Abstract
Although contrast-enhanced ultrasound (CEUS) has become a widely utilized and accepted modality in much of the world, the associated contrast agents have only recently received approval in the United States. As with all radiological techniques, image artifacts are encountered in CEUS, some of which relate to commonly encountered ultrasound artifacts, while others are unique to this technique. Image artifacts must be recognized when performing and interpreting examinations to improve technique and diagnostic accuracy. In this article, we review artifacts that may be encountered in CEUS, and where possible discuss how to minimize them or mitigate their effect on image quality and interpretation.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Contrast-enhanced ultrasound (CEUS) is a unique tool that is now widely available in the United States. Three agents have been FDA approved for use in cardiology (Definity, Lantheus Medical; Lumason, Bracco Diagnostics; Optison, GE Medical) and for characterization of focal liver lesion in adult and pediatric patients. (Lumason, Bracco Diagnostics). These agents can be used off-label for other abdominal applications. As physicians become more familiar with this examination, there will be an increasing need for users to become proficient in performing and interpreting these studies. CEUS uses multi-pulse transmission to cancel tissue signal to produce “microbubble-only” or “contrast-only” images that can be displayed side-by-side with the standard B-mode image. This imaging technique, combined with the inevitable destruction of microbubbles during the exam, results in unique artifacts that the operator and interpreter must recognize to help avoid misdiagnosis, and should understand if they wish to improve image quality. There have been relatively few publications addressing artifacts unique to CEUS studies [1,2,3]. The purpose of this report is to describe how CEUS imaging differs from standard ultrasound imaging, as well as describe the commonly encountered artifacts, their cause, and how they can be decreased or eliminated.
Current microbubble-based ultrasound contrast agents (UCAs) are composed of a relatively insoluble perfluorocarbon gas core that is encapsulated by a phospholipid shell that provides relative stability in plasma for several minutes [4]. Since these microbubbles are slightly smaller than red blood cells (1–3 µm in diameter), they pass freely throughout the pulmonary and systemic circulations and are visible in blood, providing real-time, cross-sectional angiographic images of vessels as small as 100 µm in diameter. Capillary filling results in diffuse enhancement of perfused tissues. Because microbubbles cannot exit the capillaries, there is no interstitial trapping or elimination by the kidneys. Rather, the gas within the core defuses freely across the pulmonary capillaries into the alveoli and is exhaled. Eventually, microbubbles shrink and become undetectable. The phospholipids (~ 75 µg/dose) are biodegraded and enter the body’s lipid pool. The rapid clearance of the miniscule dose (5 × 10^8 microbubbles), administered as 0.2–1.2 mL total dose depending on formulation, allows for multiple injections (up to 1 vial as set by the FDA) during a single imaging session—this provides the operator the opportunity to optimize the imaging protocol and eliminate artifacts, study multiple organs, and/or characterize multiple lesions.
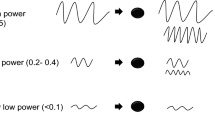
When microbubbles are exposed to an ultrasound pulse, they contract and expand with the compression and rarefaction phases of the sound wave. At resonance, when the ultrasound frequency matches microbubble size, microbubble diameter can change several folds between maximal contraction and expansion. With this oscillation, the microbubble becomes a transmitter, resulting in strong backscattered signal detectable on standard B-mode imaging [5, 6]. For most tissues, the contraction and expansion during the compression and rarefaction phases, respectively, is symmetrical. However, the oscillation of microbubbles is non-linear, meaning that velocity of microbubble contraction and expansion is asymmetrical. This non-linear behavior results in strong harmonic and ultraharmonic frequencies of the transmit frequency that are unique to microbubbles in vivo. Most manufacturers take advantage of these differences to “subtract” signal from background static tissues and produce “microbubble only” or “contrast-only” images (Fig. 1). Propriety pulse formation and post-processing includes phase and amplitude modulation and signal processing with frequency filtering to suppress much of the signal from the primary frequency, providing exquisite bubble-to-background contrast and allowing for the detection of a single microbubble in vitro and in vivo (Fig. 2) [7, 8]. In “contrast mode,” the contrast image may be displayed side-by-side with a standard B-mode image. Unfortunately, highly echogenic interfaces, particularly at gas/soft tissue margins or regions of dense calcification, may not be completely subtracted and will appear on both images.
