Abstract
Introduction
The most common reason for removal of well-fixed radial head prostheses is painful loosening. We hypothesised that short-stemmed prostheses, used for radial head arthroplasty, are not associated with an increased risk of implant loosening.
Methods
From 2002 to 2014, 65 patients were enrolled in a retrospective single-centre study. The radial head prostheses were classified as having either a long (30-mm) or short (16- to 22-mm) stem. The long-stemmed implants comprised 30 GUEPAR® DePuy Synthes (West Chester, PA, USA) and 20 Evolutive® Aston Medical (Saint-Etienne, France) devices; the short-stemmed implants comprised nine RECON and six STANDARD rHead® SBI-Stryker (Morrisville, PA, USA) devices. At last follow-up, clinical (range of motion, Disabilities of the Arm, Shoulder, and Hand score, Mayo Elbow Performance score) and radiographic (osteolysis) outcomes were assessed.
Results
At a mean follow-up of 76.78 months (24–141), the rate of painful loosening [6 (40%) vs 8 (16%), p = 0.047] and osteolysis [12 (80%) vs 23 (46%), p = 0.02] were significantly higher in patients with short-stemmed versus long-stemmed implants. Despite the significant difference in loosening between stems as groups, individual stem length was not determined.
Conclusions
Tight-fitting implants with short stems are more prone to painful loosening.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Fractures of the proximal radius account for 1.7–5.4% of all fractures and approximately one third of elbow fractures [1]. This becomes important when one considers that the proximal radius is a major contributor to elbow and forearm stability [2,3,4]. Radial head arthroplasty (RHA) has satisfactory outcomes according to recent literature [5,6,7,8]; however, tight-fitting radial head prostheses (RHP) may have inferior midterm survival than loose-fitting implants [5,6,7,8,9,10,11,12,13,14]. High complication rates have been reported after this procedure, and the most common cause of RHP removal is painful prosthetic loosening [15,16,17,18,19]. O’Driscoll and colleagues [20, 21] identified several mechanical rules to follow to obtain a well-fixed implant while minimising micromotion, with stem diameter and length being the most important factors.
Between 2002 and 2014, four different RHP designs were used in the Department of Orthopaedic Surgery at this institution to treat non-reconstructable acute radial head fractures or their post-traumatic sequela: GUEPAR® prosthesis (Depuy-Synthes, West Chester, PA, USA), Evolutive® prosthesis (Aston Medical, Saint-Etienne, France), rHead® RECON prosthesis or rHead® STANDARD prosthesis (Stryker-Small Bone Innovation). The primary aim in this study was to investigate and compare the risk of painful loosening of tight-fitting RHPs with short and long stems.
Materials and methods
This was a single-centre comparative retrospective study conducted in an academic department of orthopaedic surgery. Inclusion criteria were patients undergoing surgery for non-reconstructable fracture of the radial head fracture or sequela of trauma (including malunion, pseudarthrosis, necrosis, failure of fixation) for which an RHA was performed between 2002 and 2014, with a minimum follow-up of two years or until implant removal. Patients with follow-up of < two years, aged <18 years and prosthesis removal not due to loosening were excluded. The study was approved by the local institutional review board.
Patients
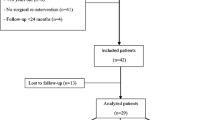
A total of 94 patients underwent RHA during this time. Sixteen patients were excluded due to: short follow-up (4 cases), removal for a reason other than painful loosening [5 cases: posterolateral instability (1); radiohumeral conflict (4)] and re-operation with implant retention [7 cases: persistent lateral posterior instabilities (5), radiohumeral conflict (1), prosthesis head dislocated from stem (1)]. Thirteen patients were lost to follow-up, and 65 were assessed [44 men, 21 women; mean age 52.46 years (20–82)]. Forty-five RHAs were performed for acute fractures and 20 for sequelae of trauma. Injury mechanisms comprised 25 falls from standing height, 17 a roof or a ladder, nine down stairs, nine traffic accidents and five sports accidents. Initial fracture evaluation showed 57 Mason type III, two Mason type II and six radial neck; 26 were isolated proximal radius fractures, 27 terrible triad, three Essex–Lopresti, four distal ulna or radius metaphyseal–epiphyseal and 11 associated transolecranon. Population details are reported in the Table 1.
