Abstract
Purpose
Osteoarticular infections due to methicillin-susceptible Staphylococcus aureus (MSSA) or its methicillin-resistant variant (MRSA) are feared due to treatment failures. According to clinical experience, Pseudomonas aeruginosa may reveal less long-term remission than S. aureus.
Methods
A case-controlled study comparing outcomes of osteoarticular infections due to P. aeruginosa vs S. aureus was performed at Geneva University Hospitals.
Results
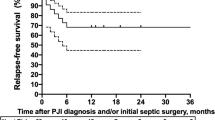
A total of 111 S. aureus (including 37 MRSA) and 20 P. aeruginosa osteoarticular infections were analysed in 131 patients: arthroplasties (n = 38), fracture fixation devices (n = 56), native joint arthritis (n = 7) and osteomyelitis without implant (n = 30). The median active follow-up time was 4 years. The patients underwent a median number of two surgical interventions for P. aeruginosa infections compared to two for S. aureus (two for MRSA), while the median duration of antibiotic treatment was 87 days for P. aeruginosa and 46 days for S. aureus infections (58 days for MRSA) (all p > 0.05). Overall, Pseudomonas-infected patients tended towards a lower remission rate than those infected with S. aureus (12/20 vs 88/111; p = 0.06). This was similar when P. aeruginosa was compared with MRSA alone (12/20 vs 30/37; p = 0.08). In multivariate logistic regression analyses adjusting for case mix, odds ratios (OR) for remission were as follows: P. aeruginosa vs S. aureus [OR 0.4, 95% confidence interval (CI) 0.1–1.2], number of surgical interventions (OR 0.6, 95% CI 0.5–1.0) and duration of antibiotic treatment (OR 1.0, 95% CI 1.0–1.0).
Conclusions
Despite a similar number of surgical interventions and longer antibiotic treatment, osteoarticular infections due to P. aeruginosa tended towards a lower remission rate than infections due to S. aureus in general or MRSA in particular.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Staphylococcus aureus comprises up to two thirds of all pathogens in osteoarticular infections [1], which are difficult to treat because of the organism’s ability to form small colony variants and to grow into biofilms [2]. In a recent report comparing the outcomes of orthopaedic implant infections due to methicillin-susceptible S. aureus (MSSA), methicillin-resistant S. aureus (MRSA) and coagulase-negative staphylococci, overall remission was worst for MRSA (57%), followed by MSSA (72%) and best for coagulase-negative staphylococci (82%) [3]. Kilgus et al. showed that hip arthroplasties infected due to MRSA were treated successfully in only 48% of cases, as compared to 81% with MSSA infection [4]. Salgado et al. attributed a ninefold higher ratio for treatment failure in prosthetic joint infections due to MRSA than due to MSSA [5]. The surgical and medical literature is full of publications regarding staphylococcal osteoarticular infections, especially for MRSA.
In contrast, Pseudomonas aeruginosa is an under-reported Gram-negative pathogen in the orthopaedic domain. P. aeruginosa is at least as clinically virulent as S. aureus and also readily forms biofilms [6, 7] using extensive quorum-sensing abilities [8], develops resistance to administered antibiotics more rapidly than S. aureus and produces a large variety of virulence factors such as type III secretion-dependent cytotoxicity [7, 8]. Unsurprisingly, P. aeruginosa is feared by orthopaedic surgeons and infectious disease specialists with experience in osteoarticular infections. Many of them would attribute a worse prognosis for pseudomonal bone and joint infections compared to S. aureus.
However, there is little literature about P. aeruginosa and osteoarticular infections. P. aeruginosa accounts for an absolute minority of all osteoarticular pathogens [1], and most reports are either case series involving around 20–30 cases [9–13] or even less [14–27]. Moreover, these reports subsume P. aeruginosa under other Gram-negative pathogens [9, 12, 13, 19], including anaerobes [10, 28], and lack adjustment for case mix [9, 10, 19, 21]. Often, outcome comparisons of osteoarticular infections due to P. aeruginosa with Gram-positive pathogens are almost non-existent [10, 28]. Hsieh et al. retrospectively compared arthroplasty infections due to Gram-positive pathogens to the same infections due to Gram-negative pathogens. They found that conservative treatment of Gram-negative infection by debridements was associated with a lower success rate than treating Gram-positive arthroplasty infections (27 vs 47% remission). However, this difference vanished when a two-stage exchange was performed [10].
