Abstract
Background
68Ga prostate specific membrane antigen PET/CT (68Ga-PSMA PET/CT) may be superior to multiparametric MRI (mpMRI) for localisation of prostate cancer tumour foci, however the concordance and differences between 68Ga-PSMA PET/CT and mpMRI when applied to all biopsied patients and potential benefit in patients with negative mpMRI is unclear.
Methods
Retrospective analysis of patients undergoing mpMRI, prostate biopsy and 68Ga-PSMA PET/CT over a 3-year period. Diagnostic performance of 68Ga-PSMA PET/CT and mpMRI were assessed using biopsy histopathology for the entire cohort and radical prostatectomy specimen in a subset of patients. Lesion concordance and additional detection of each modality were determined, including in a dedicated cohort of patients with mpMRI PIRADS 2 scans.
Results
A total of 144 patients were included in the study. Index lesion/foci detection was similar between 68Ga-PSMA PET/CT and mpMRI (sensitivity 83.1% vs 90.1%; p = 0.267), however lesions missed by mpMRI were larger (1.66 cm3 vs 0.72 cm3; p = 0.034). Lesion detection rates were similar across the biopsy histopathology and radical prostatectomy specimen subset, with a high concordance for index (80.1%) and a moderate concordance for total (67%) lesions between the 2 imaging modalities. The additional detection yield favoured 68Ga-PSMA PET/CT over mpMRI for index (13.5% vs 4.3%) and total (18.2% vs 5.4%) lesions; both modalities missed 2.1% and 12.3% of index and total lesions, respectively. 68Ga-PSMA PET/CT identified 9 of 11 patients with PIRADS 2 mpMRI but subsequently diagnosed with Gleason ≥ 3 + 4 disease.
Conclusions
Despite high concordance rates, 68Ga-PSMA PET/CT incrementally improved tumour localisation compared with mpMRI. These results suggest that 68Ga-PSMA PET/CT may have an incremental value to that of mpMRI in the diagnostic process for prostate.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Multiparametric magnetic resonance imaging (mpMRI) has changed the paradigm in the diagnosis of prostate cancer (PCa) [1,2,3]. Several studies have shown that a mpMRI-triage pathway improves diagnostic accuracy of clinically significant PCa (csPCa) (93% vs. 48%) and improved csPCa detection (38% vs 26%) while preventing unnecessary biopsies (27–28%) and decreasing insignificant PCa diagnoses by 5–13% in comparison with systematic transrectal ultrasound (TRUS) [1, 2]. These benefits have been confirmed in a day-to-day clinical practice, equating to a 47% biopsy avoidance and a 60.5% csPCa diagnosis rate in our centre [3].
Although mpMRI has demonstrated an accuracy of > 90% in identification of index lesions, its accuracy in identification of secondary foci and smaller csPCa is less well-established [4,6,7,7]. On per lesion analysis, mpMRI misses between 16 and 25.4% of csPCa tumour foci [5, 8]. Also, there is increased difficulty with transition zone tumour identification [9], and mpMRI has been shown to under estimate tumour size by as much as 3 times the actual tumour volume in comparison with whole gland histology [10]. Despite the clinical benefit of mpMRI in biopsy triage, lesion targeting and surgical planning, the limitations of mpMRI restrict its application to replace biopsy.
Prostate-specific membrane antigen (PSMA) is a transmembrane protein that has increased expression in malignant prostate cells, being expressed in > 90% of primary and secondary cases [11]. Accumulating evidence supports the utilization of PSMA positron-emission tomography (PSMA-PET) in prostate cancer staging, with most studies focusing on men who demonstrate biochemical recurrence after treatment for localised disease [11,13,14,15,16,16]. The limited evidence available suggests superior performance of PSMA-PET in comparison with mpMRI for lesion characterisation and intra-prostatic staging [17,19,20,20]. However, due to the small sample size and correlation with radical prostatectomy histopathology in these studies, it is unclear how translatable these findings are to a wider spectrum of PCa patients, including those who will undergo alternative forms of management.
The aim of this study was to evaluate PCa lesion concordance and differences according to 68Ga-PSMA PET/CT avidity and mpMRI as determined by radical prostatectomy and prostate biopsy-based PCa detection.
