Abstract
Objective
The purpose of this study is to investigate radiologic findings of struma ovarii, and to correlate both CT and MR findings.
Materials and methods
MR images of 26 cases were retrospectively reviewed. Post-contrast enhanced T1-weighted images were available in 17 patients. CT images, including seven non-contrast and eleven post-contrast studies, were available for review in 13 cases.
Results
All 26 tumors appeared as well-defined cystic tumors with solid components, which were multilobulated surfaces in 19 and smooth surfaces in seven. Twenty-four was multicystic, whereas two were unilocular. The solid components were recognized as thickened septi or walls in 23 and a mass in three tumors. On T2-weighted images, loculi of prominent low intensity were recognized in 16 tumors. On T1-weighted images, the punctuate foci of high intensity were recognized in 24 tumors in or adjacent to the solid components. Ascites was present in only one lesion. In six of seven cases with non-contrast CT images, high attenuation areas were recognized. In five of these six tumors, high attenuation areas corresponded to the areas of prominent low intensity and the solid components on T2-weighted images. In seven cases with CT, curvilinear calcifications were recognized in the solid components.
Conclusion
Struma ovarii typically presents as a lobulated multicystic lesion with solid components. The tumors frequently include loculi of low intensity on T2-weighted images and punctuate foci of high intensity on T1-weighted images. On CT, high attenuation areas and calcifications in the solid components are common findings.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Struma ovarii is a rare variant of teratoma, characterized by either entire or predominant thyroid tissue, or macroscopically recognized thyroid tissue [1, 2]. They account for 0.1–0.3% of all ovarian tumors, and approximately 3% of all mature teratomas [3], and may be associated with focal components of ordinary dermoid or carcinoid tumor (strumal carcinoid). Rarely, struma ovarii may be recognized in mucinous cystadenoma or be admixed with Brenner tumor [2]. Struma ovarii commonly occurs in reproductive women, although occasionally it has been reported in prepubery and postmenopausal females [2, 4]. The typical clinical symptom is a non-specific palpable pelvic mass. Hyperthyroidism related to struma ovarii is infrequent, and reported in only about 5% of cases [5]. Since, struma ovarii is distinctly benign, radiological preoperative diagnosis is of benefit for determining appropriate management, especially in patients of reproductive age in whom fertility preserving therapy is crucial.
CT and MRI findings in struma ovarii have already been reported. MR findings include a multicystic mass with a multilobulated surface, some solid components including thickened septi or cyst walls and cystic components showing a variety of signal intensities on both T1- and T2-weighted images [4, 6–8]. CT findings of struma ovarii are characterized by the presence of high attenuation area and calcifications [9, 10]. The purpose of this study is first to investigate the frequency of each finding in a large series of patients, and then to correlate both CT and MR findings.
Materials and methods
Patients
We retrospectively reviewed the clinical records and imaging features in 26 cases of pathologically diagnosed struma ovarii at three different institutions (Kyoto University Hospital, Shiga Medical Center for Adults and Osaka Red Cross Hospital) between April 2000 and September 2010. The age of patients ranged from 22 to 78 years old, with a mean age of 42 years old. No patients presented with clinical signs of hyperthyroidism. The interval between surgery and preoperative MR or CT examinations ranged from 2 to 90 days, with a mean of 25 days.
Images
MR images including both sagittal and axial T2-weighted images and sagittal T1-weighted images were available for review in all patients. Additional T1-weighted images with fat suppression were also available in 14 patients. Axial and sagittal contrast enhanced T1-weighted images with gadolinium-based agents were acquired in 17 patients, and fat-suppression images were applied in 14 of 17 patients. One patient was imaged on a 3.0-T scanner, while 25 patients were imaged on 1.5-T scanners.
CT images, including seven non-contrast and eleven post-contrast studies were available for review in 13 patients. Both non-contrast and post-contrast studies were acquired in five patients.
Image evaluation
CT and MR images were retrospectively reviewed by two radiologists, with experience in 16 and 13 years of gynecologic radiology, independently, regarding the following items; the size, the configuration, the signal intensity, and attenuation of solid and cystic components, and the presence of fatty components, calcifications, and ascites. On post-contrast MR studies, the presence of enhancement was evaluated by comparison with unenhanced studies.