Comparative images of a carotid artery after intravenous administration of contrast. Conventional B-mode image (A) fails to visualize microbubbles within carotid artery lumen (arrow). B-mode image with use of harmonic frequencies (B) visualizes moving microbubbles as moderately echogenic foci flowing within vessel lumen (asterisks). Contrast-mode image (C) shows relative suppression of signal originating from static tissues and offers improved visualization of microbubbles
A 48-year-old male with renal transplant underwent CEUS for suspected renal artery stenosis based on decreased urine output and equivocal Doppler findings. Contrast-mode image of the renal allograft, obtained between injections, shows single individual microbubbles (arrowheads) circulating in the bloodstream. The allograft is not well seen at this time as the few remaining circulating microbubbles are not numerous enough to completely enhance the renal parenchyma
Another unique physical property of microbubbles is that they can be destroyed when exposed to an ultrasound pulse. If insonated at relatively low acoustic pressures (low mechanical index, MI < 0.1), microbubbles oscillate as described above, and may not be destroyed; however, with each oscillation, lipid molecules can be lost from the shell, destabilizing the microbubble. At higher acoustic pressures (high mechanical index, MI > 0.5), and particularly at resonance, the lipid shell is rapidly destabilized and fractures, the gas core is released, and the bubble becomes too small to detect, resulting in loss of signal. It should be noted that MI is a mathematic estimate of acoustic pressure in tissues, and is typically not equivalent across manufacturers. Therefore, an optimal MI for one scanner may not be the same for another.
Bubble destruction is not only dependent on pulse pressure and transmit frequency, but also the number of pulses per unit time. Microbubbles flowing at a slower rate, such as in capillaries, are more easily destroyed than microbubbles flowing at higher velocity, such as in large vessels [6]. High MI imaging can be used to progressively clear an imaging plane of microbubbles, and/or accelerate microbubble elimination from the body. A short rapid succession of high-power pulses, a technique called a “flash,” clears nearly all bubbles from the field—this technique can be used to watch the refill rate and pattern of filling of lesions, and is useful to confirm that signal seen on the contrast image is indeed from microbubbles [4] (Fig. 3).
A 61-year-old male with renal transplant underwent CEUS for suspected renal artery stenosis based on decreased urine output and equivocal findings on Doppler. Contrast-mode image (A) shows enhancing parenchyma of renal allograft (arrows). A short burst of high-power pulses (“flash”) destroys bubbles within the scan plane (B). Post-flash image (C) shows loss of signal from renal parenchyma. Image obtained less than 1 s after (D) shows enhancement of segmental renal arteries from rapid microbubble refill (arrowheads). This method can be used to re-assess perfusion of a lesion, and differentiate vascularized tissue from static, non-vascular tissue
A summary of scanner settings and how they may affect the CEUS examination is shown in Table 1.
Artifacts unique to CEUS images
Technical factors and image formation
Non-linear artifacts
CEUS relies on the non-linear response of microbubbles, allowing signal from microbubbles to be distinguished from background tissues in which a linear response predominates. However, highly echogenic interfaces, and phase aberrations within some tissues, may result in signal that is not completely suppressed and therefore inseparable from signal from microbubbles. Since poorly subtracted interfaces tend to be organ capsules, vessel walls, or the diaphragm, they are useful in providing landmarks on the contrast image prior to UCA administration (Fig. 4). After contrast administration, these echogenic regions persist, though will also be present on the B-mode image simultaneously shown in the dual-display mode (Figs. 5, 6). While this artifact can be reduced by decreasing the output power (further decreasing MI) or decreasing contrast image gain (Fig. 7) [9, 10], this will also decrease microbubble signal. If not certain, a high-power flash pulse can be used to clear the imaging plane of microbubbles—any remaining echoes would then be due to ineffective subtraction of non-linear signal from background tissues (Fig. 8).
