Abstract
Objective
The aim of this study was to assess the accuracy of Ultrasound (US) and contrast-enhanced ultrasound (CEUS) in the characterization of renal nodules indeterminate on CT by identifying benign cystic lesions not requiring further examination.
Methods
72 patients with 83 indeterminate renal nodules on CT underwent baseline US and CEUS that classified lesions as benign (Bosniak I, II or IIF cysts) or potentially malignant (Bosniak III or IV cysts, solid nodules). The accuracy of US and CEUS in the differentiation between benign cysts and potentially malignant nodules was analyzed and compared with the final diagnosis obtained by histology or follow-up of at least 23 months with CEUS ± a conclusive CT/MR study.
Results
Final diagnoses comprised 50 benign complex cysts, 1 focal nephritis, 1 multilocular cystic nephroma, 3 oncocytomas, 1 transitional cell carcinoma and 27 renal cell carcinomas. Unenhanced US correctly classified 18/50 (36%) benign cysts and 17/33 (51.5%) of the potentially malignant lesions obtaining a sensitivity of 36%, specificity of 51.5%, and overall accuracy of 42.2%. The addition of CEUS allowed a correct diagnosis of 48 /50 (96%) benign cysts and of 31/33 (93.9%) nodules as potentially malignant, with a sensitivity of 96%, specificity of 93.9%, and overall accuracy of 95.2%.
Conclusion
CEUS is very useful in the differentiation between benign complex cysts and other lesions that require further investigation in non-conclusive renal nodules detected on CT, improving the accuracy of baseline US from 42.2 to 95.2%.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
The detection of incidental renal nodules is a common finding in Computed Tomography (CT) and has increased in the last years due to the proliferation of CT studies for different clinical purposes. Most of these lesions are simple cysts. Nevertheless, some benign hemorrhagic and inflammatory cysts show a complex appearance on CT that hampers their differentiation from other malignant cysts and solid lesions that require surgical management to obtain a final diagnosis [1]. The characterization of these lesions and mainly the diagnosis of renal cell carcinoma (RCC) require the intravenous administration of an iodinated contrast agent to demonstrate the enhancement secondary to the tumor neovasculature [2]. On CT, conclusive enhancement is defined when there is an increase of the attenuation >15 HU between the unenhanced and enhanced phases (although several groups consider 20 HU as the correct threshold) [3–5]. The enhancement criterion is very useful to characterize renal nodules but has several limitations. First, some low-grade RCCs and especially the papillary variant can show very slight enhancement not clearly detected by CT [6]. Second, there is the possibility of pseudo-enhancement of some small simple cysts [7]. In addition, in clinical practice most abdominal CT studies are not performed with a specific renal protocol to characterize renal nodules, thus it is common to find indeterminate renal nodules in any of the following situations: CT study including only one unenhanced phase (mainly in patients with renal failure or allergy to contrast media); CT including only enhanced phases; CT with unenhanced and enhanced phases with doubtful enhancement.
Ultrasound (US) is a very accurate technique to diagnose simple cysts [8] but has a low accuracy in the characterization of complex cysts and in the differentiation between complex cysts and solid lesions. Contrast-enhanced ultrasound (CEUS) is a very sensitive imaging technique to detect tumor microvascularity and in the kidney allows the detection of enhancement in solid tumors and the characterization of complex renal cysts using the Bosniak classification [9–11]. Recent studies have demonstrated the accuracy of CEUS to be similar compared to that of CT in the characterization of complex renal cysts [12–14] with the advantage of its price and absence of radiation and nephrotoxicity, nowadays being the main indication of CEUS in the kidney [15]. In addition, several retrospective studies of indeterminate renal masses with baseline CT, MR or US including the large report by Barr et al., [16, 17], describe the excellent accuracy of CEUS, but to date no prospective studies have assessed indeterminate CT renal nodules.
The aim of this study was to assess the accuracy of US including CEUS in the characterization of indeterminate CT renal nodules by identifying benign cystic lesions that do not require histological diagnosis.