Methods
Study population
In this retrospective analysis of a single-centre’s experience, consecutive patients from June 2014 to July 2017 with suspected PCa based on elevated serum prostate-specific antigen (PSA) levels and/or an abnormal digital rectal examination (DRE) underwent a triage mpMRI. Patients with suspicious findings or mpMRI, as well as those with ongoing clinical concern despite a normal MRI underwent a transrectal ultrasound-guided transperineal prostate biopsies (TPB). Once a cancer diagnosis was made, they were then staged using 68Ga-PSMA PET/CT imaging according to the local departmental protocol [20]. Patients were excluded from the study if they had previously received treatment with radiotherapy.
The study was approved by the Ethics Committee of the Royal Brisbane and Women’s Hospital (RBWH) Human Research Ethics Committee (approval number HREC/17/QRBW/644). The initial analysis of this dataset (n = 58), being a comparison of mpMRI, 68Ga-PSMA PET/CT with radical prostatectomy histology, has been published previously [20]. Here, we present an expanded dataset, including patients referred for radiotherapy so more applicable to the general population encountered in a urology clinic, with comparison with biopsy accuracy and a sub-analysis for patients with PCa not visible on mpMRI.
Magnetic resonance imaging protocol
Our institution utilises a mpMRI-based triage system to select patients for prostate biopsy based on favourable results from cohort and randomised studies [1, 2, 3, 21]. All mpMRI prostate scans were conducted at 3 tesla (Magnetom Skyra, Siemens) without endorectal coil using intravenous contrast (Gadovist 5–15 mL IV dynamic). The mpMRI images were interpreted by both radiologist and treating urologist with experience in prostate mpMRI interpretation of average 15 cases per month and 50 cases per month, respectively. All mpMRI scans were evaluated using the Prostate Imaging Reporting and Data System (PIRADS), transitioning from version 1 to version 2 in 2016 and considering variables such as apparent diffusion coefficient (ADC); images were subsequently reported as low-risk (PIRADS 2), equivocal (PIRADS 3) or intermediate/high-risk (PIRADS 4/5) depending on the presence and characteristics of the lesion.
Prostate biopsy protocol
The decision to biopsy was at the discretion of one of eight treating urologists, based on assessment of mpMRI result, PSA, PSA density (PSAD), PSA kinetics, age, clinical history and examination. In patients with PIRADS 3–5, cognitive directed ultrasound-guided transperineal targeted biopsies were performed through the lesion with systematic biopsies also performed in 91.7% of cases. Patients without a suspicious lesion on mpMRI (PIRADS 2) underwent systematic TPB only. Biopsies were performed under general or spinal anaesthesia, by one of eight consultant urologists or by a registrar/trainee under consultant supervision. Surgeons reviewed patient’s clinical details including PSA and imaging prior to each procedure and made final decision on proceeding to biopsy.
68Ga PSMA PET/CT protocol
The use of 68Ga PSMA PET/CT was at the discretion of the treating urologist and most commonly requested following PCa diagnosis. PET images were obtained using 68Ga-PSMA-11 (ABX AG, Germany) manufactured at the Specialised PET Services Queensland Radiopharmaceutical Laboratory. A total of 150 MBq ± 5% of tracer was injected intravenously, 60 min prior to image acquisition. Emission tomographic images were obtained from the skull vertex to thighs, and a low dose CT scan was performed during tidal respiration for attenuation correction and lesion localization. All 68Ga PSMA PET/CT images were acquired from a Biograph mCT scanner (Siemens Medical Solutions), then evaluated by two experienced nuclear medicine specialists with appropriate viewing software (Syngo.via; Siemens Medical Solutions). Each of our nuclear medicine specialists with urological imaging specialty reads approximately 200 68Ga PSMA PET/CT per year. Lesions detected with PET/CT were considered positive for PCa if they demonstrated focal 68Ga-PSMA uptake significantly higher than background prostatic uptake according to nuclear medicine specialist’s interpretation, and equivocal if uptake was only marginally higher than background according to the nuclear medicine specialist’s interpretation. The image reports were converted to a 3-point Likert scale (likely, equivocal, unlikely), as previously reported [17]. Furthermore, semi-quantitative analysis using an automated standardized maximum uptake value (SUV max) was considered for each lesion, although these values were not used to report primary lesions, in keeping with local reporting guidelines.
