with the current rates of tropical deforestation on the island of Sumatra, a large tropical forest area is rarely found in a real situation. The PPP model also showed the importance of including logging concessions and agroforestry in the overall conservation strategy for the tiger. However, the potential detrimental effect such as that from poaching pressure should be considered.
Despite the ability of acacia plantations surrounding a protected area to extend the time to extinction, due to its potential bad effect from monoculture practices, this land-use type is not recommended to be established surrounding a reserve. Both palm-oil and settlement scenarios, either on their own or combined with tropical forest do not support tiger conservation. Therefore, avoiding these land-use types from surrounding a protected area can help to reduce the probability of local extinction (Table 5).
Our findings highlight the importance of logging concessions and agroforestry surrounding a protected area for the conservation of the Sumatran tiger. Agroforestry provides ecological services through corridors and food sources for the tiger and its prey. This study shows the critical condition of the land-use composition surrounding the Tesso-Nilo National Park. A low proportion of natural areas together with a large proportion of human-affected areas are critical factors affecting the tiger in the Central Sumatran landscape. Under the assumption that the re-establishment of natural, undisturbed forests in the region is unrealistic, the most feasible way to improve this condition is to promote the practise of agroforestry systems surrounding the park, while at the same time reducing poaching.
References
Ahearn SC, Smith JLD, Joshi AR, Ding J (2001) TIGMOD: an individual-based spatially explicit model for simulating tiger/ human interaction in multiple use forests. Ecological Modelling 140:81–97
Baeza A, Estades CF (2010) Effect of the landscape context on the density and persistence of a predator population in a protected area subject to environmental variability. Biological Conservation 143:94–101
Beukema H, Danielsen F, Vincent G, Hardiwinoto S, and van Andel J. (2007) Plant and bird diversity in rubber agroforests in the lowlands of Sumatra, indonesia. Agro forest System 70. doi:10.1007/s10457-007-9037-x
Chapman NG, Furlong M, Harris S (1997) Reproductive strategies and the influence of date of birth on growth and sexual development of an aseasonally-breeding ungulate: Reeves’ muntjac (Muntiacus reevesi). Journal of Zoology, London 241:551–570
Chapman NG, Brown WAB, Rothery P (2005) Assessing the age of Reeves’ muntjac (Muntiacus reevesi) by scoring wear of the mandibular molars. Journal of Zoology, London 267:233–247. doi:10.1017/S0952836905007405
Cramer PC, Portier KM (2001) Modeling Florida panther movements in response to human attributes of the landscape and ecological settings. Ecological Modelling 140:51–80
Dinerstein E, Rijal A, Bookbinder M, Kattel B, Rajuria A (1999) Tigers as neighbours: efforts to promote local guardianship of endangered species in lowland Nepal. In: Seindensticker J, Christie S, Jackson P (eds) Riding the tiger tiger conservation in human dominated landscapes. Cambridge University Press, UK, Australia, New York
Fahrig L (2007) Non-optimal animal movement in human-altered landscapes. Functional Ecology 21:1003–1015
Franklin N, Bastoni S, Siswomartono D, Manangsang J, Tilson R (1999) Last of the indonesian tigers: a cause for optimism. In: Seindensticker J, Christie S, Jackson P (eds) Riding the tiger: tiger conservation in human dominated landscapes. Cambridge University Press, UK, Australia, New York
Fryxell JM, Hazell M, Berger L, Dalziel BD, Haydon DT, Morales JM, McIntosch T, Rosatte RC (2008) Multiple movement modes by large herbivores at multiple spatiotemporal scales. PNAS 105:19114–19119