The discrepancies in the interpretation were resolved in a consensus manner, in the presence of another radiologist with an experience of 11 years.
Signal intensities on MR images on T1-weighted images were defined as follows: low signal intensity, as signal intensity lower than that of the skeletal muscle; intermediate intensity, as signal intensity equal to that of the skeletal muscle; high intensity, as signal intensity higher than that of skeletal muscle. Signal intensities on T2-weighted images were defined as follows: low intensity, as signal intensity lower than or equal to that of the skeletal muscle; intermediate intensity, as signal intensity higher than that of the skeletal muscle but lower than that of the urine in the bladder; high intensity, as signal intensity equal to or higher than that of the urine in the bladder.
On CT images, the attenuation of the solid components was compared with that of the skeletal muscle.
Results
MRI findings
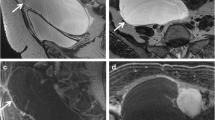
The maximum sizes of the tumors ranged from 5.2 to 27.2 cm, with a mean of 11.1 cm. All 26 tumors had well-defined margins, which were multilobulated surfaces in 19 (73%) and smooth surfaces in seven (27%) (Fig. 1). Twenty-four of 26 tumors (92%) exhibited a multicystic appearance, whereas the other two tumors (8%) were unilocular cystic lesions.
45-Year old woman with struma ovarii of the right ovary. A Axial T2-weighted image shows a multicystic lesion with a lobulated surface. Its solid components are composed with multiple thickened septa and walls of gathering cyst or loculi. The solid component shows low intensity on T2-weighted image (arrow head). A majority of the cystic components show high signal intensity. B A majority of the cystic components show variable signal intensity from low to intermediate intensity on sagittal T1-weighted image. Within or adjacent to the solid components, focal high intensity foci are scattered (arrow). C On sagittal T1-weighted image with fat suppression, these high intensity foci on T1-weighted image are not suppressed (arrow). D On Gd contrast-enhanced T1-weighted images, solid components are well enhanced.
The solid components in the cystic lesions were recognized as thickened septa or cyst walls in 23 (88%) and a mass in three tumors (12%) (Figs. 1, 2). They were well enhanced in all 17 tumors, in which post-contrast-enhanced images were available for review (Fig. 1). On T2-weighted images, the areas of prominent low intensity were recognized in 16 tumors (62%) with multilocular appearances (Fig. 3). The signal intensity in the other tumors was predominantly high. On T1-weighted images, the area of prominent low intensity on T2-weighted images displayed low in three, intermediate in seven and slightly increased intensity in six patients (Fig. 3). In 14 of 16 tumors with areas of prominent low intensity on T2-weighted images, post-contrast images were available for review. The comparison of pre- and post-contrast T1-weighted images did not reveal any enhancement in these areas of low intensity on T2-weighted images (Fig. 3). The loculi of high intensity on T2-weighted images showed various signal intensities from low to high intensity on T1-weighted images.
47-Year old woman with struma ovarii accompanied by a dermoid cyst of the left ovary. A Sagittal T2-weighted image shows a multicystic lesion with a stained glass appearance, in which some loculi display prominent low intensity (arrows). B On sagittal T1-weighted image, the loculi showing prominent low intensity on T2-weighted image display intermediate intensity (arrows). Scattered high intensity foci are also presented adjacent to the solid components. The large locule showing high intensity represents a dermoid cyst (arrowhead). C On Gd contrast-enhanced T1-weighted image, the solid components are well enhanced.
On T1-weighted images, the punctuate foci of high intensity were recognized in 24 tumors (92%) in or adjacent to the thickened septi, cyst walls, or solid mass (Fig. 1). In 14 of 24 tumors, in which fat-suppression T1-weighted images were available for review, the high intensity was not suppressed on fat-suppression images in 13 cases (Figs. 1, 2), while in the other case it was suppressed.
The signal intensity of solid components on T2-weighted images was intermediate to high in four (15%) and low to intermediate in 22 (85%) (Figs. 1, 2, 5). The signal intensity of solid components on T1-weighted images was intermediate to slightly high in nine (35%) and intermediate in 17 (65%).
