Abstract.
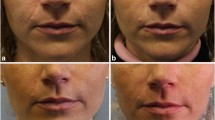
A new tissue augmentation product, made from hyaluronic acid, was clinically evaluated at three clinics in accordance with the new directive, EN 540, for medical implants. One hundred patients were fully assessed following treatments in 285 locations. The treatment was completed when the skin was levelled following one to two injections. At 6 months follow-up of all patients and at 12 months follow-up of a randomized group of the patients all showed that close to 60% of the effect was still there. No serious or permanent adverse events were noted.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Olenius, M. The First Clinical Study Using a New Biodegradable Implant for the Treatment of Lips, Wrinkles, and Folds. Aesth. Plast. Surg. 22, 97–101 (1998). https://doi.org/10.1007/s002669900172
Published:
Issue Date:
DOI: https://doi.org/10.1007/s002669900172