Abstract
Objective
The purpose of this study was to compare the clinical and radiological efficacy of autologous adipose-derived stromal vascular fraction (SVF) versus hyaluronic acid in patients with bilateral knee osteoarthritis.
Methods
Sixteen patients with bilateral symptomatic knee osteoarthritis (K-L grade II to III; initial pain evaluated at four or greater on a ten-point VAS score) were enrolled in this study, which were randomized into two groups. Each patient received 4-ml autologous adipose-derived SVF treatment (group test, n = 16) in one side of knee joints and a single dose of 4-ml hyaluronic acid treatment (group control, n = 16) in the other side. The clinical evaluations were performed pre-operatively and post-operatively at one month, three months, six months, and 12-months follow-up visit, using the ten-point visual analog scale (VAS), the Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC), and the knee range of motion (ROM). The whole-organ assessment of the knees was performed with whole-organ magnetic resonance imaging score (WORMS) based on MRI at baseline, six months and 12-months follow-up. The articular repair tissue was assessed quantitatively and qualitatively by magnetic resonance observation of cartilage repair tissue (MOCART) score based on follow-up MRI at six months and 12 months.
Results
No significant baseline differences were found between two groups. Safety was confirmed with no severe adverse events observed during 12-months follow-up. The SVF-treated knees showed significantly improvement in the mean VAS, WOMAC scores, and ROM at 12-months follow-up visit compared with the baseline. In contrast, the mean VAS, WOMAC scores, and ROM of the control group became even worse but not significant from baseline to the last follow-up visit. WORMS and MOCART measurements revealed a significant improvement of articular cartilage repair in SVF-treated knees compared with hyaluronic acid-treated knees.
Conclusion
The results of this study suggest that autologous adipose-derived SVF treatment is safe and can effectively relief pain, improve function, and repair cartilage defects in patients with knee osteoarthritis.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Osteoarthritis (OA) results from degeneration of joint cartilage and subchondral bone and is one of the leading causes of joint pain and disability [1, 2]. The knee is the most frequently involved weight-bearing joint [3]. As a “wear to tear” disease, OA is associated with significant morbidity and healthcare expenditure [4, 5]. Many treatment modalities for knee OA such as lifestyle modification, pharmaceutical, and surgery have been advocated [6]. Intra-articular injection of hyaluronic acid (HA) is effective in improving symptoms and slowing down the progression of OA [7, 8], but fail to reverse or repair the degenerative cartilage or bone [9].
Regenerative cell therapies for knee OA such as adipose-derived stromal vascular fraction (SVF) have been recently investigated [10,11,12,13,14]. Adipose-derived stromal cells (ADSC) included in SVF have the potential of differentiating into adipogenic, osteogenic, chondrogenic, and other mesenchymal lineages, and have been widely applied to knee OA research for their immunomodulatory, anti-inflammatory and paracrine effects [15, 16]. Several recent studies showed the feasibility and safety of ADSC treatments, and it should be an ideal therapeutic option for knee OA [17,18,19,20,21]. However, cell expansion greatly increases the hospitalization costs. Unlike ADSC, SVF can be readily obtained from the lipoaspirate samples without the need for any cell separation or culturing conditions, which make it more cost efficient and convenient. There is a dearth of literature in the area of SVF treatments for knee OA, few clinical trials have been performed except several case reports. In addition, most of these published clinical trials failed to blind for both the participants and the outcome assessor because of the liposuction and other additional intervention procedures [10, 13, 18, 22, 23], which would lead to a high risk of performance bias. Finally, we designed a double-blind, randomized, self-controlled trial to compare the clinical and radiological efficacy of autologous adipose-derived SVF versus hyaluronic acid treatment among patients with grade II/III knee osteoarthritis of bilateral knee.
Materials and methods
Patients and study design
This trial’s protocol was approved by Ethics Committees of Zhejiang Provincial People’s Hospital before first patient’s enrollment; all patients were provided a written informed consent voluntarily. Eligible patients were 18–70 years of age with bilateral symptomatic knee osteoarthritis of grade II to III according to Kellgren-Lawrence criteria [24] and had an initial pain evaluated at four or greater on a ten-point visual analog scale (VAS) in bilateral knee joints. More details of inclusion and exclusion criteria were listed in Table 1.
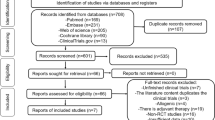
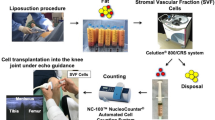
Before the study, the sample size was estimated on the basis of the results from our pilot study to obtain a power of 80% with α risk = 0.05. From January 2015 to June 2016, 16 patients (32 knees) were enrolled in this study. Three of them were male, and 13 of them were female. The completely randomization process was finished by an assistant accountant who was blinded to the patients’ data using SPSS 20.0 software (IBM Corporation, NY, US). First, we listed 1–16 serial numbers (patient serial number) in accordance with the outpatient order. Second, 16 random numbers were generated by RV.UNIFORM (0, 1) in the computer that matched number-by-number with 16 patients’ serial numbers. Third, the 16 random numbers were arrayed in ascending order; the corresponding patients of first eight random numbers were injected with 4-ml SVF in the left knee and 4-ml hyaluronic acid (SOFAST, Freda, china) in the right knee. The last eight patients were intervened with 4-ml hyaluronic acid (SOFAST, Freda, china) in the left knee and 4-ml SVF in the opposite. All SVF-treated knees formed the test group. By contrast, another 16 knees exposed with hyaluronic acid formed the control group. More details were shown in Fig. 1. All injections were done under the guidance of knee arthroscopy.
