Abstract
Kin recognition, the biased treatment of conspecific individuals based on genetic relatedness, is a widespread phenomenon in animals. The most common mechanisms mediating kin recognition are prior association (familiarity) and phenotype matching. Recognition based on prior association allows identifying familiar individuals. Recognition based on phenotype matching is an extension of prior-association-based recognition and allows identifying familiar and unfamiliar individuals due to a shared phenotypic trait. I investigated which of the two mechanisms is used by cannibalistic juvenile predatory mites, Phytoseiulus persimilis. Protonymphs that were associated with either siblings or non-kin discriminated familiar and unfamiliar larvae and preferentially cannibalized the latter irrespective of genetic relatedness. In contrast, despite previous association with either siblings or non-kin, protonymphs did not discriminate unfamiliar sibling and unfamiliar non-kin larvae. Association in the larval stage therefore mediated kin recognition based on familiarity, but not phenotype matching in cannibalistic P. persimilis protonymphs. Furthermore, in the presence of a familiar prey individual, sibling cannibalism occurred significantly sooner than non-kin cannibalism. This quick sibling cannibalism may have been the consequence of preferential association of siblings and/or may indicate the occurrence of an alternative cannibal phenotype. I discuss the adaptive significance of prior-association-based recognition for P. persimilis juveniles and emphasize the ability of P. persimilis to use multiple recognition mechanisms in dependence of the ontogeny and the ecological context.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The ability to discriminate conspecific individuals on the basis of genetic relatedness, kin recognition, has been suggested for numerous animals in a range of taxa (for reviews, Fletcher and Michener 1987; Waldman 1988; Hepper 1991; Sherman et al. 1997; Holmes 2004). As predicted by inclusive fitness theory (Hamilton 1964), kin recognition prevails in social animals. However, sociality is not an inevitable condition for the evolution of kin recognition abilities. For instance, the ability to discriminate kin and non-kin may be selectively advantageous for non-social animals that use fluctuating resources and live in conspecific aggregations (Fellowes 1998). Kin recognition has been primarily studied and found in the contexts of nepotism, parental care, mate choice, schooling behavior, and aggression-related behaviors such as cannibalism.
Kin recognition systems consist of three separate components: expression (production of the kin label), perception (the recognition mechanism), and action (kin-biased behavior) (Reeve 1989; Sherman et al. 1997; Liebert and Starks 2004; Starks 2004). Regarding the perception component, four potential mechanisms of kin recognition have been suggested: (1) context-based recognition, where templates of true genetic kin are learned from environmental features (mainly spatial) that are shared with and are exclusively available to a kin-group, (2) recognition alleles, which cause both the production and recognition of a phenotypic cue, (3) prior association (familiarity), which allows identifying familiar individuals, and (4) phenotype matching (Blaustein 1983; Waldman 1988; Halpin 1991; Mateo 2004). Phenotype matching (term coined by Holmes and Sherman 1982) is an extension of or addition to prior-association-based recognition. It allows identifying familiar and unfamiliar kin due to having formed a prototypic template of kin by learning the phenotypes from familiar individuals (Sherman et al. 1997; Hepper and Cleland 1998; Heth et al. 1998; Mateo 2004) or from self (Hauber and Sherman 2001). The latter form of phenotype matching is commonly referred to as self-referent phenotype matching or “armpit effect” (Dawkins 1982). The process leading to prior-association-based recognition has also been termed direct familiarization, whereas the process leading to phenotype matching has been termed indirect familiarization (Blaustein and Porter 1996).