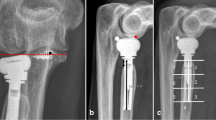
Sixty-five tight-fitting RHAs were reviewed retrospectively using four different prosthetic models: 30 GUEPAR® (bipolar implant), 20 Evolutive® (bipolar implant) six rHead® STANDARD (monopolar implant) and nine rHead® RECON prostheses (e.g. bipolar RHP) (Fig. 1). The long-stemmed implants [50 long, smooth stems (30 mm)] were GUEPAR® and Evolutive® designs, and the short-stemmed implants (15 short, roughened stems (16-22 mm)] were rHead®. A call for tenders was performed for each RHA model; one type was preselected to be used for all procedure for a limited period. Our preference changed three times for a total of four different prostheses during this time. The RHA used for each patient was dependent on our preference at the time of surgery. No RHA randomisation was performed, as only one choice was available at the time of surgery for each patient.
Surgical approach and technique
A lateral approach to the elbow was used in 58 cases, and a posterolateral approach was preferable in seven cases when there was an associated fracture of the olecranon. Particular attention was paid to preserving the radial collateral ligament if it was intact. The annular ligament (AL) was incised longitudinally (incision transverse to AL fibres). The capitellum was routinely carefully examined for the presence of cartilage lesions. Seven olecranon fractures were fixed with a plate. Four fractures of the coronoid process were fixed using retrograde screw fixation with intra-articular control of the reduction. The radial neck was divided to preserve as much bone as possible; short-stemmed components were used if it was possible to conserve radial neck length. The radial medullary canal was reamed and the prosthesis introduced such that it did not surpass the superior surface of the radial notch of the ulna. The elbow was then put through a full arc of flexion and the position was checked on extension and on anteroposterior (AP) and lateral fluoroscopic views. Low-viscosity, antibiotic-impregnated cement (PalacosGenta®) was used for fixation of all RHPs; the press-fit stability of stems with roughened surfaces was felt to be insufficient, and the short-stemmed prostheses were anchored with cement according to the manufacturers’ specifications. The final radial head components were impacted onto the neck of long-stemmed prostheses and directly onto the stem of Stryker prostheses. The radial collateral ligament was reattached to the lateral epicondyles using transosseous sutures or suture anchors in 29 patients; in the remaining patients, only the annular ligament was repaired, followed by the tendon layer. Elbow stability was then assessed. The ulnar collateral ligament was re-attached to the medial epicondyle in three patients.
Post-operative procedure
In 29 patients in whom the lateral collateral ligament (LCL) was repaired, the elbow was immobilised for 15 days in a long-arm dorsal-volar splint with the wrist in pronation. The wrist was left free in 36 patients in whom the LCL was not repaired. In five patients in whom the elbow remained unstable despite LCL reconstruction, a static external fixator was retained for two to three weeks. Between post-operative days 15 and 21, a hinged brace was used to permit extension up to −30°. By postoperative day 45, active mobilisation and physiotherapy were initiated, including active range of motion (ROM) exercises to encourage articular coaptation.
Evaluation methods
All patients were assessed by an independent reviewer at the aforementioned time points or until removal for painful loosening. Comparative clinical examination of both upper extremities (wrist and elbow) was performed. Anteroposterior and lateral radiographs were taken with the elbow in maximum extension and 90° of flexion for the 51 patients who retained the radial head prosthesis at the time of last follow-up. Pre- and post-operative clinical and radiographic data and operative details were noted from the medical records for all patients.
Primary endpoint
We reviewed pre-operative physical exams (proximal radial forearm pain reported according to O’Driscoll’s definition [18]) and operative reports (loose implant confirmed by the quality of the prosthetic sealing) to identify whether painful loosening was the cause for re-operation for each patient.
Secondary endpoint
Clinical analysis was possible for 51 patients in whom the RHP was retained at the time of the final review. Maximum ROM was measured using a goniometer. The ratio (expressed as %) of operative to non-operative elbow flexion and extension force was measured using a Kinedyn® dynamometer (Smith & Nephew, Memphis, TN, USA). Function was assessed using the Mayo Elbow Performance Score (MEPS) and the Quick Disabilities of the Arm, Shoulder, and Hand (QuickDASH) score [22, 23].
