Abstract
In monogamous bird species, male parental investment may influence offspring fitness and females may gain advantages through mating with males providing extensive paternal care. However, paternal care is a benefit that can only be assessed indirectly because mate choice precedes paternal activities. Individual quality and age, both signalled by morphological characteristics, may reflect parental abilities. Because they may reflect individual foraging abilities, carotenoid-based colorations have been proposed to honestly signal parental quality. The blackbird (Turdus merula), a socially monogamous species, exhibits biparental care and males show bills that vary from pale yellow to orange due to carotenoid pigments. In this study, we investigated whether male bill colour and age are associated with parental ability. Our results suggest that males with more orange bills and older males are better fathers. Indeed, male visit rate increased with their bill colour index independently of age, and brood condition was higher for adult males, compared to yearlings, independently of bill colour. Overall, the number of fledglings produced was positively influenced by both the age of males and the colour intensity of their bills. Males with more orange bills and adults had a greater number of fledglings and these males also had higher levels of prolactin, a hormone known to promote parental care. This latter finding suggests that prolactin may be the link between carotenoid based colorations and the intensity of paternal effort. Thus, male bill colour seems to honestly reveal male physiological adjustment to paternal activities.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
In most monogamous bird species, both males and females contribute to parental care (Burley and Johnson 2002). Both sexes are expected to adjust their expenditures on parental care in relation to their own benefit and to that of their offspring (Trivers 1972; Clutton-Brock 1991). However, even when cooperating in a joint task, male and female interests are rarely identical, and each sex should generally prefer the other to ‘work harder’, because the evolution of increased care by one sex will enable reduced care in the other (Westneat and Sargent 1996). Therefore, females may experience advantages through mating with males providing extensive paternal care (Clutton-Brock 1991; Shadd and Ritchison 1998). However, since mate choice precedes paternal activities, females cannot directly assess males’ ability to provide parental care. Therefore, they may rely on specific cues to indirectly assess male parental quality, particularly when forming a new pair-bond (Møller 1994). In this context, it has been suggested that the degree of ornamentation may signal male parental quality to females (Sundberg and Larsson 1994; Hill 2002; Senar et al. 2002).
Two models of sexual selection have addressed the relationship between male parental care and ornamentation. These models differ primarily in the kind of benefits signalled by male secondary sexual characters. First, the differential allocation hypothesis (Burley 1986, 1988; Sheldon 2000) assumes that the male trait reflects its genetic quality, and that females mated with attractive males gain indirect fitness benefits and are “willing” to contribute more to obtain and/or maintain their attractive mates (Burley 1986). In turn, attractive males can afford to reduce their parental investment and thus gain a survival advantage. This model predicts a negative relationship between male parental care and ornamentation and a positive one between female parental care and male ornamentation. Second, the good parent hypothesis (Hoelzer 1989; Price et al. 1993) stipulates that epigamic traits evolve to advertise the non-heritable quality of parental ability. When paternal care influences the viability or fertility of offspring, males can be selected to advertise honestly the quality or quantity of care they are likely to provide. Thus, females may gain direct benefits through choosing a mate with good parental abilities and, through paying attention to costly traits, females should enhance the evolution of male traits that honestly reveal parental quality. This model predicts a positive relationship between paternal care and ornamentation.
In addition, the two previous models of sexual selection do not exclude the role of important factors potentially related to male quality, parental care and reproductive success. For instance, female quality may also influence the overall breeding success (see Faivre et al. 2001), and “sexy” and older males may attract females of higher quality, leading to assortative mating (Andersson 1994). In this case, the relative influence of male and female quality on nesting output may become difficult to assess.
Male carotenoid-based colours have been shown to reflect male quality (Forslund and Pärt 1995; Blount 2004). Carotenoid-based colours are thought to be particularly reliable as signals of individual quality because birds cannot synthesize these pigments and therefore must obtain them from food (Fox and Vevers 1960; Brush and Power 1976; Gray 1996; Lozano 1994, 2001). Intraspecific variation in the red, orange and yellow colours in males may then reflect differences in the quantity or quality of carotenoids ingested, and their abilities to provide this crucial resource to their nestlings (Hill 1992; Linville and Breitwisch 1997; Hõrak et al. 2000). Hence, females may directly benefits from pairing with brightly coloured males, which may be better fathers. Besides their role as indicators of foraging ability, carotenoid-based colours have also been suggested to reflect overall genetic quality or differences in metabolisation abilities (Hamilton and Zuk 1982; Blount 2004). In this case, females paired with brightly coloured males may gain indirectly by producing offspring of higher quality. Overall, whatever the benefit females may experience (direct or indirect), they should choose highly ornamented males.
