Abstract
Purpose
Little data is available about the incidence and especially the management of hip dislocation following the implantation of modular tumor prostheses of the proximal femur. In this retrospective single-centre study we assessed the incidence of hip dislocation following implantation of a proximal femoral modular prosthesis as well as the success of the subsequent surgical or non-surgical treatment in tumor patients.
Methods
Between 1982 and 2008, 166 tumor patients received a modular prosthesis of the proximal femur at our institution. The average age at the time of surgery was 50 ± 20 years (range, six to 84 years). An additional pelvic reconstruction was done in 14 patients. An artificial band for soft tissue reconstruction of the hip was used in 19 patients. The average time of follow-up was 46 ± 64 months (range, one to 277 months).
Results
The overall dislocation rate after proximal femoral replacement was 13 % after a mean time of seven ± eight months (range, 0.3–33 months) after surgery. Between 1982 and 1986 the dislocation rate was 33 % and declined to 9 % in subsequent years (1987–2008). Patients who had received an additional pelvic reconstruction had a three fold higher dislocation rate (p <0.05). Patients with closed reduction had a 58 % (eight of 12) re-dislocation rate compared to 11 % (one of nine) for open reduction (p = 0.0357).
Conclusions
Dislocation of a modular prosthesis of the proximal femur is a common complication, especially in cases with additional pelvic resection with extensive bone and soft-tissue defects. Open surgical management may be more effective in preventing re-dislocation than closed reduction and bracing alone.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Surgical resection of primary tumours or metastatic lesions of the proximal femur can result in large bone and soft tissue resections. Modular prostheses are frequently used in the reconstruction of the resulting defects and provide a good functional outcome [1–3]. However, dislocation of the hip is a challenging complication following modular proximal femoral replacement due to concomitant soft tissue detachment, especially after tumor resection. Recent studies have shown dislocation rates of up to 20 % [1, 4–8] for these prostheses. The status of the abductor muscles, the number of prior hip operations, the position of the implant components and the surgical approach are known factors influencing the dislocation rate [6, 9].
While previous studies on proximal femoral replacements have reported on the incidence of dislocation [1, 4, 6, 7, 10–13], to date, there are only limited data with regard to the success of the subsequent surgical or non-surgical management and the respective re-dislocation rates.
In cases of acetabular reconstructions, dislocation rates are reported to be as high as 11–16 % for pelvic prostheses [14, 15] or 15 % for pedestal cups [16]. Few data are available for dislocation rates of combined proximal femoral and peri-acetabular resections (e. g. extra-articular resections of the hip).
A specific problem to proximal femoral replacements in comparison to total hip arthroplasty (THA) is the soft-tissue reattachment to the implant [17–21]. In this context, artificial bands have been used for soft-tissue reconstruction around the proximal femur [21]. Since 2002, the Ligament Advancement Reinforcement System (LARS®, Ligament Augmentation & Reconstruction System, Arc sur Tille, France), a non-absorbable polyester band with a high potential for cellular and connective tissue ingrowth has been in use at our institution [22]. We already have reported on the use of LARS® for soft tissue reconstruction around the knee joint following tumor resection, but so far experience with peri-acetabular soft tissue reconstruction is not well described [20].
Therefore, the aims of this study were: (1) to determine the incidence of hip dislocation in tumor patients following implantation of a modular prosthesis of the proximal femur; (2) to evaluate the re-dislocation rate following surgical or non-surgical treatment; (3) to evaluate the dislocation rate in patients with an additional pelvic reconstruction; and (4) to evaluate the effects of peri-acetabular soft tissue reconstructions by LARS®.
Material and methods
A retrospective cohort study was performed using prospectively collected data from the Vienna bone and soft tissue tumor registry [23]. Between June 1982 and September 2008 limb salvage surgery for a tumor around the proximal femur, using a modular endoprosthetic replacement, was performed in 170 patients. Four patients who were operated on between 1982 and 1987 were excluded from the study because of inadequate data. All other patients have been included in this retrospective single-centre study; all medical records and patient files of our database have been reviewed. Approval of the local institutional review board was obtained prior to this investigation. The mean follow-up was 46 ± 64 month (range, one to 277 months). Sixty-five patients (39 %) had a follow-up of more than two years. During the follow-up period 113 patients (68 %) died of disease.