Five investigators were included in the protocol for clinical evaluation, corresponding to pre-operation (1 week before operation; baseline), and one, three, six and 12-months post-operation respectively. At each visit, patients were carefully evaluated using the visual analog scale (VAS), Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC), as well as range of motion (ROM) measurement, and magnetic resonance imaging (MRI) examination (1 week pre-operation, baseline; 6 months and 12-months post-operation).
Except for the orthopedic surgeon, all patients, radiologists, and investigators were blind to treatment allocation of the participants. The orthopaedic surgeon who delivered the intervention did not take outcome measurements.
Preparation of SVF and cell counting
All patients were fasted of at least six hours and water deprivation of at least two hours before operation, general anaesthesia was performed in supine position after checking the patients’ information by operator, anaesthetist, and circulating nurse. Liposuction was performed by one regular skilled plastic surgeon, who was blind to patients’ information. After sterilizing on abdominal and both lower extremities skin, two small incisions about 5 mm were made around umbilicus, and a target volume of approximately 100 to 150 cc of lipoaspirate was harvested through superwet technique from the subcutaneous layer around umbilicus. The incisions were closed with sutures but not tightened to allow more drainage of the blood-tinged tumescent fluid. Abdominal binder was used after operation to prevent bruising in the surgical area.
The harvest adipose tissue was immediately put into a sterile container which was packaged in a portable cryopreservation box on the way to the laboratory. The lipoaspirate was washed twice with 37 °C phosphate buffered saline (PBS), and the residual blood cells and tissue fragments were removed by the mesh filter. Equal volume of type I collagenase (Worthington, Lakewood, NJ, USA) was added into the washed adipose tissue for digestion. The mixture was then placed in a shaking incubator at 37 °C for 30 minutes. After enzymolysis, the tube was centrifuged at 1000 g for 10 min (Eppendorf 5810R, Germany).The supernatant was discarded, and the remnant SVF pellet at the bottom was resuspended in PBS reaching a volume of 4.5-ml SVF. A 0.5-mL sample of the final product was collected for cell counting, and the cell quantity and viability was measured through an automatic cell counter (Countstar IC1000, China).
Surgical procedures and injection
While the adipose processing was going on, arthroscopic debridement was performed in bilateral knee joint by a single orthopaedic surgeon. After a standard arthroscopic examination, all unstable cartilage around the lesion was debrided to form a stabile circumstance of the cartilage. Once the SVF processing was accomplished, SVF and HA were injected under arthroscopic guidance, after the arthroscopic fluid was drained. In the test group, about 4 ml of SVF suspension was injected into the cartilage lesion surface. The contralateral knee received 4 ml of HA injection. Incisions subcuticular suture and pressure dressing after injection were confirmed. All the procedures were done under general anesthesia that the patients themselves were blind about the injection allocation.
Post-operative protocol
All patients were instructed to be non-weight bearing for one day after operation and were discharged two days post-operation with the same health propaganda. Regular daily activities were allowed during follow-up period, and all participants should contact the doctor in charge immediately once there was any sign of adverse event, including fever; cutaneous pruritus, and erythra; swelling, pyorrhea, or fissuration of the incisions. Additionally, a dosage of 200-mg Celebrex twice daily for 2 days was applied as a discharge medication, when patients complained about incision pain with an evaluation over five on a VAS scale on the discharged day. These patients were followed via telephone until the incision pain was relieved.
Clinical evaluation
Pain and functional limitation were evaluated using VAS and WOMAC questionnaire. The WOMAC measures five items for pain (score range 0–20), two for stiffness (score range 0–8), and 17 for functional limitation (score range 0–68) with a total score range from 0 (slightest) to 96 (worst). While functional limitation cannot be scored per joint, pain and stiffness were measured per joint separately by two copies of the questionnaires. In addition, ROM of bilateral knee joints was also recorded.
MRI assessment
The protocol required three MRI scan: baseline (1 week before operation), six months, and 12 months of follow-up. Each MRI was performed using SIEMENS 3.0 T Skyra MRI device, with the 15-channel knee coil. The patients lay supine 30 mintes to reduce the influence of the knee motion and weight bearing to the results of scanning. The following sequences were applied: PDWI-FS images in the sagittal, coronal, and transverse planes; T1 W1 images in the sagittal planes. All data were transmitted to Siemens post-processing workstation, two trained radiologists blinded to each other completed the measurement and recording, and finally obtained a consensus conclusion. The whole-organ assessment of the knees was performed by whole-organ magnetic resonance imaging score (WORMS) [25]. The cartilage repair tissue was assessed by magnetic resonance observation of cartilage repair tissue (MOCART) score (include 9 variables) [26].