In this clinical case-control study, we investigate the hypothesis of lower remission risks of pseudomonal osteoarticular infections compared to S. aureus (the most frequent pathogen) and MRSA (the pathogen with the most frequent treatment relapses known so far) by adjusting for case mix of the underlying complex patient population.
Methods
Setting and data collection
The Orthopedic Service of Geneva University Hospitals is a tertiary centre with a septic subunit consisting of 22 acute care beds. A dedicated team of orthopaedic surgeons, infectious disease specialists, diabetologists, nurses and physiotherapists treat patients with osteoarticular infections [29].
Prospectively registered databases from the arthroplasty cohort and septic orthopaedic cohort were searched for osteoarticular infections among adult orthopaedic patients from January 1996 to November 2009. Patients were followed up to 31 July 2011. Forty-two variables for each episode were assessed related to demographic characteristics, immunosuppression, microbiology, surgical and antibiotic treatment modalities and outcomes. The quantitative microbiological procedures were unchanged during the study period and based on the Clinical and Laboratory Standards Institute (CLSI) guidelines [30] and in-house MRSA identification methods [3]. No typing was performed. Since clinical specimens had not been stored, no retrospective analyses of minimal inhibitory concentrations could be performed. The study was supported by the local Ethics Committee (No. 08-017R; 08-029R).
Criteria and definitions
Definition of osteoarticular infection required local signs of infection such as heat, erythema, pus or functional impairment; a medical report; a targeted antibiotic treatment; and the presence of the same pathogen in at least three intra-operative samples. In order to enhance specificity, only cultures which were grown on plates were considered. Co-pathogens were accepted only if S. aureus or P. aeruginosa outnumbered them by at least threefold in intra-operative microbiological specimens, for instance coagulase-negative staphylococci (103 cfu/ml) in one specimen and P. aeruginosa in all four specimens. Only the first episode of bone and joint infection per patient was included; patients presenting with relapsing episodes were excluded. Further exclusion criteria were paediatric patients (≤ 16 years), cure by amputation, involvement of fingers or toes only and a minimal active follow-up of less than 18 months after end of treatment.
Remission was defined as complete clinical and laboratory resolution of the former infection. Immunosuppressed patients were considered those with active neoplasm, insulin-dependent diabetes mellitus, Child class C cirrhosis, dialysis or those requiring chronic steroid therapy (prednisolone equivalence of 15 mg/day). Multi-resistance among P. aeruginosa strains meant resistance to at least four classes of antibiotic agents.
Statistical analyses
Group comparisons were performed using the Pearson χ2 test, Fisher’s exact test or the Wilcoxon rank sum test, as appropriate. Logistic regression analyses determined associations with remission. Independent variables with a p value ≤0.2 in univariate analysis were added stepwise in the multivariate analysis to adjust for case mix. We included five to ten predictor variables per outcome event [31]. The following variables were introduced into the final model independently of their association in univariate analysis: P. aeruginosa vs S. aureus, age, duration of antibiotic treatment and number of surgical interventions. Since comparison of P. aeruginosa with S. aureus (MSSA & MRSA) might be different from the comparison of P. aeruginosa with MRSA alone, all analyses were repeated for S. aureus and MRSA separately. Key variables were checked for collinearity and interaction, the latter by Mantel-Haenszel estimates. A p value ≤ 0.05 (two-tailed) was considered significant. Stata software (9.0, StataCorp., College Station, TX, USA) was used.
Results
Infections and patients
A total of 131 episodes of osteoarticular infections in 131 adult orthopaedic patients were retrieved. The median age was 63 years and 53 were women. Thirty-seven patients (28%) were immunocompromised: 17 due to diabetes mellitus, nine by advanced cirrhosis due to alcoholism, one by dialysis, one by cancer and nine by chronic steroid medication. The infected implants in the arthroplasty group included total hip (n = 22) and total knee (n = 16) prostheses. In the fracture fixation devices group, the infected implants included plates (n = 28), intramedullary nails (n = 14), external fixation devices (n = 7), hip screws (n = 3), other screws (n = 2), patellar cerclage wire (n = 1) and spondylodesis material (n = 1). Additionally, we retrieved seven episodes of septic native joint arthritis and 30 episodes of osteomyelitis without implant. Crude group comparisons of demographic and treatment characteristics according to the pathogen are displayed in Table 1.