Prior-association-based recognition and phenotype matching are the mechanisms most often assessed and found. Among vertebrates, phenotype matching was revealed in mammals (e.g. ground squirrels, Heth et al. 1998; Holmes and Sherman 1982; Mateo 2002; hamsters, Mateo and Johnston 2003; beavers, Sun and Müller-Schwarze 1997; primates, Smith et al. 2003), birds (e.g., peacocks, Petrie et al. 1999) and fish (e.g., trout, Brown et al. 1993). However, the mechanism most often found in these taxa is prior association (for mammals, see e.g., Holmes and Sherman 1982; Halpin 1991; Pillay 2002; for fish, see review by Ward and Hart 2003; and for birds, e.g., Choudhury and Black 1994; Komdeur et al. 2004; van der Jeugd et al. 2002; Sharp et al. 2005). Among invertebrates, it is mainly social hymenoptera that have been shown to use recognition mechanisms based on prior association and/or phenotype matching (e.g., Buckle and Greenberg 1981 for sweat bees; Provost 1991 for ants; Getz and Smith 1986 for honey bees; Gamboa 2004 for review on social wasps). In contrast, studies examining the kin recognition mechanisms used by non-social arthropods are lacking. Knowing which mechanism, familiarity-based recognition or phenotype matching, is mediated by association is fundamental for interpreting any form of intraspecific interaction and identifying analogies/differences in life history and behavior among different organisms.
If it is usually related individuals that grow up in the same site (e.g., nesting species or non-social species laying their eggs in clutches), association may be used as a reliable indicator of kin (Blaustein and O’Hara 1986). In the predatory mite Phytoseiulus persimilis Athias-Henriot, the focal animal of this study, the probability of kin groups to occur is determined by the patchy distribution and life history of its prey, the herbivorous two-spotted spider mite Tetranychus urticae Koch (Gerson 1985; Sabelis 1985a; Sabelis and Dicke 1985). P. persimilis is a highly specialized predator of tetranychid mites such as the polyphagous T. urticae (for review, McMurtry and Croft 1997). T. urticae primarily lives on the abaxial leaf surfaces where the mites create distinct patches (Gerson 1985). Gravid P. persimilis females forage and deposit their eggs in the spider mite patches. Each predator female may lay multiple egg clutches, the size of which depends on the prey density within patches (e.g., Nagelkerke et al. 1996). Due to the high reproductive rate of P. persimilis (up to five eggs per female per day), offspring are often aggregated in prey patches (e.g., Sabelis 1985b). When prey within a patch diminishes, food competition intensifies, and the likelihood of cannibalism increases (Polis 1981; Elgar and Crespi 1992). Both adult and juvenile P. persimilis are cannibalistic (Schausberger and Croft 2000, 2001; Schausberger 2003, 2004), yet adult females tend to leave the patch before the spider mites are locally extinct (Vanas et al. 2006). Juvenile individuals are less dispersive and may cannibalize each other close to or after prey extinction (Sabelis 1981; Pels and Sabelis 1999; Schausberger 2003). Depending on the number and relatedness of predator females that colonized a spider mite patch, juveniles within a patch may be only kin—which is the majority of cases—or a mixture of kin and non-kin (e.g., Nagelkerke et al. 1996). Such circumstances seem ideal to promote the evolution of kin recognition abilities.
P. persimilis develops through four successive life stages before reaching adulthood: egg, larva, protonymph, and deutonymph. Larvae do not feed and are particularly vulnerable to cannibalism by older and/or larger life stages such as protonymphs (Schausberger and Croft 1999; Schausberger 2003, 2004, 2005). Previous studies on recognition of kin and/or familiar larvae by cannibalistic P. persimilis protonymphs (Schausberger 2004, 2005) suggested that (1) larvae learn the cues of associated individuals and later on treat familiar individuals as kin, (2) egg/larval associations and interactions may be manipulated by the ovipositing female via egg placement, which may affect the cannibalism risk for offspring and their performance as potential cannibals later in life, (3) cannibalistic protonymphs discriminate familiar and unfamiliar larvae, irrespective of genetic relatedness, and avoid eating the former. Association with conspecifics early in life has significance for who is treated as kin by juvenile cannibalistic P. persimilis later in life, but the mechanism used is not known. The aforementioned observations would be consistent with both prior association and phenotype matching, but not with the other two of the four possible mechanisms. This study therefore aims at distinguishing the two mechanisms.