Statistical analysis
All data are presented as means ± SD. We used SPSS software (version 20.0, IBM Corporation, NY, US) for all data calculation. Within group analysis of follow-up statistics (VAS, WOMAC score, ROM, and WORMS) were compared with baseline using the paired t test, and the independent t test was used to compare data at same follow-up time point between groups. The discrete data were analyzed by chi-square test. Differences with P < 0.05 were considered statistically significant.
Results
Patient characteristics
A total of 32 knees from 16 patients with bilateral knee OA were randomly allocated to the group test (knee received SVF treatment) and group control (knee received HA treatment) (Fig. 1). The patients characteristics showed no significant difference in age, gender distribution, and BMI, and preferred leg distribution between patients received SVF therapy in the left knee and patients received SVF therapy in the right knee (Table 2). No relevant baseline differences in symptom duration time, Kellgren-Lawrence OA grade, VAS score, WOMAC pain and stiffness, knee ROM, and WORMS between two groups were observed (Tables 3 and 5). In addition, there was no significant difference in preferred leg proportion between the group test, and group control showed (P > .05), which diminished the influence of preferred leg in the treatment and follow-up.
Safety
Four patients (25%) complained about pain of the abdomen, like muscle soreness after strenuous exercise, sustained about one week after liposuction. Six patients (37.5%) reported pain and swelling in bilateral knee joints that continued for a few days after knee surgery and all resolved within two weeks. The pain reported above all responded well to Celebrex. There were no other adverse events related to the knee surgery (including infection, allergy, and poor wound healing) and adipose harvest (including deformity and severe ecchymosis).
Clinical outcome
Mean changes of clinical scores from baseline to one month, three months, six months, and 12 months were summarized in Fig. 2 and Table 4. In the test group, all scores including VAS, WOMAC pain, WOMAC stiffness, and knee ROM significantly improved at one month, three months, six months, and 12-months follow-up visits as compared with baseline (Fig. 2). The mean VAS, WOMAC pain, WOMAC stiffness, and ROM in the test group improved by 3.19 ± 0.98, 8.00 ± 4.77, 2.25 ± 2.11, and 19.06 ± 7.76, respectively, between baseline and last follow-up (Table 4). In the control group, pain (VAS score) was significantly relieved by one month and three months after HA injection, but was amplified again at six and 12-months visits, from 5.75 ± 1.24 to 5.81 ± 1.33 (P = 0.791) and 5.81 ± 1.83 (P = 0.835) (Fig. 2a). Functional improvement of ROM was significant at one month after HA therapy (P < 0.001). However, this trend even took a turn for the worse after three months post-operation in the control group (decreased by 1.88 ± 6.40 from baseline to last follow-up, not significantly) (Fig. 2b). Unlike the SVF treated group, the general tendency of WOMAC pain and stiffness subscores towards worsening in the control group showed significant differences compared with the test group, as showed in Fig. 2c and Fig. 2d.
Changes of VAS, WOMAC score, and knee ROM in two groups during 12-months follow-up. Values in graphs are expressed as mean ± SD in vertical bars, **P < 0.01, ***P < 0.001, ns, non-significant (P > 0.05). All values were compared with baseline. a VAS score. b Knee ROM. c WOMAC pain. d WOMAC stiffness
Radiologic evaluation
The whole-organ assessment of the knees was performed with WORMS based on MRI at baseline, six months and 12-months follow-up (Tables 5 and 6). In the test group, WORMS showed an important improvement that the mean WORMS decreased by 11.38 ± 24.89 (P = 0.088) and 15.44 ± 21.95 (P < 0.05) from baseline to six and 12 months, respectively. By contrast the consequence in the control group was poor, WORMS deteriorated by 12.81 ± 12.66 (P < 0.01) and 15.50 ± 14.65 (P < 0.01) from baseline to six and 12 months, respectively. The repair of the articular cartilage defects was measured by MOCART system based on the MRI results at six and 12-months follow-up, details were shown in Table 7. In the test group, the mean MOCART score was 54.06 ± 11.58 at six months visit and was 62.81 ± 8.16 at 12-months follow-up, showing a significant improvement (P < 0.01). However, the mean MOCART, in the control group was poor in both six months (19.38 ± 9.64) and 12 months (19.06 ± 7.79), showed no improvement from six months to 12 months in the HA treated group (P = 0.924). It is remarkable that the MOCART in the test group was significantly better than that in the control group, both at six and 12-months MRI follow-up (P < 0.001). In addition, in the test group, there were 11(69%) knees that showed complete or hypertrophic repair tissue filling of the defect compared with only one (6%) knee in the control group, seven (44%) knees in the test group showed complete integration with adjacent cartilage, and the value in the control group is only one (6%) (Fig. 3).
Discussion
In this paper, we reported our findings comparing SVF versus HA treatment for 16 pairs of knees with K-L grade II-III osteoarthritis, with 12-months follow-up. Our data demonstrated that SVF could provide effective improvements in both radiological (WORMS and MOCART), and clinical (include VAS, WOMAC pain and stiffness, knee ROM) outcomes which was significantly superior to HA treatment (single dose of 40 mg) for bilateral knee joints with osteoarthritis at II-III stage (K-L grade). In a multi-centre analysis among 2372 patients underwent MSC treatment, the major adverse event was pain post-procedure [27]. Except pain and swelling after liposuction and operation, there was no severe adverse event in the whole process of our study.
