Abstract
Negotiation models of biparental care predict that parents should partially compensate for a reduction in the level of care that their partner provides. A number of studies, mostly with monogamous birds, have tested this prediction by removing or manipulating the level of effort of one parent and then measuring the response of the other parent. In this study, we test this prediction with the burying beetle Nicrophorus orbicollis, which breeds on small vertebrate carcasses and provides biparental care to their young. We reduced the effort of either the male or female parent by attaching a weight to their elytron and evaluated the effect that their reduction in time spent providing care had on their partner’s level of effort during the carcass preparation stage of reproduction. After their partner was weighted, males increased the time spent preparing the carcass by 28 %. Females were already spending nearly 100 % of their time working and observed changes in effort were negligible. Our results show that parents directly monitor their partner’s level of effort in the carcass preparation stage of reproduction. This is the first handicapping study in an invertebrate species to provide support for the negotiation model of intersexual conflict over parental investment.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
In species that provide biparental care, conflict occurs regarding the level of investment that each parent provides. This occurs because the fitness of each parent is determined by the combined effort of both. However, the cost of care to each is a result of their effort alone (Parker 2006). Thus, any investment shifted from one parent to the other results in increased fitness for the former at the expense of the latter (Johnstone and Hinde 2006; Harrison et al. 2009). The result is sexual conflict over the level of parental investment each parent provides.
A game theoretical approach has been used to evaluate the outcome of sexual conflict. An early model, referred to as the sealed bid model (Houston and Davies 1985), assumes that parents do not exhibit plasticity in the amount of parental investment that they put into offspring as their partner’s level of investment changes. Instead, the level of effort that each parent contributes is independently and evolutionarily determined. This model predicts that biparental care is evolutionarily stable when a reduction in effort by one parent results in partial but not full compensation by the other parent. However, this response is realized only over an evolutionary time scale.
A second model, the negotiation model, proposes a system where parents can behaviorally adjust the level of parental investment that they provide depending on their partner’s level of investment (McNamara et al. 1999). The response of one parent to changes in the other parent’s effort is predicted to occur in real time with the degree of change varying from partial compensation to matching of investment levels.
Several studies have tested this prediction of the negotiation model by removing one parent or manipulating the level of effort of one parent and then measuring the response of the other parent. Most studies have used monogamous birds as a model system, and results of these studies vary. Removal experiments typically show partial compensation by the remaining parent (Hunt and Simmons 2002; Lendvai and Chastel 2008; Lendvai et al. 2009). Manipulation studies where the effort of one parent is reduced experimentally show results that range from partial compensation (Schwagmeyer et al. 2005; Lendvai et al. 2009) to matching (Hinde 2006), to full compensation (Sanz et al. 2000; Stoehr and Hill 2000), and to no compensation (Mazuc et al. 2003). In a study where parental effort was increased, the other parent responded, as predicted, by reducing its level of effort (Kosztolányi et al. 2009).
The effect of experimentally removing parents may be different from experimentally reducing their level of parental care. McNamara et al. (2003) show that, if the level of parental care is a result of a negotiation, an experimental reduction in effort by one parent should result in corresponding partial compensation. If one parent is experimentally removed, the remaining parent is predicted to respond as if the removed parent is providing zero effort. Supporting this prediction, a recent meta-analysis of intersexual conflict studies in birds found that the partners of birds that were removed increased feeding effort more than did the partners of birds that were manipulated to decrease their effort (Harrison et al. 2009).
Relatively few studies have been done using alternative model systems, especially insects (see Houston et al. 2005; Harrison et al. 2009). In the study presented here, we evaluate parental compensation in the burying beetle Nicrophorus orbicollis. Burying beetles use small vertebrate carcasses as a food resource for offspring, and both parents provide extensive parental care. Burying beetles share a number of similarities with birds, which make them a valuable model system to contrast with birds. Parental care in both groups involves distinct stages of reproduction: carcass preparation and larvae provisioning in burying beetles, and nest preparation, incubation, and nestling provisioning in birds. As a result, burying beetle parental investment behavior can be compared to parallel reproductive behaviors in birds.