Materials and methods
Study species, populations, and individuals
P. persimilis used in experiments were offspring of females withdrawn from five distinct populations (EL, ER, PANF, OR, and G). Populations EL and ER were founded with individuals originating from a mass production company of beneficial mites and insects (Biohelp, Vienna); population PANF was founded with individuals collected in Sicily, population OR with individuals collected in Oregon (USA), and population G with individuals collected in Greece. Populations were founded with 30 to 100 individuals each and maintained in the laboratory for about 0.5 to 3.5 years before starting the experiments. Each population was maintained on an artificial arena consisting of a plastic tile (15 × 15 cm) resting on a water-saturated foam cube in a plastic box and surrounded by water-saturated tissue paper. The predators were fed mixed stages of two-spotted spider mite, T. urticae, by adding infested bean leaves on to the arenas in 2 to 3-day intervals. Rearing units of different populations were kept in separate places to avoid cross contamination. Environmental conditions were 20 to 25°C, 60 to 80% RH, and 16:8 h L/D photoperiod.
Recognition mechanism experiments
To assess the ability to recognize familiar individuals and distinguish between recognition based on familiarity and recognition based on phenotype matching, I performed two experiments. In experiment 1, I tested whether cannibalistic P. persimilis protonymphs that were associated with either siblings or non-kin in the larval stage discriminate familiar and unfamiliar larvae. In experiment 2, I tested whether cannibalistic P. persimilis protonymphs that were associated with either sibling or non-kin referents in the larval stage discriminate unfamiliar sibling larvae and unfamiliar non-kin larvae. In either treatment of experiment 2, one of the two prey larvae was a sibling of the referents (Table 1). Each experiment consisted of three parts: obtaining sibling eggs, the association phase, and the choice test.
To obtain sibling eggs, gravid females were randomly withdrawn from populations and singly placed on egg laying arenas. Each egg laying arena consisted of a leaf placed adaxial surface down on a water-saturated foam cube in a small plastic box (5 × 5 cm) half filled with water. Strips of moist tissue paper were folded over the edges of the leaf to prevent the mites from escaping. Eggs were collected in 24-h intervals and stored in the refrigerator at 8°C until they were used in experiments.
Acrylic cages were used for the association phase and the choice tests. Each cage consisted of a cylindrical cavity (1.5-cm diameter) drilled in an acrylic plate (0.5-cm-thick). The cavity was covered with a mesh on the lower side and closed on the upper side with a microscope slide (Schausberger and Croft 2001). The association phase proceeded as follows: an egg (the prospective cannibal) was placed in an acrylic cage. Then, four to seven eggs (the prospective referents, either siblings or non-kin of the prospective cannibal) were added after 12 to 16 h. Referents are those individuals that provide the reference an internal template is based upon (Tsutsui 2004). Non-kin referents were laid by a female from a different population than the cannibal. Moreover, a further four to seven eggs, sibling of the referents, and thus, either sibling or non-kin of the prospective cannibal, were placed in a separate cage. They were used as unfamiliar siblings or unfamiliar non-kin in experiment 1 and unfamiliar siblings of the referents in experiment 2. Cages were then monitored for hatching larvae at irregular intervals. The prospective cannibal was more advanced in its development and hatched 12 to 16 h earlier than the referents. The prospective cannibal molted from the larva to the protonymph while the referents were still in the larval stage, and was thereafter subjected to a choice test.
To start a choice test, the prospective cannibal (a protonymph) and two prospective prey (two larvae) were transferred to a cage. In experiment 1, cannibals, previously associated with siblings, were caged with a familiar sibling larva and an unfamiliar non-kin larva; cannibals, previously associated with non-kin, were caged with a familiar non-kin larva and an unfamiliar sibling larva (Table 1). Each treatment of experiment 1 was replicated 27 times. In experiment 2, cannibals, previously associated with siblings, were caged with an unfamiliar sibling larva and an unfamiliar non-kin larva; cannibals, previously associated with non-kin, were caged with an unfamiliar sibling larva and an unfamiliar non-kin larva where the latter was a sibling of the non-kin referent (Table 1). Treatments of experiment 2 were replicated 21 and 27 times. Before transferring the prospective prey to the choice cages, larvae were marked by red and blue watercolor dots on the dorsal shield to make them distinguishable. In each choice situation, colors were randomly assigned to familiar and unfamiliar larvae and sibling and non-kin larvae, respectively. Cages were monitored at irregular intervals of 10 to 25 min until cannibalism on one of both larvae occurred. Status of the cannibalized larva (sibling or non-kin to the cannibal—experiments 1 and 2; familiar or unfamiliar to the cannibal—experiment 1; sibling or non-kin to the referents—experiment 2) and time elapsed until cannibalism occurred (experiments 1 and 2) were recorded. Egg laying arenas, association cages, and choice cages were stored at 23 to 25°C.