Pathogens
According to study definitions, we included osteoarticular infections due to S. aureus and P. aeruginosa only. With 111 infectious episodes, S. aureus was overwhelmingly predominant compared to P. aeruginosa (20 episodes). In 37 cases, S. aureus was resistant to methicillin. In 21 S. aureus infections, there was a co-pathogen with at minimum a threefold lesser quantitative growth in cultures: coagulase-negative staphylococci (four), streptococci (seven), Proteus spp. (five), Klebsiella spp. (two), Escherichia coli (one), Enterobacter cloacae (one) and Enterococcus faecalis (one). Co-pathogens were seen in six pseudomonal infections: coagulase-negative staphylococci (four), Enterobacter spp. (one) and Klebsiella spp. (one). With the exception of two multi-resistant P. aeruginosa episodes, all pseudomonal infections were susceptible to most antipseudomonal antibiotic agents, 11 of which were susceptible to every antipseudomonal antibiotic tested.
Among the S. aureus cases, all but two were surgical site infections, while the origin of P. aeruginosa infections was heterogeneous: open fractures (seven), surgical site infections (five), haematogenous infections (two; one of which was in a patient with sickle cell anaemia), IV drug abuse (one), unknown (two) and pressure ulcer (three). Two patients had prior empiric antibiotic therapy before diagnosis. We failed to establish epidemiological links between the different cases of pseudomonal or MRSA infections.
Surgical treatment
All patients underwent surgical treatment with a median of two surgical interventions in the operating theatre (range one to six surgeries). Pseudomonal and staphylococcal surgical treatments did not differ in the number of interventions (median two vs two interventions, p = 0.43) (Table 1).
Antibiotic treatment
All patients received systemic antibiotic therapy. No antibiotics or detergents [32] were added to the irrigating solutions. Gentamicin beads were used in six cases (three for S. aureus and three for P. aeruginosa) for a median duration of three weeks. The median duration of total antibiotic therapy was eight weeks (range six–14 weeks) for the entire study population. The median duration of antibiotic treatment tended to be longer for P. aeruginosa (87 days), compared to 46 days for S. aureus infections, and 58 days for MRSA, although the differences were not significant (all p > 0.05) (Table 1).
There were no clear preferences for the choice of antibiotic agents. For staphylococcal parenteral therapy, flucloxacillin, amoxicillin/clavulanic acid and vancomycin were the most frequently used agents and ciprofloxacin/rifampicin and clindamycin for oral treatment. In 44 staphylococcal infections (40%), rifampin was used in combination therapy at a median dose of 1,200 mg/day. The choices were culture based.
For pseudomonal infection, parenteral therapy consisted of ceftazidime (n = 5), cefepime (n = 4), imipenem (n = 2), piperacillin (n = 1) and polymyxin B (n = 1). Seven episodes were treated by oral ciprofloxacin 500–750 mg b.i.d. from the start. Thirteen episodes revealed a combined initial parenteral antimicrobial treatment for P. aeruginosa. Including rifampin treatment for S. aureus, 50 episodes (38%) were treated with a combination therapy during the majority of the antibiotic administration time. Three recurrent P. aeruginosa strains (3/20; 15%) became resistant to one of the antimicrobial agents used during initial treatment (twice to ciprofloxacin and once to imipenem). Except for one case there was no ciprofloxacin resistance among pseudomonal isolates at the beginning of therapy.
Outcomes
Overall, 100 episodes (76%) remained in remission after a median active follow-up time of 4.0 years (range 1.5–13 years). Stratified by the pathogen, a tendency for a lower remission rate was found for pseudomonal infections as compared to S. aureus (12/20 vs 88/111; p = 0.06). This was similar when P. aeruginosa was compared with MRSA alone (12/20 vs 30/37; p = 0.08) (Table 1).