In addition, age, or experience, may also explain a great part of variation in reproductive success between individuals (Clutton-Brock 1988). For instance, fledging success is influenced by an increased foraging efficiency with increasing age of parents (Forslund and Pärt 1995). Thus, females paired with older males may gain direct benefits from their higher paternal abilities, as well as indirect ones from the higher genetic quality of their mates (Forslund and Pärt 1995; Brooks and Kemp 2001). In such case, age may act as a potential confounding factor for carotenoid-based colorations if colour increases with age. For instance, Sundberg and Larsson (1994) have shown that older males exhibited a more yellow plumage in the yellowhammer (Emberiza citrinella).
From a mechanistic point of view, incubation and brood care are under hormonal control (Murton and Westwood 1977). Each aspect of parental care (nest defence, provisioning, fecal removal) is likely to be influenced by a common physiological mechanism such as prolactin level elevation. Thus, prolactin level measurement may be a more accurate assessment of an individual motivational state to take care of the brood than the measurement of a single aspect of parental behaviour (Duckworth et al. 2003). Prolactin has been shown to mediate paternal behaviour in birds and to act in different ways to ensure sufficient provisioning of the young during the period of parental feeding dependence (Buntin 1996). For instance, Schoech et al. (1996) found a positive relationship between male feeding rate and their level of prolactin in Florida scrub-jays, (Aphelocoma c. coerulescens). Thus, it can be easily suggested that females paired with males with high prolactin levels may benefit from a higher contribution of their mates in nestling care. In addition, if male ornamentation reflects parental quality, we predict that prolactin concentration should correlate with ornamentation in males. Surprisingly, apart from one reported relationship between male prolactin levels and their plumage colour (Duckworth et al. 2003), no study has dealt with the link between male prolactin levels and the degree of expression of their secondary sexual characters.
Here, we assess the relationship between male parental ability and male bill colour and age in the blackbird (Turdus merula), a sexually dichromatic and socially monogamous species (Snow 1958; Desrochers and Magrath 1996). Plumage is entirely black in males while it is brownish and relatively cryptic in females. In addition, males exhibit coloured bills that vary from pale yellow to orange (Cramp 1988) due to carotenoid pigments (Faivre et al. 2003). Blackbirds exhibit biparental care, although the male’s contribution is the greatest after hatching, as incubation duties are essentially performed by the female (Snow 1958; Chamberlain et al. 1999). Recent studies indicate that bill colour is a sexually selected trait in blackbirds. Male bill colour has been shown to play a role in intrasexual selection: during interactions between males, birds with orange bills seem to represent a greater threat than those with yellow ones (Bright and Waas 2002). In addition, Faivre et al. (2001) found a positive correlation between female body condition (a crucial determinant of breeding success) and their mate’s bill colour. However, benefits that females may experience from pairing with orange-billed males remain poorly understood.
In the present study, we examine the relationship between male parental ability and male bill colour and age in the blackbird. Parental ability is determined directly from observations of the provisioning of chicks by both parents, and indirectly through the measurement of prolactin levels.
Methods
General field procedures
The study was conducted over five consecutive breeding seasons, from 1998 to 2002, on an urban population of colour-ringed blackbirds in Dijon (Burgundy, eastern France). There, blackbirds are habituated to human presence and thus can be watched easily without any disturbance. Birds were trapped with mist-nets during weekly capture sessions distributed through each year. All captured birds were ringed with one aluminium ring (under ringing licence from the Museum National d’Histoire Naturelle—CRBPO) and a unique combination of plastic colour rings. For each captured bird, a blood sample (0.2–0.3 ml) was taken from the brachial vein using heparinized capillary tube. The blood was centrifugated (4,000 rpm, 4°C) and the plasma stored at −20°C for prolactin assessment.
Breeding pairs were monitored each day during each breeding season from March to July. Nests were found by following females that were carrying nesting material and by weekly systematic searches for nests in bushes, hedges and trees in the park. Nests were inspected every 2 days for eggs and nestlings. For each breeding pair and breeding attempt, we recorded (1) the date of the first egg laying, (2) clutch size, (3) hatchling date, (4) brood size (number of nestlings during parental care measurements) and (5) fledging success (number of fledglings produced per brood).
Parental nest visits
For each breeding pair from 2000 to 2002, we estimated the visit rate of the male and the female attending first-brood nestlings aged 6–9 days for two 1-h periods during the 3 h following dawn and the 3 h before dusk. When breeding pairs attended several broods in the same year, only the first one was considered. Two observers positioned themselves at 20 m from the nest and used binoculars to record male and female visits to the nest. Time of nest-arriving and time of nest-departure were recorded. We defined male and female visit rate as the number of visits per hour. Relative visit rate was defined as female visit rate divided by total visit rate.