Methods
Patients
This prospective study was approved by the Ethical Committee of our hospital and all patients gave their informed consent to participate. The study began on April 2009 and included 92 consecutive patients (61 men, 31 women, mean age 64.9 years, range 34–92 years) who presented an indeterminate renal nodule on a CT performed in our department. Eleven patients had 2 indeterminate nodules, thus a total of 103 nodules were included. All patients underwent a baseline US and CEUS within the next month, and the results were compared with the final diagnosis obtained by histology or follow-up with US/CT/magnetic resonance (MR) that were considered conclusive. Nevertheless, 20 patients with 20 nodules were excluded because of a lack of conclusive diagnosis (they did not complete the follow-up or have a histological diagnosis).
Therefore, we included a total of 72 patients (50 men, 22 women, mean age: 64.2 years, age range: 34–85 years) with 83 indeterminate nodules of a mean size of 20.7 mm, range 5–65 mm, 39 on the right kidney and 44 on the left kidney.
Imaging techniques
CT studies were performed with Duo emotion or Sensation 64 Siemens equipment following different protocols depending on the clinical indication, including at least one of the following phases: unenhanced, arterial, venous, and delayed excretory phases. The enhanced phases were acquired after the intravenous bolus injection of iodinated contrast material (Visipaque 400, GE, 320 mg I/mL or Ultravist 300, Bayer, 300 mg I/mL, 2–5 mL/s at 4 mL/kg followed by 50 mL of saline flush). All CT studies included reconstructions of all phases of 5 mm at intervals of 5 mm, and some studies included reconstructions at intervals of 1 or 2 mm. CT attenuation of the renal nodules was calculated placing region-of-interest (ROI) cursors over the renal nodules excluding the adjacent kidney parenchyma in all phases to evaluate the presence and magnitude of enhancement. All imaging and reconstruction parameters remained constant throughout all the phases. The clinical indications of CT were different cancers follow-up (34 patients), kidney transplant evaluation (9), renal mass diagnosis (8 patients), hematuria (5), other causes (16 patients). The reasons to consider a nodule as indeterminate included:
-
1.
Study with only an unenhanced phase with a nodule >20 HU; 10 nodules, mean size of 18.5 mm, range 10–34 mm;
-
2.
Study without an unenhanced phase, with only one enhanced phase (13 nodules) or with several enhanced phases with a difference of enhancement <20 HU between each (27 nodules); total 40 nodules; mean size of 22.5 mm, range 6–60 mm;
-
3.
Study with an unenhanced and at least 2 enhanced phases with a nodule >20 HU and a difference of enhancement <20 HU between the unenhanced and enhanced phases; 33 nodules; mean size of 19.1 mm, range 5–65 mm.
US studies were performed by one of the radiologists of the Radiology Department with at least 8 years of experience in US, using Sequoia 512 or S2000 equipment (Siemens Acuson Mountain View, CA). First of all, a baseline US of the kidneys was performed in fundamental mode, using greyscale, with a multifrequency 4C1 convex array probe with harmonics in order to identify the nodule, and color Doppler was performed to evaluate the intratumoral vascularity. After the baseline study, a dynamic CEUS of the renal nodule was performed using the specific contrast software Cadence contrast pulse sequencing technology (CPS), which allows real-time evaluation of contrast agents with minimum bubble destruction at low MI power levels. CPS was performed with the same convex array probe using a low mechanical index (<0.2 at Sequoia 512, <0.009 at S2000) in order to avoid microbubble disruption. CEUS studies were performed after the administration of 2.4 mL of Sonovue as a bolus using a 21 gauge peripheral intravenous cannula followed by a 5-mL saline flush. Nodules were studied up to 3 min. Images and cine-loops of baseline, arterial and venous phases were selected on digital cine-loops by the same radiologists who performed the US studies and stored at the picture archiving and communication system (PACS) of the Radiology Department for off-line analysis. As the aim of the study was the characterization of renal nodules using CEUS, CEUS was performed independently of the results of the grey-scale US study.