In the test group treated with SVF, the knee joints showed statistically significant improvements in the mean VAS, ROM, WOMAC pain, and stiffness compared with baseline after 12-months follow-up, but the mean VAS score of 12-months visit increased significantly (p = 0.015) compared with that of six months. We found these patients with increased VAS score of 12 months in the test group; all had a gradually aggravating the VAS score of the knee in the control group. When checking the history, we found that these patients were used to load more weight on the milder knee rather than the most severe knee, which may explain the worsening trend of the VAS score from six months to 12 months in the test group. From the previous literature, we knew that HA treatment was effective in ameliorating pain and symptoms for OA studied and often served as a control [28, 29]. In our study, we used a single dose of 40-mg hyaluronic acid (SOFAST, Freda) injection in the control group for a better blind and variable control, but the outcome indicated that the therapeutic effect of one-single dose of 40-mg HA injection (SOFAST, Freda) was not obvious in the intermediate and long-term follow-up. This result was different from the study of Vega et al. [28]. They used a single dose of hyaluronic acid (60 mg in 3 mL; Durolane) as control, and the VAS score was significantly improved at 12-months follow-up in the control group. More research comparing SVF and adequate course of HA treatment for knee OA is needed in the future.
The MRI follow-up showed a significant improvement of the WORMS in knees treated with SVF. Particularly notable was the reduction in the cartilage and marrow abnormality subscores, which decreased by 12 ± 21.55 (P < 0.05) and 2.50 ± 2.00 (P < 0.001) from baseline to 12-months MRI. The radiological outcome of MOCART proved that the test group had a statistically significant superior articular cartilage repair both at six months (mean MOCART 54.06 ± 11.58 in the test group and 19.38 ± 9.64 in the control group, P < 0.001) and 12-months (mean MOCART 62.81 ± 8.16 in the test group and 19.06 ± 7.79 in the control group, P < 0.001) MRI follow-up, compared with the control group (Table 7). In the group treated with SVF, four knees had a MOCART score of less than 60 at last follow-up; all accompanied with a poor subchondral lamina and bone as well as a large area of cartilage defect on baseline MRI, suggesting that SVF injection provided a less satisfactory outcome in relatively large cartilage defects. Different from the test group, the MRI outcome in the control group was poor, as the previous literature indicated that hyaluronic acid played a limited role in the repair of damaged cartilage. Furthermore, several other researches studied the relationship between cell dose and therapeutic efficacy of ADSC [18,19,20,21], but came to contradictory results. In the two year follow-up study of Jo CH et al.[18, 19], significant improvement was found mainly in the high-dose group (1 × 108), and the outcomes in the low and medium dose groups tended to deteriorate after one year; whereas, those in the high-dose group plateaued until two years. Interestingly, in another clinical trial of ADIPOA [21], significant improvement was detected only in the low-dose (2 × 106) ASCs-treated patients. In another pilot study treated with repeated injections of ADMSCs, the dosage of 5 × 107 showed the highest improvement [20]. In our study, we failed to find an actual association between SVF cell density, cell viability, and outcomes that we need more studies to explore the cell dose effect in the future. There are multiple sources of stem cells for orthopedic conditions [30,31,32]. Since adipose tissue-derived stem cells (ADSCs) were first characterized by Zuk et al. in 2001 [16], ADSCs have been widely studied for their regenerative and therapeutic potential. Recently, several researches indicated that the regenerative potential was also found in the SVF [33,34,35], a mixture of ADSCs, endothelial precursor cells (EPCs), endothelial cells (ECs), macrophages, smooth muscle cells, lymphocytes, pericytes, and pre-adipocytes [36, 37]. Traditionally, SVF is isolated by enzymatic processing from lipoaspirate. The advantages of SVF over ADSCs consist of the following parts. Firstly, unlike ADSCs, SVF is readily accessible from the lipoaspirate without the requirement for any cell separation or cell culture. Secondly, SVF therapy is much cheaper and faster than ADSCs because of the absence of culturing procedures. Thirdly, besides the similarities in immunomodulation, anti-inflammatory, and angiogenesis, the characteristic, heterogeneous cellular components of SVF may explain the better therapeutic effect observed in some animal studies [36, 38].
As far as we know, this was the first prospective, randomized, double-blind, and self-controlled clinical trial studying autologous adipose-derived stromal vascular fractions injection for bilateral human knee osteoarthritis. The study was designed according to the principle of completely random, minimizing the distinctions between two groups and reducing the interference of the preferred leg. The setting of self-control between bilateral knees ensured the consistency of sample size between groups during the follow-up process. All procedures were performed under general anaesthesia, minimizing the pain of the patients. Furthermore, adequate blinding was guaranteed in our study, all patients, radiologists, and investigators were blind of treatment allocation, and the orthopedic surgeon who delivered the intervention did not take outcome measurements, reducing the performance bias of the study.