Data analyses
Replicates at which one of both prey larvae molted to a protonymph before the occurrence of cannibalism or at which no cannibalism occurred within 6 h after starting the experiment were excluded from analyses. Log-linear analyses were used to test the influence of the victim’s relatedness and familiarity to the cannibal (experiment 1) and the victim’s relatedness to the cannibal and to the referents (experiment 2) on the choice of the cannibal. A separate analysis was performed for each explaining variable, and prior association with siblings or non-kin was used as a covariate. The influence of the victim’s familiarity and relatedness to the cannibal (experiment 1) and the influence of the victim’s relatedness to the cannibal and to the referents (experiment 2), respectively, on the time elapsed until cannibalism occurred were analyzed by univariate analyses of variance (ANOVA).
Results
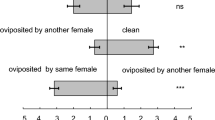
In experiment 1, P. persimilis protonymphs discriminated familiar and unfamiliar larvae and preferentially cannibalized the latter when given a choice. Familiarity had a significant effect on the choice of the cannibals (log-linear analysis, Z = 2.63, p = 0.009), whereas genetic relatedness between cannibal and victim had not (log-linear analysis, Z = −0.27, p = 0.787; Fig. 1). Moreover, cannibalism on sibling larvae occurred significantly sooner than cannibalism on non-kin larvae regardless of familiarity (Tables 2 and 3). In experiment 2, protonymphs did not discriminate unfamiliar sibling larvae and unfamiliar non-kin larvae, although they were familiar with siblings of one of the two stimulus larvae (log-linear analysis, relatedness between cannibal and victim Z < 0.01, p > 0.992; relatedness between referents and victim Z = 0.29, p = 0.772; Fig. 2). Protonymphs given a choice between two unfamiliar larvae cannibalized siblings as early as they cannibalized non-kin, irrespective whether previously associated with siblings or non-kin (Tables 4 and 5).
Discussion
Association early in life (in the larval stage) mediated recognition based on familiarity, but not phenotype matching in juvenile predatory mites, P. persimilis. In the larval stage, prospective cannibals learned the cues of associated conspecific larvae. After molting to protonymphs, cannibals treated familiar and unfamiliar larvae differently in that they preferentially cannibalized unfamiliar larvae irrespective of whether sibling or non-kin. Cannibals, however, did not discriminate unfamiliar larvae that differed in genetic relatedness, sibling and non-kin, although they were familiar with siblings of one of the two stimulus larvae.
The perceptual distinction among recognition mechanisms, in particular between recognition based on familiarity and phenotype matching, is a controversial issue (see, e.g., Tang-Martinez 2001; Mateo 2004). The main problem is that the neural process cannot be observed directly, but is inferred from the behavioral responses of the animals, i.e., the action component. Tang-Martinez (2001) argued there exists only a single genuine recognition mechanism. A more widely accepted view is that familiarity-based recognition and phenotype matching are different perceptual mechanisms (Holmes 2004; Mateo 2004). The former requires an exact match to an individual template, whereas the latter generalizes from individual templates that have been formed from individuals sharing phenotypic traits to a common representation of kin (Mateo 2004). Differential treatment of familiar and unfamiliar larvae by P. persimilis protonymphs reared under the same conditions may hence indicate individual recognition using genetically determined cues.
