Abstract
Improved understanding of the anatomy and physiology of the aging face has laid the foundation for adopting an earlier and more comprehensive approach to facial rejuvenation, shifting the focus from individual wrinkle treatment and lift procedures to a holistic paradigm that considers the entire face and its structural framework. This article presents an overview of a comprehensive method to address facial aging. The key components to the reported strategy for improving facial cosmesis include, in addition to augmentation of volume loss, protection with sunscreens and antioxidants; promotion of epidermal cell turnover with techniques such as superficial chemical peels; microlaser peels and microdermabrasion; collagen stimulation and remodeling via light, ultrasound, or radiofrequency (RF)-based methods; and muscle control with botulinum toxin. For the treatment of wrinkles and for the augmentation of pan-facial dermal lipoatrophy, several types of fillers and volumizers including hyaluronic acid (HA), autologous fat, and calcium hydroxylapatite (CaHA) or injectable poly-l-lactic acid (PLLA) are available. A novel bimodal, trivector technique to restore structural facial volume loss that combines supraperiosteal depot injections of volume-depleted fat pads and dermal/subcutaneous injections for panfacial lipoatrophy with PLLA is presented. The combination of treatments with fillers; toxins; light-, sound-, and RF-based technologies; and surgical procedures may help to forestall the facial aging process and provide more natural results than are possible with any of these techniques alone.
Level of Evidence V
This journal requires that authors assign a level of evidence to each article. For a full description of these Evidence-Based Medicine ratings, please refer to the Table of Contents or the online Instructions to Authors www.springer.com/00266.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Increased understanding of the complex and dynamic changes that occur with facial aging has laid the foundation for adopting an earlier and more holistic interventional approach to facial rejuvenation. Although preventive strategies are commonplace in many medical arenas, facial aging prophylaxis, particularly the early treatment of aging signs before they progress, is a relatively new concept in cosmetic dermatology that attempts to delay or prevent surgical intervention.
We currently understand that age-related changes occur in all tissues and structures of the face and that changes in one area may cause a cascading effect in adjacent areas [27]. Additionally, there is growing recognition of the complex interplay between craniofacial structural remodeling, volume loss, and age-related skin changes. Consequently, methods that consider the entire face and its structural framework by using multiple available technologies and treatments can provide a more holistic approach and may forestall further deterioration.
This article provides an overview of age-related changes associated with the skin and the underlying structural framework of the face, and also describes the devices and techniques that can be used both prophylactically and for the reversal of facial aging, including a novel bimodal, trivector technique for volume replacement (Table 1).
The Aging Face
Facial aging is characterized by a myriad of features including upper eyelid dermatochalasis, dyschromia, wrinkles, protruding eyelid fat, brow ptosis, and volume changes [41]. These features result from a complex interplay of anatomic, histologic, and physiologic changes that affect the bone, ligaments, fat, and skin in the upper, middle, and lower portions of the face (Fig. 1) [16, 25].
Age-related changes in facial tissues. NLFs nasolabial folds. Adapted from [16]
Alterations in Volume and Structural Support
Volume loss in the aging face results from both craniofacial remodeling and atrophy of superficial and deep fat [25]. Recent studies have highlighted the significant loss of craniofacial skeleton volume [52, 63], and a recent review of the craniofacial remodeling literature found consistent age-related contour changes of the orbit, anterior maxilla, and mandibular body, as well as decreased dimensions of the glabellar, pyriform, and maxillary angles [62].
Because the bony elements of the face provide the framework on which the soft tissues rest [64], craniofacial changes have a substantial impact on facial features and overall aesthetics. For instance, the increase in mandibular angle observed with aging may cause blunting or loss of definition of the lower border of the face [64], and an increase in the pyriform aperture can lead to an appearance of nose elongation and a drooping nasal tip [63]. Additionally, midfacial bone loss may exacerbate the nasolabial fold by reorienting the malar fat pad medially and inferiorly [63].
In addition to craniofacial remodeling, growing evidence shows that remodeling of facial fat occurs with time and contributes to the appearance of the aging face [34, 57]. In the youthful face, deep and superficial fat are balanced, creating an even distribution of facial fullness [17]. As the face ages, redistribution and descent of facial fat pads contribute to the older appearance (Fig. 1) [34, 57].
Groundbreaking studies have recently elucidated the partitioning of superficial and deep facial fat into discrete anatomic compartments, or fat pads, confirming that facial fat is not one confluent mass [54, 56, 57]. Deep fat is the foundation for overlying subcutaneous fat compartments [57], and age-related changes in the volume and positioning of these compartments can lead to predictable changes in the appearance of the face [54]. For example, volume loss in the deep medial cheek fat pads diminishes anterior projection of tissue, leading to “pseudoptosis” and the appearance of nasolabial folds [54]. This redistribution of facial fat causes atrophy in certain areas and hypertrophy in others [17]. Atrophy occurs in the periorbital, forehead, buccal, temporal, and perioral areas, leading to sagging due to the relative excess of remaining skin [17].
Redistribution or loss of volume in the temporal, suborbital, and buccal fat pads leads to age-related changes in multiple facial areas including the cheeks, temples, and nasolabial folds. Superficial and deep volume loss can lead to the manifestation of wrinkles, folds, and lines on the surface of the aging face, and methods aimed at restoring the lost volume in both the superficial and deep structural supportive features may provide benefit in impeding the facial aging process.