The two stages of parental care in burying beetles include distinctly different behaviors and associated costs. In the carcass preparation stage, there are energetic and physiological costs associated with moving, manipulating, and preserving the carcass. For example, carcass preparation is costly and results in a significant reduction in survivorship and subsequent female fecundity and shortens adult lifespan (Cotter et al. 2011). The consequences of reduced parental care could be the complete loss of the carcass to another burying beetle or other scavenger. The larval provisioning stage involves carcass defense, regurgitation of digested carcass to the offspring, and carcass maintenance. As such, there are significant costs associated with nutritional limitation (Creighton et al. 2009). The consequences of reduced parental care during this phase could be reduced survivorship and/or growth of the offspring.
Previous tests of the negotiation model with N. orbicollis (Fetherston et al. 1994; Rauter and Moore 2004), Nicrophorus vespilloides (Smiseth et al. 2005), and Nicrophorus quadripunctatus (Suzuki and Nagano 2009) have focused on the larval provisioning stage and have primarily been parent removal experiments. The results of these tests have been a bit mixed but have generally found that males but not females compensate for mate removal or mate reduction in effort. In this study on N. orbicollis, we test for compensation during the carcass preparation stage in response to experimentally decreased parental investment. According to the sealed bid model, there should be no change in reproductive effort (because artificial handicaps have not been part of their evolutionary past), but according to the negotiation model, individuals should exhibit compensatory parental effort in response to decreased partner effort. In addition, we test for differences in patterns of parental care and compensation between sexes. We compare our results to mate removal and manipulation experiments done on burying beetles during the larvae provisioning stage and similar experiments done with birds.
Methods
Natural history of burying beetles
N. orbicollis parents use small vertebrate carcasses as a food resource for larvae (Scott 1998). Both parents prepare the carcass, which involves removing hair, rolling the carcass into a ball, applying anal and oral secretions that prevent decay, and burial of the prepared carcass. Both sexes are capable of performing all parental duties in the absence of the other (Wilson and Fudge 1984). The female lays eggs in the soil around the carcass, which hatch approximately 5–7 days after carcass preparation begins. After hatching, the larvae move to a small hole in the carcass made by the parents. Larvae feed directly from the carcass as well as by receiving regurgitated, partially digested carcass from both parents (Fetherston et al. 1990; Rauter and Moore 1999). The male typically abandons the brood 2–5 days after the larvae hatch, but the female remains with the brood for approximately 7 days, after which the larvae disperse into the soil to pupate (Trumbo 1991; Scott 1998). Both parents regulate brood size through filial cannibalism (Bartlett 1987; Scott and Traniello 1990; Trumbo 2006) producing a positive correlation between offspring number and carcass size (Bartlett 1987; Scott and Traniello 1990; Creighton 2005; Creighton et al. 2009).
Source of burying beetles
Burying beetles used in our experiments were captured in central Wisconsin (June 2006) using pitfall traps baited with aged chicken. Wild-caught pairs were placed on a 30-g mouse carcass and allowed to breed to generate the laboratory population. At eclosion, all first generation laboratory-reared beetles were placed individually in small plastic containers (15.6 × 11.6 × 6.7 cm) with ad libitum raw chicken liver and maintained on a 14:10 h light/dark cycle.
Experimental design
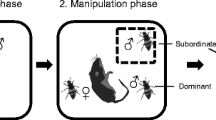
We randomly paired 21-day-old virgin males and females. Each beetle was weighed and their pronotum width was measured. Pairs were then placed on a 20-g (±1.0 g) mouse carcass in a small plastic container filled with approximately 6 cm of commercially purchased top soil. Each pair was randomly assigned to one of three treatment groups: impaired male, impaired female, or control.