We further stratified remission rate according to the type of infection. In all three groups of infection, P. aeruginosa tended towards a lower remission rate than S. aureus, but none of these results were statistically significant: 2/5 vs 20/25 [odds ratio (OR) 0.2, p = 0.1] for osteomyelitis without implant, 2/3 vs 3/4 (OR 0.7, p = 1.0) for septic native arthritis and 8/12 vs 61/78 (OR 0.5, p = 0.5) for implant-related infections as a group. Of note, among the seven episodes of pseudomonal infections treated with oral ciprofloxacin from the start, five were cured and two failed to respond. This proportion was not different from P. aeruginosa episodes treated with initial parenteral therapy (5/7 vs 6/13; p = 0.58).
Multivariate analysis
Because of the heterogeneity of the study population and differences in crude group comparison between MRSA and P. aeruginosa infections (Table 1), we performed two multivariate logistic regression analyses to adjust for this case mix. The first analysis compared P. aeruginosa with S. aureus as a group and the second P. aeruginosa with MRSA.
No demographic or therapeutic variable showed a significant association with remission in either of the multivariate analyses, although pseudomonal infection tended towards a lower remission rate with an OR of 0.4 (p = 0.08)(Table 2). While the number of surgical interventions tended to be less associated with remission, the duration of antibiotic treatment did not influence outcome. Less than six weeks of total antibiotic treatment had the same outcome as six–12 weeks [OR 0.8, 95% confidence interval (CI) 0.3–2.6] or more than 12 weeks (OR 0.4, 95% CI 0.1–1.7). Our statistical model yielded a non-significant goodness-of-fit test (p = 0.4; p = 0.2 for MRSA comparison) and a receiver-operating characteristic (ROC) curve value of 0.72 (ROC curve value 0.83 for MRSA comparison), highlighting an acceptable appropriateness of our final models.
Discussion
Formally, we report a trend towards a lower remission rate for orthopaedic infections due to P. aeruginosa compared to S. aureus or to MRSA with an adjusted OR of 0.4 for remission (2.5 for failure) in multivariate analysis. Although this result was formally statistically insignificant, we detected this tendency in all analyses performed.
Our failure risk of 24% is congruent with the 20% [12], 21% [13], 25% [27], 27% [14] or 30% [9] failure rates reported for pseudomonal osteoarticular infections, although lower (5% [11]; 7% [21]) or higher failure rates ranging from 33% [23], 35% [24], 38% [22] to 39% [15] also exist in small case series from the 1980s. Development of resistance among failure cases to the antimicrobial agents used during therapy occurred in 15%, which is congruent with the 13–24% risk after a prolonged antibiotic monotherapy during three–four months [9, 24]. Hence, we exclude a major setting bias. The only particularity of our study population is the relatively small proportion of haematogenous infections due to S. aureus (2 of 111), which has been reported more frequently in another tertiary centre of Switzerland [33].
Of note, infections due to P. aeruginosa benefited from the same median number of surgical interventions and an enhanced median duration of 87 days of antibiotic administration compared to 46 days for S. aureus and still tended towards more treatment failures. Conversely, duration of post-debridement antibiotic administration was unrelated to remission. Less than six weeks of antibiotic treatment yielded the same remission as six–12 or more than 12 weeks. Our data suggest that the tendency towards a lower remission rate in pseudomonal osteoarticular infections cannot be compensated for by simple prolongation of post-debridement antibiotic therapy. This finding is congruent with recent retrospective data suggesting that a total antibiotic duration of maximally six weeks post-debridement [3, 16, 20, 34], or regimens with an early switch to oral antibiotics with good oral bioavailability and bone penetration [11], are as effective as prolonged parenteral regimens [14, 35]. Longer treatment durations, e.g. for more than three months [9, 14, 20, 33] or longer [13], did not enhance remission when compared in the literature. Dan et al. treated 22 cases of pseudomonal osteomyelitis with oral ciprofloxacin with various durations ranging from four to 12 weeks. Remission rates remained the same or were even enhanced with short regimens (95%) [11].