Histological examination and lesion concordance
Histopathological analysis and reporting was performed according to the International Society of Urological Pathology (ISUP) standard protocols by four experienced uropathologists, and structured according to the 2014 ISUP Gleason Grading Guidelines [22]. Clinically significant disease (csPCa) was defined as PCa with Gleason score 3 + 4 = 7 or greater [23]. Index tumours were defined as the tumour focus with highest Gleason score; if there were two foci with identical tumour grade, the larger focus would be designated as index [24].
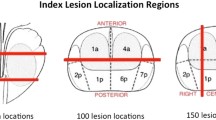
For concordance analysis, both whole-gland radical prostatectomy (RP) histology and prostate biopsy histology were analysed. For whole-gland prostate analysis, tumour foci locations were determined according to the sextant-based descriptions included in structured pathology reports [20]. To compare lesions with biopsy histology, biopsy location was described by the operating surgeon and was correlated with both operation and pathology reports. Tumour foci locations, from both the biopsy and RP histopathology, were then compared with lesion locations on each imaging modality. To assess for bias, subgroup analysis was carried out on the biopsy results of patients whom underwent RP.
Tumour foci were allocated to one of four concordance groups, both imaging modalities were concordant with histopathology (concordant; group C), only 68Ga-PSMA PET/CT was concordant (group P), only mpMRI was concordant (group M), and neither imaging modality was concordant (invisible; group I). All lesions described within the imaging reports for 68Ga-PSMA PET/CT and mpMRI were considered, as previously described [20].
Statistical analysis
Relevant patient clinical, imaging and histopathological data was extracted from the hospital database and stored confidentially in Excel. All statistical analysis was performed using the SPSS Statistics v25 (IBM; Armonk, NY, USA). Patient demographics and baseline clinical characteristics were summarized using descriptive statistics. Frequency distribution with percentages were used when summarizing categorical variables, and medians with interquartile range was used to summarize continuous variables. Data was curated into dichotomous or categorical variables for most comparative analyses, except for continuous variables, including prostate volume (PV), PSA, PSAD, SUVmax, ADC and age. Categorical values were compared using Fisher’s test, while continuous variables were compared with Mann-Whitney test as most comparisons involved one or more datasets that were not normally distributed.
Results
Demographics
From 653 patients undergoing mpMRI, 344 proceeded to biopsy, of which 144 had a 68Ga-PSMA PET/CT and were included in the study population (Table 1). 68Ga-PSMA PET/CT was predominantly used in higher risk men, based on serum PSA (8.60, IQR 6.00–12.75 ng/mL vs 6.40, IQR 4.60–9.45 ng/ml; p < 0.001), PSAD (0.242, IQR 0.153–0.322 ng/mL2 vs 0.120, IQR 0.090–0.190; p < 0.001) and proportion of patients with Gleason score ≥ 4 + 3 csPCa (47.2% vs 21.20%; p < 0.001) when compared with those who did not undergo 68Ga-PSMA PET/CT. Most mpMRI lesions were PIRADS 4/5 (125; 86.7%). RP was the most common treatment method (49%; Table 1), followed by radiation therapy (36 patients; 25.9%), androgen deprivation therapy (8.6%), active surveillance (6.5%) and watchful waiting (2.9%), while 9 (6.5%) had incomplete medical documentation at time of publication. The radiation therapy cohort was older, with higher PSA, PSAD and Gleason scores than men who underwent prostatectomy (Table 1). The 3 patients without PCa on biopsy underwent routine PSA surveillance. Index lesion Gleason grade according to RP histopathology were mostly Gleason 3 + 4 = 7 (n = 41; 58%), 4 + 3 = 7 (n = 18; 25%), and ≥ 4 + 4 = 9 (n = 12; 17%).
Imaging diagnostic performance according to RP histopathology
The RP subset was initially considered as the “gold standard” for histopathological accuracy of index lesions (Table 2). All 71 patients who underwent RP had whole-gland histopathology demonstrating csPCa. For detection of the index lesion, the sensitivities of mpMRI and 68Ga-PSMA PET/CT were 83.1% vs 90.1% (p = 0.267) when equivocal or likely lesions were considered, and 80.3% vs 87.3% (p = 0.267) when only likely lesions were considered. Furthermore, the median size of index tumour foci missed by mpMRI was larger than those missed by 68Ga-PSMA PET/CT (1.66 cm3; IQR 0.79–2.53 cm3 vs 0.72 cm3; IQR 0.36–1.0 cm3; p = 0.034) despite similar serum PSA, prostate volume and PSAD (p > 0.05).