There were 81 males and 85 females with an average age of 50 ± 20 years (range, six to 84 years) at the time of surgery. Seventy-one patients (43 %) had a primary malignant tumor (20 osteosarcomas, 15 chondrosarcomas, 15 Ewing’s sarcomas and 21 other bone and soft tissue sarcomas). Eight patients (5 %) had either myeloma or lymphoma and one (<1 %) had eosinophilic granuloma. The average age at time of surgery was 36 ± 21 years (range, six to 84 years). In 86 patients (52 %) a proximal femur replacement was used in severe cases with metastatic disease of the femur. The average age at time of surgery was 60 ± 12 years (range, 18–83 years) for cases with metastatic disease.
The Kotz Modular Femur and Tibia Reconstruction System (KMFTR, Howmedica GmbH, Kiel, Germany) or the Howmedica Modular Reconstruction System (HMRS, Howmedica GmbH, Kiel, Germany) were implanted in 144 patients (87 %) since 1982 [2]. More recently the Global Modular Replacement System (GMRS, Stryker Corp., Mahwah, NJ) was implanted in 22 patients (13 %). A cemented stem fixation with a gentamycin loaded bone cement (Palacos®R + G; Heraeus Medical, Hanau, Germany) was performed in 12 patients (7 %). All other 158 patients had a cementless fixation of the stem. The mean length of the prosthesis was 165 ± 56 mm (range, 70–390 mm). A unipolar or bipolar head was used in 152 patients (92 %). In 14 patients (8 %), an additional pelvic reconstruction was performed due to tumor involvement of the acetabulum and pelvis including a pedestal cup (Schoellner cup, Zimmer, Warsaw, IN, USA) in four, a cage/acetabular roof reinforcement ring in four, a custom-made pelvic prosthesis in four, or a saddle prosthesis in two. In cases of pelvic reconstruction, used head sizes were 32 mm in ten patients, 36 mm in two patients and 28 mm in two patients. An “enhanced tendon attachment system” (ETA®; Stryker-Howmedica Inc.) for trochanteric fixation of gluteal structures was used in 13 patients. Nineteen patients were treated with a LARS® ligament [20] for soft tissue reconstruction between 2002 and 2008. In contrast to the LARS® ligament for ACL (anterior cruciate ligament) reconstruction, the implant used in this series of patients is designed as bands or tubes with an overall length of 40 cm and a width of 6 cm. The non-resorbable band consists of 90 non-woven longitudinal polyester fibres, which are interlinked at a molecular level with a minimal rupture level of 4000 N and an elongation rate of less than 7 % of its original length. This implant has been used for several indications in peri-articular soft tissue reconstruction and reattachment, such as augmentation or complete reconstruction of the extensor mechanism of the knee joint or prosthetic fixation of the proximal humerus or proximal femur by reconstruction of an artificial pseudo-capsule.
Patient mobilization was allowed from the first postoperative day. Initially, all patients had splints for six weeks after surgery. Subsequently all patients were advised to wear abduction braces with slow increases in range of hip motion for up to six months depending on the surgeons advice. Since the introduction of LARS® in 2002, splints or braces were only in cases of an additional pelvic reconstruction.
In case of dislocation all affected patients were either initially treated at our institution or immediately transferred to our hospital. At admittance a closed reduction under sedation was attempted in all patients as primary treatment. If relocation was possible and the hip was found to be stable, the patient was immobilized in an abduction brace for six weeks. If relocation was impossible or the hip was found to be unstable, open reduction was performed and further surgical revision was indicated depending on intra-operative findings.
Statistical analysis
Statistical analyses of the data focused on dislocations after implantation of a proximal femur modular prosthesis as the primary endpoint. Descriptive summary statistics included means and frequencies. Age and time intervals were regarded as continuous variables; all other covariates were modeled as categorical variables. Differences between means and proportions were tested with the chi-square test for categorical variables and the t-test for continuous variables. Actuarial survival was estimated using the Kaplan–Meier method. All statistical tests were two-sided. A P-value of <0.05 was considered significant. All statistical calculations and graphical visualization were made with the GraphPad Prism® (GraphPad Software Inc.; version 5.00c, 2009) software package.
Results
Overall data
The overall patient survival was 65 % at one year, 30 % at five years and 25 % at ten years (Fig. 1). A total of 52 prosthesis-related complications were observed and 21 (49 %) of these were dislocations. Corresponding implant survival rate until first dislocation was 88 % at one year and 83 % at five years (Fig. 2).