Pairs that were assigned to either the impaired male or impaired female treatment groups had a small weight attached to the male or female, respectively. A small aluminum weight was cut down to approximately half the mass of the individual and was glued to the anterio-central portion of the beetle’s right elytron using gel super glue (cyanoacrolate) 6–12 h after a pair was placed on a carcass. Pairs assigned to the control treatment were handled briefly to control for the effect of handling independent of the weighting treatment. Although experimental handicapping has been used successfully in a number of sexual conflict studies, it does potentially alter the perceived quality of the manipulated mate. This perception could potentially be a confounding factor in determining reproductive allocation by the unmanipulated mate. However, the predicted change would be in the opposite direction as predicted by the negotiation model.
Observations for each pair were done under red light for 30 min before impairment, followed by a 2-h adjustment period after being weighted or handled, which was followed by another 30 min observation period. An observation period of 30 min was sufficient to observe a range of behaviors without adversely affecting the beetles. During the observation periods, scan samples were taken every 60 s and the behavior of each parent was recorded. The first observation period was initiated between 9:00 pm and 12:00 am. The entire observational period corresponds to a period when beetles are active. After the observation periods were finished, the weights were gently removed from the beetles.
Behaviors were separated into two categories: parental and non-parental. Parental behaviors included (1) excavating, which consisted of behavior where the parent moves soil from underneath or around the carcass; (2) removing fur, which consisted of behavior where the parent strips fur from the carcass and moves it to the edges of the burial site; (3) applying oral and anal secretions, which consisted of the parent spreading secretions over the carcass to preserve it; and (4) manipulating carcass, which consisted of behavior where the parent moves the carcass or manipulates it such as attempting to roll the carcass into a ball. Non-parental behaviors included (1) feeding on the carcass, (2) copulating, and (3) stationary (when the parent remains motionless). Twenty replicates were initiated in each treatment, but only pairs that successfully prepared the carcass and subsequently produced larvae were included in statistical analyses. Subsequent sample sizes ranged from 17 to 20 across treatments.
Statistical analyses
We asked three questions to evaluate the extent of compensation in response to reduced effort by a mate and differences between sexes. First, we assessed whether males and females allocate equal effort to carcass preparation under natural, nonmanipulated conditions. Second, we asked whether weight manipulations have a direct effect on parental effort and whether there is a difference in the size of the effect between the sexes. Third, we assessed whether the unweighted member of the pair compensates for reduced effort by the weighted member and if these differences vary between sexes. We used a mixed model analysis of variance approach to evaluate these questions.
To answer the first question, we compared beetles in control replicates only (n = 18 males and 18 females). Proportion of time spent in carcass preparation (including all parental behaviors listed above) was the response variable (arcsin-square root transformed to ensure normality) in all tests. Sex and time (first and second observation period) were predictor variables, and the interaction between sex and time was included. Pair number (unique number to identify each pair) and individual identification number were included as random effects because of the non-independence of pairs and the repeated measure on each individual (before and after) in all tests. A significant difference between sexes or a significant interaction between sex and time would suggest that sexes spend different proportions of time on parental care under control conditions (unmanipulated).
To answer the second question (direct effect of weighting), we compared beetles in control replicates (n = 18 males and 18 females) to weighted individuals in manipulated replicates (n = 17 males and 20 females) through time. Sex, time, and treatment (control or weighted) were predictor variables, and all interactions between main effects were included. Random effects were the same as in the previous test. This comparison validates the effectiveness of the weighting manipulation in reducing parental effort, and tests for differences between sexes in the effect of the weighting. A significant time by treatment interaction would indicate an effect of the weighting treatment for both sexes. Differences in the effect size of the weighting treatment between sexes would be indicated by a significant three-way interaction (sex by time by treatment).
To answer the third question (indirect effects of weighting), we compared beetles in the control replicates (n = 18 males and 18 females) to the same-sex unweighted individual in manipulated replicates (n = 20 males and 17 females). Sex, time, and treatment (control or unweighted member of manipulated pair) were predictor variables, and all interactions between main effects were included. Random effects were the same as in the other tests. This comparison tests for the extent of compensation and differences between sexes in response to reduced effort by the weighted mate. A significant time by treatment interaction would indicate an indirect effect (possible compensation) for both sexes. Differences in the effect size of the indirect treatment between sexes would be indicated by a significant three-way interaction (sex by time by treatment). All analyses were run with PROC MIXED in SAS version 9.3 (SAS Institute Inc., Cary, NC, USA).