Why does association mediate recognition by familiarity but not phenotype matching in cannibalistic P. persimilis protonymphs? In general, early in life, a flexible and reversible recognition mechanism seems more favorable than a static one (Mateo 2004). Individual templates may be stored and used for limited time periods and then vanish again, whereas prototypic templates may be irreversible and stored throughout life (Tang-Martinez 2001; Holmes 2004; Mateo 2004). Errors in template learning and formation would thus have more dramatic consequences with prototypic than with individual templates. Prior-association-based recognition allows constant updating and modification of templates, generalizing from individual templates to form a prototype and extending the mechanism to phenotype matching later in life (Fletcher 1987; Liebert and Starks 2004; Mateo 2004). Familiarity-based recognition seems selectively advantageous for P. persimilis juveniles because recognition of larvae is relevant for only a short period of time, and a few larvae provide enough nutrients to proceed with development (Walzer and Schausberger 1999). Moreover, it allows mothers to reduce the risk of offspring cannibalism when colonizing a patch that harbors eggs from another female via selective egg placement (Schausberger 2005).
Time elapsed until sibling/kin cannibalism occurs seems context-dependent. Consistent with recent experiments (Schausberger 2004, 2005) sibling/kin cannibalism occurred significantly earlier than non-kin cannibalism when the cannibals were offered familiar and unfamiliar prey. Possible proximate and ultimate reasons for the occurrence of quick sibling cannibals have been put forward previously. They include preferential association of siblings/kin and consequently earlier sibling/kin cannibalism without representing a true preference and two types of cannibals with differing preferences (Schausberger 2004, 2005). Exceptionally quick sibling/kin cannibalism could also be interpreted as selfish behavior if it benefits direct fitness at the detriment of indirect fitness (Hamilton 1964, 1970; Mock and Parker 1997). As previously suggested (Schausberger 2004, 2005), those quick sibling cannibals appear to use self-referent phenotype matching to discriminate siblings/kin and non-kin because cannibals unfamiliar with siblings/kin still recognized “true” kin (inferred from time until cannibalism occurred), but treated non-kin as if kin (inferred from cannibalism avoidance). Scrutinizing the circumstances that promote the occurrence of quick sibling cannibals and/or make them apparent and determining their fate later in life remain intriguing tasks for future studies.
This study corroborates recent findings that cannibalistic P. persimilis protonymphs treat any familiar conspecific larva as kin irrespective of genetic relatedness (Schausberger 2005). Familiarity as the sole mechanism to determine kin and used as a proxy of relatedness has been shown for diverse organisms and contexts: Association attractiveness among salmon varies with familiarity and odor concentration (Courtenay et al. 2001), guppies choose shoal mates based on familiarity (Griffiths and Magurran 1999), familiar piglets show less aggressiveness than unfamiliar ones irrespective of genetic relatedness (Stookey and Gonyou 1998), juvenile sticklebacks compete less with familiar individuals without any indication that genetic relatedness plays a role (Utne-Palm and Hart 2000), and penguin chicks discriminate between familiar and unfamiliar calls but not between calls of familiar kin and non-kin (Nakagawa et al. 2001).
Accumulating evidence suggests that prior association is the mechanism more frequently encountered than phenotype matching (e.g., Komdeur and Hatchwell 1999; Mateo 2004). The two mechanisms may be distinct, or phenotype matching may be an extension of prior association. In no case do the two mechanisms exclude each other. There exist several examples that one and the same organism may use both mechanisms. Precedents are Belding’s ground squirrels (Holmes and Sherman 1982; Mateo and Johnston 2003; Mateo 2004), lambs (Ligout and Porter 2003), social paper wasps (Gamboa et al. 1986; Bura and Gamboa 1994; Gamboa 2004), and the focal animal of this study. P. persimilis may use recognition based on self-referent phenotype matching (Schausberger 2004; Enigl and Schausberger 2004), phenotype matching (Schausberger 2005), and prior association (Schausberger 2004, 2005, present study).