In conclusion, our results indicates that autologous adipose-derived SVF treatment is safe and can effectively relief pain, improve function, and repair cartilage defects in patients with K-L grade II-III knee osteoarthritis. It is therefore believed that adipose tissue may be a good cell source for cartilage regenerative engineering.
Limitations of the study
We must acknowledge that there were several limitations in this study. First, the follow-up period seemed short (12 months); we need more follow-up time to determine the long-term effects of SVF. Second, the sample size was small because the incidence of bilateral knee osteoarthritis was lower than unilateral knee OA. Third, second-look arthroscopy and pathological biopsy of newborn cartilage tissue is the gold standard for evaluating cartilage repair; however, arthroscopy and biopsy are invasive and inconvenient for dynamic follow-up, and therefore difficult to carry out in China. Fourth, we could not find a clinical rating index aiming at unilateral knee joint that patients should complete two same questionnaires focusing on the individual characteristics with different sides of knees. Fifth, it is unknown, whether SVF injection in one knee could influence the contralateral knee. Sixth, we did not find an actual association between SVF cell density, cell viability, and outcomes, more studies are needed to explore the cell dose effect of SVF treatment.
References
Arden N, Nevitt MC (2006) Osteoarthritis: epidemiology. Best Pract Res Clin Rheumatol 20(1):3–25. https://doi.org/10.1016/j.berh.2005.09.007
Buckwalter JA, Martin J, Mankin HJ (2000) Synovial joint degeneration and the syndrome of osteoarthritis. Instr Course Lect 49:481–489
Vos T, Flaxman AD, Naghavi M, Lozano R, Michaud C, Ezzati M, Shibuya K, Salomon JA, Abdalla S, Aboyans V, Abraham J, Ackerman I, Aggarwal R, Ahn SY, Ali MK, Alvarado M, Anderson HR, Anderson LM, Andrews KG, Atkinson C, Baddour LM, Bahalim AN, Barker-Collo S, Barrero LH, Bartels DH, Basanez MG, Baxter A, Bell ML, Benjamin EJ, Bennett D, Bernabe E, Bhalla K, Bhandari B, Bikbov B, Bin Abdulhak A, Birbeck G, Black JA, Blencowe H, Blore JD, Blyth F, Bolliger I, Bonaventure A, Boufous S, Bourne R, Boussinesq M, Braithwaite T, Brayne C, Bridgett L, Brooker S, Brooks P, Brugha TS, Bryan-Hancock C, Bucello C, Buchbinder R, Buckle G, Budke CM, Burch M, Burney P, Burstein R, Calabria B, Campbell B, Canter CE, Carabin H, Carapetis J, Carmona L, Cella C, Charlson F, Chen H, Cheng AT, Chou D, Chugh SS, Coffeng LE, Colan SD, Colquhoun S, Colson KE, Condon J, Connor MD, Cooper LT, Corriere M, Cortinovis M, de Vaccaro KC, Couser W, Cowie BC, Criqui MH, Cross M, Dabhadkar KC, Dahiya M, Dahodwala N, Damsere-Derry J, Danaei G, Davis A, De Leo D, Degenhardt L, Dellavalle R, Delossantos A, Denenberg J, Derrett S, Des Jarlais DC, Dharmaratne SD, Dherani M, Diaz-Torne C, Dolk H, Dorsey ER, Driscoll T, Duber H, Ebel B, Edmond K, Elbaz A, Ali SE, Erskine H, Erwin PJ, Espindola P, Ewoigbokhan SE, Farzadfar F, Feigin V, Felson DT, Ferrari A, Ferri CP, Fevre EM, Finucane MM, Flaxman S, Flood L, Foreman K, Forouzanfar MH, Fowkes FG, Franklin R, Fransen M, Freeman MK, Gabbe BJ, Gabriel SE, Gakidou E, Ganatra HA, Garcia B, Gaspari F, Gillum RF, Gmel G, Gosselin R, Grainger R, Groeger J, Guillemin F, Gunnell D, Gupta R, Haagsma J, Hagan H, Halasa YA, Hall W, Haring D, Haro JM, Harrison JE, Havmoeller R, Hay RJ, Higashi H, Hill C, Hoen B, Hoffman H, Hotez PJ, Hoy D, Huang JJ, Ibeanusi SE, Jacobsen KH, James SL, Jarvis D, Jasrasaria R, Jayaraman S, Johns N, Jonas JB, Karthikeyan G, Kassebaum N, Kawakami N, Keren A, Khoo JP, King CH, Knowlton LM, Kobusingye O, Koranteng A, Krishnamurthi R, Lalloo R, Laslett LL, Lathlean T, Leasher JL, Lee YY, Leigh J, Lim SS, Limb E, Lin JK, Lipnick M, Lipshultz SE, Liu W, Loane M, Ohno SL, Lyons R, Ma J, Mabweijano J, MacIntyre MF, Malekzadeh R, Mallinger L, Manivannan S, Marcenes W, March L, Margolis DJ, Marks GB, Marks R, Matsumori A, Matzopoulos R, Mayosi BM, McAnulty JH, McDermott MM, McGill N, McGrath J, Medina-Mora ME, Meltzer M, Mensah GA, Merriman TR, Meyer AC, Miglioli V, Miller M, Miller TR, Mitchell PB, Mocumbi