68Ga-PSMA PET/CT demonstrated 90 lesions (equivocal or likely), of which 85 were concordant with csPCa per whole-gland histopathology (specificity 94.4%; Fig. 1a; Table 3). Although mpMRI detected similar total (n = 74) and concordant (n = 68) lesions compared with 68Ga-PSMA PET/CT, with specificity not reaching statistical significance (90.7%; p = 0.55). In the more likely lesions there was also no statistically significant difference in specificity (Table 3; 97.4% vs 93.8%; p > 0.05) between 68Ga-PSMA PET/CT and MRI.
Imaging lesion concordance analysis comparing PSMA and mpMRI according to whether the lesions were equivocal or likely (> = Equivocal) or likely only. Percentage of lesions concordant with respective histopathology tumour foci is described. a Comparison of PSMA and mpMRI lesions to radical prostatectomy (RP) histopathology. b Comparison to biopsy histopathology within radical prostatectomy (RP) cohort. c Comparison to biopsy histopathology in entire cohort
Imaging performance according to biopsy histopathology in patients who underwent RP
When biopsy histopathology of the subset of patients who underwent RP was considered (Fig. 1b), the specificity of MRI in detecting csPCa was unchanged, despite identifying fewer lesions (both likely and equivocal or likely). The specificity of 68Ga-PSMA PET/CT was slightly reduced to 88.9% in equivocal or likely and 94.8% for likely only lesions. The specificities of 68Ga-PSMA PET/CT (p = 0.570) or mpMRI (p = 0.807) were similar in lesion detection according to RP and biopsy (among those who underwent RP) histopathology.
Imaging diagnostic performance according to biopsy histopathology in entire cohort
When the entire cohort was considered, there were 141 patients (97.9%) with biopsies positive for PCa, with 136 (94.4%) demonstrating csPCa (Table 2). Considering lesions that were equivocal or likely with corresponding biopsy histopathology, mpMRI detected significantly fewer cases of csPCa than 68Ga-PSMA PET/CT (117 vs 129 patients; sensitivity 86.0% vs 94.9%; p = 0.017). When looking specifically at the likely lesions, the difference in csPCa detection between mpMRI and 68Ga-PSMA PET/CT was not statistically significant (115 vs 124 patients; sensitivity 84.6% vs 91.2%, p = 0.064). Both mpMRI and 68Ga-PSMA PET/CT had high specificity for csPCa, with equivocal or likely lesions detected with specificity of 93.8% and 92.8% and likely only with specificity of 95.2% and 95.4%, respectively.
When all lesions on mpMRI and 68Ga-PSMA PET/CT were considered (Table 3), mpMRI detected less lesions than 68Ga-PSMA PET/CT. For likely only lesions, mpMRI detected 142 lesions, of which 6 (4.2%) were benign and 3 (2.1%) were Gleason 3 + 3, while 68Ga-PSMA PET/CT detected 164 lesions, of which 11 (6.7%) were benign and 3 (1.8%) were Gleason 3 + 3. For equivocal lesions, Gleason 3 + 4 (n = 7) and Gleason 3 + 3 (n = 1) were detected with both mpMRI and 68Ga-PSMA PET/CT, while mpMRI harboured a case of Gleason 4 + 3 (n = 1) and the remainder were benign (mpMRI n = 1, 68Ga-PSMA PET/CT n = 11).
Concordance group analysis
Considering index lesions (Fig. 2a), group “C” was most common, with both imaging modalities detecting 80.1% of the cancerous biopsy regions and 77.5% of RP tumour foci. mpMRI detected an additional 4.3% and 5.6% foci compared with 68Ga-PSMA PET/CT (group “M”), however 68Ga-PSMA PET/CT detected an additional 13.5% (p = 0.005) and 12.7% (p = 0.121) index foci/lesions than mpMRI (group “P” patients) according to biopsy and RP histopathology, respectively. Both imaging modalities missed 2.1% and 4.2% of index foci, which remained “invisible” (group “I”). Considering the RP cohort, SUVmax of group “I” patients were 5.2, 2.9 and 4.22 with histopathology demonstrating Gleason’s 4 + 4, 4 + 4 and 4 + 3, with tumour volumes of 1.0 cc, 3.8 cc and 0.2 cc, respectively.