Alterations in the Skin
Although the cellular and molecular mechanisms involved in skin aging are complex and the processes are influenced by a range of both intrinsic and extrinsic factors, sun exposure (photoaging) has been identified as a key cause, with degradation of the collagen matrix as a key mechanism.
The importance of maintaining collagen stability was recently reviewed by Fisher et al. [21]. Briefly, as the skin ages, the structural integrity of the dermis is impaired due to enzymatic degradation of the collagen by matrix metalloproteinases [21]. The resulting fragmented collagen can no longer provide the structural support needed for fibroblast attachment, causing the fibroblasts to collapse [21]. The balance between collagen synthesis and production of collagen-degrading enzymes shifts toward degradation, and the aging process continues to advance in a sustained cycle of collagen loss and fibroblast collapse [21]. The described process results clinically in dynamic and static wrinkles, dermal atrophy, and elastosis [28]. Further signs of aging are dyschromic manifestations such as senile lentigines, patchy hyperpigmentation, telangiectasia, and erythema, as well as rough skin and enlarged pores [39, 73].
Approaches to Facial Aging Prophylaxis
Given the role of collagen degeneration, fibroblast collapse, and volume loss in the progression of the aging face, an approach to prophylaxis should seek to protect against environmental exposure, stimulate collagen synthesis and cell turnover, and address dyschromia, wrinkles, and both superficial and deep structural volume loss before they become significant. Hence, the key elements of our approach to facial aging treatment are protection, promotion of cell turnover, collagen stimulation/remodeling, muscle control, and volume replacement. To that end, an abundance of treatments are available, with the choice of individual and combination therapies influenced by patient considerations, desired clinical effect, ease of use, reliability, safety, and cost. These options are summarized in Fig. 2 and described in the following sections.
Topical Protection and Stimulation
Protection against the cumulative effects of ultraviolet (UV) exposure and other extrinsic factors is paramount to preserving the appearance of youthful skin. Stable broad-spectrum sunscreens are a mainstay for protecting the skin from UVA and UVB radiation. In addition, topical formulations containing antioxidants such as vitamins C and E [12], resveratrol [1], idebenone [45], coffeeberry [6], or superoxide dismutase [42] may help to protect the skin by scavenging free radicals. They can provide important benefits for the skin, including protection against UV-induced erythema, hyperpigmentation, and photoaging. Cell regulatory molecules such as retinoids have been shown to inhibit the induction of matrix metalloproteinases and may therefore protect against collagen fiber degradation [20–22]. Topically applied actives such as aminoguanidine may protect the skin from the formation of age-related advanced glycation end products [50] that lead to discoloration [46] and collagen damage [49, 50]. Additionally, topical peptides have shown benefit in reducing facial lines and wrinkles, possibly by stimulating collagen production [38, 55].
Muscle Control
Botulinum toxin type A is an established treatment for the lines, furrows, soft tissue malposition, and degeneration of facial shape associated with facial aging by controlling hyperdynamic muscle activity through local chemodenervation [19]. Furthermore, botulinum toxin type A also may aid in facial line prevention if initiated at an earlier age and if regular treatments are sustained [7, 14]. The toxin prevents the release of the neurotransmitter acetylcholine at the peripheral nerve endings, generating a temporary denervation and relaxation of muscles for 16–24 weeks [29].
Cell Turnover
Epidermal turnover is essential for resurfacing and improving skin pigmentation and textural changes. Chemical peels are effective resurfacing tools that can treat superficial to moderate rhytides and dyschromia by decreasing corneocyte adhesion [36]. Microdermabrasion is an effective alternative, particularly for patients who have not yet experienced profound degenerative changes of aging [37]. This technique improves the appearance of fine wrinkles and dilated pores while increasing epidermal thickness and improving elastosis [37].
Laser micropeeling is a new method for superficial ablation up to 50 μm. A recent randomized clinical study based on a variable-pulse erbium:yttrium–aluminium-garnet (Er:YAG) laser with a wavelength of 2,940 nm has demonstrated a reduction in dyschromia and only minimal postprocedure erythema [66]. The benefits of this method are its tolerability and high patient satisfaction with limited downtime [15].
Collagen Stimulation and Remodeling
Dermal changes and collagen loss can be addressed via a number of devices based on a range of physical methods including light, radiofrequency (RF), and more recently, ultrasound. Additional devices include the combined use of these technologies.
Whereas devices for collagen stimulation target the fibrous network of the papillary dermis to correct collagen loss as well as glycosaminoglycan and elastin disorganization, devices for skin tightening involve heat-induced structural changes in the deep dermis and subcutis that cause the fibers to contract while retaining tensile strength. The heat-modified tissues undergo remodeling via fibroplasia, extracellular matrix production, and new collagen deposition, leading to skin tightening and gradual dermal thickening [59].
Collagen-stimulating devices are mainly premised on laser-based platforms and can be divided further into ablative and nonablative as well as fractional and nonfractional categories [2]. With fractional laser devices, the fractionated laser light causes thermal tissue damage and necrosis in targeted microscopic treatment zones while sparing the surrounding tissue [9]. Healing begins within 1 week, and replacement of the epithelial cells as well as expulsion of necrotic tissue ensues with a minimal inflammatory reaction [9].