Different perceptual mechanisms may be used in different social contexts, and the ability to switch between mechanisms may have an ontogenetic or developmental component. As suggested for Belding’s ground squirrels, the ability to prior-association-based recognition is expected to precede an ability to phenotype-matching-based recognition (Mateo 2004). The mechanisms used by P. persimilis seem to have an ontogenetic component and are context-dependent: Prior-association-based recognition is used by protonymphs reared in association with conspecifics (Schausberger 2004, 2005, present), self-referent phenotype matching is used by protonymphs reared in isolation (Schausberger 2005) and virgin females in mate choice (Enigl and Schausberger 2004), and phenotype-matching-based recognition (self-referent and/or after learning the cues of own eggs) is used by ovipositing females (Schausberger and Croft 2001; Schausberger 2005). Determination of the chronological order, hierarchy, and context dependence of the kin recognition mechanisms that may operate in one and the same P. persimilis individual are intriguing tasks for future research.
References
Blaustein AR (1983) Kin recognition mechanisms: phenotypic matching or recognition alleles? Am Nat 121:749–754
Blaustein AR, O’Hara RK (1986) Kin recognition in tadpoles. Sci Am 254:108–116
Blaustein AR, Porter RH (1996) The ubiquitous concept of recognition with special reference to kin. In: Bekoff M, Jamieson D (eds) Readings in animal cognition. MIT, Cambridge, pp 169–184
Brown G, Brown J, Crosbie A (1993) Phenotype matching in juvenile rainbow trout. Anim Behav 46:863–871
Buckle GR, Greenberg L (1981) Nestmate recognition in sweat bees Lasioglossum zephyrum: does an individual recognize its own odor or only odors of its nestmates? Anim Behav 29:802–809
Bura EA, Gamboa GJ (1994) Kin recognition by social wasps—asymmetric tolerance between aunts and nieces. Anim Behav 47:977–979
Choudhury S, Black JM (1994) Barnacle geese preferentially pair with familiar associates from early life. Anim Behav 48:81–88
Courtenay SC, Quinn TP, Dupuis HMC, Groot C, Larkin PA (2001) Discrimination of family-specific odours by juvenile coho salmon: roles of learning and odour concentration. J Fish Biol 58:107–125
Dawkins R (1982) The extended phenotype. WH Freeman, San Francisco, CA
Elgar M, Crespi BJ (1992) Cannibalism: ecology and evolution among diverse taxa. Oxford University Press, Oxford
Enigl M, Schausberger P (2004) Mate choice in the predaceous mite Phytoseiulus persimilis: evidence of self-referent phenotype matching? Entomol Exp Appl 112:21–28
Fellowes MDE (1998) Do non-social insects get the (kin) recognition they deserve? Ecol Entomol 23:223–227
Fletcher DJC (1987) The behavioral analysis of kin recognition: perspectives on methodology and interpretation. In: Fletcher DJC, Michener CD (eds) Kin recognition in animals. Wiley, New York, pp 19–54
Fletcher DJC, Michener CD (eds) (1987) Kin recognition in animals. Wiley, New York
Gamboa GJ (2004) Kin recognition in eusocial wasps. Ann Zool Fenn 41:789–808
Gamboa GJ, Reeve HK, Ferguson ID, Wacker TL (1986) Nestmate recognition in social wasps: the origin and acquisition of recognition odours. Anim Behav 34:685–695
Gerson U (1985) Webbing. In: Helle W, Sabelis MW (eds) Spider mites, their biology, natural enemies and control, vol 1 A. Elsevier, Amsterdam, pp 223–232
Getz WM, Smith KB (1986) Honey-bee kin recognition—learning self and nestmate phenotypes. Anim Behav 34:1617–1626
Griffiths SW, Magurran AE (1999) Schooling decisions in guppies (Poecilia reticulata) are based on familiarity rather than kin recognition by phenotype matching. Behav Ecol Sociobiol 45:437–443