Considering total lesions, or all cancer foci (Fig. 2b), group “C” was most common across each of the cohorts, identifying 64.0% of the cancerous biopsy regions in the entire cohort and 67% of RP tumour foci. mpMRI detected an additional 5.4% and 5.2% foci compared with 68Ga-PSMA PET/CT (group “M”), however 68Ga-PSMA PET/CT detected an additional 18.2% (p < 0.001) and 19.6% (p = 0.004) total foci than mpMRI (group “P” patients) according to biopsy and RP histopathology, respectively. Both imaging modalities missed 12.3% and 8.2% of total foci, which remained “invisible” (group “I”). There were no significant differences between cohorts for any of the concordance groups (“C” p = 0.388, “P” p = 0.528, “M” = 0.445 and “I” p = 0.321). Overall, 68Ga-PSMA PET/CT detected significantly more lesions than mpMRI for the entire cohort according to both biopsy (p = 0.004) and RP histopathology (p = 0.02) (p = 0.004 and p = 0.020, respectively).
PIRADS 2 subgroup analysis
Of 15 patients who had PIRADS 2 mpMRI reports but proceeded to biopsy due to ongoing suspicion of PCa, which was subsequently followed by 68Ga-PSMA PET/CT in most cases (93.3%), 13 had a positive biopsy, and 11 (73%) had csPCa (Supplementary Table 1). 68Ga-PSMA PET/CT detected 10 of 13 (76.92%) cases of PCa and 9 of 11 (81.82%) cases of csPCa in patients with PIRADS 2 MRI. Those with PIRADS 2 mpMRI and csPCa had a PSAD of 0.262 (interquartile range; IQR 0.131–0.289) versus PSAD 0.105 (interquartile range; IQR 0.094–0.126) in those without csPCa (p = 0.138). Nine of these 11 patients underwent RP, and 5 were found to have Gleason 3 + 4, 3 had Gleason 4 + 3 and 1 had Gleason 4 + 5; median tumour volume was 1.88 cc (IQR 1.01–3.23 cc).
Discussion
Our data suggests that while both 68Ga-PSMA PET/CT and mpMRI modalities have excellent rates of prostate cancer index lesion detection, 68Ga-PSMA PET/CT is superior, especially in detecting secondary cancer foci and smaller lesions, providing additional detection. This superiority was consistent across the entire biopsy cohort and RP subset. These data have significant implications for PCa detection with the increasing use of imaging prior to biopsy and may provide insight into the future of the “PSMA-era”.
While previous studies provide conflicting reports as to the superiority of 68Ga-PSMA PET/CT based on RP specimen when compared with mpMRI [17, 20, 25,27,27], the current study confirms higher sensitivity for csPCa detection than mpMRI (94.85% vs 86.03%; p = 0.022) and incremental benefit of 68Ga-PSMA PET/CT within a wider population with both RP and biopsy histopathology. Overall, in the biopsy cohort 68Ga-PSMA PET/CT led to an additional yield of 13.5% on a per patient basis and 18.2% in a per lesion basis. Thus, 68Ga-PSMA PET/CT was significantly more sensitive than mpMRI while maintaining high specificity.
Within this cohort, in which a mpMRI-based triage approach was used, an 80% index lesion detection rate was observed for the RP and biopsy cohorts, consistent with substantial local and international evidence supporting the use of mpMRI in improving efficiency of PCa detection [1, 3, 21, 28, 29]. However, concern persists over limitations in mpMRI failure to detect csPCa in 5–15% of patients [1, 21, 30], while up to a third of men will have a secondary focus of csPCa missed [31]. Indeed, a high detection rate of index lesions is favourable given the evidence that the index lesion is often rich in genomic alterations and potentially the basis for aggressive disease and metastatic spread [32].
Here, the improved sensitivity of 68Ga-PSMA PET/CT resulted in additional detection yield of 13.5% on a per patient basis and 18.2% in a per lesion basis, while 2–4% of index lesions were missed with both imaging modalities. When all lesions were considered, approximately 60% concordance was observed, while 68Ga-PSMA PET/CT and mpMRI contributed an additional approximately 20% and 5% of lesions, respectively. This is consistent with reports on simultaneous 68Ga-PSMA HBED-CC PET/MRI, where AUC values of 0.83, 0.73 and 0.88 were reported, respectively [33]. Furthermore, 68Ga-PSMA PET/CT has high sensitivity for detection of Gleason 3 + 4 and 4 + 3 disease, as seen here, as well as by the others [34].