Ablative fractional lasers have a higher affinity for water and ablate micro-columns of tissue including the stratum corneum, whereas nonablative fractional lasers result in column-like denaturation of the dermis and epidermis and an intact stratum corneum [10].
Good results in collagen stimulation can be expected with the 1,064- [68], 1,320- [67], and 1,440-nm neodymium (Nd):YAG lasers or the 1,410- and 1,550-nm Er:glass fiber lasers [60]. Multiple clinical studies show improvement in rhytides when high-power micropulses, high repetition, and large spot size are used to obtain bulk heating.
To treat deeper wrinkles and skin laxity, light-based devices need wavelengths that are strongly absorbed by tissue water, such as the 10,600-nm carbon dioxide (CO2), the 2,940-nm Er:YAG, or the 2,790-nm erbium:yttrium scandium gallium garnet (Er:YSGG) laser [48]. These have the ability to penetrate deep into the dermis and thermally ablate controlled layers of tissue [48]. Compared with CO2 lasers, Er:YAG [4] and Er:YSGG lasers have less severe side effects and faster overall healing times due to their more precise ablative quality with minimal thermal damage to the surrounding tissues.
Ablative laser rejuvenation often results in postoperative hyper- and hypopigmentation as well as scars [40]. Therefore, fractional, partly ablative resurfacing has become more popular because of its shorter recovery time and lower risk for these adverse events.
Unipolar, bipolar, and multipolar RF devices deliver electrical energy directly to the skin, causing heat due to impedance of the tissue, which results in collagen contraction and thereby immediate skin tightening [5, 59]. Subsequent remodeling, reorientation of collagen bundles, and formation of new collagen due to wound healing [74] are achieved during the months after the treatment [11]. Known complications include discomfort, persistent erythema, nodularity/panniculitis, burns, and dermal adhesions.
Electromagnetic fields also have been shown to stimulate collagen synthesis [65], and a new device combines multipolar RF with magnetic pulses. The benefits of this platform are lower overall energy, increased safety, and no need for pretreatment preparation and topical cooling agents. Although experience with this approach is limited, results from a small pilot study suggest that RF-based technologies may be safely combined with volume replacement.
Another new device uses focused intense ultrasound energy to create small inverted cone-shaped zones of thermal coagulation within the ultrasound beam, leaving the surrounding tissue unaffected [3, 31]. The ultrasound field vibrates tissues, causing friction between molecules and secondary generation of heat, resulting in histologic evidence of collagen denaturation in the dermis and sparing the overlying epidermal layer [3, 31]. A study investigating the safety and efficacy of this device for facial skin tightening found that a single treatment of the forehead produced an average brow height elevation of nearly 2 mm and that it is a safe system with very little discomfort [3]. Adverse events include transient redness and swelling immediately after treatment and, more rarely, elevated white linear wheals [3].
Volume Replacement
As described earlier, volume is lost both in the dermis and in the deep structural and supportive elements of the face. Consequently, it is important to address both of these layers in a holistic approach to facial aging. Strategies for augmenting lost volume in the soft tissues include the use of injectable fillers such as hyaluronic acid (HA) derivatives, autologous fat transfer, or injectable biostimulatory fillers such as calcium hydroxylapatite (CaHA), injectable poly-l-lactic acid (PLLA), and polymethyl methacrylate (PMMA).
Hyaluronic acid is glycosaminoglycan and a natural compound of the extracellular matrix. Clinicians can differentiate HA fillers by their mechanical properties and duration as a function of their cross-linking density [18].The resulting strength of the gel mainly determines the clinical application of the filler. Strong gels have a high lifting capacity and can be used for deep wrinkles and folds, whereas weak gels have a low viscosity and can be used for very fine wrinkles and mesotherapeutic approaches.
As a nonantigenic, nonirritating, nontoxic agent, CaHA is identical to the inorganic constituent of teeth and bone in its chemical composition. After injection, the small CaHA particles are fixed in place via thin connective tissue without causing a reaction in the surrounding area, evidence of migration, or evidence of heterotopic bone growth [69]. After 18 months, the microspheres begin to degrade into metabolites consisting of calcium and phosphate ions [43]. In addition to use in facial augmentation, CaHA is particularly suitable for the treatment of volume loss in the hands because it produces a smooth and natural-looking result with only some temporary swelling as a side effect [30].
Poly-l-lactic acid is a biocompatible, biodegradable, immunologically inert, semipermanent soft tissue filler. Placed into the reticular dermis and subcutaneous tissue planes, where it lasts up to 2 years or more, PLLA stimulates local fibroblasts to promote neocollagenesis [32]. Its efficacy has been shown for the treatment of facial and nonfacial areas including the hands, neck, and chest [44, 51]. Common adverse events associated with PLLA use are ecchymoses, edema, pain, pruritus, inflammation, nodules, and hematomas [53].