Side-by-side contrast-mode (left) and B-mode (right) image. Highly echogenic interfaces seen on contrast image, typically representing abdominal wall fascial planes organ capsules (arrowheads), and vessel walls (open arrowhead), can be compared with B-mode image and used for localization prior to contrast administration
Side-by-side B-mode (left) and contrast-mode (right) images from CEUS examination in a patient with internal carotid artery stenosis. Bright region (arrowhead on right) can be seen with plaque near the surface, which could be mistaken for superficial ulceration. However, comparison with B-mode image indicates that this focus represents a highly echogenic calcified component of the plaque (arrowhead on left) and not true enhancement. This type of artifact can be differentiated from true enhancement by comparing pre-contrast and B-mode images, and noting that such echoes are static, whereas echoes representing microbubbles are seen moving
A 68-year-old female with cirrhosis and remote history of radiofrequency ablation for hepatocellular carcinoma (HCC), found to have recurrence adjacent to ablation sight. Side-by-side contrast-mode (left) and B-mode (right) images during late arterial phase following contrast administration (A) show hyperenhancement of recurrent HCC (arrowheads). The gallbladder containing several shadowing gallstones (asterisk) is partly visualized. Standard B-mode image taken during microwave ablation (B) shows microwave needle (arrows) and formation of gas within ablation site (arrowheads). Side-by-side, post-contrast image following microwave ablation (C) shows zone of devascularized hepatic parenchyma encompassing site of HCC (arrowheads). Linear area of increased signal within the central ablation cavity on contrast image (left, arrow) could be mistaken for residual viable tissue. Corresponding B-mode image (right) confirms that this echogenicity is not related to injected bubbles, but rather, echogenic gas remaining in the ablation cavity. Pre-contrast images, and a high-power flash, can also be used to distinguish contrast enhancement from incomplete subtraction
Side-by-side contrast-mode (left) and B-mode (right) images of a renal angiomyolipoma (A) before injection of contrast demonstrate high signal in the angiomyolipoma (arrows). After UCA administration, it may be difficult to distinguish signal from microbubbles from that of high intrinsic signal present pre-contrast. Gain was therefore decreased, and another image before injection of contrast (B) shows decreased signal from the lesion. This signal is often attributed to the high intrinsic echogenicity and phase aberrations caused by fat/water interfaces, resulting in incomplete signal subtraction
A 79-year-old female with chronic kidney disease underwent CEUS for evaluation of multiple renal masses. Standard B-mode image (A) shows an exophytic, complex cystic renal mass with echogenic mural nodularity (arrowheads). Side-by-side contrast-mode (left) and B-mode (right) image following contrast administration (B) shows signal within this nodularity on contrast image, suggesting the presence of vascularized tissue. Short burst of high-power pulses (“flash”) is applied to destroy bubbles within the scan plane (C). Post-flash image (D) shows loss of signal from mural nodule, confirming that signal on pre-flash contrast image was indeed from microbubbles. Histopathology at partial nephrectomy revealed cystic renal cell carcinoma
Pseudoenhancement
Tissue subtraction is highly effective within fairly homogeneous organ parenchyma such as liver and spleen. Phase aberrations that occur in some tissues will result in less effective subtraction—this is commonly seen as moderate to high echogenicity of the subcutaneous fat, and can be seen in the setting of fatty liver (Fig. 9). In addition, if gain or output power are inappropriately increased in an attempt to improve signal from the far field or increase signal from microbubbles in the late phase after contrast injection, the signal from the non-linear harmonics produced by background tissues may be unintentionally amplified, resulting in “pseudoenhancement” (Figs. 7, 9) [11]. This artifact can be minimized by choosing appropriate power and gain settings prior to contrast administration—at baseline, signal from most tissues are suppressed and appear black, aside from the bright echogenic interfaces described above. After contrast injection, care must be taken not to reflexively increase the contrast image gain. If issues arise in differentiating true vs. artifactual enhancement, a high-power flash can be used—for truly enhanced tissues, signal will be lost and reperfusion may be seen, whereas artifactual enhancement will appear the same prior to and after the flash (Fig. 8). This technique is useful when poorly subtracted echoes are not associated with linear interfaces, particularly in the setting of calcifications, echogenic lesions like angiomyolipomas, and large bubbles caused by radiofrequency or microwave ablation.