Dislocation rate
The dislocation rate over the entire period was 13 % (21 of 166) and the dislocation occurred at an average six ± eight months (range, one to 33 months) after surgery. We found a three-fold higher dislocation rate (33 % compared to 9 %, p = 0.003) in patients who were operated between 1982 and 1986 compared to the years 1987 to 2008. We found no statistical difference in patients with primary bone tumours compared to patients with metastases or others diseases considering the dislocation rate. We found no statistical difference in patients with primary bone tumours and with metastases with and without radiotherapy or chemotherapy considering the dislocation rate.
Five of 14 patients (36 %) with additional pelvic reconstruction dislocated compared to 16 of 152 patients (11 %) without additional pelvic reconstruction resulting in a three-fold higher dislocation rate (p = 0.0219).
Six of 16 patients (38 %) without additional pelvic reconstruction dislocated within six weeks after surgery. In comparison, no patient who had an additional pelvic reconstruction dislocated within six weeks after surgery (p = 0.2923).
Re-dislocation rate
The re-dislocation rate was 43 % (nine of 21) after a mean time of 39 ± 61 months (range, one to 193 months) after surgery and a mean time of 29 ± 63 months (range, one to 192 months) after first dislocation.
Five of nine patients (56 %) who dislocated twice sustained a third dislocation after a mean time of 65 ± 89 months (range, three to 220 months) after surgery and a mean time of seven ± ten months (range, one to 28 months) after dislocating for the second time.
Two of five patients (40 %) who dislocated three times sustained a fourth dislocation after a mean time of 130 ± 133 months (range, 35–224 months) after surgery and after a mean time of 12 ± 11 months (range, four to 20 months) after dislocating for the second time.
Closed reduction was performed in 12 of 21 patients (57 %) who sustained a dislocation, and an open reduction was performed in nine of 21 patients (43 %). In all cases, capsulorraphy was performed using PDS® II or Ethibond® (ETHICON Products, Norderstedt, Germany) sutures. Additionally, the implant length was reduced because of initial over-lengthening in one patient. LARS® was implanted in a second patient. Finally a complete exchange of the prosthesis including the re-fixation of on ETA® was performed in a third patient.
Additional pelvic reconstruction
Out of 16 dislocation cases without pelvic reconstruction, nine (56 %) were treated by closed reduction and 7 (44 %) were treated by open reduction. One patient (14 %) treated by open reduction sustained a re-dislocation and was finally treated by open reduction whereas six of nine patients (67 %) treated by closed reduction sustained a re-dislocation. These three were treated by open reduction and no further dislocation occurred. Three were treated by closed reduction and dislocated a third time and were repeatedly treated by closed reduction. One of them dislocated a fourth time and was again treated by closed reduction.
Out of five patients with an additional pelvic reconstruction who had sustained a dislocation, two patients were treated with open reduction and three were treated with closed reduction. None of the patients treated by open reduction had a re-dislocation. Two of three patients (67 %) treated conservatively had a re-dislocation. One patient was treated by closed reduction and sustained a further dislocation before being successfully treated by open reduction. The other patient was treated by open reduction twice and revision of the acetabular cup was performed due to aseptic loosening of the cup. This patient dislocated a fourth time and underwent another revision due to an inadequate cup position.
Overall, the re-dislocation rate was 58 % (eight of 12) after initial closed reduction compared to 11 % (one of nine) after primary treatment with an open reduction (p = 0.0357).
There was no significant correlation between implant size, cup size or head size and the dislocation and re-dislocation rates for patients with or without pelvic reconstruction (Figs. 3 and 4).
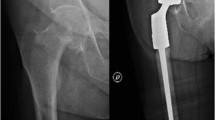
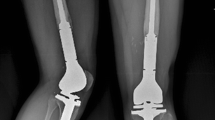
Radiological imaging of a 64-year-old male patient with history of renal cell carcinoma and bone metastasis to the right proximal femur and acetabulum with pathological subtrochanteric fracture. The patient underwent resection of the proximal femur and extended curettage of the acetabular defect. Reconstruction was performed by a modular endoprosthesis of the proximal femur (GMRS®, Stryker Corp, Mahwah, NJ) and a pedestal cup (Schoellner cup, Zimmer, Warsaw, IN). The patient suffered dislocation three months after surgery and was treated by closed reduction and bracing for six weeks without further dislocation until death of disease. a AP radiograph of the proximal femur preoperative. b Frontal plain MRI of the lesion. c and d Postoperative AP radiograph. e AP radiograph of the dislocated prosthesis. f AP radiograph after closed reduction and bracing
Artificial band for soft tissue reconstruction
Two of 19 patients (11 %) with an additional soft tissue reconstruction by LARS® sustained a dislocation, and one had a pelvic reconstruction.