Halpin ZT (1991) Kin recognition cues of vertebrates. In: Hepper PG (ed) Kin recognition. Cambridge University Press, Cambridge, pp 220–258
Hamilton WD (1964) The genetical evolution of social behaviour I. J Theor Biol 7:1–52
Hamilton WD (1970) Selfish and spiteful behaviour in an evolutionary model. Nature 228:1218–1220
Hauber ME, Sherman PW (2001) Self-referent phenotype matching: theoretical considerations and empirical evidence. Trends Neurosci 24:609–616
Hepper PG (1991) Kin recognition. Cambridge University Press, Cambridge
Hepper PG, Cleland J (1998) Developmental aspects of kin recognition. Genetica 104:199–205
Heth G, Todrank J, Johnston RE (1998) Kin recognition in golden hamsters: evidence for phenotype matching. Anim Behav 56:409–417
Holmes WG (2004) The early history of Hamiltonian-based research on kin recognition. Ann Zool Fenn 41:691–711
Holmes WG, Sherman PW (1982) The ontogeny of kin recognition in two species of ground-squirrels. Am Zool 22:491–517
Komdeur J, Hatchwell BJ (1999) Kin recognition: function and mechanism in avian societies. Trends Ecol Evol 14:237–241
Komdeur J, Richardson DS, Burke T (2004) Experimental evidence that kin discrimination in the Seychelles warbler is based on association and not on genetic relatedness. Proc R Soc Lond B 271:963–969
Liebert AE, Starks PT (2004) The action component of recognition systems: a focus on the response. Ann Zool Fenn 41:747–764
Ligout S, Porter RH (2003) Social discrimination in lambs: the role of indirect familiarization and methods of assessment. Anim Behav 65:1109–1115
Mateo JM (2002) Kin-recognition abilities and nepotism as a function of sociality. Proc R Soc Lond B 269:721–727
Mateo JM (2004) Recognition systems and biological organization: the perception component of social recognition. Ann Zool Fenn 41:729–745
Mateo JM, Johnston RE (2003) Kin recognition by self-referent phenotype matching: weighing the evidence. Anim Cogn 6:73–76
McMurtry JA, Croft BA (1997) Life-styles of phytoseiid mites and their role in biological control. Annu Rev Entomol 42:291–321
Mock DA, Parker GA (1997) The evolution of sibling rivalry. Oxford University Press, New York
Nagelkerke CJ, van Baalen M, Sabelis MW (1996) When should a female avoid adding eggs to the clutch of another female? A simultaneous oviposition and sex allocation game. Evol Ecol 10:475–497
Nakagawa S, Waas J, Miyazaki M (2001) Heart rate changes reveal that little blue penguin chicks Eudyptula minor can use vocal signatures to discriminate familiar from unfamiliar chicks. Behav Ecol Sociobiol 50:180–188
Pels B, Sabelis MW (1999) Local dynamics, overexploitation and predator dispersal in an acarine predator–prey system. Oikos 86:573–583
Petrie M, Krupa A, Burke T (1999) Peacocks lek with relatives even in the absence of social and environmental cues. Nature 401:155–157
Pillay N (2002) Father–daughter recognition and inbreeding avoidance in the striped mouse, Rhabdomys pumilio. Mamm Biol 67:212–218
Polis G (1981) The evolution and dynamics of intraspecific predation. Annu Rev Ecol Syst 12:225–251
Provost E (1991) Non-nestmate kin recognition in the ant Leptothorax lichtensteini: evidence that genetic factors regulate colony recognition. Behav Genet 21:151–167
Reeve HK (1989) The evolution of conspecific acceptance thresholds. Am Nat 133:407–435
Sabelis MW (1981) Biological control of two-spotted spider mites using phytoseiid predators, Part I: modelling the predator–prey interaction at the individual level. Centre of Agricultural Publishing and Documentation, Wageningen
Sabelis MW (1985a) Reproductive strategies. In: Helle W, Sabelis MW (eds) Spider mites, their biology, natural enemies and control, vol 1A. Elsevier, Amsterdam, pp 265–278