We cannot comment on the effect of combination antibiotic therapy for pseudomonal bone and joint infections, since few of our episodes had been treated with monotherapy. Although a more favourable outcome of combined regimens is still under debate for severe pseudomonal infections such as bacteraemia [7] or pneumonia [7], so far no study could really provide convincing evidence for the benefits of a targeted combined regimen for these infections once the results of the susceptibility testing became available, let alone for osteoarticular infections. Indeed, orthopaedic and infectious diseases studies reporting a dual therapy for P. aeruginosa [13, 14] seem to have similar remission incidences than reports with single-agent treatment [15, 26]. In one randomised comparative trial, seven cases of biopsy-proven P. aeruginosa osteomyelitis were treated with oral ciprofloxacin, and six patients received a standard parenteral therapy including an aminoglycoside plus an antipseudomonal beta-lactam during six–eight months. Although it appeared that patients on standard therapy did better (two vs four failures), the difference was statistically insignificant and numbers were too small to permit a valid evaluation [13, 25]. In another randomised study comparing oral ciprofloxacin to parenteral ceftazidime and amikacin, Gentry and Rodriguez reported equal efficacy of the regimens (77% remission vs 79%) [27].
Our study has limitations: (1) It was a retrospective, single-centre study with a small and heterogeneous population. The small sample size may hide differences that would have been seen if the analyses had been performed with a larger patient population. However, such a sample size has never been achieved for osteoarticular infections due to P. aeruginosa to the best of our knowledge. According to our estimation, a study population of at least 120 P. aeruginosa patients would be required. (2) Recurrences of osteomyelitis after several years, if not decades, have been reported [36] and there is no internationally accepted minimal follow-up duration. In the literature, minimal follow-up times range from three [16] to six months [21, 25, 26] or one [12, 22, 23] to two years [9, 10]. Hence, we consider our individual minimal and median follow-up times of 1.5 and four years as a strong point of our study. (3) Patients with failures treated in another hospital may have been undetected. However, Geneva University Hospitals are by far the largest and only public hospitals in the area; we therefore consider this possibility as low. (4) The completeness of initial surgical debridement as well as the consideration of tissue vascularity and bone necrosis are of paramount importance [23]. Surgeons know how extensively they had performed this debridement. There are no retrospective possibilities to estimate the completeness of such a debridement. (5) Staphylococcal small colony variants are associated with persistence of bone infections [2]. We could not assess their presence, since our specimens had not been stored. (6) Despite adjustment for case mix, we cannot completely exclude therapeutic decision bias. Patients, who were doing less well, might have deserved a longer antibiotic treatment. Likewise, patients infected due to P. aeruginosa may have special circumstances that explain this unusual pathogen. Indeed, a large part of our Pseudomonas cases were due to open fractures. (7) The period of data collection is from 1996 to 2009. During this period, surgical technique and knowledge about osteoarticular infections might have evolved.
Conclusions
Despite a similar number of surgical interventions and a trend towards longer antibiotic treatment, osteoarticular infections due to P. aeruginosa may tend towards a lower remission rate than infections due to S. aureus or MRSA. This assumption has existed for 20 years but has not yet been proven [37]. Our single-centre study warrants confirmation in large multicentre cohorts. If such larger databases confirm a worse outcome for P. aeruginosa osteoarticular infections, specific research on treatment strategies and prevention is needed. In today’s recommendations, only the choice of the antimicrobial agent [38] is pathogen dependent, but not its duration or the surgical approach, with a few exceptions for tuberculosis, fungal infection or nocardiosis [39].