Whereas HA, CaHA, and PLLA are temporary dermal fillers, PMMA is a nonresorbable, permanent filler. Composed of 20 % PMMA microspheres in 3.5 % bovine collagen, PMMA is FDA approved in the United States for the treatment of nasolabial folds. Off-label use of PMMA has been reported for several other facial lines including glabellar frown lines, upper lip lines, and mouth corners [35]. Although the collagen component is degraded over time, the PMMA microspheres are permanent and cannot be reversed without excision [33]. Common side effects are contour irregularity or nodules shortly after injection, as well as granulomatous reactions months or years after injection [13].
Consensus guidelines and details regarding the selection and application of these products can be found in recently published reviews [23, 24]. Whereas HAs, CaHA, and PMMA are primarily used for wrinkle and local volume augmentation, injectable PLLA is used for panfacial volume augmentation and general rejuvenation, although it is not well suited to address fine rhytides or lip volume. Additionally, HAs, CaHA, and PLLA are appropriate for nonfacial usage, including rejuvenation of the hands and chest.
To address volume loss in craniofacial support, alloplastic implants have traditionally been used but are not free of risk because bleeding, hematoma, seroma, fistula, pain, and persistent inflammation can occur. Moreover, infections appear in 1–3.8 % of patients receiving silicone implants [8, 58].
Nevertheless, the biostimulatory filler, injectable PLLA, has recently been applied to augment craniofacial support through the use of supraperiosteal injections [26, 72]. Injectable PLLA is a biocompatible volumizer well suited for panfacial volume restoration, with duration of up to 36 months and natural-appearing results [61, 70]. Injectable PLLA replaces lost collagen by increasing fibroblastic activity and stimulating collagen synthesis, which thereby begins gradually to restore volume, leading to the refinement and smoothing of shallow to deep facial wrinkles and folds [32, 71].
Injectable PLLA is versatile and can be used in multiple ways. Deep dermal/subcutaneous injections can be used to strengthen the dermis and address dermal lipoatrophy. It also can be used to mimic lost fat pad volume, or if placed supraperiosteally, it can be used to simulate bone volume [26]. Based on these versatile properties of injectable PLLA to correct the underlying causes of facial aging such as collagen loss and both superficial and deep structural volume depletion, we developed a bimodal technique for restoring facial volume that uses supraperiosteal injections to address depletion of the deep structural and supportive tissues of the face together with deep dermal/subcutaneous injections to treat dermal lipoatrophy of the more superficial tissues. This novel approach, described in the following sections, uses a trivector injection method for supraperiosteal product deposition.
The Bimodal Technique for Panfacial Volume Restoration
The first step to the bimodal technique involves supraperiosteal injections of injectable PLLA for the purpose of restoring lost volume in the deep structural layers of the upper, middle, and lower face to provide a structural platform. The supraperiosteal placement is used to mimic the volume lost in the supportive temporal, preauricular lateral cheek, mid-cheek, and mandibular fat pads and to address musculoskeletal atrophy.
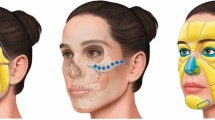
The novel technique minimizes patient discomfort through the use of three injection access points: (1) the temporal fossa to address forehead atrophy and brow ptosis, (2) the midface to treat the tear-trough, infranasal, and malar fat pad areas, and (3) the jaw line to address the mandibular angle, maxilla, and marionette lines (Fig. 3) [56].
Trivector injection approach for structural volume restoration [56]. Three injection access points are used to minimize patient discomfort during supraperiosteal placement of injectable poly-l-lactic acid (PLLA) for restoration of lost volume in the deep structural layers of the upper, middle, and lower face. Green-shaded areas indicate subcutaneous fat compartments. Adapted with permission from [56]
The technique is termed the “trivector approach” to structural volume restoration in the face. The amount of product may vary for the depot injections, but in general, a small face with moderate volume loss requires a 1–2-ml temple, a 2–4-ml cheek, and a 1–2-ml perioral area [26]. The total amount of product can be divided into aliquots for the depot injections [26]. In fanning from an injection point, it is important to avoid multiple deposits at the apex of the fan [27].
Once the structural platform has been reinforced using the trivector approach, areas of dermal lipoatrophy are treated via deep dermal/subcutaneous injections in the next step of the bimodal technique. The goal of these injections is full customization of the volumetric filling in accordance with individualized desired outcomes and patient preferences.
The injections should be performed using a 27-gauge needle (0.5, 1.0, or 1.5 in.) at a 30–40° entry angle. A reflux maneuver to avoid blood vessels and a tunneling technique should be applied in a cross-hatch pattern in the lower face [26]. Injectable PLLA (0.1–0.2 ml per injection site) should be deposited at the subdermal junction, avoiding areas above hypermobile muscles. To ensure even product distribution, the injected area should be massaged after every two or three injections and at the end of treatment, and patients should be instructed to massage the injected areas for 5 min five times per day for 5 days after the procedure [27].
Augmentation of the temple area should use the periosteal, depot injection technique, and although injections around the orbits also use periosteal placement, they should not be attempted without sufficient training. In the lower face, combined supraperiosteal and subcutaneous injections are used via perioral injection for correction of the nasolabial folds and marionette lines, and injections to the malar eminence serve to restructure the fat pad. The malar augmentation technique involves placement immediately below the zygomatic bony prominence, directly above the periosteum over the maxillary bone, with linear threading and retrograde deposition of product.