For all cases, radiographic analysis of the elbow (lateral/AP views) was used to asses for signs of periprosthetic osteolysis, overstuffing (on lateral radiograph according to the method described by Van Riet et al. [24]) and presence or absence of river delta sign (asymmetric humero-ulnar joint space, wider on the radial side according to Gauci et al. [9]). Assessment included heterotopic ossification according to Brooker classification and capitellar wear. These were noted at each post-operative review.
Statistical analysis
A single-sample chi-square analysis was used to compare sex, associated lesions, prosthesis use in an acute or delayed fashion, osteolysis and overstuffing between long- and short-stemmed prostheses. The Mann–Whitney U test, also known as the Wilcoxon rank-sum test, was used to compare age, QuickDASH, MEPS, ROM and force with the healthy contralateral side. Odds ratios (OR) were used to analyse the predictive link between stem size (long or short) and loosening, the predictive link between overstuffing, and loosening or capitellar wear. The survival rate of long- versus short-stemmed prostheses was determined according to the Kaplan–Meier method. Comparative analysis between survival curves of long and short stems was performed using the log-rank test (Mantel–Cox). Confidence intervals (CI) were fixed at 95% and statistical significance at p < 0.05. Results are described according to their mean or percentage (with absolute number) and range. Normality of data was assessed, and all data was determined to be non-normally distributed (P < 0.01, Kolmogorov–Smirnov test) requiring the use of non-parametric tests.
Results
Mean follow-up was 76.78 months (24–141) for 51 patients. At the time of data collection, mean follow-up for the short-stemmed implants was 58.01 (44–69) and 82.42 (24–141) months for long-stemmed implants. The remaining patients were censored due to prosthesis removal prior to this time point. Clinical and radiographic results are reported in Table 2.
Primary endpoint
A total of 14 cases of painful aseptic loosening were reported in ten men and four women, mean age 51.1 years, with seven GUEPAR®, one Evolutive®, five rHead® RECON and one rHead® STANDARD. There was a statistically significantly difference in loosening between short- and long-stemmed implants [6 (40%) vs 8 (16%), respectively; p = 0.047] (Table 2). Despite the significant difference in loosening between stems as groups, individual stem length was not determined [OR 3.5 (0.9–12.59); p = 0.055]. The loosened implants were removed at a mean of 23.28 months (6–36) postoperatively, with no significant difference between groups (24.7 versus 23.1 months; p = 0.75). Comparative analysis demonstrated greater survival for long stems, though this difference was not significant [log-rank (Mantel–Cox); p = 0.0506] (Fig. 2).
Secondary endpoint
Secondary endpoints of clinical and radiological results are shown in Table 2.
Clinical results
No significant difference in results between short- and long-stemmed implants was found. The average reported MEP score was 87.4 points (92 versus 86.8, respectively; p = 0.35), and mean quickDASH score was 16.57 points (12.8 versus 17.7, respectively; p = 0.38).
Radiological results
Periprosthetic osteolysis was noted in 35 cases; it was located around the neck of the radius in 11 cases, circumferentially around the stem in 27 cases and limited to the lateral or medial side of the stem in four and three cases, respectively. Overstuffing was identified in 30 cases at a frequency of 47% (7) for short and 46% (23) for long stems; (p = 0.96). A river delta sign [9] was reported in 11 cases (3 short and 8 long). There was no significant association between implant overstuffing and loosening [OR 0.96 (0.28–3.27); p = 0.95] or osteolysis [OR 0.78 (0.30–2.02); p = 0.62]. Additionally, overstuffing was not associated with negative radiographic findings, such as osteolysis [OR 0.78 (0.30–2.02); p = 0.62], capitellar wear [OR 0.76 (0.27–2.08); p = 0.59] or painful loosening [OR 0.96 (0.28–3.27); p = 0.95). Analysis of humeroulnar joint space narrowing revealed capitellar wear in 27 prostheses. Heterotopic periprosthetic ossification was found in 24 cases and classified according to Brooker: grade 0 in 41, grade 1 in 15, grade 2 in four, and grade three in five cases.