Male characters
Bill colour was assessed for each captured male, using a coloration index derived from the Yolk Colour Fan (ROCHE), and thus could range from 1 (pale yellow) to 15 (orange) (Faivre et al. 2001). Measurements of bill colour were highly repeatable within and between observers (see Faivre et al. 2003). Compared to plumage colour, bill colour is a dynamic character that may change over the breeding season (Ligon 1999). Thus, bill colour index determined during systematic captures within the framework of population monitoring were included in the analyses only if male capture occurred within 3 weeks of his nest visit observation. When males were not caught within this time interval, we performed localised capture around their nest just after parental visit observations in order to avoid disturbance in parental activities measurement. Yearling males (i.e. individuals that were born on the previous year) and adults (i.e. older birds) were determined using plumage characteristics. Indeed, primaries, primary coverts and secondary coverts are black in adults whereas these feathers are dark brown in yearlings (Cramp 1988; Svensson 1992).
Brood condition and fledging success
Nestling characteristics were determined after nest parental visit observation and, when possible, after male capture, between 8 and 11 days old. Nestlings were weighed using a balance (Ohaus, HSC 4010) with a precision of 0.1 g, and tarsus length was measured to the nearest 0.02 mm using callipers. Nestling body condition corresponded to the ratio between nestling mass and nestling tarsus length (Hatchwell et al. 2001). Both tarsus length and body mass increase slightly between 8 and 11 days old in blackbird nestlings (see Magrath 1992). Therefore, it is unlikely that ratio between nestling mass and tarsus length was strongly influenced by nestling age. Brood condition corresponded to the mean nestling conditions of the brood.
Fledging success was defined as the number of nestlings that fledged. Predation was an external factor that affected fledging success in several pairs. Such pairs were not included in the analyses in order to test only the influence of male characters on fledging success.
Prolactin assays
In male blackbirds, prolactin levels vary during the breeding cycle (mean ± SD = 25.648±8.5, n=17) and are twice higher during the period of care of offspring compared to the non-parental phase (mean ± SD = 10.974±12.026, n=65; t test: t=4.72, p<0.0001). From 1998 to 2001, 13 paired males were captured while provisioning nestlings or fledglings. For these 13 males, blood was collected within 30 min following their capture. Unlike others hormones (e.g. corticosterone) prolactin levels do not increase in response to an acute stressor (Schoech et al. 1996) such that time elapsed between capture and blood sampling has no influence on measurements. Plasma concentration of prolactin was determined by radioimmunoassays (see Lormée et al. 1999, 2000 for method) to investigate whether it correlated with male age and bill colour. In addition, the relationship between date of capture (expressed as the number of days following 1 January) and prolactin levels in males provisioning their young was checked to determine if prolactin levels varied with time.
Statistical procedures
Non parametric statistics (Siegel and Castellan 1988; Sokal and Rohlf 1995) were used for low sample sizes (n<15) and when data did not fit normality. Otherwise, parametric tests were performed. Multiple regressions were performed to assess the influence of male bill color and male age on visit rate and brood condition. Effects on fledging success were assessed using ordinal logistic regressions (Agresti 1990). Kendall rank order partial correlation tests (Siegel and Castellan 1988) were performed to assess the relationship between prolactin levels and fledging success, independently of bill colour.
Results
Parental nest visits
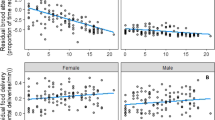
We observed parental effort in a total of 32 breeding pairs (12 in 2000, 9 in 2001 and 11 in 2002). As found by Chamberlain et al. (1999), there was no evidence for an effect of the observation period (morning vs evening) on the visit rate of either sex (Wilcoxon signed rank test: males: 2000: z=0.667, n=12, p=0.504; 2001: z=0.524, n=9, p=0.600; 2002: z=0.445, n=11, p=0.657; females: 2000: z=0.592, n=12, p=0.554; 2001: z=0.118, n=9, p=0.906; 2002: z=1.47, n=11, p=0.141). Hence, the mean visit rate is used hereafter for both males and females. Visit rate did not differ between years (Kruskal-Wallis test: males: H=1.31, n=32, p=0.519; females: H=0.18, n=32, p=0.913). Therefore, the 3 years were pooled for subsequent analyses. However, male and female visit rate increased with brood size (linear regression: males: r2=0.169, F1,30=6.13, p=0.019; females: r2=0.170, F1,30=6.16, p=0.018). To control for brood size effect, residual visit rates corresponded to the residuals taken from these linear regressions in the subsequent analyses. Male and female residual visit rates were correlated within pair (Fig. 1, Pearson correlation: r=0.573, n=32, p<0.001).