A 49-year-old male with pancreatic cancer underwent screening CEUS examination for liver metastases. Due to chemotherapy-induced hepatic steatosis, ultrasound exam was challenging. Side-by-side contrast-mode (left) and B-mode (right) images of the right lobe of liver, in long, following the intravenous administration of contrast (A) show poor signal from hepatic parenchyma, and poor acoustic penetration into the far field. In an attempt to improve image quality, gain was increased (B), and parenchymal signal improved. However, it should be noted that signal from right portal vein did not increase (arrows, A and B), indicating the absence of significant microbubbles in the bloodstream. Increasing gain amplifies noise and the small component of non-linear harmonic signal from the liver, resulting in “pseudoenhancement” of the parenchyma. Image obtained with higher-dose injection (C) shows expected signal within the portal vein (arrows)
Near-field signal loss
Unexpected loss of contrast signal may be encountered with unintentional bubble destruction. Acoustic pressure is not uniform across the imaging field, and is greatest in the near field and at the focal zone. A commonly encountered phenomenon is progressive loss of signal in the near field, resulting in a band of low or no signal in tissues close to the transducer [1]. This is accentuated with a higher MI, higher scanning frequencies, and at higher frame rates, leading to accelerated bubble destruction, or “burn off,” in the near field (Fig. 10).
Side-by-side contrast-mode (left) and B-mode (right) image of the right lobe of liver, following contrast administration, shows a band of decreased signal in the near field (arrows), or “bubble burn off,” resulting from disproportionate power deposition in the superficial tissues, and prolonged scan time at one location. This artifact can be reduced by decreasing MI, reducing frame rate, or utilizing intermittent imaging or sweeping through the region in order to limit bubble destruction
Image plane signal loss
Similarly, large areas of no or decreased contrast signal may be encountered when either sweeping through a region previously imaged, or when alternating between orthogonal imaging planes—as shown in Figures 11 and 12, this affect can be dramatic. This phenomenon occurs when the transducer is held in one fixed location for an extended length of time, resulting in accelerated bubble destruction within a specific imaging plane relative to adjacent tissues where bubbles are not being insonated. When the transducer is moved from that plane, contrast signal significantly changes, and when turned to an orthogonal plane, a band of low signal in the plane that had just been imaged may be seen. This can be mitigated by ensuring low-power imaging (low MI), adjusting settings to decrease the frame rate, utilizing intermittent imaging, and sweeping through an area instead of remaining fixed in one location during image acquisition [1, 2, 12, 13]. Intermittent imaging can be achieved by forcing an ultrasound device to acquire a limited number of images per unit time (eg. Pulse triggering every 1000 ms) or by acquiring representative images at specific times, with intermediate interruptions in scanning (“freezing” in the intervals between).
A 64-year-old female with incidental hypervascular lesion on prior ultrasound performed for pancreatitis. Contrast-mode image during the portal-venous phase of enhancement (A) shows a solidly enhancing hypervascular lesion (arrowheads) with greater echogenicity relative to background. Make note of a feeding artery (arrow). After a deep breath several seconds later (B), image is obtained through the solid lesion at a slightly different location—background parenchyma is now iso-echoic relative to the solid lesion. This artifact is due to prolonged, continuous imaging performed at one image location (in A), resulting in accelerated bubble destruction within the background parenchyma in that plane—the hypervascular lesion remained hyperechoic due to rapid bubble refill relative to surrounding liver. Image during the same exam, in an orthogonal plane relative to A and B, shows wide area of bubble “burn-out” from prolonged continuous scanning in one location (arrowheads, C)
Similar to Figure 11, a band of low/no enhancement is seen through the liver when the transducer is turned to an orthogonal plane after prolonged, continuous scanning at one location (arrows)
Pseudo-washout
A related artifact is the unintended destruction of bubbles in tissues with altered perfusion relative to background, such as the slow flow in a hepatic hemangioma [14, 15]. If bubbles progress more slowly through the scan plan, such as in a slow-flow lesion, their risk of destruction is increased, thus leading to greater than intended bubble loss. Since washout is one perfusion characteristic used for lesion characterization, particularly in the liver [11, 16], this “pseudo-washout” may lead to diagnostic dilemmas or even misdiagnoses (Figs. 11, 13, 14). Again, decreasing the frame rate, using intermittent imaging, making sure the focal zone is well beyond the area of interest, and sweeping through a lesion as opposed to remaining in a stationary position, may help eliminate this artifact (Fig. 12).