Discussion
This study shows that the rate of painful loosening was significantly higher in tight-fitting RHPs with short stems (p < 0.05). Despite satisfactory mid- to long-term outcomes (mean quickDASH and MEP scores of 16.57 and 87.4, respectively), tight-fitting RHP may have inferior mid-term survival than loose-fitting implants according to recent literature [5,6,7,8,9,10,11,12,13,14,15]. Painful loosening of tight-fitting implants is the most common cause of RHA failure [15,16,17,18,19, 25, 26]. Our study did find a decreased implant survival time for short- versus long-stemmed implants (Fig. 2); however, demonstrating a cumulative survival difference (p = 0.0506) and an increased risk of loosening (p = 0.055) according to stem length would have required larger sample size. According to Shukla et al. [21], the risk of instability is dependent on the ratio of RHP length divided by total implant length (Fig. 1). When this ratio is >0.4, the risk of instability is significantly higher due to increased stem micromotion. The increased ratio in short-stemmed RHAs could explain the significantly increased rate of loosening (p = 0.047) and osteolysis (p = 0.02) found in our study. For all short-stemmed implants, intra-operative press-fit was insufficient, and cement was required to obtain a satisfactory fixation. Since a layer of cement could be added, it follows that the diameter of these prostheses was smaller than the maximal and submaximal diameter needed to fill the canal. Moon et al. [20], found that implants of submaximal size had micromotion (>250 μm) that exceeded the threshold needed for bone ingrowth and initial stability. Lastly, the level of comfort with the surgical technique could play a role in the high failure rate. We speculate that the increased rate of overstuffing [30 (46%), regardless of stem length; (p = 0.96)] was due to difficulties in obtaining stable fixation. This may predispose the surgeon to favour stability over implant position. We speculate overstuffing could theoretically contribute to the risk of stem micromotion by increasing the extramedullary portion of the prosthesis. However, our study did not show that overstuffing was a risk factor for painful loosening (p = 0.95) or osteolysis (p = 0.62) [21, 27,28,29]. The rate of early capitellar wear in our series was high (>30%) and did not vary with stem length (p = 0.07); overstuffing was not a predictive factor of capitellar wear (p = 0.59). We speculate that cup hypermobility and repeated episodes of posterolateral subluxation of bipolar implants, as well as higher radiocapitellar contact pressures with monopolar implants, could explain the high rate of early capitellar wear [30,31,32].
The implant selected for each patient did not depend on soft tissue integrity. Only one RHA design was available at the time of each operation for all patients in this series. We recognize that this is a weakness of the study, as the bipolar implant is clearly recommended only when there is malalignment of the proximal radius with respect to the capitellum. Other limitations of our study relate to its retrospective, single-centre nature and small sample size. The retrospective design inherently leads to more data loss and bias. The rate of loss to follow-up was elevated, at (13.9%); however, this rate is justified in light of the retrospective design and the requirement of a midterm clinical visit dedicated specifically to the study. The small sample size of short-stemmed implants did not allow us to find statistically significant risk factors for painful loosening. We considered only tight-fitting RHAs and did not include loose-fitting RHAs or designs. We analysed RHPs with a variety of associated lesions that were not accounted for by comparative analysis in the follow-up period. However, characteristics of the study population were not significantly different between implant groups (Table 1); most importantly, the rates of associated lesions (p = 0.69) and use of the prosthesis in a delayed or acute fashion (p > 0.38) were not different. Surgeon training in elbow surgery, particularly in RHA, was variable; which may influence results [17]. Since the mean follow-up for short-stemmed implants was less than for long-stemmed implants, the true rate of painful loosening and negative radiographic outcomes may have been lower in the short-stemmed implant group.
Conclusions
Tight-fitting RHPs with short stems are more prone to painful loosening. An additional study with a larger cohort of patients focused on fixed RHP malpositioning and sequential painful loosening will be necessary to further understand this association.