Among the 32 breeding pairs observed, male age and bill colour was determined for 23 pairs (9 in 2000, 6 in 2001 and 8 in 2002; 14 adults and 9 yearlings). Bill colour didn’t differ between years (H=3.95, n=23, p=0.140) nor between months (H=0.094, n=23, p=0.993) nor between male age classes (Mann-Whitney: U=57, n=23, p=0.680). Male age and age-bill colour interaction did not affect male residual visit rate, but male residual visit rate tended to correlate with bill colour (Table 1). Female residual visit rate was significantly influenced by the age and the bill colour of their mate and by their interaction (Table 1). Indeed, females showed higher visit rates when they were mated with adult males or with males exhibiting a higher bill colour index. However, the positive relationship between female visit rate and male bill colour was much stronger within yearling males (Fig. 2). Female relative visit rate did not vary with male age or bill colour.
Brood condition and fledging success
Among the 32 observed broods, two partially overlapping subsamples of 25 broods were described for nestling characteristics and fledging success respectively. Nestling description was impossible in 7 broods: two nests were inaccessible, while predation and nestling death suppressed respectively 4 and 1 broods between the completion of parental care observations and nestling description. Brood predation between parental care observations and fledging occurred in 7 nests (4 previously mentioned + 3 after nestlings measurement) thus reducing the analysis of fledging success to 25 broods.
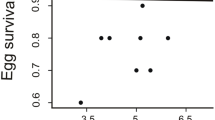
Among the 25 broods where nestling characteristics were recorded, male traits were known for 19 (12 adults and 7 yearlings). Brood size did not affect brood condition (H=2.11, n=25, p=0.551). Brood condition increased with male and female residual visit rates (Pearson correlations, males: r=0.422, n=25, p=0.036, females: r=0.555, n=25, p=0.004). Brood condition was affected by male age, but not by their bill colour (Table 2).
Indeed, broods reared by adult males were in better condition than broods reared by yearling males (Fig. 3).
Among the 25 broods not depredated before fledging, brood condition was known for 22 broods and male characters were assessed for 18 (12 adults and 6 yearlings). Fledging success was not significantly related to male and female residual visit rates (Ordinal logistic regression: males: r2=0.0018, χ2=0.155, n=25, p=0.694, females: r2=0.0124, χ2=1.05, n=25, p=0.307), neither to brood condition (r2=0.001, χ2=0.066, n=22, p=0.798). To analyse the effects of male age and bill colour on fledging success independently of brood size, brood size was included as a factor in the multiple ordinal logistic regression. Fledging success varied with male age and bill colour (Table 3). Indeed, adult and orange-billed males produced a larger number of fledglings (Fig. 4).
Prolactin levels
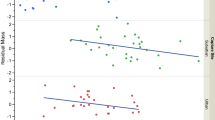
Among the 13 males analysed for prolactin levels, nest visit rate was known for 9, and brood size, brood condition and fledging success were known for 11. Prolactin levels in males provisioning their young did not vary with date of capture (Spearman correlation: rs=−0.311, n=13, p=0.300, Power=0.29). Male prolactin levels were not significantly related to brood size (rs=0.181, n=11, p=0.595, Power=0.15), neither to brood condition (rs=−0.173, n=11, p=0.612, Power=0.14) nor to male residual visit rate (rs=0.092, n=9, p=0.813, Power=0.13). Because male bill colour influenced fledging success (see before), this variable was held constant to assess the relationship between prolactin levels and fledging success. Fledging success did not vary with male prolactin levels (Kendall partial correlation: τ=0.186, n=11, p=0.460). Male age affected prolactin levels (Fig. 5a, U=4, n=13, p=0.031): adults (n=9) showed higher levels of prolactin than yearling males (n=4). In addition, prolactin levels increased significantly with male bill colour index (Fig. 5b, rs=0.582, n=13, p=0.037) independently of age since bill colour did not differ according to age (U=17, n=13, p=0.873).
Discussion
Our results show that male residual visit rate increased with bill colour index, and that female residual visit rate was positively related to their mate’s bill colour index and age. In addition, male and female residual visit rates were positively correlated, and both affected brood condition which was also higher for adult males. Finally, orange-billed and adult males had a greater number of fledglings and showed a higher level of prolactin.