Considerations of the Bimodal Technique
Hands-on examinations are helpful before beginning or continuing a treatment program and should take into consideration the totality of the age-related changes in three dimensions, including superficial changes as well as deeper volume loss. These evaluations should begin with recognition of regional anatomic landmarks including musculoskeletal structures and fat pads. Then a careful assessment should be made of the changes in skeletal width and height, bone loss, both deep and superficial fat loss, and overall tissue quality. Palpation of the soft tissue can be useful in provoking weaknesses and folds or creases, thereby aiding in identifying key areas for correction [26].
An understanding of the anatomic location and age-related changes of the facial fat compartments can be used as a “roadmap” to guide clinicians as they evaluate the need for deep support through deeply placed facial injectables [57]. A detailed map of the key areas to be addressed should then be used to guide both the deeper supraperiosteal injections and the more superficial subcutaneous injections.
Because injectable PLLA acts gradually by replacing lost collagen, multiple treatments are used to yield incremental, natural correction. The process follows the paradigm of treat, wait, and assess [27]. Therefore, the treatment sessions are spaced 4–6 weeks apart to prevent overcorrection. After the first two treatments, an optional third treatment may be needed, depending on the progress of the results and the individual’s desired degree of correction. Later treatment sessions may use less product than earlier treatment sessions. It is worth noting that to avoid overcorrection, the amount of product used at the initial treatment session is based primarily on the two-dimensional area to be addressed, with subsequent treatments used to gradually attain the ultimately desired degree of correction in the third dimension of depth or overall volume [27].
As an added benefit with the use of a facial aging treatment method, improvements in skin quality have been noted, in our practices and by others, after injections of PLLA [47]. A possible explanation for this phenomenon, which currently is under investigation, could be the collagen stimulation and dermal thickening that occur with this treatment.
In preparation for use, the product should be reconstituted and stored at room temperature at least 24 h before treatment, with gentle agitation immediately before injection to ensure even product distribution. Each vial is reconstituted to a final volume of 9 ml with sterile water plus 1–2 % lidocaine with or without epinephrine, if desired [27].
Figure 4 shows an example of global facial rejuvenation with injectable PLLA. Combining injectable PLLA treatment with other methods such as laser and RF devices or botulinum toxin injections can yield very natural-appearing results (Fig. 5). Our clinical experience has shown the following between-treatment intervals to be necessary after injectable PLLA treatment: 48 h before IPL treatment; 1 week before IR, RF, or ultrasound treatments; and 1 month before fractional laser treatment.
Global facial rejuvenation with injectable poly-l-lactic acid (PLLA). a, b A 49-year-old woman treated twice with one 6 ml vial of injectable PLLA spaced 4 weeks apart. a Before treatment. b After treatment. c–e A 66-year-old woman. c Before treatment. d After four treatments with injectable PLLA spaced 4 weeks apart. Two 6-ml vials of injectable PLLA were used in the first and second treatments. One 6-ml vial of injectable PLLA was used in the third, fourth, and fifth treatments. e View 3 months after the fifth treatment with injectable PLLA. Photographs courtesy of Dr. N. Sadick
Treatment with unipolar radiofrequency (RF) and injectable poly-l-lactic acid (PLLA) for a 52-year-old woman. The left panel shows the appearance of facial tissue before treatment. The right photo was taken 8 weeks after RF treatment. The patient received three treatments with one 6-ml vial of injectable PLLA per treatment. All injections were periosteal. The treatments were spaced 4 weeks apart. The patient also was treated with unipolar RF treatment (1.5 cm2, 200 pulses total) 2 weeks after the last injectable PLLA treatment. Photographs courtesy of Dr. N. Sadick
The described bimodal technique for panfacial volume restoration makes injectable PLLA one of the most useful tools in reversing the increased angularity and the loss of aesthetically pleasing ratios, smoothness, softness, and plumpness in the aging face. The keys to treatment success include supraperiosteal and deep dermal/subcutaneous injections, recognition of regional anatomic landmarks including fat pad compartments as guides for correction, minimized number of injections via the trivector approach, immediate and postprocedure massage, individualized treatment, and adequate dilution and incubation of injectable PLLA to ensure uniform product suspension and distribution.
Individualized Treatment and Earlier Intervention
Age-related changes of the face include the totality of the alterations in the superficial and deeper structural features. Because these changes vary from individual to individual, so must the approach in the choice of which age-related changes should be addressed and when as well as which techniques should be used and when. Although nearly any intervention may be effective in young patients with early morphologic changes, an approach that makes use of multiple treatment methods is likely to be necessary for older patients, in whom advanced changes have already occurred. For these older patients, varied treatment techniques in combination with surgical approaches can be very beneficial for an optimal outcome. Good synergistic effects can be seen by combining the discussed noninvasive and minimally invasive treatments with many surgical procedures including blepharoplasty, rhytidectomy, neck-lift, or brow-lift.
It is assumed that by beginning these less-invasive programs earlier, the facial aging process can be slowed down, but this remains to be evaluated, and prospective studies are needed.