Discussion
Dislocation of the hip is a challenging complication in tumor patients following modular proximal femoral replacement. In this study we present the dislocation rates as well as the success rates of the subsequent treatment of a large consecutive cohort of modular endoprosthesis of the proximal femur.
The overall dislocation rate in our cohort was 13 % during this 30-year timespan. We found a three-fold higher dislocation in patients treated between 1982 and 1986 compared to the remaining time period (33 % vs. to 9 %, p = 0.003), indicating a surgical learning curve with this type of implant. We also found that an additional pelvic reconstruction led to a three-fold higher dislocation rate (36 % [five of 14] compared to 11 % [16/152]; p = 0.0219). Several reports have addressed the incidence of dislocation after modular reconstruction of the proximal femur. Zehr et al. [7] reported a dislocation rate of 18 % (three of 17), and Kabukcuoglu et al. [4] reported a dislocation rate of 11 % (six of 54) in tumor patients who were treated between 1972 and 1992. Menendez et al. [6] reported a dislocation rate of 10 % (ten of 96) in patients who were treated between 1992 and 2003. Chandrasekar et al. [1] presented a dislocation rate of 6 % (six of 100) in patients who were treated between 2001 and 2006. He also reported that three of six patients (50 %) who sustained a dislocation had an additional pelvic reconstruction. This suggests that an additional bone and soft tissue loss in the pelvic region is a severe risk factor for hip dislocation in patients with proximal femoral replacement.
While previous studies only reported on the incidence of dislocation alone, they did not address the success or failure rate of the subsequent treatment or the exact timing of the dislocation. Here, we were able to show that most dislocations occur within four months after surgery (13 of 21). Interestingly, patients with additional pelvic reconstruction dislocated later than patients without additional pelvic reconstruction. Yet, patients with additional pelvic reconstruction had to wear abduction braces for a considerable time period and were mobilized slower than patients without an additional pelvic reconstruction. This may be the reason why these patients dislocated later. Compared to primary THA or hemiarthroplasty [24] the dislocation rates in proximal femur reconstruction are significantly higher but are rather similar to those of patients who underwent revision arthroplasty or had primary total hip arthroplasty for neoplastic disease [25, 26].
Our results indicate that non-surgical management of hip dislocation in proximal femoral replacements may be inferior to surgical management with regard to re-dislocation. Eight of 12 patients (58 %) who were treated with closed reduction sustained a re-dislocation. Only one of nine patients (11 %) treated with open reduction after their first dislocation sustained a second dislocation. Additionally the use of the LARS® has shown that dislocation was slightly reduced but not avoided. Moreover, in cases of dislocation with an artificial ligament in situ, closed reduction might be impossible due to its impingement around the acetabulum. Yet, further studies with larger sample sizes are needed to address the topic of reconstruction with an artificial ligament. Mendendez et al. [6] reported that two of ten patients were treated by closed reduction and five of ten patients were treated by open reduction. In three of ten a revision of the implant was necessary. All three of the revisions for instability were in patients with THA head segments, which may suggest that the use of a bipolar endoprosthesis may decrease the risk of this complication. Other reports support this theory [27]. Surprisingly, we could not find any significant difference between head sizes in patients who sustained a dislocation and patients who did not. Furthermore we did not find a statistically significant difference considering the sizes between patients who had a dislocation with or without pelvic reconstruction. These variables are within the control of the surgeon and can be adjusted differently with modular designs of proximal femur megaprosthesis. In another study by Chandrasekar et al. [1], six postoperative dislocations occurred, of which three patients had an acetabular reconstruction. The other three dislocations occurred in the 91 patients with a unipolar femoral heads.
Alberton et al. [9] described the dislocation of 115 hips with an overall dislocation rate of 7 % from 1980 to 1989 for patients with revision hip arthroplasty. The initial management of dislocation was non-operative in 103 of the 115 (90 %) hips. Sixty-seven of the 103 (65 %) patients had subsequent instability and 38 of the 67 (66 %) patients had to have surgery. Our data indicates that surgical treatment appears to be superior to prevent re-dislocation, especially in cases with additional pelvic reconstruction. Although surgery involves several unpredictable risks, open reduction seems to be more effective if a patient sustains recurrent dislocations. However, open revision was in almost all cases just a soft-tissue procedure, where the present scar tissue was reinforced and sutured with strong slowly-absorbable or non-absorbable sutures. Moreover we cannot exclude a certain selection bias since all irreducible dislocations were operated and this may be a different patient cohort than the ones who are reducible under sedation.