A 44-year-old male with known hepatic steatosis and elevated liver function tests. Standard B-mode image of left lobe of liver (A) reveals a slightly geographic lesion with decreased echogenicity relative to background liver (arrowheads). Following contrast administration, contrast-mode images in early arterial (B), late arterial (C), and portal-venous (D) phases show a central vascular nidus and centrifugal spoke wheel enhancement characteristic of focal nodular hyperplasia. Generally, these lesions are isoenhancing to background liver in portal-venous and delayed phases of enhancement. However, with prolonged continuous imaging, unintentional bubble destruction may result in “pseudo-washout” (arrowheads, E), particularly when suboptimal tissue subtraction is present because of phase aberrations caused by surrounding hepatic steatosis. Note the associated near-field bubble destruction (arrows). Reduced MI, decreased frame rate, and intermittent imaging, or increasing contrast dose can be implemented to limit this artifact
A 68-year-old female with cirrhosis and known HCC. Contrast-mode image in early (A) and late arterial (B) phases of enhancement after contrast administration shows lesion with hyperenhancement and pseudocapsule (arrowheads, A and B, respectively) characteristic of HCC. However, at 70 s, washout was encountered (arrowheads, C). Rapid washout is atypical for HCC, and may result in a diagnostic dilemma as this finding is generally attributed to cholangiocarcinoma and metastases. Decreased frame rate and intermittent imaging were implemented during a second injection, and image at 70 s (D), same as in image C, shows lesion to be iso-echoic to background liver. Reducing MI and sweeping through the lesion, as opposed to continuous stationary imaging, may also reduce unintentional bubble destruction
Unintentional effects of microbubbles
Signal saturation
Depending on the scanner’s output power and gain settings, and the contrast dose, signal received may exceed the display range on the contrast-only image (as seen by a glare of uniform bright echoes at the top range of the color scale spectrum) [1, 2]. This is most evident during angiographic applications of CEUS, or in highly perfused tissues such as the kidneys (Fig. 15). Although this may not affect qualitative assessment when determining the presence or absence of vascularity, detecting subtle differences in enhancement in highly perfused lesions may be affected. In addition, this artifact is important to consider when attempting to quantify perfusion (washin and washout) of a lesion—if the image brightness reaches its greatest pixel value, perfusion curve analysis will show a plateau and peak enhancement may be underestimated. Decreasing pulse power, decreasing contrast image gain, and decreasing contrast dose on subsequent injections will minimize this effect.
A 75-year-old male with chronic kidney disease undergoing evaluation of an incidental renal mass. Shortly after contrast administration there is avid parenchymal enhancement of the kidney and an exophytic, solid renal mass. Several areas of perfused tissue appear extremely bright (arrows), corresponding to highest color value on adjacent reference scale (arrowhead). Once high signal is mapped to the highest color pixel value, any increased signal beyond this value will not be appreciated (saturation artifact). Although usually not an issue for qualitative assessment, this artifact may significantly affect quantitative perfusion analysis, during which the signal in these regions will plateau. In order to limit this artifact, decrease contrast dose, increase output power, or decrease gain for subsequent injections. Note the decreased signal from the far field resulting from attenuation from a high concentration microbubbles in these tissues
Shadowing
When large concentrations of bubbles are present in a localized area they may cause shadowing similar to that seen with intraluminal bowel gas (Figs. 15, 16). A dense concentration of bubbles is highly attenuating, causing shadowing in the far field. When this occurs, the shadowing will decrease in intensity as the bubble concentration decreases in the near field. This artifact may be eliminated by administering a smaller dose of microbubbles with subsequent injections, increasing the MI, or bringing the focal zone to the near field to hasten microbubble destruction in intervening tissues.