These results are consistent with the “good parent hypothesis” (Hoelzer 1989) since males with more orange bills performed better during a breeding attempt: they showed an increased visit rate and had a greater number of fledglings. Despite the positive relationship between female visit rate and male bill colour, the “differential allocation hypothesis” is poorly supported by our results. Indeed, we found no evidence for a decrease of male visit rate with bill colour, nor for a negative correlation between male and female parental care. In our study, females may obtain direct benefits from choosing males with more orange bills. Indeed, carotenoid dependent colorations have been suggested to reflect foraging ability (Hill 1992; Senar and Escobar 2002) or individual health (Lozano 1994, 2001; Hatchwell et al. 2001; Faivre et al. 2003; Blount 2004). Hence, the higher fledging success of males with more orange bills may be a direct consequence of their greater ability at feeding nestlings. Contrary to other studies (Hill 1991; Palokangas et al. 1994; Keyser and Hill 2000; Siefferman and Hill 2003), we found no significant relationship between male coloration and visit rate. However, visit rate may not be the sole indicator of parental quality since it does not reflect load size or food quality (Sundberg and Larsson 1994; Saetre et al. 1995; Senar et al. 2002). In addition, contrary to Senar et al. (2002), male coloration and brood condition were not directly linked. However, we have shown an indirect link because males with more orange bills visited their chicks at a higher rate than yellow-billed males, and higher male visit rate led to higher brood condition.
A positive relationship between age and reproductive success has been previously discussed in term of increased paternal care with age. For instance, Woodard and Murphy (1999) showed that the differences found between experienced and inexperienced males eastern kingbirds (Tyrannus tyrannus) in provisioning rates of nestlings might be age-related. In addition, the ability to find and collect food constrains the number of offspring a pair can rear (Clutton-Brock 1991). In our study, adult males produced nestlings in better condition than yearling males and had a higher fledging success. This may be due to a greater foraging efficiency in adults, resulting in differences in the total amount of food provided, in the ability to exploit alternative resources during hard weather, or in the quality of food (Desrochers 1992; Sasvari et al. 2000). However, it is difficult to dissociate age and individual quality effects, particularly if quality is related to survival (Forslund and Pärt 1995; Møller et al. 2000).
Our study has shown that female visit rate was strongly dependent on their mate bill colour and age. This is not in accordance with the differential allocation hypothesis because we found no relationship between female relative visit rate and male bill colour, while male visit rate was positively linked with their bill colour. Other hypotheses that do not refute the good parent hypothesis could explain the observed situation. First, the assortative mating hypothesis suggested that pairing does not occur randomly and that male and female of the same age or of the same quality are likely to pair together (Burley 1983). The observed positive relationship between male and female visit rate supports this point of view (Filliater and Breitwisch 1997). In addition, Faivre et al. (2001) found that males having more orange bills were paired to females in better body condition. This suggests that the higher visit rate observed for females paired with adult males and males with more orange bills could be directly explained by their intrinsic quality. Second, the observed pattern could also result from a difference in territory quality. In several species, adult males and males with brighter carotenoid colorations acquired territory of higher quality (Forslund and Pärt 1995; Wolfenbarger 1999; Pryke et al. 2001), and the abundance of food near the nest could probably affect the cost of provisioning. In our case, blackbird females paired with adult and orange-billed males may have benefited from increased food availability and, thus, visited their offspring at a higher rate. Interestingly, a recent study has linked bill colour and territorial abilities in male blackbirds (Bright and Waas 2002). Third, females paired with yearling males showing lower bill colour index had a lower nest visit rate, a lower brood condition and a lower fledging success. It has been suggested that females not allowed to mate with preferred males and “forced” to pair with unattractive males produce fewer offspring of lower quality by reduction of their own reproductive effort (de Lope and Møller 1993). This may occur in blackbirds if females prefer adult males with more orange bills (Faivre et al. 2001; but see Préault et al. 2002; Bright and Waas 2002). Interestingly, the positive relationship between female feeding frequency and male bill colour index was stronger in yearling than in adult males. That suggests that bill colour may be particularly important to signal individual quality within inexperienced males.
From a more general point of view, our results do not exclude the good gene hypothesis (Searcy 1982). If male age and bill colour reflect differences in heritable male quality, then females paired with older and orange-billed males indirectly benefit by rearing higher quality offspring. The good parent hypothesis and the good genes hypothesis are thus not mutually exclusive and male ornament may act as an indicator of both heritable and direct benefits (Iwasa and Pomiankowski 1999).
Relationships between male characters and nesting output have been reported on several occasions (Norris 1990; Sundberg and Larsson 1994; Saetre et al. 1995; Siefferman and Hill 2003). In this study, nesting output was not an exact measurement of male reproductive success because extrapair fertilisations (EPF) were not considered whereas they may occur in blackbirds (Creighton 2000). Further studies are still required to investigate how EPF may modulate male fitness and female benefits in relation to parental care.