Grimm V, Railsback SF (2005) Individual-based Modelling and Ecology, Princeton Series in theorical and computational biology, vol 1. Princeton University Press, Princeton and Oxford
Grimm V, Wissel C (2004) The intrinsic mean team to extinction: a unifying approach to analysing persistence and viability of populations. OIKOS 105:501–511
Grimm V, Berger U, Bastiansen F, Eliassen S, Ginot V, Giske J, Goss-Custard J, Grand T, Heinz SK, Huse G, Huth A, Jepsen JU, Jorgensen C, Mooij WM, Mueller B, Pe’er G, Piou C, Railsback SF, Robbins AM, Robbins MM, Rossmanith E, Rüger N, Strand E, Souissi S, Stillman RA, Vabø R, Visser U, DeAngelis DL (2006) A standard protocol for describing individual based and agent-based models. Ecological Modelling 198:115–126
Gurung B, Smith JLD, McDougal C, Karkic JB, Barl A (2008) Factors associated with human-killing tigers in Chitwan national park, Nepal. Biological Conservation 141:3069–3078
Johnson A, Vongkhamheng C, Hedemark M, Saithongdam T (2006) Effect of human-carnivore conflict on tiger (Panthera tigris) and prey population in Lao PDR. Animal Conservation 9:421–430
Karanth UK, Stith BM (1999) Prey depletion as critical determinant of tiger population viability. In: Seindensticker J, Christie S, Jackson P (eds) Riding the tiger: tiger conservation in human dominated landscapes. Cambridge University Press, UK, Australia, New York
Karanth UK, Nichols JD, Kumar NS, Link WA, Hines JE (2004) Tiger and their prey: predicting carnivore densities from prey abundance. PNAS: 4854–4858
Kenney JS, Smith JLD, Starfeld AM, McDougal CW (1995) The long-term effects of tiger poaching on population viability. Conservation Biology 9:1127–1133
Kerley LL, Goodrich JM, Miquelle DG, Smirnov EN, Quigley HB, Hornocker MG (2002) Effects of roads and human disturbance on Amur tigers. Conservation Biology 16:97–108
Kinnaird MF, Sanderson EW, O’Brien TG, Wibisono HT, Woolmer G (2003) Deforestation trends in a tropical landscape and implications for endangered large mammals. Conservation Biology 17:245–257
Kramer-Schadt S, Revilla E, Wiegand T, Breitenmoser U (2004) Fragmented landscapes, road mortality and patch connectivity: modeling influences on the dispersal of Eurasian lynx. Journal of Applied Ecology 41:711–723
Kusters K, P’erez MR, de Foresta H, Dietz T, Ros-Tonen M, Belcher B, Manalu P, Nawir A, Wollenberg E (2008) Will agroforests vanish? The case of damar agroforests in Indonesia. Human Ecology 36:357–370
Linkie M, Martyr DJ, Holden J, Yanuar A, Hartana AT, Sugardjito J, Williams NL (2003) Habitat destruction and poaching threaten the Sumatran tiger in Kerinci Seblat national park, Sumatra. Oryx 37:41–48
Linkie M,I, Haidir A, Nugroho A, Dinata Y (2008) Conserving tigers panthera tigris in selectively logged sumatran forests. Biological Conservation 141:2410–2415 (Research Note)
Maddox T, Priatna D, Gemita E, Salampessy A (2007) The conservation of tigers and other wildlife in oil palm plantations Jambi Province, Sumatra, Indonesia. ZSL Conservation Report No. 7 The Zoological Society of London, London
Meijaard E, Sheil D (2008) The persistence of conservation of borneo’s mammals in lowland rain forests managed for timber: observation, overview and opportunities. Ecological Research 23:21–34
Michon G, Mary F, Bompard J (1986) Multistoried agroforest system garden system in west Sumatra, Indonesia. Agrorofestry sytem 4:315–338
Miquelle DG, Smirnov EN, Merril TW, Myslenkov AE, Quigley HB, Hornocker MG, Schleyer B (1999) Hierrachical spatial analysis of Amur tiger relationships to habitat and prey. In: Seindensticker J, Christie S, Jackson P (eds) Riding the tiger: tiger conservation in human dominated landscapes. Cambridge University Press, UK, Australia, New York
Nugen G, Fraser KW, Asher GW, Tustin KG (2001) Advances in New Zealand mammalogy 1990–2000: Deer. Journal of The Royal Society of New Zealand 31:263–298