AO, Moffitt TE, Mokdad AA, Monasta L, Montico M, Moradi-Lakeh M, Moran A, Morawska L, Mori R, Murdoch ME, Mwaniki MK, Naidoo K, Nair MN, Naldi L, Narayan KM, Nelson PK, Nelson RG, Nevitt MC, Newton CR, Nolte S, Norman P, Norman R, O’Donnell M, O’Hanlon S, Olives C, Omer SB, Ortblad K, Osborne R, Ozgediz D, Page A, Pahari B, Pandian JD, Rivero AP, Patten SB, Pearce N, Padilla RP, Perez-Ruiz F, Perico N, Pesudovs K, Phillips D, Phillips MR, Pierce K, Pion S, Polanczyk GV, Polinder S, Pope CA 3rd, Popova S, Porrini E, Pourmalek F, Prince M, Pullan RL, Ramaiah KD, Ranganathan D, Razavi H, Regan M, Rehm JT, Rein DB, Remuzzi G, Richardson K, Rivara FP, Roberts T, Robinson C, De Leon FR, Ronfani L, Room R, Rosenfeld LC, Rushton L, Sacco RL, Saha S, Sampson U, Sanchez-Riera L, Sanman E, Schwebel DC, Scott JG, Segui-Gomez M, Shahraz S, Shepard DS, Shin H, Shivakoti R, Singh D, Singh GM, Singh JA, Singleton J, Sleet DA, Sliwa K, Smith E, Smith JL, Stapelberg NJ, Steer A, Steiner T, Stolk WA, Stovner LJ, Sudfeld C, Syed S, Tamburlini G, Tavakkoli M, Taylor HR, Taylor JA, Taylor WJ, Thomas B, Thomson WM, Thurston GD, Tleyjeh IM, Tonelli M, Towbin JA, Truelsen T, Tsilimbaris MK, Ubeda C, Undurraga EA, van der Werf MJ, van Os J, Vavilala MS, Venketasubramanian N, Wang M, Wang W, Watt K, Weatherall DJ, Weinstock MA, Weintraub R, Weisskopf MG, Weissman MM, White RA, Whiteford H, Wiersma ST, Wilkinson JD, Williams HC, Williams SR, Witt E, Wolfe F, Woolf AD, Wulf S, Yeh PH, Zaidi AK, Zheng ZJ, Zonies D, Lopez AD, Murray CJ, MA AM, Memish ZA (2012) Years lived with disability (YLDs) for 1160 sequelae of 289 diseases and injuries 1990–2010: a systematic analysis for the Global Burden of Disease Study 2010. Lancet (London, England) 380(9859):2163–2196. https://doi.org/10.1016/s0140-6736(12)61729-2
Global, regional, and national incidence, prevalence, and years lived with disability for 310 diseases and injuries, 1990–2015: a systematic analysis for the Global Burden of Disease Study 2015 (2016). Lancet (London, England) 388 (10053):1545–1602. doi:https://doi.org/10.1016/s0140-6736(16)31678-6
Leardini G, Salaffi F, Caporali R, Canesi B, Rovati L, Montanelli R (2004) Direct and indirect costs of osteoarthritis of the knee. Clin Exp Rheumatol 22(6):699–706
Zhang W, Moskowitz RW, Nuki G, Abramson S, Altman RD, Arden N, Bierma-Zeinstra S, Brandt KD, Croft P, Doherty M, Dougados M, Hochberg M, Hunter DJ, Kwoh K, Lohmander LS, Tugwell P (2007) OARSI recommendations for the management of hip and knee osteoarthritis, part I: critical appraisal of existing treatment guidelines and systematic review of current research evidence. Osteoarthr Cartil 15(9):981–1000. https://doi.org/10.1016/j.joca.2007.06.014
Baier Leach J, Bivens KA, Patrick CW Jr, Schmidt CE (2003) Photocrosslinked hyaluronic acid hydrogels: natural, biodegradable tissue engineering scaffolds. Biotechnol Bioeng 82(5):578–589. https://doi.org/10.1002/bit.10605
Pagnano M, Westrich G (2005) Successful nonoperative management of chronic osteoarthritis pain of the knee: safety and efficacy of retreatment with intra-articular hyaluronans. Osteoarthr Cartil 13(9):751–761. https://doi.org/10.1016/j.joca.2005.04.012
Maricar N, Callaghan MJ, Felson DT, O’Neill TW (2013) Predictors of response to intra-articular steroid injections in knee osteoarthritis--a systematic review. Rheumatology (Oxford, England) 52(6):1022–1032. https://doi.org/10.1093/rheumatology/kes368
Koh YG, Kwon OR, Kim YS, Choi YJ, Tak DH (2016) Adipose-derived mesenchymal stem cells with microfracture versus microfracture alone: 2-year follow-up of a prospective randomized trial. Arthroscopy 32(1):97–109. https://doi.org/10.1016/j.arthro.2015.09.010
Fodor PB, Paulseth SG (2016) Adipose derived stromal cell (ADSC) injections for pain management of osteoarthritis in the human knee joint. Aesthet Surg J 36(2):229–236. https://doi.org/10.1093/asj/sjv135
Koh YG, Choi YJ, Kwon SK, Kim YS, Yeo JE (2015) Clinical results and second-look arthroscopic findings after treatment with adipose-derived stem cells for knee osteoarthritis. Knee Surg Sports Traumatol Arthrosc 23(5):1308–1316. https://doi.org/10.1007/s00167-013-2807-2