The proximal mechanism underlying such a relationship remains unclear. We suggest that prolactin may be the link between carotenoid-based coloration and intensity of paternal effort. Indeed, in our study, prolactin level was positively related to bill colour index and was higher in adult males. An age effect on prolactin levels has been shown in male dark-eyed juncos (Junco hyemalis) (Deviche et al. 2000). However, Duckworth et al. (2003) have shown that redder male house finches (Carpodacus mexicanus) had low levels of prolactin while dull males, which provisioned more frequently, showed high levels of prolactin. Unlike feathered ornaments, bills are continuously being replaced as they are worn away (Rawles 1960). Bill colour seems to be more sensitive to the environment compared to plumage colour and may therefore be a better signal of male condition or male current physiological state (Dale 2000; Bennett and Owens 2002; Blount et al. 2003; Faivre et al. 2003). Thus, although the link between prolactin and bill colour remains unclear, bill colour seems to honestly reveal male physiological adjustment to paternal activities during the parental phase.
We found no evidence of a direct link between paternal behaviour and prolactin levels in male blackbirds. At first sight, this result may be surprising, but currently the relation between prolactin level and parental care in birds remains unclear (Buntin 1996). This relationship may be age dependent because reproductive experience may lead to a transition from a hormonal to a nonhormonal mode of parental behaviour control (Buntin 1996). Thus, stimuli from young or cues associated with parent-young interactions may become behavioural regulators (Buntin 1996). Unfortunately, our sample size was too small to test this hypothesis. Furthemore, prolactin levels may be linked to another aspect of parental care such as quantity of food delivered to the nestlings, or nest defence and vigilance (Buntin 1996).
Further studies are thus required to detect the relative contribution of male quality and its signalling of brood condition and fledging success in blackbirds. In particular, a more detailed knowledge of the link between physiological control of paternal abilities and the expression of secondary sexual traits would be a key step to understand a crucial mechanism in sexual selection. For instance, future studies could focus the relationship between prolactin and carotenoid-based colours because (1) prolactin may control parental care, (2) carotenoids have signalling functions, and (3) both carotenoids and prolactin are related to immune defences (Bendich 1993; Morell 1995; Olson and Owens 1998). Although testosterone has been frequently invoked to mediate the link between the level of expression of male secondary sexual characters and immunocompetence, other hormones, such as prolactin, may also be involved. Furthermore, it could be particularly relevant to investigate factors determining parental care and the expression of secondary sexual traits showing short term variations because they seem more able to mirror male condition and abilities.
References
Agresti A (1990) Categorical Data Analysis. Wiley, New York
Andersson M (1994) Sexual Selection. Princeton University Press, Princeton
Bendich A (1993) Biological functions of Dietary Carotenoids. Ann NY Acad Sci 691:61–67
Bennett PM, Owens IPF (2002) Evolutionary ecology of birds: life histories, mating systems, and extinction. Oxford University Press, Oxford
Blount JD, Metcalfe NB, Birkhead TR, Surai PF (2003) Carotenoid modulation of immune function and sexual attractiveness in zebra finches. Science 300:125–127
Blount JD (2004) Carotenoids and life-history evolution in animals. Arch Biochem Biophys 430:10–15
Bright A, Waas JR (2002) Effects of bill pigmentation and UV reflectance during territory establishment in blackbirds. Anim Behav 64:207–213
Brooks R, Kemp DJ (2001) Can older males deliver the good genes? Trends Ecol Evol 16:308–313
Brush AH, Power DM (1976) House finch pigmentation: carotenoid metabolism and the effect of diet. Auk 93:725–739
Buntin JD (1996) Neural and hormonal control of parental behavior in birds. Adv Stud Behav 25:161–213
Burley NT (1983) The meaning of assortative mating. Ethol Sociobiol 4:191–203
Burley NT (1986) Sexual selection for aesthetic traits in species with biparental care. Am Nat 127:415–445
Burley NT (1988) The differential-allocation hypothesis: an experimental test. Am Nat 132:611–628
Burley NT, Johnson K (2002) The evolution of avian parental care. Phil Trans R Soc Lond B 357:241–250