Sabelis MW (1985b) Sex allocation. In: Helle W, Sabelis MW (eds) Spider mites, their biology, natural enemies and control, vol 1A. Elsevier, Amsterdam, pp 83–94
Sabelis MW, Dicke M (1985) Long-range dispersal and searching behaviour. In: Helle W, Sabelis MW (eds) Spider mites, their biology, natural enemies and control, vol 1A. Elsevier, Amsterdam, pp 141–160
Schausberger P (2003) Cannibalism among phytoseiid mites: a review. Exp Appl Acarol 29:173–191
Schausberger P (2004) Ontogenetic isolation favours sibling cannibalism in mites. Anim Behav 67:1031–1035
Schausberger P (2005) The predatory mite Phytoseiulus persimilis manipulates imprinting among offspring through egg placement. Behav Ecol Sociobiol 58:53–59
Schausberger P, Croft BA (1999) Activity, feeding, and development among larvae of specialist and generalist phytoseiid mite species (Acari: Phytoseiidae). Environ Entomol 28:322–329
Schausberger P, Croft BA (2000) Cannibalism and intraguild predation among phytoseiid mites: are aggressiveness and prey preference related to diet specialization? Exp Appl Acarol 24:709–725
Schausberger P, Croft BA (2001) Kin recognition and larval cannibalism by adult females in specialist predaceous mites. Anim Behav 61:459–464
Sharp SP, McGowan A, Wood MJ, Hatchwell BJ (2005) Learned kin recognition cues in a social bird. Nature 434:1127–1130
Sherman PW, Reeve HK, Pfennig DW (1997) Recognition systems. In: Krebs JR, Davies NB (eds) Behavioural ecology, an evolutionary approach, 4th edn. Blackwell Science, Oxford, pp 69–96
Smith K, Alberts SC, Altmann J (2003) Wild female baboons bias their social behaviour towards paternal half-sisters. Proc R Soc Lond B 270:503–510
Starks PT (2004) Recognition systems: from components to conservation. Ann Zool Fenn 41:689–690
Stookey JM, Gonyou HW (1998) Recognition in swine: recognition through familiarity or genetic relatedness. Appl Anim Behav Sci 55:291–305
Sun LX, Müller-Schwarze D (1997) Sibling recognition in the beaver: a field test for phenotype matching. Anim Behav 54:493–502
Tang-Martinez Z (2001) The mechanisms of kin discrimination and the evolution of kin recognition in vertebrates: a critical re-evaluation. Behav Processes 53:21–40
Tsutsui ND (2004) Scents of self: the expression component of self/non-self recognition systems. Ann Zool Fenn 41:713–727
Utne-Palm AC, Hart PJB (2000) The effects of familiarity on competitive interactions between threespined sticklebacks. Oikos 91:225–232
Vanas V, Enigl M, Walzer A, Schausberger P (2006) The predatory mite Phytoseiulus persimilis adjusts patch-leaving to own and progeny prey needs. Exp Appl Acarol 39:1–11
van der Jeugd HP, van der Veen IT, Larsson K (2002) Kin clustering in barnacle geese: familiarity or phenotype matching? Behav Ecol 13:786–790
Waldman B (1988) The ecology of kin recognition. Annu Rev Ecol Syst 19:543–571
Walzer A, Schausberger P (1999) Cannibalism and interspecific predation in the phytoseiid mites Phytoseiulus persimilis and Neoseiulus californicus: predation rates and effects on reproduction and juvenile development. BioControl 43:457–468
Ward AJW, Hart PJB (2003) The effects of kin and familiarity on interactions between fish. Fish Fish 4:348–358
Acknowledgments
The study was primarily financed by the Austrian Academy of Sciences (APART fellowship). I thank J. S. Blackwood and M. Enigl for comments on a previous version of the manuscript and D. Hoffmann and M. Enigl for technical assistance.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by N. Wedell
Rights and permissions
About this article
Cite this article
Schausberger, P. Kin recognition by juvenile predatory mites: prior association or phenotype matching?. Behav Ecol Sociobiol 62, 119–125 (2007). https://doi.org/10.1007/s00265-007-0444-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00265-007-0444-9