Conclusion
Understanding the dynamic changes associated with the anatomy and physiology of facial aging as well as appreciating the degree to which these various changes have occurred in an individual patient allows for a customized treatment plan that combines multiple treatment methods such as light, RF- or ultrasound-based devices, injections of botulinum toxin, and volume augmentation with a filler such as HA, CaHA, or PLLA. A promising approach for volume restoration is the bimodal technique presented in this article that uses both deep periosteal injections and more superficial subcutaneous injections.
The discussed overall approach to facial aging treatment can lead to natural-appearing results, minimal downtime, a high level of patient satisfaction, increased cost effectiveness, and safety with minimal complications. A combination approach with invasive surgical procedures can enhance the effect and, in many cases, may be the best solution for the aging face.
References
Afaq F, Mukhtar H (2006) Botanical antioxidants in the prevention of photocarcinogenesis and photoaging. Exp Dermatol 15:678–684
Alam M, Dover JS, Arndt KA (2011) To ablate or not: a proposal regarding nomenclature. J Am Acad Dermatol 64:1170–1174
Alam M, White LE, Martin N, Witherspoon J, Yoo S, West DP (2010) Ultrasound tightening of facial and neck skin: a rater-blinded prospective cohort study. J Am Acad Dermatol 62:262–269
Alexiades-Armenakas MR, Dover JS, Arndt KA (2008) The spectrum of laser skin resurfacing: nonablative, fractional, and ablative laser resurfacing. J Am Acad Dermatol 58:719–737
Atiyeh BS, Dibo SA (2009) Nonsurgical nonablative treatment of aging skin: radiofrequency technologies between aggressive marketing and evidence-based efficacy. Aesthetic Plast Surg 33:283–294
Baumann L, Woolery-Lloyd H, Friedman A (2009) “Natural” ingredients in cosmetic dermatology. J Drugs Dermatol 8(6 Suppl):s5–s9
Binder WJ (2006) Long-term effects of botulinum toxin type A (Botox) on facial lines: a comparison in identical twins. Arch Facial Plast Surg 8:426–431
Binder WJ (2011) Facial rejuvenation and volumization using implants. Facial Plast Surg 27:86–97
Bodendorf MO, Grunewald S, Wetzig T, Simon JC, Paasch U (2009) Fractional laser skin therapy. J Dtsch Dermatol Ges 7:301–308
Bogdan Allemann I, Kaufman J (2010) Fractional photothermolysis: an update. Lasers Med Sci 25:137–144
Bogle MA, Ubelhoer N, Weiss RA, Mayoral F, Kaminer MS (2007) Evaluation of the multiple pass, low fluence algorithm for radiofrequency tightening of the lower face. Lasers Surg Med 39:210–217
Burke KE (2007) Interaction of vitamins C and E as better cosmeceuticals. Dermatol Ther 20:314–321
Carruthers A, Carruthers JD (2005) Polymethylmethacrylate microspheres/collagen as a tissue augmenting agent: personal experience over 5 years. Dermatol Surg 31:1561–1564
Carruthers JD, Glogau RG, Blitzer A (2008) Advances in facial rejuvenation: botulinum toxin type a, hyaluronic acid dermal fillers, and combination therapies—consensus recommendations. Plast Reconstr Surg 121(5 Suppl):5S–30S
Christian MM (2003) Microresurfacing using the variable-pulse erbium: YAG laser: a comparison of the 0.5- and 4-ms pulse durations. Dermatol Surg 29:605–611
Coleman SR, Grover R (2006) The anatomy of the aging face: volume loss and changes in 3-dimensional topography. Aesthet Surg J 26(1 Suppl):S4–S9
Donofrio LM (2000) Fat distribution: a morphologic study of the aging face. Dermatol Surg 26:1107–1112
Edsman K, Nord LI, Ohrlund A, Larkner H, Kenne AH (2012) Gel properties of hyaluronic acid dermal fillers. Dermatol Surg 38:1170–1179
Fagien S (2003) Botulinum toxin type A for facial aesthetic enhancement: role in facial shaping. Plast Reconstr Surg 112:6S–18S
Fisher GJ, Datta SC, Talwar HS, Wang ZQ, Varani J, Kang S, Voorhees JJ (1996) Molecular basis of sun-induced premature skin ageing and retinoid antagonism. Nature 379:335–339
Fisher GJ, Varani J, Voorhees JJ (2008) Looking older: fibroblast collapse and therapeutic implications. Arch Dermatol 144:666–672
Fisher GJ, Wang ZQ, Datta SC, Varani J, Kang S, Voorhees JJ (1997) Pathophysiology of premature skin aging induced by ultraviolet light. N Engl J Med 337:1419–1428
Fitzgerald R, Graivier MH, Kane M, Lorenc ZP, Vleggaar D, Werschler WP, Kenkel JM (2010) Appropriate selection and application of nonsurgical facial rejuvenation agents and procedures: panel consensus recommendations. Aesthet Surg J 30(Suppl):36S–45S
Fitzgerald R, Graivier MH, Kane M, Lorenc ZP, Vleggaar D, Werschler WP, Kenkel JM (2010) Nonsurgical modalities to treat the aging face. Aesthet Surg J 30(Suppl):31S–35S