Imaging analysis
Two radiologists, each with at least 10 years of experience in interpreting CEUS studies, independently reviewed the US studies off-line using the RAIM ALMA software version 4.1.4.0002. First, they evaluated and classified the unenhanced US and then they evaluated and classified the CEUS studies. Both were blinded to the clinical histories, histological results, and other imaging findings, and classified all nodules as solid, cystic or indeterminate. On baseline US, anechoic lesions with hair-line thin wall without septa or solid components were defined as simple cysts, while those cysts with few thin septa (≤1 mm) were considered benign complex cysts [17]. Lesions with thickened septa or wall or solid component showing color Doppler signal were defined as potentially malignant complex cysts or solid lesions. The remaining cystic lesions with intracystic hyperechoic content or lesions with solid appearance, both without color Doppler signal were classified as indeterminate (not diagnostic) by US (Table 1). On CEUS, solid lesions were defined as those which were hypo-iso or hyperechoic nodules with intratumoral enhancement and cysts were defined as simple or complex following the Bosniak classification adapted to CEUS [11] depending on the presence of the following characteristics: thickening of septa and wall (>1 mm), enhanced septa and wall, intracystic enhancing nodules. Lesions with no conclusive enhancement were classified as indeterminate (Table 2). US and CEUS diagnoses of all discrepant results were established by consensus. Finally, and depending on these findings, the lesions were classified as benign complex cysts (Bosniak I, II, IIF) or potentially malignant (Bosniak III, IV or solid lesions). As defined in the aim of the study we classified the lesions depending on the management of the patients separating benign cystic lesions not requiring further investigation (Bosniak I or II cysts) or imaging follow-up (Bosniak IIF) from the remaining nodules usually requiring histology to achieve a final diagnosis [17].
Reference standards for diagnosis
Findings at baseline US and CEUS were correlated with the final diagnoses obtained by histology or a follow-up of at least 23 months including a definitive diagnosis by US or a dedicated renal CT/MR study performed before and after the intravenous injection of contrast material and imaging. Follow-up by US was only accepted when the lesions met the criteria of a simple Bosniak I cyst by US and CEUS (considered worldwide as conclusive for the diagnosis of simple cyst). In all other cases, dedicated CT/MR/histology were required.
Statistical analysis
Statistical analyses were performed using a computer software package (SPSS 17.0 Inc. 1989–1995, Chicago, IL). The baseline characteristics of the patients and renal nodules were expressed as mean and range. For US and CEUS, the sensitivity, specificity, positive predictive value, negative predictive value, and overall accuracy in determining the benign or potentially malignant nature of the lesions were calculated using the reference standard and analyzed with the Fisher exact test. For the estimation of sensitivity, indeterminate lesions on baseline US or CEUS were classified as false negatives (potentially malignant). On the contrary, for the estimation of specificity, uncertain results on US and CEUS were classified as false positives (benign complex cysts). A value of p < 0.05 was considered statistically significant.
Results
Figure 1 summarizes the final diagnosis of the 83 lesions (Fig. 1). All 50 benign cysts were diagnosed within at least 23 months of follow-up (range 23–41 months) using CEUS ± dedicated CT/MR. The other 28 malignant lesions (1 transitional cell carcinoma and 27 RCC, with 14 papillary, 9 clear cell, 2 chromophobe, 2 non-defined subtypes), and 4 benign lesions (1 multilocular cystic nephroma and 3 oncocytomas) were diagnosed by histology. The last lesion, a focal nephritis was diagnosed and followed with enhanced CT and had disappeared at the 1-year follow-up CT.
Baseline grey-scale US was able to correctly classify 18/50 (36%) Bosniak I–II or IIF benign cysts and 17/33 (51.5%) of the potentially malignant lesions (Tables 1, 3). The remaining 48 lesions (57.8%) were classified as indeterminate on baseline US. Thus, baseline US obtained a sensitivity of 36%, specificity of 51.5%, and overall accuracy of 42.2% to discriminate between benign cysts and other lesions.