In addition, foci of fat intensity were present in twelve tumors (46%) (Fig. 3). A large amount of ascites was present in only one lesion (4%).
CT findings and correlated MR findings
In seven cases with non-contrast CT images, foci of high attenuation were recognized in six tumors (86%) (Figs. 4, 5). In five of these six tumors, the foci of high attenuation corresponded to the areas of prominent low intensity and the solid components on T2-weighted images. In one of six cases which had no areas of prominent low intensity, they corresponded to the solid components. On T1-weighted images, they showed intermediate to slightly high signal intensity. One tumor with no foci of high attenuation on non-contrast CT did not show any areas of prominent low intensity on T2-weighted images. The solid components in this tumor showed the attenuation equal to that of the skeletal muscle on non-contrast CT images.
The low attenuation areas on non-contrast CT images corresponded to the cystic areas showing high intensity on T2-weighted images.
In seven of 13 cases with CT, curvilinear calcifications were recognized in the thickened septi or cyst walls (54%) (Fig. 6).
Discussion
Struma ovarii typically appears as a multicystic mass with lobulated surface and some solid components including thickened septic or cyst walls, representing thyroid tissue with markedly dilated thyroid follicles [4, 6–8]. On transvaginal ultrasonography, struma ovarii typically appears as cystic tumors containing well circumscribed roundish solid areas with smooth contour named as “struma pearls”, although struma ovarii can show variable sonographic features [11]. CT findings in struma ovarii on non-contrast image are characterized by the presence of high attenuation area and calcification [9, 10, 12]. The presence of areas of very low intensity on T2-weighted images (62%) and the punctuate foci of high intensity on T1-weighted images (91%) are common findings in our series, although the cystic components show a variety of signal intensities on MR images. The areas of very low intensity on T2-weighted images were thought to be cystic, because they did not reveal any enhancement on contrast-enhanced MR studies.
In the correlation between CT and MR images, all areas of prominent low intensity on T2-weighted images corresponded to the foci of high attenuation on non-contrast CT images. The high attenuation on non-contrast CT images and low intensity on T2-weighted images can be simultaneously explained by the thyroid follicles that contain highly gelatinous colloid material containing thyroid hormones with iodine compounds [4, 6, 9]. On the other hand, cystic components of decreased attenuation on CT, which shows increased intensity on T2-weighted images and decreased intensity on T1-weighted images, are considered to contain fluid of diluted thyroid hormones.
On T1-weighted images, the high intensity foci in struma ovarii is mostly punctuate, and their causes are either tiny cysts of viscous material, hemorrhage, or focal fatty tissue representing dermoid cysts admixed in the struma [6, 8]. When fat-suppression MR images or CT are obtained, a focus of dermoid cyst shows suppressed signal or decreased attenuation on CT similar to subcutaneous fat tissue.
The solid components in struma ovarii were frequently observed as focal thickening in the septi of the cystic tumor. These solid components show high attenuation on non-contrast CT images, and are well enhanced on contrast-enhanced MR or CT studies. These components may correspond to sonographic “strumal pearls”, which appears vascularized on Doppler images [11]. Pathologically, the solid components in struma are composed of mature thyroid tissue, abundant blood vessels, and fibrous tissue [1, 6, 11]. The high attenuation of solid components on non-contrast CT images reflects the thyroid tissues with small follicles filled with colloid material similar to the thyroid gland.
On CT images, calcifications along the thickened septa or cyst walls were frequent findings (54%). A previous study on CT findings of struma ovarii reports similar frequency (60%) of this finding [12]. This finding may be one of helpful clues for the diagnosis of struma ovarii.