Fitzgerald R, Graivier MH, Kane M, Lorenc ZP, Vleggaar D, Werschler WP, Kenkel JM (2010) Update on facial aging. Aesthet Surg J 30(Suppl):11S–24S
Fitzgerald R, Vleggaar D (2009) Using poly-l-lactic acid (PLLA) to mimic volume in multiple tissue layers. J Drugs Dermatol 8(10 Suppl):s5–s14
Fitzgerald R, Vleggaar D (2011) Facial volume restoration of the aging face with poly-l-lactic acid. Dermatol Ther 24:2–27
Friedman O (2005) Changes associated with the aging face. Facial Plast Surg Clin North Am 13:371–380
Gadhia K, Walmsley AD (2009) Facial aesthetics: Is botulinum toxin treatment effective and safe? A systematic review of randomised controlled trials. Br Dent J 207:E7–E9
Gargasz SS, Carbone MC (2010) Hand rejuvenation using Radiesse. Plast Reconstr Surg 125:259e–260e
Gliklich RE, White WM, Slayton MH, Barthe PG, Makin IR (2007) Clinical pilot study of intense ultrasound therapy to deep dermal facial skin and subcutaneous tissues. Arch Facial Plast Surg 9:88–95
Gogolewski S, Jovanovic M, Perren SM, Dillon JG, Hughes MK (1993) Tissue response and in vivo degradation of selected polyhydroxyacids: Polylactides (PLA), poly(3-hydroxybutyrate) (PHB), and poly(3-hydroxybutyrate-co-3-hydroxyvalerate) (PHB/VA). J Biomed Mater Res 27:1135–1148
Goldberg DJ (2009) Breakthroughs in US dermal fillers for facial soft tissue augmentation. J Cosmet Laser Ther 11:240–247
Gosain AK, Klein MH, Sudhakar PV, Prost RW (2005) A volumetric analysis of soft tissue changes in the aging midface using high-resolution MRI: implications for facial rejuvenation. Plast Reconstr Surg 115:1143–1152
Haneke E (2004) Polymethyl methacrylate microspheres in collagen. Semin Cutan Med Surg 23:227–232
Herbig K, Trussler AP, Khosla RK, Rohrich RJ (2009) Combination Jessner’s solution and trichloroacetic acid chemical peel: technique and outcomes. Plast Reconstr Surg 124:955–964
Hernandez-Perez E, Ibiett EV (2001) Gross and microscopic findings in patients undergoing microdermabrasion for facial rejuvenation. Dermatol Surg 27:637–640
Kaczvinsky JR, Griffiths CE, Schnicker MS, Li J (2009) Efficacy of anti-aging products for periorbital wrinkles as measured by 3-D imaging. J Cosmet Dermatol 8:228–233
Lagarde JM, Rouvrais C, Black D (2005) Topography and anisotropy of the skin surface with ageing. Skin Res Technol 11:110–119
Laubach HJ, Tannous Z, Anderson RR, Manstein D (2006) Skin responses to fractional photothermolysis. Lasers Surg Med 38:142–149
Lewis CD, Perry JD (2009) A paradigm shift: volume augmentation or “inflation” to obtain optimal cosmetic results. Curr Opin Ophthalmol 20:389–394
Lods LM, Dres C, Johnson C, Scholz DB, Brooks GJ (2000) The future of enzymes in cosmetics. Int J Cosmet Sci 22:85–94
Marmur ES, Phelps R, Goldberg DJ (2004) Clinical, histologic, and electron microscopic findings after injection of a calcium hydroxylapatite filler. J Cosmet Laser Ther 6:223–226
Mazzuco R, Hexsel D (2009) Poly-l-lactic acid for neck and chest rejuvenation. Dermatol Surg 35:1228–1237
McDaniel DH, Neudecker BA, DiNardo JC, Lewis JA, Maibach HI (2005) Clinical efficacy assessment in photodamaged skin of 0.5 % and 1.0 % idebenone. J Cosmet Dermatol 4:167–173
Ogura Y, Kuwahara T, Akiyama M, Tajima S, Hattori K, Okamoto K, Okawa S, Yamada Y, Tagami H, Takahashi M, Hirao T (2011) Dermal carbonyl modification is related to the yellowish color change of photo-aged Japanese facial skin. J Dermatol Sci 64:45–52
Onesti MG, Troccola A, Scuderi N (2009) Volumetric correction using poly-l-lactic acid in facial asymmetry: Parry Romberg syndrome and scleroderma. Dermatol Surg 35:1368–1375
Paasch U, Haedersdal M (2011) Laser systems for ablative fractional resurfacing. Expert Rev Med Devices 8:67–83
Pageon H (2010) Reaction of glycation and human skin: the effects on the skin and its components, reconstructed skin as a model. Pathol Biol Paris 58:226–231
Pageon H, Techer MP, Asselineau D (2008) Reconstructed skin modified by glycation of the dermal equivalent as a model for skin aging and its potential use to evaluate anti-glycation molecules. Exp Gerontol 43:584–588
Palm MD, Woodhall KE, Butterwick KJ, Goldman MP (2010) Cosmetic use of poly-l-lactic acid: a retrospective study of 130 patients. Dermatol Surg 36:161–170
Pecora NG, Baccetti T, McNamara JA Jr (2008) The aging craniofacial complex: a longitudinal cephalometric study from late adolescence to late adulthood. Am J Orthod Dentofacial Orthop 134:496–505
Peterson JD, Goldman MP (2011) Rejuvenation of the aging chest: a review and our experience. Dermatol Surg 37:555–571