The combination of both US and CEUS was able to correctly classify 48/50 (96%) benign cysts and 31/33 (93.9%) potentially malignant nodules, obtaining a sensitivity of 48/50 (96%), specificity of 31/33 (93.9%), positive predictive value of 48/50 (96%) and negative predictive value of 31/33 (93.9%) with an overall accuracy of 79/83 (95.2%) (Tables 2, 4) for the differentiation between benign cysts and potentially malignant lesions. The use of contrast media improved the accuracy of US by 53% (42.2–95.2%) (p < 0.005).
When we evaluated the group of 48 lesions that remained indeterminate on baseline US, CEUS obtained a sensitivity of 30/32 (93.7%), specificity of 14/16 (87.5%), positive predictive value of 30/32 (93.7%), negative predictive value of 14/16 (87.5%) and an overall accuracy of 44/48 (91.7%) to discriminate between benign cysts and other lesions (Table 5).
The type of original indeterminate CT study did not make any difference regarding the accuracy of US with CEUS, as the accuracy in the group of 50 non-conclusive nodules due to non-complete CT evaluation (nodules only studied by unenhanced phase or by only enhanced-phases) (Figs. 2, 3, 4) with a sensitivity of 94.1% (32/34), specificity of 93.7% (15/16) and accuracy of 94% (47/50) was the same as in the group of 33 nodules with unenhanced and enhanced phases and non-conclusive enhancement, with a sensitivity of 100% (16/16), specificity of 94.1% (16/17) and accuracy of 96.9% (32/33) (Figs. 5, 6).
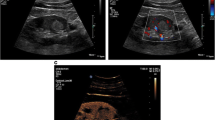
43-year-old man with chronic renal disease on waiting list for kidney transplantation. An unenhanced CT was performed to evaluate the presence of vascular calcifications. A An homogeneous mass of 34 mm at the upper pole of the right kidney was detected with attenuation greater than 20 HU (ROI = 52 HU). B Baseline US showed a cystic lesion with echoes within (arrow) C CEUS demonstrated the presence of a simple cystic lesion without septa or enhancing nodules (arrow). A follow-up dedicated CT with unenhanced, arterial and nephrographic phases demonstrated the absence of enhancement, being compatible with a benign cyst. The cyst was followed over 36 months and remained stable.
75-year-old man with CT follow-up performed on portal phase due to prostate cancer. A A well defined, homogeneous renal mass was identified on the upper third of the right kidney. A ROI within the mass showed attenuation of 57 HU. B Baseline US (right arrow) showed a cystic lesion with echoes within and a doubtful septum. C CEUS in dual mode (left) showed a simple cyst without septa or enhancing nodules (left arrow). The 30-months follow-up included a conclusive CT with absence of enhancement and stability.
65-year-old woman referred to our center for a CT to evaluate an adnexal mass. A CT with portal and excretory phases was performed. A, B The portal and excretory phases showed a homogeneous nodule. ROIs demonstrated attenuation of 66 and 61 HU, respectively (difference of attenuation <20 HU). C Baseline US showed suspicion of complex cyst (arrow). D CEUS showed a hypovascular nodule with slight intratumoral enhancement (arrow) on arterial phase. D, E with washout in early and late venous phase compatible with hypovascular solid mass. Final diagnosis obtained by histology was papillary RCC.
36-year-old woman with suspicion of renal mass who was referred to our center. A CT with unenhanced and enhanced phases was performed. A, B The unenhanced and nephrographic phases showed a homogeneous mass with attenuation of 32 and 46 HU (ROI), respectively with enhancement <20 HU. C Baseline US identified an echogenic mass without color Doppler within it that was considered indeterminate because of the absence of color Doppler signal. D CEUS confirmed the solid nature of the mass which was hypervascular in arterial phase (arrow), E with a quick wash-out (arrow) compatible with solid mass. The histological diagnosis obtained after surgery was chromophobe RCC.