Clearly there are several limitations to our study. It is a retrospective study overlooking a vast time span of treatment, during which the quality of diagnostic and therapeutic means have considerably changed and therefore may have influenced outcomes. Obviously, surgical or non-surgical treatment was not randomized. The evaluation of hip stability during initial closed reduction as well as the indication for surgical revision was thus subjective and several surgeons were involved in this process. Moreover, we did not precisely measure the acetabular component position, which must be regarded as a detrimental risk factor for dislocation. However, only 14 of 166 patients (8 %) had a pelvic reconstruction or an acetabular cup, while all other patients had a hemiprosthetic implant, not affecting the bony pelvic anatomy. The vast study period explains historically missing data on the amount of soft tissue resection during tumor surgery and postoperative function. Since a large number of these parameters are missing and hence could not be reviewed, we decided to exclude these parameters from our study rather than presenting inconclusive data with potential bias. Moreover the sample size for patients with an artificial ligament may be too small to draw conclusions regarding the efficacy of this treatment.
In conclusion, we found a high dislocation rate in proximal femur replacement. Patients with a combined pelvic reconstruction are at a higher risk for dislocation. Our results indicate that surgical treatment might result in a lower re-dislocation rate than closed reduction, yet large randomized multicenter studies are needed to further evaluate this clinically important matter.
References
Chandrasekar CR, Grimer RJ, Carter SR, Tillman RM, Abudu AT, Buckley L (2009) Modular endoprosthetic replacement for tumours of the proximal femur. J Bone Joint Surg Br 91:108–112. doi:10.1302/0301-620X.91B1.20448
Kotz RI (2014) Progress in musculoskeletal oncology from 1922–2012. Int Orthop 38:1113–1122. doi:10.1007/s00264-014-2315-0
Tunn PU, Pomraenke D, Goerling U, Hohenberger P (2008) Functional outcome after endoprosthetic limb-salvage therapy of primary bone tumours–a comparative analysis using the MSTS score, the TESS and the RNL index. Int Orthop 32:619–625. doi:10.1007/s00264-007-0388-8
Kabukcuoglu Y, Grimer RJ, Tillman RM, Carter SR (1999) Endoprosthetic replacement for primary malignant tumors of the proximal femur. Clin Orthop Relat Res 358:8–14
Gosheger G, Gebert C, Ahrens H, Streitbuerger A, Winkelmann W, Hardes J (2006) Endoprosthetic reconstruction in 250 patients with sarcoma. Clin Orthop Relat Res 450:164–171. doi:10.1097/01.blo.0000223978.36831.39
Menendez LR, Ahlmann ER, Kermani C, Gotha H (2006) Endoprosthetic Reconstruction for Neoplasms of the Proximal Femur. Clin Orthop Relat Res 450:46–51. doi:10.1097/01.blo.0000229332.91158.05
Zehr RJ, Enneking WF, Scarborough MT (1996) Allograft-prosthesis composite versus megaprosthesis in proximal femoral reconstruction. Clin Orthop Relat Res 322:207–223
Malkani AL, Settecerri JJ, Sim FH, Chao EY, Wallrichs SL (1995) Long-term results of proximal femoral replacement for non-neoplastic disorders. J Bone Joint Surg Br 77:351–356
Alberton GM, High WA, Morrey BF (2002) Dislocation after revision total hip arthroplasty : an analysis of risk factors and treatment options. J Bone Joint Surg Am 84-A:1788–1792
Parvizi J, Tarity TD, Slenker N, Wade F, Trappler R, Hozack WJ, Sim FH (2007) Proximal femoral replacement in patients with non-neoplastic conditions. J Bone Joint Surg Am 89:1036–1043. doi:10.2106/JBJS.F.00241
Parvizi J, Sim FH (2004) Proximal femoral replacements with megaprostheses. Clin Orthop Relat Res 420:169–175
Finstein JL, King JJ, Fox EJ, Ogilvie CM, Lackman RD (2007) Bipolar proximal femoral replacement prostheses for musculoskeletal neoplasms. Clin Orthop Relat Res 459:66–75. doi:10.1097/BLO.0b013e31804f5474