Nyhus P, Tilson R (2004) Agroforesry, elephants, and tigers: balancing conservation theory and practice in human dominated landscape of southeast Asia. Agriculture, Ecosystem and Environment 104:87–97
O’Brien TG, Kinnaird MF, Wibisono HT (2003) Crouching tigers, hidden prey: Sumatran tiger and prey populations in a tropical forest landscape. Animal Conservation 6:131–139
Philpott SM, Bichier P, Rice RA, Greenberg R (2008) Biodiversity conservation, yield, and alternative products in caffe agroecosystems in Sumatra, Indonesia. Biodiversity Conservation 17:1805–1820. doi:10.1007/s10531-007-9267-2
Ramakrishnan U, Coss RG, Pelkey NW (1999) Tiger decline caused by reduction of large ungulate prey: evidence from a study of leopard diets in southern India. Biological Conservation 89:113–120
Reddy HS, Srinivasulu C, Rao KT (2004) Prey selection by Indian tiger (Phantera tigris tigris) in Nagarjunasagar Srisailam tiger reserve, India. Mammalian Biology 69:384–391
Ryall KL, Fahrig L (2006) Response of predators to loss and fragmentation of prey habitat: a review of theory. Ecology 87:1086–1093
Seindensticker J, Christie S, Jackson P (1999) Tiger ecology: understanding and encouraging landscape patterns and condition where tigers can persist. In: Seindensticker J, Christie S, Jackson P (eds) Riding the tiger: tiger conservation in human dominated landscapes. Cambridge University Press, UK, Australia, New York
Semiadi G, Muir PD, Barry TN (1994) General biology of sambar deer (Cervus unicolor) in captivity. New Zealand Journal of Agricultural Research 37:79–85
Smith JLD (1993) The role of dispersal in structuring the Chitwan tiger population. Behaviour 124:169–195
Sunquist ME (1981) The Social Organization of Tigers (Phantera tigris) in Royal Chitawan National Park, Nepal. Smitsonian contribution to zoology, 336th edn. Smitsonian Institution Press, Washington
Sunquist M (2010) What is a tiger? Ecology and behavior. In: Tilson R, Nyhus R (eds) Tigers of the world the science, politics and conservation of Panthera tigris, 2nd ed. Academic Press, London, Chap. 1, pp 16–34
Sunquist M, Karanth KU, Sunquist F (1999) Ecology, behaviour and resilience of the tiger and its conservation needs. In: Seindensticker J, Christie S, Jackson P (eds) Riding the tiger: tiger conservation in human dominated landscapes. Cambridge University Press, UK, Australia, New York
Wilensky U (1999) NetLogo. Center for Connected Learning and Computer-Based Modeling Northwestern University, Evanston, IL. NetLogo 4.1RC3 eds. http://ccl.northwestern.edu/netlogo/
Acknowledgments
We are very grateful for the support provided by the Gadjah Mada University, Gesellschaft für TU Dresden and Deutscher Akademischer Austauschdienst (DAAD / the German Academic Exchange Service) that provided funds for field work and Balai Taman Nasional Tesso Nilo for issuing us with a research permit. Furthermore, we thank Conservation International for providing travel support to present this article on the World Agroforestry Congress in Kenya. We also thank Karmila Parakkasi and Sunarto from WWF-Riau for providing land covers maps and satellite images and for their support during field work, Arief Wijaya for providing assistantship of GIS work, Yustina and Eggy for assisting the field work. We thank also for comments given by editors, three anonymous reviewers, and Nabiul Khan. Finally, we thank Janine Murphy and Sarah Gwillym-Margianto for proof-reading an earlier version of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Appendix 1
Rights and permissions
About this article
Cite this article
Imron, M.A., Herzog, S. & Berger, U. The Influence of Agroforestry and Other Land-Use Types on the Persistence of a Sumatran Tiger (Panthera tigris sumatrae) Population: An Individual-Based Model Approach. Environmental Management 48, 276–288 (2011). https://doi.org/10.1007/s00267-010-9577-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00267-010-9577-0