Results
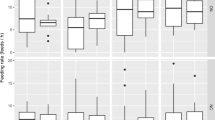
Sexes differed in their level of parental care under control conditions. Parental care in the control treatment as evidenced by proportion of scan samples spent preparing a carcass differed by sex (Table 1). Control females spent nearly all of their time preparing the carcass during both time periods, whereas control males decreased time spent preparing the carcass by about one third from the first time period to the second (Fig. 1).
Weighting caused a significant decline in parental effort. Proportion of time spent preparing a carcass differed significantly between weighted individuals and controls, between sexes, and between time periods. Both the sex by treatment and the time by treatment interaction were significant (Table 1). Weighting decreased parental effort during the second time period to about one fifth in males (Fig. 1a) and to about one third in females (Fig. 1b) compared to the first period.
Individuals compensated for decreased effort by their partner. The indirect effect of weighting on parental effort of carcass preparation differed between sexes, and the time by treatment interaction, and the three-way interaction of sex by time by treatment were significant (Table 1). Males and females exhibited higher levels of effort in response to the decline in effort by their mate (95 % confidence intervals do not overlap between first and second time periods). However, female change was minimal, given their high, initial level of effort (Fig. 1).
Discussion
The sexes differed in their level of care with females spending significantly more time than did males preparing the carcass. After they were handicapped (i.e., weighted), both males and females reduced the proportion of time spent working on the carcass, confirming that our manipulation worked. Male partners of handicapped females responded to the decreased effort by increasing their level of effort. Males exhibited partial compensation, consistent with the negotiation model. However, females were already spending the vast majority of their time working on the carcass (as evidenced by the control treatments and the before time periods in Fig. 1b). Thus, despite a statistically significant increase, there was minimal opportunity for increasing their overall level of effort (i.e., “ceiling effect;” Kosztolányi et al. 2009).
An alternative explanation for our results is that affixing a weight to one parent may have altered the perceived quality of that individual, which could affect their partner’s level of effort. In this instance, we would expect the unmanipulated parent to decrease their effort in response to a lower quality partner (Burley 1986; Sheldon 2000). However, both males and females increased their effort in response to their partners’ being handicapped, so it is unlikely that they perceived the weighted partner as a lower quality individual.
Our result that males compensate during the carcass preparation stage is consistent with removal studies conducted with burying beetles during the offspring provisioning stage. Removal studies conducted during this stage found males but not females increased provisioning rates after mate removal in N. vespilloides (Smiseth et al. 2005, 2006), N. orbicollis (Rauter and Moore 2004; but see Fetherston et al. (1994) for example of compensation by both sexes), and N. quadripunctatus (Suzuki and Nagano 2009). As with females during the carcass preparation stage, females during the provisioning stage may be working near their maximum effort and thus may not be able to compensate after males are removed (Smiseth and Moore 2004; Smiseth et al. 2005).
Mate removal studies have been shown (at least in birds) to illicit a higher level of compensation than handicapping studies (Harrison et al. 2009). Thus, a comparison of handicapping studies may be most useful. Two handicapping experiments testing the negotiation model have now been conducted on burying beetles (this study and Suzuki and Nagano 2009 with N. quadripunctatus). In contrast to our results, Suzuki and Nagano (2009) found that neither sex increased their level of effort to compensate for the reduction in effort by their partner during the provisioning stage. Differences between these two studies may reflect the relative importance of biparental care during these two periods of parental care. During the carcass preparation stage, both parents participate in burial and preservation of the carcass. Investment by both sexes may increase the likelihood that the carcass is secured underground and preserved against potential microbial competitors (Scott 1998). Additionally, biparental females gain relatively more mass prior to larval arrival than do uni-parental females (Jenkins et al. 2000). Thus, biparental care potentially results in a higher fitness payoff. Under these circumstances, partial compensation (Houston and Davies 1985; McNamara et al. 1999) or even full compensation (Jones et al. 2002) for a partner’s reduced effort is predicted. In contrast, during the offspring provisioning stage, the benefits of biparental care are not clear (Müller et al. 1998). Biparental care does not result in any measurable increase in fitness-related traits—brood size and offspring body size do not increase (Trumbo and Fernandez 1995; Sakaluk et al. 1998; Smiseth et al. 2005; Trumbo 2006) nor does development time change (Creighton, unpublished data). In addition, biparental care results in reduced fitness for females compared to uni-parental care (Boncoraglio and Kilner 2012), including when males are allowed to disperse freely (Smith, unpublished data). Therefore, the burying beetle system, in the offspring provisioning stage, may not meet the basic assumptions of the negotiation model where an increase in costly parental care by one parent results in an increase in fitness of the other.