A 65-year-old male with history of a renal transplant, presenting with allograft dysfunction, undergoing CEUS to exclude renal artery stenosis. Side-by-side contrast-mode (left) and B-mode (right) images of the main renal artery (A) show posterior acoustic shadowing (arrowheads) from the high concentration of bubbles in the renal artery (closed arrow) and adjacent iliac artery (open arrow). Image from a second patient (B) showing shadowing from high concentration of bubbles in upper renal pole (arrows). To minimize this artifact, a lower dose of contrast should be considered for subsequent injections
Shadowing may be more commonly seen during some off-label uses of UCAs, particularly intracavitary administration. This can be seen in the urinary bladder or collecting system during pediatric voiding cystourethrograms, or during attempted abscess aspiration and drainage. This is due to the high concentration of microbubbles administered or because of the introduction of air bubbles. Therefore, careful dilution of contrast is needed, and introduction of air bubbles should be avoided.
Effects on color Doppler
Microbubbles affect Doppler displays in multiple ways. First, overall signal in both vessels and adjacent perfused tissues is increased. Since color Doppler has been optimized for signals received from red blood cells, and since microbubbles produce dramatically greater signal, signal from slow flow that may have been previously undetected or filtered may become visible. Since the color pixel size is determined as the full width at half maximum of the decorrelated signal from moving blood cells, the overall increase in single produces larger color pixels. Both of these effects result in blooming artifacts (Fig. 17) [1, 3], which can be ameliorated but not eliminated by decreasing color Doppler gain or increasing pulse repetition frequency[3, 13]. In addition, the increase in Doppler bandwidth may be falsely interpreted as increased turbulence (Fig. 18), a hallmark of an vascular stenosis. Finally, microbubble destruction between the first and second Doppler pulse may result in a decorrelation error, generating a mosaic of red and blue pixels. This is likely to occur in tissues with very slow flow. Imaging with color Doppler prior to the arrival of contrast can resolve most of these interpretive dilemas.
Color Doppler image of the proximal internal carotid artery before (A) and after contrast administration (B). Microbubbles greatly increase color Doppler signal, causing blooming artifact with color outside the vascular lumen. This artifact can be decreased by reducing color gain or setting a higher pulse repetition frequency. Aliasing (as seen in B) could be misinterpreted as turbulent flow associated with vessel stenosis
Color Doppler image of the common carotid artery (A) showing a normal common carotid artery waveform. Color Doppler image after contrast administration (B) shows the presence of short high-intensity spikes (arrowheads) projecting over Doppler waveform. Note the presence of microbubbles being visualized as moving echogenic foci on B-mode image (asterisk)
Effects on pulse-wave Doppler
The administration of UCAs has been attributed to changes in the Doppler waveform. Early reports suggested an increase of peak velocity by as much as 45%, attributed in one study to the limited system dynamic range and non-linear conversion of backscattered signal [3], and in another to the improved signal at very high velocities that were too weak to detect prior to the intravenous administration of a UCA [17]. This is likely related to the increase in signal at all velocities—increased bandwidth, including the small components flowing at high velocity that now contains microbubbles. This phenomenon is more commonly observed with older systems as this observation has not been confirmed in more recent studies. It has been reported that the mean, peak velocity, and spectral Doppler indices of human common carotid arteries do not differ significantly before and after contrast administration [18]. Another study concluded that there is no difference in the resistive index on spectral Doppler analysis in blood vessels of metacarpophalangeal joints of healthy subjects, before and after administration of a UCA [19]. Nevertheless, based on the risk of falsely grading vascular pathology, duplex Doppler parameters are best evaluated prior to contrast administration. Following contrast however, vascular stenoses can be directly measured as luminal diameter narrowing, as is done with catheter angiography, or cross-sectional area narrowing which more accurately describes the degree of narrowing. Both of these measures are accurate and have been validated in patients [20].
In addition to the increase in bandwidth, high-intensity transient signals may appear on a pulse-wave Doppler waveform after contrast administration (Fig. 18) [3, 13]. These sharp spikes can also be heard as crackling sounds on the audio output. This artifact is caused by cavitation of microbubbles along the sampling line during Doppler interrogation and results in a broadband pulse that contains many frequencies.