Donati D, Zavatta M, Gozzi E, Giacomini S, Campanacci L, Mercuri M (2001) Modular prosthetic replacement of the proximal femur after resection of a bone tumour a long-term follow-up. J Bone Joint Surg Br 83:1156–1160
Windhager R, Karner J, Kutschera HP, Polterauer P, Salzer-Kuntschik M, Kotz R (1996) Limb salvage in periacetabular sarcomas: review of 21 consecutive cases. Clin Orthop Relat Res 331:265–276
Rudert M, Holzapfel BM, Pilge H, Rechl H, Gradinger R (2012) Partial pelvic resection (internal hemipelvectomy) and endoprosthetic replacement in periacetabular tumors. Oper Orthop Traumatol 24:196–214. doi:10.1007/s00064-012-0161-z
Fisher NE, Patton JT, Grimer RJ, Porter D, Jeys L, Tillman RM, Abudu A, Carter SR (2011) Ice-cream cone reconstruction of the pelvis: a new type of pelvic replacement: early results. J Bone Joint Surg Br 93:684–688. doi:10.1302/0301-620X.93B5.25608
Masterson EL, Ferracini R, Griffin AM, Wunder JS, Bell RS (1998) Capsular replacement with synthetic mesh: effectiveness in preventing postoperative dislocation after wide resection of proximal femoral tumors and prosthetic reconstruction. J Arthroplast 13:860–866
Gottsauner-Wolf F, Egger EL, Giurea A, Antosch M, Olsen D, Rock MG, Sim FH (1999) Biologic attachment of an allograft bone and tendon transplant to a titanium prosthesis. Clin Orthop Relat Res 358:101–110
Gottsauner-Wolf F, Egger EL, Schultz FM, Sim FH, Chao EY (1994) Tendons attached to prostheses by tendon-bone block fixation: an experimental study in dogs. J Orthop Res 12:814–821. doi:10.1002/jor.1100120609
Dominkus M, Sabeti M, Toma C, Abdolvahab F, Trieb K, Kotz RI (2006) Reconstructing the extensor apparatus with a new polyester ligament. Clin Orthop Relat Res 453:328–334. doi:10.1097/01.blo.0000229368.42738.b6
Gosheger G, Hillmann A, Lindner N, Rödl R, Hoffmann C, Bürger H, Winkelmann W (2001) Soft tissue reconstruction of megaprostheses using a trevira tube. Clin Orthop Relat Res 393:264-271
Trieb K, Blahovec H, Brand G, Sabeti M, Dominkus M, Kotz R (2004) In vivo and in vitro cellular ingrowth into a new generation of artificial ligaments. Eur Surg Res 36:148–151. doi:10.1159/000077256
Kotz R, Dominkus M, Zettl T, Ritschl P, Windhager R, Gadner H, Zielinski C, Salzer-Kuntschik M (2002) Advances in bone tumour treatment in 30 years with respect to survival and limb salvage. A single institution experience. Int Orthop 26:197–202. doi:10.1007/s00264-002-0365-1
Meek RM, Allan DB, McPhillips G, Kerr L, Howie CR (2008) Late dislocation after total hip arthroplasty. Clin Med Res 6:17–23. doi:10.3121/cmr.2008.770
Mahomed NN, Barrett JA, Katz JN, Phillips CB, Losina E, Lew RA, Guadagnoli E, Harris WH, Poss R, Baron JA (2003) Rates and outcomes of primary and revision total hip replacement in the United States medicare population. J Bone Joint Surg Am 85-A:27–32
Schneiderbauer MM, Sierra RJ, Schleck C, Harmsen WS, Scully SP (2005) Dislocation rate after hip hemiarthroplasty in patients with tumor-related conditions. J Bone Joint Surg Am 87:1810–1815. doi:10.2106/JBJS.D.02830
Howie DW, Holubowycz OT, Middleton R (2012) Large femoral heads decrease the incidence of dislocation after total hip arthroplasty: a randomized controlled trial. J Bone Joint Surg Am 94:1095–1102. doi:10.2106/JBJS.K.00570
Conflict of interest
The authors did not receive any outside funding or grants in support of their research for this study. Neither they nor a member of their immediate families received payments or other benefits or a commitment or agreement to provide such benefits from a commercial entity.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Puchner, S.E., Funovics, P.T., Hipfl, C. et al. Incidence and management of hip dislocation in tumour patients with a modular prosthesis of the proximal femur. International Orthopaedics (SICOT) 38, 1677–1684 (2014). https://doi.org/10.1007/s00264-014-2376-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00264-014-2376-0