References
Hedstrom SA (1997) Septic bone and joint infections. Curr Opin Rheumatol 9:317–320
Sendi P, Rohrbach M, Graber P, Frei R, Ochsner PE, Zimmerli W (2006) Staphylococcus aureus small colony variants in prosthetic joint infection. Clin Infect Dis 43:961–967
Teterycz D, Ferry T, Lew D, Stern R, Assal M, Hoffmeyer P, Bernard L, Uçkay I (2010) Outcome of orthopedic implant infections due to different staphylococci. Int J Infect Dis 14:e913–e918
Kilgus DJ, Howe DJ, Strang A (2002) Results of periprosthetic hip and knee infections caused by resistant bacteria. Clin Orthop Relat Res 404:116–124
Salgado CD, Dash S, Cantey JR, Marculescu CE (2007) Higher risk of failure of methicillin-resistant Staphylococcus aureus prosthetic joint infections. Clin Orthop Relat Res 461:48–53
Neut D, Hendriks JG, van Horn JR, van der Mei HC, Busscher HJ (2005) Pseudomonas aeruginosa biofilm formation and slime excretion on antibiotic-loaded bone cement. Acta Orthop 76:109–114
van Delden C (2007) Pseudomonas aeruginosa bloodstream infections: how should we treat them? Int J Antimicrob Agents 30:S71–S75
Köhler T, Buckling A, van Delden C (2009) Cooperation and virulence of clinical Pseudomonas aeruginosa populations. Proc Natl Acad Sci U S A 106:6339–6344
Galanakis N, Giamarellou H, Moussas T, Dounis E (1997) Chronic osteomyelitis caused by multi-resistant Gram-negative bacteria: evaluation of treatment with newer quinolones after prolonged follow-up. J Antimicrob Chemother 39:241–246
Hsieh PH, Lee MS, Hsu KY, Chang YH, Shih HN, Ueng SW (2009) Gram-negative prosthetic joint infections: risk factors and outcome of treatment. Clin Infect Dis 49:1036–1043
Dan M, Siegman-Igra Y, Pitlik S, Raz R (1990) Oral ciprofloxacin treatment of Pseudomonas aeruginosa osteomyelitis. Antimicrob Agents Chemother 34:849–852
Martínez-Pastor JC, Muñoz-Mahamud E, Vilchez F, García-Ramiro S, Bori G, Sierra J, Martínez JA, Font L, Mensa J, Soriano A (2009) Outcome of acute prosthetic joint infections due to gram-negative bacilli treated with open debridement and retention of the prosthesis. Antimicrob Agents Chemother 53:4772–4777
Legout L, Senneville E, Stern R, Yazdanpanah Y, Savage C, Roussel-Delvalez M, Rosele B, Migaud H, Mouton Y (2006) Treatment of bone and joint infections caused by Gram-negative bacilli with a cefepime-fluoroquinolone combination. Clin Microbiol Infect 12:1030–1033
Lucht RF, Fresard A, Berthelot P, Farizon F, Aubert G, Dorche G, Bousquet G (1994) Prolonged treatment of chronic Pseudomonas aeruginosa osteomyelitis with a combination of two effective antibiotics. Infection 22:276–280
Lesse AJ, Freer C, Salata RA, Francis JB, Scheld WM (1987) Oral ciprofloxacin therapy for gram-negative bacillary osteomyelitis. Am J Med 82:247–253
Antón E (2010) Pseudomonas arthritis in an elderly woman. J Emerg Med Jul 23
Walton K, Hilton RC, Sen RA (1985) Pseudomonas arthritis treated with parenteral and intra-articular ceftazidime. Ann Rheum Dis 44:499–500
Keynes SA, Due SL, Paul B (2009) Pseudomonas arthropathy in an older patient. Age Ageing 38:245–246
Cordero-Ampuero J, Esteban J, García-Rey E (2010) Results after late polymicrobial, gram-negative, and methicillin-resistant infections in knee arthroplasty. Clin Orthop Relat Res 468:1229–1236
Elliott SJ, Aronoff SC (1985) Clinical presentation and management of Pseudomonas osteomyelitis. Clin Pediatr (Phila) 24:566–570
Brouqui P, Rousseau MC, Stein A, Drancourt M, Raoult D (1995) Treatment of Pseudomonas aeruginosa-infected orthopedic prostheses with ceftazidime-ciprofloxacin antibiotic combination. Antimicrob Agents Chemother 39:2423–2425
Greenberg RN, Kennedy DJ, Reilly PM, Luppen KL, Weinandt WJ, Bollinger MR, Aguirre F, Kodesch F, Saeed AM (1987) Treatment of bone, joint, and soft-tissue infections with oral ciprofloxacin. Antimicrob Agents Chemother 31:151–155
Scully BE, Neu HC, Parry MF, Mandell W (1986) Oral ciprofloxacin therapy of infections due to Pseudomonas aeruginosa. Lancet 1:819–822