Our results fall within the spectrum of results from manipulation experiments conducted with other species, although there does not seem to be a general consensus among these studies as to what pattern of compensation is observed. In birds, compensation by both sexes (Wright and Cuthill 1989; Wright and Cuthill 1990a; Wright and Cuthill 1990b; Wiebe 2010) or female only compensation (Slagsvold and Lifjeld 1990; Sanz et al. 2000) has been documented. Female compensation for decreased effort, but not male compensation, is also seen in other vertebrates, such as the cichlid fish Eretmodus cyanostictus (Steinegger and Taborsky 2007).
The negotiation model assumes that parents can monitor the efforts of their partner and adjust their level of care, accordingly. Partners can potentially monitor each other directly or indirectly, for example, through the begging rates of their offspring. Offspring were not present during our experiment, but partners could potentially monitor each other indirectly through the rate of carcass preparation. However, carcass preparation in N. orbicollis occurs over approximately a 4-day time period. The short time frame over which our observations were made (30 min in each time period) precludes much actual progress on carcass burial and preservation. Instead, our results suggest that burying beetles directly monitor the level of care provided by their partner during this stage of care, although the exact behavioral mechanism remains to be determined. Two studies on species of Nicrophorus have evaluated monitoring during the offspring provisioning stage. Smiseth and Moore (2004), using path analysis, found that males directly respond to changes in female effort in N. vespilloides. However, Suzuki and Nagano (2009) concluded that N. quadripunctatus parents were sensitive to their partner’s presence but not their level of effort because compensation was observed in their removal but not their handicapping experiment. Whether these results reflect species-specific differences, or are due to the focus on different periods of care, is unclear. A similar mix of results has been observed in birds where the direct monitoring of partner care has been proposed in a number of bird species (Hinde 2006; Hinde and Kilner 2007; Johnstone and Hinde 2006; Kosztolányi et al. 2009) but not in others (Beaulieu et al. 2009; Schwagmeyer et al. 2002; Tajima and Nakamuara 2003).
Our study suggests that intersexual conflict plays an important role in determining the level of parental care provided by N. orbicollis during the carcass preparation phase. Males, during this stage, appear to directly monitor the level of care provided by their partner and respond to a reduction in effort by increasing their own level of care. These results are consistent with removal experiments done during the offspring provisioning stage when males responded but not with a handicapping experiment conducted during this stage. This difference may reflect the relative importance and type of care provided during these two time periods as well as the relative fitness payoff of biparental care during these two stages of burying beetle reproduction.