B-mode ultrasound artifacts in contrast images
Through transmission and shadowing
Commonly encountered ultrasound artifacts such as posterior acoustic enhancement and shadowing also occur in the contrast-only images on CEUS [21]. In general, spatial compounding and frame averaging are turned off in contrast mode to further reduce microbubble destruction. This results in more sharply demarcated through transmission or shadowing artifact on the contrast-only images compared to optimized B-mode images. Similar to non-enhanced ultrasound, increased signal may be received deep to a lesion with lower than expected attenuation (such as a simple cyst) (Fig. 19). Similarly, signal may not be received deep to an attenuating structure, resulting in posterior acoustic shadowing (Fig. 20).
Loss of contrast in the far field
In patients with attenuating abdominal walls, or when there is attenuation from a steatotic liver with fatty infiltration, contrast signal may be very weak from the far field, an issue exacerbated by the use of low MI imaging (Fig. 21). Increasing MI may improve signal from the far field at the expense of signal loss from microbubble destruction in the near field. Changing to a lower transmit frequency (lowest frequency probe available, or lowest frequency setting on the transducer available) also improves tissue penetration on contrast images (Fig. 21). Another solution is to reposition the patient to decrease the distance between the probe and the area of interest. Ultimately, a balance is needed between the rate of bubble loss in the near field, and the acoustic power needed to image at a certain depth.
Reflection and refraction artifacts
Reflection and refraction artifacts are typically seen at curved interfaces separating two tissues with differences in acoustic impedance, resulting in a triangular hypoechoic band emanating from the specular reflector (“edge shadow”). This is also visible on contrast images. Again, as image compounding is deactivated for CEUS, this artifact may be more accentuated. In order to prevent this artifact from affecting the area of interest, patient repositioning may be needed in order to image the target from a different orientation (Fig. 22).
A 63-year-old male undergoing evaluation of an indeterminate renal mass. Contrast-mode image of left kidney and spleen (A) reveals a hypoenhancing endophytic renal mass at lateral upper pole (arrow). A large dark band extends from the interface between spleen and upper renal pole (refractive edge shadow). By reposition patient and changing probe orientation, refraction artifact is no longer visible
Mirror image, reverberation, and other artifacts
Misregistration artifacts caused by sound reflection or reverberation are also visible on contrast images. A classic mirror image artifact caused by a highly reflective surface such as the diaphragm is shown in Figure 23. Similarly, reverberation artifacts caused by closely apposed highly reflective surfaces, often found in the abdominal wall, will also be seen on contrast images, as shown in Figure 24. Other commonly encountered B-mode artifacts, such as those caused by side lobes, beam width, and elevation plane volume averaging, can also be seen.
A 78-year-old female with indeterminate renal lesion. Side-by-side contrast-mode (left) and B-mode (right) images of the left kidney (A) show a partly exophytic renal cyst in which sequential echogenic lines are seen in the near field (arrowheads), indicating reverberation artifact from interfaces in the abdominal wall
Conclusions
Similar to other imaging modalities, artifacts may be encountered during contrast-enhanced ultrasound studies. Some of these artifacts are similar to those commonly seen in standard B-mode imaging, while others are unique to the contrast-mode images. In particular, echoes within the “contrast-only” image may result from incomplete tissue subtraction, while an apparent lack of enhancement may be due to inappropriate settings and greater than expected bubble destruction. Identifying these artifacts and learning how to account for or eliminate them with appropriate scanning techniques will improve a radiologist’s confidence, and the diagnostic performance of this emerging modality.
References
Dietrich CF, et al. (2014) Artifacts and pitfalls in contrast-enhanced ultrasound of the liver. Ultraschall Med 35(2):108–125; quiz 126–127.