61-year-old woman with suspicion of right pyelonephritis. A CT with unenhanced, nephrographic and excretory phases was performed, detecting an incidental hyperattenuating nodule of 25 mm at the lower pole of the left kidney. A–C. ROI in the unenhanced, nephrographic and excretory phases showed an attenuation of 49, 61 and 67 HU, respectively, with a difference of enhancement between 10 and 20 HU. D Baseline US showed a cystic lesion with suspicion of focal wall thickening (arrow). E CEUS confirmed the presence of a cyst without intracystic masses compatible with a Bosniak II cyst. Benignancy was confirmed with a follow-up of 30 months which included a conclusive MR confirming the absence of enhancement.
Regarding the 4 nodules not correctly classified by CEUS, 3 were considered as indeterminate lesions because of their medial location or small size (8, 11, and 18 mm) which hampered evaluation of possible enhancement. The false-positive of a benign cystic lesion in our study was a nodule classified as Bosniak II by CEUS which, on the follow-up performed by dedicated CT, intratumoral nodular enhancement was observed, being compatible with a cystic RCC as was demonstrated by histology.
Discussion
In the present study CEUS demonstrated a very high accuracy in the differentiation between benign cysts and potentially malignant nodules which are indeterminate on CT, with an important impact on the management of these nodules since benign cysts do not require histology or further immediate investigations, and only Bosniak IIF cysts require imaging follow-up that can also be performed by CEUS [11]. The characterization of renal nodules by CT usually requires the performance of unenhanced and enhanced phases. Even in this condition, the evaluation of enhancement can be difficult depending on the technique employed, the possibility of pseudo-enhancement described in some small cysts, or the slight enhancement of some RCCs [6, 7, 18, 19]. In addition, in clinical practice several CT studies are performed only in baseline conditions (patients with renal failure or allergy to iodinated contrast media) or more commonly without an unenhanced phase (for example in acute abdomen or follow-up of several cancers). Independently of the reason for not obtaining a conclusive diagnosis by CT, US can be used to diagnose benign cysts that do not require further examinations or require only follow-up imaging as occurred in 42.2% of the nodules in our study using grey-scale US with an improvement of the accuracy to 95.2% when CEUS was used. We obtained similar results to those of other studies also describing high accuracies for CEUS characterization of complex cysts [13, 20] or indeterminate renal masses [17]. The recent guidelines of the European Federation of Societies for Ultrasound in Medicine and Biology (EFSUMB) consider the characterization of complex cysts as one of the main indications for CEUS [15]. Similar to the study by Cazals X et al [16] we obtained a high accuracy in this characterization which may be explained by the high capacity of CEUS to detect microvasculature. In this way, recent studies have reported a higher accuracy of CEUS than CT in the detection of wall and septa microvasculature in complex cysts [12, 20, 21] with the known advantage of absence of radiation. In the study by Clevert et al. including 32 complex cysts detected on unenhanced US, CEUS showed more thin septa and wall thickening with an upgrade from II to IIF in 5 patients (1/3 of Bosniak II cysts), and with additional Bosniak IV cysts in 2 patients. In the study of Quaia et al. including 40 complex cystic renal nodules, CEUS was found to be better than CT in the diagnosis of malignancy. As found in 42.2% of the nodules in this study, unenhanced US may, in some cases, be enough to characterize indeterminate CT lesions, especially in the case of anechoic simple cysts without septa or intracystic nodules [22]. This low accuracy of unenhanced US in our study can be explained by the characteristics of the lesions. None of the lesions was liquid on CT and did not present clear enhancement, thus the presence of a high number of cysts with echogenic content even using harmonics and the presence of lesions with solid appearance without color Doppler signal that were classified as indeterminate lesions were expected on unenhanced US, as happened in our study [11].
Regardless of whether unenhanced US identifies a simple cyst, the use of contrast media allows the detection of enhancement of the wall, septa, intracystic nodules or of the entire nodule. On the basis of our results, CEUS may play an important role in demonstrating blood flow within hypovascular tumors not correctly detected by CT, but no less important, converting indeterminate CT lesions into benign Bosniak I /II cysts that do not require further examination as occurred in 44 out of 83 (53%) lesions in our study with the advantage of the absence of radiation and low cost. Nevertheless, the effectiveness of CEUS can decrease if the lesion cannot be adequately identified by US, (as happened in 3 of our cases), and this limitation depends on the features of the patients and size and location of lesions, being more common in obese patients, bowel gas interposition and small lesions on the medial aspect of the kidney. However, in our study we did not evaluate the influence of the size or location of the lesions on the accuracy of CEUS.