References
Bartlett J (1987) Filial cannibalism in burying beetles. Behav Ecol Sociobiol 21:179–183
Beaulieu M, Raclot T, Dervaux A, Le Maho Y, Ropert-Coudert Y, Ancel A (2009) Can a handicapped parent rely on its partner? An experimental study within Adélie penguin pairs. Anim Behav 78:313–320
Boncoraglio G, Kilner RM (2012) Female burying beetles benefit from male desertion: sexual conflict and counter-adaptation over parental investment. PLoS ONE 7:e31713. doi:10.1371/journal.pone.0031713
Burley N (1986) Sexual selection for aesthetic traits in species with biparental care. Am Nat 127:415–445
Cotter SC, Ward RJS, Kilner RM (2011) Age-specific reproductive investment in female burying beetles: independent effects of state and risk of death. Funct Ecol 25:652–660
Creighton JC (2005) Population density, body size, and phenotypic plasticity of brood size in a burying beetle. Behav Ecol 16:1031–1036
Creighton JC, Heflin ND, Belk MC (2009) Cost of reproduction, resource quality, and terminal investment in a burying beetle. Am Nat 174:673–684
Fetherston IA, Scott MP, Traniello JFA (1990) Parental care in burying beetles: the organization of male and female brood-care behavior. Ethology 85:177–190
Fetherston IA, Scott MP, Traniello JFA (1994) Behavioral compensation for mate loss in the burying beetle Nicrophorus orbicollis. Anim Behav 47:777–785
Harrison F, Barta Z, Cuthill I, Székely T (2009) How is sexual conflict over parental care resolved? A meta-analysis. J Evolution Biol 22:1800–1812
Hinde CA (2006) Negotiation over offspring care?—a positive response to partner-provisioning rate in great tits. Behav Ecol 17:6–12
Hinde CA, Kilner RM (2007) Negotiations within the family over the supply of parental care. Proc R Soc B 274:53–60
Houston AI, Davies NB (1985) The evolution of co-operation and life history in the dunnock, Prunella modularis. In: Sibly RM, Smith RH (eds) Behavioural ecology: ecological consequences of adaptive behaviour. Blackwell Scientific, Oxford, pp 471–487
Houston AI, Székely T, McNamara JM (2005) Conflict between parents over care. Trends Ecol Evol 20:33–38
Hunt J, Simmons LW (2002) Behavioural dynamics of biparental care in the dung beetle Onthophagus taurus. Anim Behav 64:65–75
Jenkins EV, Morris C, Blackman S (2000) Delayed benefits of paternal care in the burying beetle Nicrophorus vespilloides. Anim Behav 60:443–451
Johnstone RA, Hinde CA (2006) Negotiation over offspring care—how should parents respond to each other’s efforts? Behav Ecol 17:818–827
Jones KM, Ruxton GD, Monaghan P (2002) Model parents: is full compensation for reduced partner nest attendance compatible with stable biparental care? Behav Ecol 13:838–843
Kosztolányi A, Cuthill IC, Székely T (2009) Negotiation between parents over care: reversible compensation during incubation. Behav Ecol 20:446–452
Lendvai AZ, Chastel O (2008) Experimental mate-removal increases the stress response of female house sparrows: the effects of offspring value? Horm Behav 53:395–401
Lendvai AZ, Barta Z, Chastel O (2009) Conflict over parental care in house sparrows: do females use a negotiation rule? Behav Ecol 20:651–656
Mazuc J, Chastel O, Sorci G (2003) No evidence for differential maternal allocation to offspring in the house sparrow (Passer domesticus). Behav Ecol Sociobiol 14:340–346
McNamara JM, Gasson CE, Houston AI (1999) Incorporating rules for responding into evolutionary games. Nature 401:368–371
McNamara JM, Houston AI, Barta Z, Osorno JL (2003) Should young ever be better off with one parent than with two? Behav Ecol 14:301–310
Müller JK, Eggert AK, Sakaluk SK (1998) Carcass maintenance and biparental brood care in burying beetles: are males redundant? Ecol Entomol 23:195–200
Parker GA (2006) Sexual conflict over mating and fertilization: an overview. Philos T Roy Soc B 361:235–259
Rauter CM, Moore AJ (1999) Do honest signaling models of offspring solicitation apply to insects? P Roy Soc Lond B Bio 266:1691–1696
Rauter CM, Moore AJ (2004) Time constraints and trade-offs among parental care behaviours: effects of brood size, sex, and loss of mate. Anim Behav 68:695–702
Sakaluk SK, Eggert AK, Müller JK (1998) The ‘widow effect’ and its consequences for reproduction in burying beetles. Ethology 104:553–564