Reece EM, Pessa JE, Rohrich RJ (2008) The mandibular septum: anatomical observations of the jowls in aging: Implications for facial rejuvenation. Plast Reconstr Surg 121:1414–1420
Robinson LR, Fitzgerald NC, Doughty DG, Dawes NC, Berge CA, Bissett DL (2005) Topical palmitoyl pentapeptide provides improvement in photoaged human facial skin. Int J Cosmet Sci 27:155–160
Rohrich RJ, Pessa JE (2007) The fat compartments of the face: anatomy and clinical implications for cosmetic surgery. Plast Reconstr Surg 119:2219–2227
Rohrich RJ, Pessa JE, Ristow B (2008) The youthful cheek and the deep medial fat compartment. Plast Reconstr Surg 121:2107–2112
Rubin JP, Yaremchuk MJ (1997) Complications and toxicities of implantable biomaterials used in facial reconstructive and aesthetic surgery: a comprehensive review of the literature. Plast Reconstr Surg 100:1336–1353
Sadick N (2008) Tissue tightening technologies: fact or fiction. Aesthet Surg J 28:180–188
Sadick NS, Smoller B (2009) A study examining the safety and efficacy of a fractional laser in the treatment of photodamage on the hands. J Cosmet Laser Ther 11:29–33
Salles AG, Lotierzo PH, Gimenez R, Camargo CP, Ferreira MC (2008) Evaluation of the poly-l-lactic acid implant for treatment of the nasolabial fold: 3-Year follow-up evaluation. Aesthetic Plast Surg 32:753–756
Sharabi SE, Hatef DA, Koshy JC, Hollier LH Jr, Yaremchuk MJ (2010) Mechanotransduction: the missing link in the facial aging puzzle? Aesthetic Plast Surg 34:603–611
Shaw RB Jr, Kahn DM (2007) Aging of the midface bony elements: a three-dimensional computed tomographic study. Plast Reconstr Surg 119:675–681
Shaw RB Jr, Katzel EB, Koltz PF, Kahn DM, Girotto JA, Langstein HN (2010) Aging of the mandible and its aesthetic implications. Plast Reconstr Surg 125:332–342
Soda A, Ikehara T, Kinouchi Y, Yoshizaki K (2008) Effect of exposure to an extremely low-frequency electromagnetic field on the cellular collagen with respect to signaling pathways in osteoblast-like cells. J Med Invest 55:267–278
Somoano B, Hantash BM, Fincher EF, Wu P, Gladstone HB (2011) The erbium micropeel: a prospective, randomized trial of the effects of two fluence settings on facial photoaging. J Drugs Dermatol 10:179–185
Trelles MA, Allones I, Luna R (2001) Facial rejuvenation with a nonablative 1,320-nm Nd:YAG laser: a preliminary clinical and histologic evaluation. Dermatol Surg 27:111–116
Trelles MA, Alvarez X, Martin-Vazquez MJ, Trelles O, Velez M, Levy JL, Allones I (2005) Assessment of the efficacy of nonablative long-pulsed 1,064-nm Nd:YAG laser treatment of wrinkles compared at 2, 4, and 6 months. Facial Plast Surg 21:145–153
Tzikas TL (2004) Evaluation of the radiance FN soft tissue filler for facial soft tissue augmentation. Arch Facial Plast Surg 6:234–239
Vleggaar D (2006) Soft tissue augmentation and the role of poly-l-lactic acid. Plast Reconstr Surg 118(3 Suppl):46S–54S
Vleggaar D, Bauer U (2004) Facial enhancement and the European experience with Sculptra (poly-l-lactic acid). J Drugs Dermatol 3:542–547
Vleggaar D, Fitzgerald R (2008) Dermatological implications of skeletal aging: a focus on supraperiosteal volumization for perioral rejuvenation. J Drugs Dermatol 7:209–220
Yaar M, Eller MS, Gilchrest BA (2002) Fifty years of skin aging. J Investig Dermatol Symp Proc 7:51–58
Zelickson BD, Kist D, Bernstein E, Brown DB, Ksenzenko S, Burns J, Kilmer S, Mehregan D, Pope K (2004) Histological and ultrastructural evaluation of the effects of a radiofrequency-based nonablative dermal remodeling device: a pilot study. Arch Dermatol 140:204–209
Acknowledgments
Editorial support for this article was provided by Peloton Advantage, LLC, funded by Sanofi–Aventis U.S. LLC, a Sanofi company. The authors were fully responsible for the content, editorial decisions, and opinions expressed in the current article. No author received an honorarium related to the development of this manuscript.
Disclosure
Neil Sadick is involved in workshops and serves as an injector trainer for Sanofi–Aventis U.S. S. Manhas-Bhutani has no competing financial interests or pertinent disclosures or known conflicts of interest to declare. N. Krueger has no competing financial interests or pertinent disclosures or known conflicts of interest to declare.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sadick, N.S., Manhas-Bhutani, S. & Krueger, N. A Novel Approach to Structural Facial Volume Replacement. Aesth Plast Surg 37, 266–276 (2013). https://doi.org/10.1007/s00266-012-0052-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00266-012-0052-6