This study has other limitations. First, we did not obtain a histological diagnosis of benign cystic lesions. Even with a lengthy follow-up (minimum of 23 months in this study) some RCC may have a very slow growth, and a follow-up of at least 3 years is recommended to rule-out malignancy in patients managed without surgery. In our study one lesion characterized as a benign cyst using CEUS showed nodular enhancement at a follow-up CT that corresponded to a RCC. Nevertheless, due to the interval of time from the CEUS to the CT study, it is impossible to ensure whether CEUS had not identified the intracystic nodule or the nodule had appeared between the CEUS and follow-up CT studies. Second, we did not evaluate the real usefulness of CEUS to differentiate between benign and malignant lesions. In the group of benign cystic lesions we included Bosniak IIF cysts with a 5–10% estimated probability of malignancy, and in the group of potentially malignant lesions we included some benign lesions, which were 3 oncocytomas and 1 multicystic nephroma. However, the aim of the study was to evaluate whether CEUS is helpful in determining the management of indeterminate lesions and identifying lesions with features on CEUS that do not require further assessment or only require follow-up imaging. It has been well demonstrated, that the absence of enhancement after microbubble injection implies no further workup, whereas evidence of thickened septations or mural nodules that enhance after microbubble injection is considered a reliable criterion for malignancy [11, 20]. Moreover, in clinical practice most solid benign lesions (except the typical angiomyolipoma) and benign Bosniak III cysts cannot be distinguished from malignant lesions using imaging techniques based on contrast enhancement and require histology to achieve a final diagnosis [23–25].
In conclusion, the present study demonstrates that in renal nodules which are indeterminate on CT, CEUS is very useful in the differentiation between benign complex cysts and other lesions that require further investigation, improving the accuracy of unenhanced US from 42.2 to 95.2%.
References
Israel GM, Bosniak MA (2005) An update of the Bosniak renal cyst classification system. Urology 66:484–488. doi:10.1016/j.urology.2005.04.003
Israel GM, Silverman SG (2011) The incidental renal mass. Radiol Clin North Am 49:369–383. doi:10.1016/j.rcl.2010.10.007
Israel GM, Hindman N, Bosniak MA (2004) Evaluation of cystic renal masses: comparison of CT and MR imaging by using the Bosniak classification system. Radiology 231:365–371. doi:10.1148/radiol.2312031025
Silverman SG, Israel GM, Herts BR, Richie JP (2008) Management of the incidental renal mass. Radiology 249:16–31. doi:10.1148/radiol.2491070783
Patel NS, Poder L, Wang ZJ, et al. (2009) The characterization of small hypoattenuating renal masses on contrast-enhanced CT. Clin Imaging 33:295–300. doi:10.1016/j.clinimag.2008.12.002
Egbert ND, Caoili EM, Cohan RH, et al. (2013) Differentiation of papillary renal cell carcinoma subtypes on CT and MRI. AJR Am J Roentgenol 201:347–355. doi:10.2214/AJR.12.9451
Tappouni R, Kissane J, Sarwani N, Lehman EB (2012) Pseudoenhancement of renal cysts: influence of lesion size, lesion location, slice thickness, and number of MDCT detectors. AJR Am J Roentgenol 198:133–137. doi:10.2214/AJR.10.6057
Weber TM (2006) Sonography of benign renal cystic disease. Radiol Clin North Am 44:777–786. doi:10.1016/j.rcl.2006.10.013
Xu Z-F, Xu H-X, Xie X-Y, et al. (2010) Renal cell carcinoma: real-time contrast-enhanced ultrasound findings. Abdom Imaging 35:750–756. doi:10.1007/s00261-009-9583-y