Chamberlain DE, Hatchwell BJ, Perrins CM (1999) Importance of feeding ecology to the reproductive success of blackbirds Turdus merula nesting in rural habitats. Ibis 141:415–427
Clutton-Brock TH (1988) Reproductive Success. University of Chicago Press, Chicago
Clutton-Brock TH (1991) The Evolution of Parental Care. Princeton University Press, Princeton
Cramp S (1988) Birds of the western Palearctic, vol 5. Oxford University Press, Oxford
Creighton E (2000) Reproductive Strategies in the European Blackbird, Turdus merula. PhD thesis, Milton Keynes: The Open University
Dale J (2000) Ornamental plumage does not signal male quality in red-billed queleas. Proc R Soc Lond B 267:2143–2149
de Lope F, Møller AP (1993) Female reproductive effort depends on the degree of ornamentation of their mates. Evolution 47:1152–1160
Desrochers A (1992) Age and foraging success in European blackbirds: variation between and within individuals. Anim Behav 43:885–894
Desrochers A, Magrath RD (1996) Divorce in the European Blackbird: seeking greener pastures? In: Black JM (ed). Partnerships in birds. Oxford University Press, Oxford, pp 177–191
Deviche P, Wingfield JC, Sharp PJ (2000) Year-class differences in the reproductive system, plasma prolactin and corticosterone concentrations, and onset of prebasic molt in male Dark-eyed Juncos (Junco hyemalis) during the breeding period. Gen Comp Endocrinol 118:425–435
Duckworth R, Badyaev A, Parlow A (2003) Elaborately ornamented males avoid costly parental care in the house finch (Carpodacus mexicanus): a proximate perspective. Behav Ecol Sociobiol 55:176–183
Faivre B, Préault M, Théry M, Secondi J, Patris B, Cézilly F (2001) Breeding strategies and morphological characters in an urban population of blackbirds, Turdus merula. Anim Behav 61:969–974
Faivre B, Grégoire A, Préault M, Cézilly F, Sorci G (2003) Immune activation rapidly mirrored in a carotenoid-based secondary sexual trait. Science 300:103
Filliater TS, Breitwisch R (1997) Nestling provisioning by the extremely dichromatic northern cardinal. Wilson Bull 109:145–153
Forslund P, Pärt T (1995) Age and reproduction in birds-hypotheses and tests. Trends Ecol Evol 10:374–378
Fox DL, Vevers G (1960) The Nature of Animal Color. University of Washington Press, Seattle
Gray DA (1996) Carotenoids and sexual dichromatism in North American passerine birds. Am Nat 148:453–480
Hamilton WD, Zuk M (1982) Heritable true fitness and bright birds: a role for parasites? Science 218:384–387
Hatchwell BJ, Wood MJ, Ali Anwar M, Chamberlain DE, Perrins CM (2001) The haematozoan parasites of common Blackbirds Turdus merula: associations with host condition. Ibis 143:420–426
Hill GE (1991) Plumage coloration is a sexually selected indicator of male quality. Nature 350:337–339
Hill GE (1992) Proximate basis of variation in carotenoid pigmentation in male house finches. Auk 109:1–12
Hill GE (2002) A red bird in a brown bag: the function and evolution of colorful plumage in the House Finch. Oxford University Press, Oxford
Hoelzer GA (1989) The good parent process of sexual selection. Anim Behav 38:1067–1078
Hõrak P, Vellau H, Ots I, Møller AP (2000) Growth conditions affect carotenoid-based plumage coloration of great tit nestlings. Naturwissenschaften 87:460–464
Iwasa Y, Pomiankowski A (1999) Good parent and good genes models of handicap evolution. J Theor Biol 200:97–109
Keyser AJ, Hill GE (2000) Structurally based plumage coloration is an honest signal of quality in male blue grosbeaks. Behav Ecol 11:202–209
Ligon JD (1999) The Evolution of Avian Breeding Systems. Oxford University Press, Oxford
Linville SU, Breitwisch R (1997) Carotenoid availability and plumage coloration in a wild population of northern cardinals. Auk 114:796–800
Lormée H, Jouventin P, Chastel O, Mauget R (1999) Endocine correlates of parental care in an antarctic winter breeding seabird, the Emporor Penguin, Aptenodytes forsteri. Horm Behav 35:9–17
Lormée H, Jouventin P, Lacroix A, Lallemand J, Chastel O (2000) Reproductive endocrinology of tropical seabirds: sex-specific patterns in LH, steroids, and prolactin secretion in relation to parental care. Gen Comp Endocrinol 17:413–426
Lozano GA (1994) Carotenoids, parasites, and sexual selection. Oikos 70:309–311
Lozano GA (2001) Carotenoids, immunity, and sexual selection: comparing apples and oranges? Am Nat 158:200–203
Magrath RD (1992) The effect of egg mass on the growth and survival of blackbirds: a field experiment. J Zool Lond 227:639–653
Møller AP (1994) Sexual Selection and the Barn Swallow. Oxford University Press, Oxford
Møller AP, Biard C, Blound JD, Houston DC, Ninni P, Surai PF (2000) Carotenoïd-dependent signals: Indicators of foraging efficiency, immunocompetence or detoxification ability? Poult Avian Biol Rev 11:137–159