When struma ovarii is accompanied by foci of fatty tissue, they can be considered as areas of dermoid cyst associated with strumal ovarii [13]. In these circumstances, the presence of dermoid cyst can be strong clue to suspect struma ovarii. Although, fat tissue may be seen in immature teratoms, immature teratoma occurs in very young patients, and contains scattered fat tissues throughout tumors, unlike struma ovarii [13, 14]. When struma ovarii consists of cystic components without fat, the differential diagnosis includes a wide variety of multilocular cystic ovarian tumors, including serous and mucinous cystic tumors, metastatic ovarian tumors, and endometrial cysts. The loculi of very low intensity on T2-weighted images can be seen in all of these cystic tumors above, and are not useful findings for differentiation. However, the surfaces of most of these tumors are generally smooth, whereas struma ovarii shows a lobulated appearance. Although, endometrial cysts may show a multicystic appearance with decreased intensity on T2-weighted images, the cystic components usually show entirely high intensity on T1-weighted images. When the diagnosis is indeterminate on MR images, the presence of high attenuation areas and calcifications on non-contrast CT images are suggestive findings for struma ovarii.
Conclusion
Struma ovarii typically presents as a multicystic lesion with a lobulated surface and some degree of solid components including thickened septi or cyst walls and occasionally has a solid component like a mass.
The cystic components occasionally have loculi of low intensity on T2-weighted images and frequently have punctuate foci of high intensity in or adjacent to solid components on T1-weighted images, which may be of great diagnostic value.
The high attenuation of cystic lesions, solid components and calcifications along the cyst walls may be characteristic findings on CT images.
Recognition of these manifestations is helpful in the diagnosis of this tumor.
References
Kurman RJ (2002) Germ cell tumors of the ovary. Blaustein’s pathology of the female genital tract, 5th edn. New York: Springer
Scully RE, Young RH, Clement PB (1998) Monodermal teratomas. Tumors of the ovary, maldeveloped gonads, fallopian tube, and broad ligament. Atlas of tumor pathology. Third Series, Fascicle 23. Armed Forces Institute of Pathology, Washington DC
Outwater EK, Siegelman ES, Hunt JL (2001) Ovarian teratomas: tumor types and imaging characteristics. Radiographics 21(2):475–490
Joja I, Asakawa T, Mitsumori A, et al. (1998) Struma ovarii: appearance on MR images. Abdom Imaging 23(6):652–656
Kempers RD, Dockerty MB, Hoffman DL, Bartholomew LG (1970) Struma ovarii—ascitic, hyperthyroid, and asymptomatic syndromes. Ann Intern Med 72(6):883–893
Kim JC, Kim SS, Park JY (2000) MR findings of struma ovarii. Clin Imaging 24(1):28–33
Matsuki M, Kaji Y, Matsuo M, Kobashi Y (2000) Struma ovarii: MRI findings. Br J Radiol 73(865):87–90
Dohke M, Watanabe Y, Takahashi A, et al. (1997) Struma ovarii: MR findings. J Comput Assist Tomogr 21(2):265–267
Jung SI, Kim YJ, Lee MW, et al. (2008) Struma ovarii: CT findings. Abdom Imaging 33(6):740–743
Matsumoto F, Yoshioka H, Hamada T, Ishida O, Noda K (1990) Struma ovarii: CT and MR findings. J Comput Assist Tomogr 14(2):310–312
Savelli L, Testa AC, Timmerman D, Paladini D, et al. (2008) Imaging of gynecological disease (4): clinical and ultrasound characteristics of struma ovarii. Ultrasound Obstet Gynecol 32(2):210–219
Shen J, Xia X, Lin Y, Zhu W, Yuan J (2010) Diagnosis of struma ovarii with medical imaging. Abdom Imaging 36(5):627–631
Saba L, Guerriero S, Sulcis R, et al. (2009) Mature and immature ovarian teratomas: CT, US and MR imaging characteristics. Eur J Radiol 72(3):454–463
Yamaoka T, Togashi K, Koyama T, et al. (2003) Immature teratoma of the ovary: correlation of MR imaging and pathologic findings. Eur Radiol 13(2):313–319
Acknowledgments
We would like to acknowledge Dr. Shunsuke Minami, Dr. Girou Toudou, and Dr. Thai Akasaka for their many helpful advices for drafting this manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ikeuchi, T., Koyama, T., Tamai, K. et al. CT and MR features of struma ovarii. Abdom Radiol 37, 904–910 (2012). https://doi.org/10.1007/s00261-011-9817-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00261-011-9817-7