References
Morrey BF, Tanaka S, An KN (1991) Valgus stability of the elbow. A definition of primary and secondary constraints. Clin Orthop 187–195
Van Glabbeek F, van Riet RP, Baumfeld JA et al (2005) The kinematic importance of radial neck length in radial head replacement. Med Eng Phys 27:336–342. https://doi.org/10.1016/j.medengphy.2004.04.011
Mason ML (1954) Some observations on fractures of the head of the radius with a review of one hundred cases. Br J Surg 42:123–132
Captier G, Canovas F, Mercier N et al (2002) Biometry of the radial head: biomechanical implications in pronation and supination. Surg Radiol Anat SRA 24:295–301. https://doi.org/10.1007/s00276-002-0059-9
Allavena C, Delclaux S, Bonnevialle N et al (2014) Outcomes of bipolar radial head prosthesis to treat complex radial head fractures in 22 patients with a mean follow-up of 50 months. Orthop Traumatol Surg Res OTSR 100:703–709. https://doi.org/10.1016/j.otsr.2014.06.019
Berschback JC, Lynch TS, Kalainov DM et al (2013) Clinical and radiographic comparisons of two different radial head implant designs. J Shoulder Elb Surg 22:1108–1120. https://doi.org/10.1016/j.jse.2013.02.011
Dou Q, Yin Z, Sun L, Feng X (2015) Prosthesis replacement in Mason III radial head fractures: a meta-analysis. Orthop Traumatol Surg Res OTSR 101:729–734. https://doi.org/10.1016/j.otsr.2015.06.015
Flinkkilä T, Kaisto T, Sirniö K et al (2012) Short- to mid-term results of metallic press-fit radial head arthroplasty in unstable injuries of the elbow. J Bone Joint Surg Br 94:805–810. https://doi.org/10.1302/0301-620X.94B6.28176
Gauci M-O, Winter M, Dumontier C et al (2016) Clinical and radiologic outcomes of pyrocarbon radial head prosthesis: midterm results. J Shoulder Elb Surg 25:98–104. https://doi.org/10.1016/j.jse.2015.08.033
Giannicola G, Sacchetti FM, Antonietti G et al (2014) Radial head, radiocapitellar and total elbow arthroplasties: a review of recent literature. Injury 45:428–436. https://doi.org/10.1016/j.injury.2013.09.019
Katthagen JC, Jensen G, Lill H, Voigt C (2013) Monobloc radial head prostheses in complex elbow injuries: results after primary and secondary implantation. Int Orthop 37:631–639. https://doi.org/10.1007/s00264-012-1747-7
Marsh JP, Grewal R, Faber KJ et al (2016) Radial head fractures treated with modular metallic radial head replacement: outcomes at a mean follow-up of eight years. J Bone Joint Surg Am 98:527–535. https://doi.org/10.2106/JBJS.15.00128
Moro JK, Werier J, MacDermid JC et al (2001) Arthroplasty with a metal radial head for unreconstructible fractures of the radial head. J Bone Joint Surg Am 83–A:1201–1211
Shore BJ, Mozzon JB, MacDermid JC et al (2008) Chronic posttraumatic elbow disorders treated with metallic radial head arthroplasty. J Bone Joint Surg Am 90:271–280. https://doi.org/10.2106/JBJS.F.01535
Laumonerie P, Ancelin D, Reina N et al (2017) Causes for early and late surgical re-intervention after radial head arthroplasty. Int Orthop 41:1435–1443. https://doi.org/10.1007/s00264-017-3496-0
Duckworth AD, Wickramasinghe NR, Clement ND et al (2014) Radial head replacement for acute complex fractures: what are the rate and risks factors for revision or removal? Clin Orthop 472:2136–2143. https://doi.org/10.1007/s11999-014-3516-y
Kachooei AR, Claessen FMAP, Chase SM et al (2016) Factors associated with removal of a radial head prosthesis placed for acute trauma. Injury 47:1253–1257. https://doi.org/10.1016/j.injury.2016.02.023
O’Driscoll SW, Herald JA (2012) Forearm pain associated with loose radial head prostheses. J Shoulder Elb Surg 21:92–97. https://doi.org/10.1016/j.jse.2011.05.008
van Riet RP, Sanchez-Sotelo J, Morrey BF (2010) Failure of metal radial head replacement. J Bone Joint Surg Br 92:661–667. https://doi.org/10.1302/0301-620X.92B5.23067