Sanz JJ, Kranenbarg S, Tinbergen JM (2000) Differential response by males and females to manipulation of partner contribution in the great tit (Parus major). J Anim Ecol 69:74–84
Schwagmeyer PL, Mock DW, Parker GA (2002) Biparental care in house sparrows: negotiation or sealed bid? Behav Ecol 13:713–721
Schwagmeyer PL, Schwabl HG, Mock DW (2005) Dynamics of biparental care in house sparrows: hormonal manipulations of paternal contributions. Anim Behav 69:481–488
Scott MP (1998) The ecology and behavior of burying beetles. Annu Rev of Entom 43:595–618
Scott MP, Traniello JFA (1990) Behavioural and ecological correlates of male and female parental care and reproductive success in burying beetles (Nicrophorus spp.). Anim Behav 39:274–283
Sheldon BC (2000) Differential allocation: tests, mechanisms and implications. Trends in Ecol and Evol 15:397–402
Slagsvold T, Lifjeld JT (1990) Influence of male and female quality on clutch size in tits (Parus spp.). Ecology 71:1258–1266
Smiseth PT, Moore AJ (2004) Behavioral dynamics between caring males and females in a beetle with facultative biparental care. Behav Ecol 15:612–628
Smiseth PT, Dawson C, Varley E, Moore AJ (2005) How do caring parents respond to mate loss? Differential response by males and females. Anim Behav 69:551–559
Smiseth PT, Musa S, Moore AJ (2006) Negotiation between parents: does the timing of mate loss affect female compensation in Nicrophorus vespilloides? Behaviour 143:293–301
Steinegger M, Taborsky B (2007) Asymmetric sexual conflict over parental care in a biparental cichlid. Behav Ecol Sociobiol 61:933–941
Stoehr AM, Hill GE (2000) Testosterone and the allocation of reproductive effort in male house finches (Carpodacus mexicanus). Behav Ecol Sociobiol 48:407–411
Suzuki S, Nagano M (2009) To compensate or not? Caring parents respond differentially to mate removal and mate handicapping in the burying beetle, Nicrophorus quadripunctatus. Ethology 115:1–6
Tajima K, Nakamuara M (2003) Response to manipulation of partner contribution: a handicapping experiment in the barn swallow. Ornithol Sci 2:65–72
Trumbo ST (1991) Reproductive benefits and the duration of paternal care in a biparental burying beetle, Nicrophorus orbicollis. Behaviour 117:82–105
Trumbo ST (2006) Infanticide, sexual selection and task specialization in a biparental burying beetle. Anim Behav 72:1159–1167
Trumbo ST, Fernandez AG (1995) Regulation of brood size by male parents and cues employed to assess resource size by burying beetles. Ethol, Ecol and Evol 7:113–122
Wiebe KL (2010) Negotiation of parental care when the stakes are high: experimental handicapping of one partner during incubation leads to short-term generosity. J Anim Ecol 79:63–70
Wilson DS, Fudge J (1984) Burying beetles: intraspecific interactions and reproductive success in the field. Ecol Entomol 9:195–203
Wright DS, Cuthill I (1989) Manipulations of sex differences in parental care. Behav Ecol Sociobiol 25:171–181
Wright DS, Cuthill I (1990a) Biparental care: short-term manipulation of partner contribution and brood size in the starling, Sturnus vulgaris. Behav Ecol 1:116–124
Wright DS, Cuthill I (1990b) Manipulations of sex differences in parental care: the effect of brood size. Anim Behav 40:462–471
Acknowledgments
This research was funded in part by an undergraduate research grant from Purdue University Calumet. We thank D. Mock, A. Moore, S. Sakaluk, and S. Trumbo for their constructive reviews of our manuscript. We also thank N. Heflin and S. Zlotorzynski for assistance in maintaining laboratory animals and J. Shoup and S. Shoup for allowing us to collect beetles on their property.
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethical standards
The experiments described herein comply with the current laws of the USA, the country in which they were performed.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by S. Sakaluk
Rights and permissions
About this article
Cite this article
Creighton, J.C., Smith, A.N., Komendat, A. et al. Dynamics of biparental care in a burying beetle: experimental handicapping results in partner compensation. Behav Ecol Sociobiol 69, 265–271 (2015). https://doi.org/10.1007/s00265-014-1839-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00265-014-1839-z