Koh YG, Kwon OR, Kim YS, Choi YJ (2014) Comparative outcomes of open-wedge high tibial osteotomy with platelet-rich plasma alone or in combination with mesenchymal stem cell treatment: a prospective study. Arthroscopy: the journal of arthroscopic & related surgery: official publication of the Arthroscopy Association of North America and the International Arthroscopy Association 30(11):1453–1460. https://doi.org/10.1016/j.arthro.2014.05.036
Bansal H, Comella K, Leon J, Verma P, Agrawal D, Koka P, Ichim T (2017) Intra-articular injection in the knee of adipose derived stromal cells (stromal vascular fraction) and platelet rich plasma for osteoarthritis. J Transl Med 15(1):141. https://doi.org/10.1186/s12967-017-1242-4
Gimble J, Guilak F (2003) Adipose-derived adult stem cells: isolation, characterization, and differentiation potential. Cytotherapy 5(5):362–369. https://doi.org/10.1080/14653240310003026
Zuk PA, Zhu M, Mizuno H, Huang J, Futrell JW, Katz AJ, Benhaim P, Lorenz HP, Hedrick MH (2001) Multilineage cells from human adipose tissue: implications for cell-based therapies. Tissue Eng 7(2):211–228. https://doi.org/10.1089/107632701300062859
Spasovski D, Spasovski V, Bascarevic Z, Stojiljkovic M, Vreca M, Andelkovic M, Pavlovic S (2018) Intra-articular injection of autologous adipose-derived mesenchymal stem cells in the treatment of knee osteoarthritis. The Journal of Gene Medicine 20(1). https://doi.org/10.1002/jgm.3002
Jo CH, Lee YG, Shin WH, Kim H, Chai JW, Jeong EC, Kim JE, Shim H, Shin JS, Shin IS, Ra JC, Oh S, Yoon KS (2014) Intra-articular injection of mesenchymal stem cells for the treatment of osteoarthritis of the knee: a proof-of-concept clinical trial. Stem cells (Dayton, Ohio) 32(5):1254–1266. https://doi.org/10.1002/stem.1634
Jo CH, Chai JW, Jeong EC, Oh S, Shin JS, Shim H, Yoon KS (2017) Intra-articular injection of mesenchymal stem cells for the treatment of osteoarthritis of the knee: a 2-year follow-up study. Am J Sports Med 45(12):2774–2783. https://doi.org/10.1177/0363546517716641
Song Y, Du H, Dai C, Zhang L, Li S, Hunter DJ, Lu L, Bao C (2018) Human adipose-derived mesenchymal stem cells for osteoarthritis: a pilot study with long-term follow-up and repeated injections. Regen Med 13(3):295–307. https://doi.org/10.2217/rme-2017-0152
Pers YM, Rackwitz L, Ferreira R, Pullig O, Delfour C, Barry F, Sensebe L, Casteilla L, Fleury S, Bourin P, Noel D, Canovas F, Cyteval C, Lisignoli G, Schrauth J, Haddad D, Domergue S, Noeth U, Jorgensen C (2016) Adipose mesenchymal stromal cell-based therapy for severe osteoarthritis of the knee: a phase I dose-escalation trial. Stem Cells Transl Med 5(7):847–856. https://doi.org/10.5966/sctm.2015-0245
Koh YG, Choi YJ (2012) Infrapatellar fat pad-derived mesenchymal stem cell therapy for knee osteoarthritis. Knee 19(6):902–907. https://doi.org/10.1016/j.knee.2012.04.001
Koh YG, Jo SB, Kwon OR, Suh DS, Lee SW, Park SH, Choi YJ (2013) Mesenchymal stem cell injections improve symptoms of knee osteoarthritis. Arthroscopy 29(4):748–755. https://doi.org/10.1016/j.arthro.2012.11.017
Kellgren JH, Lawrence JS (1957) Radiological assessment of osteo-arthrosis. Ann Rheum Dis 16(4):494–502
Peterfy CG, Guermazi A, Zaim S, Tirman PF, Miaux Y, White D, Kothari M, Lu Y, Fye K, Zhao S, Genant HK (2004) Whole-organ magnetic resonance imaging score (WORMS) of the knee in osteoarthritis. Osteoarthr Cartil 12(3):177–190. https://doi.org/10.1016/j.joca.2003.11.003
Marlovits S, Striessnig G, Resinger CT, Aldrian SM, Vecsei V, Imhof H, Trattnig S (2004) Definition of pertinent parameters for the evaluation of articular cartilage repair tissue with high-resolution magnetic resonance imaging. Eur J Radiol 52(3):310–319. https://doi.org/10.1016/j.ejrad.2004.03.014
Centeno CJ, Al-Sayegh H, Freeman MD, Smith J, Murrell WD, Bubnov R (2016) A multi-center analysis of adverse events among two thousand, three hundred and seventy two adult patients undergoing adult autologous stem cell therapy for orthopedic conditions. Int Orthod 40(8):1755–1765. https://doi.org/10.1007/s00264-016-3162-y