Moon J-G, Berglund LJ, Domire Z et al (2009) Stem diameter and micromotion of press fit radial head prosthesis: a biomechanical study. J Shoulder Elb Surg 18:785–790. https://doi.org/10.1016/j.jse.2009.02.014
Shukla DR, Fitzsimmons JS, An K-N, O’Driscoll SW (2012) Effect of stem length on prosthetic radial head micromotion. J Shoulder Elb Surg 21:1559–1564. https://doi.org/10.1016/j.jse.2011.11.025
Cusick MC, Bonnaig NS, Azar FM et al (2014) Accuracy and reliability of the Mayo elbow performance score. J Hand Surg 39:1146–1150. https://doi.org/10.1016/j.jhsa.2014.01.041
Hudak PL, Amadio PC, Bombardier C (1996) Development of an upper extremity outcome measure: the DASH (disabilities of the arm, shoulder and hand) [corrected]. The upper extremity collaborative group (UECG). Am J Ind Med 29:602–608. https://doi.org/10.1002/(SICI)1097-0274(199606)29:6<602::AID-AJIM4>3.0.CO;2-L
van Riet RP, van Glabbeek F, de Weerdt W et al (2007) Validation of the lesser sigmoid notch of the ulna as a reference point for accurate placement of a prosthesis for the head of the radius: a cadaver study. J Bone Joint Surg Br 89:413–416. https://doi.org/10.1302/0301-620X.89B3.18099
Delclaux S, Lebon J, Faraud A et al (2015) Complications of radial head prostheses. Int Orthop 39(5):907–913. https://doi.org/10.1007/s00264-015-2689-7
Neuhaus V, Christoforou DC, Kachooei AR et al (2015) Radial head prosthesis removal: a retrospective case series of 14 patients. Arch Bone Jt Surg 3:88–93
Chanlalit C, Shukla DR, Fitzsimmons JS et al (2012) Stress shielding around radial head prostheses. J Hand Surg 37:2118–2125. https://doi.org/10.1016/j.jhsa.2012.06.020
Athwal GS, Frank SG, Grewal R et al (2010) Determination of correct implant size in radial head arthroplasty to avoid overlengthening: surgical technique. J Bone Joint Surg Am 92(Suppl 1 Pt 2):250–257. https://doi.org/10.2106/JBJS.J.00356
Van Glabbeek F, Van Riet RP, Baumfeld JA et al (2004) Detrimental effects of overstuffing or understuffing with a radial head replacement in the medial collateral-ligament deficient elbow. J Bone Joint Surg Am 86–A:2629–2635
Chanlalit C, Shukla DR, Fitzsimmons JS et al (2011) Radiocapitellar stability: the effect of soft tissue integrity on bipolar versus monopolar radial head prostheses. J Shoulder Elb Surg 20:219–225. https://doi.org/10.1016/j.jse.2010.10.033
Sahu D, Holmes DM, Fitzsimmons JS et al (2014) Influence of radial head prosthetic design on radiocapitellar joint contact mechanics. J Shoulder Elb Surg 23:456–462. https://doi.org/10.1016/j.jse.2013.11.028
Moungondo F, El Kazzi W, van Riet R et al (2010) Radiocapitellar joint contacts after bipolar radial head arthroplasty. J Shoulder Elb Surg 19:230–235. https://doi.org/10.1016/j.jse.2009.09.015
Funding
All the authors (Pierre Laumonerie, Nicolas Reina, Claudia Gutierrez, Stephanie Delclaux, Meagan E Tibbo, Nicolas Bonnevialle, Pierre Mansat) and their immediate family, and any research foundation with which they are affiliated, did not receive any financial payments or other benefits from any commercial entity related to the subject of this article.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclaimer
Not applicable.
Conflict of interest
All the authors (Pierre Laumonerie, Nicolas Reina, Claudia Gutierrez, Stephanie Delclaux, Meagan E Tibbo, Nicolas Bonnevialle, Pierre Mansat) report no conflict of interest concerning the materials or methods used in this study or the findings specified in this paper.
Rights and permissions
About this article
Cite this article
Laumonerie, P., Reina, N., Gutierrez, C. et al. Tight-fitting radial head prosthesis: does stem size help prevent painful loosening?. International Orthopaedics (SICOT) 42, 161–167 (2018). https://doi.org/10.1007/s00264-017-3644-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00264-017-3644-6