Gerst S, Hann LE, Li D, et al. (2011) Evaluation of renal masses with contrast-enhanced ultrasound: initial experience. AJR Am J Roentgenol 197:897–906. doi:10.2214/AJR.10.6330
Nicolau C, Bunesch L, Sebastia C (2011) Renal complex cysts in adults: contrast-enhanced ultrasound. Abdom Imaging 36:742–752. doi:10.1007/s00261-011-9727-8
Ascenti G, Mazziotti S, Zimbaro G, et al. (2007) Complex cystic renal masses: characterization with contrast-enhanced US. Radiology 243:158–165. doi:10.1148/radiol.2431051924
Park BK, Kim B, Kim SH, et al. (2007) Assessment of cystic renal masses based on Bosniak classification: comparison of CT and contrast-enhanced US. Eur J Radiol 61:310–314. doi:10.1016/j.ejrad.2006.10.004
Peng Y, Jia L, Sun N, et al. (2010) Assessment of cystic renal masses in children: comparison of multislice computed tomography and ultrasound imaging using the Bosniak classification system. Eur J Radiol 75:287–292. doi:10.1016/j.ejrad.2010.05.035
Piscaglia F, Nolsøe C, Dietrich CF, et al. (2012) The EFSUMB Guidelines and Recommendations on the Clinical Practice of Contrast Enhanced Ultrasound (CEUS): update 2011 on non-hepatic applications. Ultraschall Med 33:33–59. doi:10.1055/s-0031-1281676
Cazals X, Bleuzen A, Tranquart F, et al. (2011) Value of contrast-enhanced ultrasound in the evaluation of indeterminate renal cysts on CT. J Radiol 92:1081–1090. doi:10.1016/j.jradio.2011.09.008
Barr RG, Peterson C, Hindi A (2014) Evaluation of indeterminate renal masses with contrast-enhanced US: a diagnostic performance study. Radiology 271:133–142. doi:10.1148/radiol.13130161
Curry NS, Cochran ST, Bissada NK (2000) Cystic renal masses: accurate Bosniak classification requires adequate renal CT. AJR Am J Roentgenol 175:339–342. doi:10.2214/ajr.175.2.1750339
Johnson PT, Horton KM, Fishman EK (2010) How not to miss or mischaracterize a renal cell carcinoma: protocols, pearls, and pitfalls. AJR Am J Roentgenol 194:W307–W315. doi:10.2214/AJR.09.3033
Quaia E, Bertolotto M, Cioffi V, et al. (2008) Comparison of contrast-enhanced sonography with unenhanced sonography and contrast-enhanced CT in the diagnosis of malignancy in complex cystic renal masses. AJR Am J Roentgenol 191:1239–1249. doi:10.2214/AJR.07.3546
Clevert D-A, Minaifar N, Weckbach S, et al. (2008) Multislice computed tomography versus contrast-enhanced ultrasound in evaluation of complex cystic renal masses using the Bosniak classification system. Clin Hemorheol Microcirc 39:171–178
Hélénon O, Correas JM, Balleyguier C, et al. (2001) Ultrasound of renal tumors. Eur Radiol 11:1890–1901. doi:10.1007/s003300101077
Choudhary S, Rajesh A, Mayer NJ, et al. (2009) Renal oncocytoma: CT features cannot reliably distinguish oncocytoma from other renal neoplasms. Clin Radiol 64:517–522. doi:10.1016/j.crad.2008.12.011
Rosenkrantz AB, Hindman N, Fitzgerald EF, et al. (2010) MRI features of renal oncocytoma and chromophobe renal cell carcinoma. AJR Am J Roentgenol 195:W421–W427. doi:10.2214/AJR.10.4718
Pedrosa I, Sun MR, Spencer M, et al. (2008) MR imaging of renal masses: correlation with findings at surgery and pathologic analysis. Radiographics 28:985–1003. doi:10.1148/rg.284065018
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nicolau, C., Buñesch, L., Paño, B. et al. Prospective evaluation of CT indeterminate renal masses using US and contrast-enhanced ultrasound. Abdom Imaging 40, 542–551 (2015). https://doi.org/10.1007/s00261-014-0237-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00261-014-0237-3