Morell V (1995) Zeroing in on how hormones affect immune system. Science 269:773–775
Murton RK, Westwood NJ (1977) Avian breeding cycles. Clarendon, Oxford
Norris K (1990) Female choice and quality of parental care in the great tit Parus major. Behav Ecol Sociobiol 27:275–281
Olson VA, Owens IPF (1998) Costly sexual signals: are carotenoids rare, risky or required? Trends Ecol Evol 13:510–514
Palokangas P, Korpimäki E, Hakkarainen H, Huhta E, Tolonen P, Alatalo RV (1994) Female kestrels gain reproductive success by choosing ornamented males. Anim Behav 47:443–448
Préault M, Deregnaucourt S, Sorci G, Faivre B (2002) Does beak coloration of male blackbirds play a role in intra and/or intersexual selection? Behav Process 58:91–96
Price T, Schluter D, Heckman NE (1993) Sexual selection when the female directly benefits. Biol J Linn Soc 48:187–211
Pryke SR, Lawes MJ, Andersson S (2001) Agonistic carotenoid signalling in male red-collared widowbirds: aggression related to the colour signal of both the territory owner and model intruder. Anim Behav 62:695–704
Rawles ME (1960) The integumentary system. In: Marshall AJ (ed). Biology and Comparative Physiology of Birds. Academic Press, New York, pp 189–240
Saetre GP, Fossnes T, Slagsvold T (1995) Food provisionning in the pied flycatcher: do female gain direct benefits from choosing bright-coloured males? J Anim Ecol 64:21–30
Sasvari L, Hegyi Z, Csörgö T, Hahn I (2000) Age-dependent diet change, parental care and reproductive cost in tawny owls Strix aluco. Acta Oecol 21:267–275
Schoech SJ, Mumme RL, Wingfield JC (1996) Prolactin and helping behaviour in the cooperatively breeding Florida scrub-jay, Aphelocoma c. coerulescens. Anim Behav 52:445–456
Searcy WA (1982) The evolutionary effects of mate selection. Annu Rev Ecol Syst 13:57–85
Senar JC, Escobar D (2002) Carotenoid derived plumage coloration in the siskin Carduelis spinus is related to foraging ability. Avian Sci 2:19–24
Senar JC, Figuerola J, Pascual J (2002) Brighter yellow blue tits make better parents. Proc R Soc Lond B 269:257–261
Shadd CA, Ritchison G (1998) Provisioning of nestlings by male and female yellow-breasted chats. Wilson Bull 110:398–402
Sheldon BC (2000) Differential allocation: tests, mechanism and implications. Trends Ecol Evol 15:397–402
Siefferman L, Hill GE (2003) Structural and melanin coloration indicate parental effort and reproductive success in male eastern bluebirds. Behav Ecol 14:855–861
Siegel S, Castellan NJ (1988) Nonparametric Statistics for the Behavioral Sciences. McGraw-Hill, New-York
Snow DW (1958) A Study of Blackbirds. Allen & Unwin, London
Sokal RR, Rohlf FJ (1995) Biometry. Freeman, New York
Sundberg J, Larsson C (1994) Male coloration as an indicator of parental quality in the yellowhammer Emberiza citrinella. Anim Behav 48:885–892
Svensson L (1992) Identification guide to european passerines. British Trust for Ornithology, Stockholm
Trivers RL (1972) Parental investment and sexual selection. In: Campbell B (ed). Sexual Selection and the Descent of Man,1871–1971. Heinemann, London, pp 136–179
Westneat DF, Sargent C (1996) Sex and parenting: the effects of sexual conflict and parentage on parental strategies. Trends Ecol Evol 11:87–91
Wolfenbarger L (1999) Red coloration of male northern cardinals correlates with male quality and territory quality. Behav Ecol 10:80–90
Woodard JD, Murphy MT (1999) Sex roles, parental experience and reproductive success of eastern kingbirds, Tyrannus tyrannus. Anim Behav 57:105–115
Acknowledgements
We thank the C.R.B.P.O., the ville de Dijon and Jardin Botanique de la ville de Dijon for permissions. This study was supported by the GDR CNRS 2155 “Ecologie Comportementale”. André Lacroix has done an invaluable technical work. We would like to thank Manuel Soler and the two anonymous referees for valuable comments that improved the mansucript. This work would not have been possible without technical assistance in the field from Mathias Barot, Romain Barot, Marco Barroca, Anne Besson, Claire Carvin, Claire Chalopin, Séverine Clair, Arnaud Grégoire, Luc Guillaume and Laurence Torcel. This work complies with French laws governing bird capture and experiments on animals
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by M. Soler
Rights and permissions
About this article
Cite this article
Préault, M., Chastel, O., Cézilly, F. et al. Male bill colour and age are associated with parental abilities and breeding performance in blackbirds. Behav Ecol Sociobiol 58, 497–505 (2005). https://doi.org/10.1007/s00265-005-0937-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00265-005-0937-3