Dietrich CF, et al. (2011) Pitfalls and artefacts using contrast enhanced ultrasound. Z Gastroenterol 49(3):350–356
Forsberg F, et al. (1994) Artifacts in ultrasonic contrast agent studies. J Ultrasound Med 13(5):357–365
Denham SL, Alexander LF, Robbin ML (2016) Contrast-enhanced ultrasound: practical review for the assessment of hepatic and renal lesions. Ultrasound Q 32(2):116–125
Girard MS, et al. (1999) Assessment of liver and kidney enhancement with a perfluorocarbon vapor-stabilized US contrast agent. Acad Radiol 6(5):273–281
Sirlin CB, et al. (1999) Effect of acquisition rate on liver and portal vein enhancement with microbubble contrast. Ultrasound Med Biol 25(3):331–338
Cui W, et al. (2013) Neural progenitor cells labeling with microbubble contrast agent for ultrasound imaging in vivo. Biomaterials 34(21):4926–4935
Klibanov AL, et al. (2004) Detection of individual microbubbles of ultrasound contrast agents: imaging of free-floating and targeted bubbles. Invest Radiol 39(3):187–195
ten Kate GL, et al. (2012) Far-wall pseudoenhancement during contrast-enhanced ultrasound of the carotid arteries: clinical description and in vitro reproduction. Ultrasound Med Biol 38(4):593–600
Ten Kate GL, et al. (2013) Current status and future developments of contrast-enhanced ultrasound of carotid atherosclerosis. J Vasc Surg 57(2):539–546
Jo PC, et al. (2017) Integration of contrast-enhanced US into a multimodality approach to imaging of nodules in a cirrhotic liver: how i do it. Radiology 282(2):317–331
Harvey CJ, et al. (2000) Hepatic malignancies: improved detection with pulse-inversion US in late phase of enhancement with SH U 508A-early experience. Radiology 216(3):903–908
Quaia E (2005) In: Quaia E, (ed) Artefacts from microbubble-based agents, in contrast media in ultrasonography basic principles and clinical applications. New York: Springer, pp. 25–29
Dietrich CF, et al. (2007) Contrast-enhanced ultrasound of histologically proven liver hemangiomas. Hepatology 45(5):1139–1145
Dietrich CF, et al. (2012) Liver tumor characterization–review of the literature. Ultraschall Med 33(Suppl 1):S3–S10
Malhi H, Grant EG, Duddalwar V (2014) Contrast-enhanced ultrasound of the liver and kidney. Radiol Clin North Am 52(6):1177–1190
Gutberlet M, et al. (1997) Contrast agent enhanced duplex ultrasonography: visualization of the hepatic artery after orthotopic liver transplantation. Rofo 166(5):411–416
Gutberlet M, et al. (1998) Do ultrasonic contrast agents artificially increase maximum Doppler shift? In vivo study of human common carotid arteries. J Ultrasound Med 17(2):97–102
Terslev L, et al. (2005) Doppler ultrasound findings in healthy wrists and finger joints before and after use of two different contrast agents. Ann Rheum Dis 64(6):824–827
Kono Y, et al. (2004) Carotid arteries: contrast-enhanced US angiography–preliminary clinical experience. Radiology 230(2):561–568
Hindi APC, Barr RG (2013) Artifacts in diagnostic ultrasound. Reports in Medical. Imaging 6:29–48
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosures
DTF has a research agreement with Philips Healthcare, and is on the speaker’s bureau for Philips Healthcare. VR has no disclosures. CLP is on the speaker’s bureau for Bracco Diagnostics. EGG has a research grant from GE. RFMPS has no disclosures. RGB has research grants from Siemen’s Ultrasound, Philips Ultrasound, B and K Ultrasound, GE Ultrasound, Bracco Diagnostics, and SuperSonic Imagine. He is on the speaker’s bureau for Philips Ultrasound, Lantheus Medical and Bracco Diagnostics. He is on the advisory of Bracco Diagnostics and Lantheus Medical. He also receives royalties from Thieme Publishers.
Funding
No funding was received for this work.
Ethical Approval
This article does not include data from human research subjects. Images were acquired from clinical patients for education purposes.
Rights and permissions
About this article
Cite this article
Fetzer, D.T., Rafailidis, V., Peterson, C. et al. Artifacts in contrast-enhanced ultrasound: a pictorial essay. Abdom Radiol 43, 977–997 (2018). https://doi.org/10.1007/s00261-017-1417-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00261-017-1417-8