Vega A, Martin-Ferrero MA, Del Canto F, Alberca M, Garcia V, Munar A, Orozco L, Soler R, Fuertes JJ, Huguet M, Sanchez A, Garcia-Sancho J (2015) Treatment of knee osteoarthritis with allogeneic bone marrow mesenchymal stem cells: a randomized controlled trial. Transplantation 99(8):1681–1690. https://doi.org/10.1097/tp.0000000000000678
Lisi C, Perotti C, Scudeller L, Sammarchi L, Dametti F, Musella V, Di Natali G (2018) Treatment of knee osteoarthritis: platelet-derived growth factors vs. hyaluronic acid. A randomized controlled trial. Clin Rehabil 32(3):330–339. https://doi.org/10.1177/0269215517724193
Kubosch EJ, Heidt E, Niemeyer P, Bernstein A, Sudkamp NP, Schmal H (2017) In-vitro chondrogenic potential of synovial stem cells and chondrocytes allocated for autologous chondrocyte implantation - a comparison: synovial stem cells as an alternative cell source for autologous chondrocyte implantation. Int Orthod 41(5):991–998. https://doi.org/10.1007/s00264-017-3400-y
Cuti T, Antunovic M, Marijanovic I, Ivkovic A, Vukasovic A, Matic I, Pecina M, Hudetz D (2017) Capacity of muscle derived stem cells and pericytes to promote tendon graft integration and ligamentization following anterior cruciate ligament reconstruction. Int Orthod 41(6):1189–1198. https://doi.org/10.1007/s00264-017-3437-y
Xia P, Wang X, Lin Q, Li X (2015) Efficacy of mesenchymal stem cells injection for the management of knee osteoarthritis: a systematic review and meta-analysis. Int Orthod 39(12):2363–2372. https://doi.org/10.1007/s00264-015-2785-8
Nguyen A, Guo J, Banyard DA, Fadavi D, Toranto JD, Wirth GA, Paydar KZ, Evans GR, Widgerow AD (2016) Stromal vascular fraction: a regenerative reality? Part 1: current concepts and review of the literature. Journal of plastic, reconstructive & esthetic surgery: JPRAS 69(2):170–179. https://doi.org/10.1016/j.bjps.2015.10.015
Chung MT, Zimmermann AS, Paik KJ, Morrison SD, Hyun JS, Lo DD, McArdle A, Montoro DT, Walmsley GG, Senarath-Yapa K, Sorkin M, Rennert R, Chen HH, Chung AS, Vistnes D, Gurtner GC, Longaker MT, Wan DC (2013) Isolation of human adipose-derived stromal cells using laser-assisted liposuction and their therapeutic potential in regenerative medicine. Stem Cells Transl Med 2(10):808–817. https://doi.org/10.5966/sctm.2012-0183
Atalay S, Coruh A, Deniz K (2014) Stromal vascular fraction improves deep partial thickness burn wound healing. Burns 40(7):1375–1383. https://doi.org/10.1016/j.burns.2014.01.023
Bora P, Majumdar AS (2017) Adipose tissue-derived stromal vascular fraction in regenerative medicine: a brief review on biology and translation. Stem Cell Res Ther 8(1):145. https://doi.org/10.1186/s13287-017-0598-y
Riordan NH, Ichim TE, Min WP, Wang H, Solano F, Lara F, Alfaro M, Rodriguez JP, Harman RJ, Patel AN, Murphy MP, Lee RR, Minev B (2009) Non-expanded adipose stromal vascular fraction cell therapy for multiple sclerosis. J Transl Med 7:29. https://doi.org/10.1186/1479-5876-7-29
You D, Jang MJ, Kim BH, Song G, Lee C, Suh N, Jeong IG, Ahn TY, Kim CS (2015) Comparative study of autologous stromal vascular fraction and adipose-derived stem cells for erectile function recovery in a rat model of cavernous nerve injury. Stem Cells Transl Med 4(4):351–358. https://doi.org/10.5966/sctm.2014-0161
Funding
This study was supported by grants from National Natural Science Foundation of China (81672769) and Medical Science and Technology Foundation of Zhejiang Province (CN) (2017KY016).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethical approval
All procedures performed in the studies involving human participants were in accordance with the ethical standards of Ethics Committee of the Zhejiang Provincial People’s Hospital and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. This study was registered at Chinses Clinical Trial Registry with identifier ChiCTR1800015125.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Hong, Z., Chen, J., Zhang, S. et al. Intra-articular injection of autologous adipose-derived stromal vascular fractions for knee osteoarthritis: a double-blind randomized self-controlled trial. International Orthopaedics (SICOT) 43, 1123–1134 (2019). https://doi.org/10.1007/s00264-018-4099-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00264-018-4099-0