Gilbert DN, Tice AD, Marsh PK, Craven PC, Preheim LC (1987) Oral ciprofloxacin therapy for chronic contiguous osteomyelitis caused by aerobic gram-negative bacilli. Am J Med 82:254–258
Greenberg RN, Tice AD, Marsh PK, Craven PC, Reilly PM, Bollinger M, Weinandt WJ (1987) Randomized trial of ciprofloxacin compared with other antimicrobial therapy in the treatment of osteomyelitis. Am J Med 82:266–269
Giamarellou H, Galanakis N, Dendrinos C, Stefanou J, Daphnis E, Daikos GK (1986) Evaluation of ciprofloxacin in the treatment of Pseudomonas aeruginosa infections. Eur J Clin Microbiol 5:232–235
Gentry LO, Rodriguez GG (1990) Oral ciprofloxacin compared with parenteral antibiotics in the treatment of osteomyelitis. Antimicrob Agents Chemother 34:40–43
Uçkay I, Bernard L (2010) Gram-negative versus gram-positive prosthetic joint infections. Clin Infect Dis 50:795
Uçkay I, Vernaz-Hegi N, Harbarth S, Stern R, Legout L, Vauthey L, Ferry T, Lübbeke A, Assal M, Lew D, Hoffmeyer P, Bernard L (2009) Activity and impact on antibiotic use and costs of a dedicated infectious diseases consultant on a septic orthopaedic unit. J Infect 58:205–212
Clinical and Laboratory Standards Institute (2007) Performance standards for antimicrobial susceptibility testing; 17th informational supplement, M100-S17
Vittinghoff E, McCulloch CE (2007) Relaxing the rule of ten events per variable in logistic and Cox regression. Am J Epidemiol 165:710–718
Anglen JO, Gainor BJ, Simpson WA, Christensen G (2003) The use of detergent irrigation for musculoskeletal wounds. Int Orthop 27:40–46
Sendi P, Banderet F, Graber P, Zimmerli W (2011) Periprosthetic joint infection following Staphylococcus aureus bacteremia. J Infect 63:17–22
Bernard L, Legout L, Zürcher-Pfund L, Stern R, Rohner P, Peter R, Assal M, Lew D, Hoffmeyer P, Uçkay I (2010) Six weeks of antibiotic treatment is sufficient following surgery for septic arthroplasty. J Infect 61:125–132
Cordero-Ampuero J, Esteban J, García-Cimbrelo E (2009) Oral antibiotics are effective for highly resistant hip arthroplasty infections. Clin Orthop Relat Res 467:2335–2342
Uçkay I, Assal M, Legout L, Rohner P, Stern R, Lew D, Hoffmeyer P, Bernard L (2006) Recurrent osteomyelitis caused by infection with different bacterial strains without obvious source of reinfection. J Clin Microbiol 44:1194–1196
Loty B, Postel M, Evrard J, Matron P, Courpied JP, Kerboull M, Tomeno B (1992) One stage revision of infected total hip replacements with replacement of bone loss by allografts. Study of 90 cases of which 46 used bone allografts. Int Orthop 16:330–338
Zimmerli W, Trampuz A, Ochsner PE (2004) Prosthetic-joint infections. N Engl J Med 351:1645–1654
Uçkay I, Bouchuiguir-Wafa K, Ninet B, Emonet S, Assal M, Harbarth S, Schrenzel J (2010) Posttraumatic ankle arthritis due to a novel Nocardia species. Infection 38:407–412
Acknowledgments
All authors state that they have read and approved the manuscript. It has not been published elsewhere nor is it under consideration for publication elsewhere.
We are indebted to Dr. Tristan Ferry, Dr. Dorota Teterycz, Dr. Anne Lübbeke and Christophe Baréa for data retrieval. We thank the Orthopedic Service and the Laboratory of Microbiology for support.
Conflict of interest
The authors received no financial support, grants or royalties and have no financial interests that could lead to a conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Parts of the manuscript were presented at the Annual Swiss National Orthopaedic Congress, Lausanne, Switzerland, June 2011.
Rights and permissions
About this article
Cite this article
Seghrouchni, K., van Delden, C., Dominguez, D. et al. Remission after treatment of osteoarticular infections due to Pseudomonas aeruginosa versus Staphylococcus aureus: a case-controlled study. International Orthopaedics (SICOT) 36, 1065–1071 (2012). https://doi.org/10.1007/s00264-011-1366-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00264-011-1366-8