A contributor to the improved sensitivity was detection of index lesions by 68Ga-PSMA PET/CT in 81.26% of patients with no suspicious lesion on mpMRI (PIRADS 2) but later found to have csPCa on biopsy. While this population is selected toward a diagnosis of PCa, as 68Ga-PSMA PET/CT was performed after a confirmed diagnosis in most cases, most patients (73%) demonstrated intraprostatic lesions on 68Ga-PSMA PET/CT. This group also demonstrated other concerning features that would prompt biopsy despite a negative MRI, such as PSA density. A negative 68Ga-PSMA PET/CT in men with equivocal or negative mpMRI with other concerning features prompting biopsy (e.g. elevated PSA density), 68Ga-PSMA PET/CT prior to biopsy may facilitate targeted biopsy to improve diagnostic accuracy similar to mpMRI; 68Ga-PSMA PET/CT used in this context may reduce unnecessary biopsies and their complications [35], as well as decrease the diagnosis of low grade, indolent disease. Emerging evidence suggests PSMA may be a prognostic tool, with PSMA expression on biopsy and RP immunohistochemistry reported to be predictive of disease recurrence [36], as well as lymph node avidity and PSA persistence [37] following surgery. However, prospective studies in the primary diagnostic setting of prostate cancer are required to validate the role of 68Ga-PSMA PET/CT, above the results already obtained with a MRI-triage approach.
This study has several limitations. Firstly, the order of testing may have affected our result. mpMRI was used as a triage test and for cognitive targeting of biopsies, which may have overestimated its accuracy. Patients underwent 68Ga-PSMA PET/CT after a diagnosis of prostate cancer and had the mpMRI available for review at the time. While the complete biopsy histology results were not routinely provided on the PSMA PET/CT request form, general comments about histology results on the request form may also have assisted nuclear medicine specialists in identifying lesions, overestimating its sensitivity. Conversely, the true specificity of 68Ga-PSMA PET/CT may be even higher than reported in the overall cohort because (1) the biopsies were performed with cognitive MRI targeting but not with 68Ga-PSMA PET/CT; (2) it is well established that systematic biopsies have suboptimal sensitivity in detecting all foci of csPCa and it follows that some foci detected on imaging will only be detected in the radical prostatectomy specimen. However, the differences across the entire cohort and radical prostatectomy subset were small, supporting the validity of the findings.
In conclusion, our data suggest that while both modalities have excellent rates of prostate cancer index lesion detection, 68Ga-PSMA PET/CT demonstrates superior detection of both index and secondary cancer foci compared with mpMRI, a finding that is consistent across biopsy and RP histopathology. There is increasing evidence supporting the use of 68Ga-PSMA PET/CT in primary staging following diagnosis [16] as well as re-staging in men who demonstrate biochemical recurrence. The superior diagnostic ability and multifocal tumour identification of 68Ga-PSMA PET/CT suggests a potential role in the diagnostic pathway, especially in patients with ongoing clinical concern of prostate cancer despite negative mpMRI. The promising results presented here support a PSMA-guided biopsy approach, and the results of prospective trials (ACTRN12618001640291, ClinicalTrials.gov Identifier: NCT03471650) are eagerly awaited.
References
Ahmed HU, El-Shater Bosaily A, Brown LC, Gabe R, Kaplan R, Parmar MK, et al. Diagnostic accuracy of multi-parametric MRI and TRUS biopsy in prostate cancer (PROMIS): a paired validating confirmatory study. Lancet. 2017;389(10071):815–22. https://doi.org/10.1016/s0140-6736(16)32401-1.
Kasivisvanathan V, Rannikko AS, Borghi M, Panebianco V, Mynderse LA, Vaarala MH, et al. MRI-targeted or standard biopsy for prostate-cancer diagnosis. N Engl J Med. 2018;378(19):1767–77. https://doi.org/10.1056/NEJMoa1801993.
Donato P, Morton A, Yaxley J, Teloken PE, Coughlin G, Esler R, et al. Improved detection and reduced biopsies: the effect of a multiparametric magnetic resonance imaging-based triage prostate cancer pathway in a public teaching hospital. World J Urol. 2019. https://doi.org/10.1007/s00345-019-02774-y.
Baco E, Rud E, Eri LM, Moen G, Vlatkovic L, Svindland A, et al. A randomized controlled trial to assess and compare the outcomes of two-core prostate biopsy guided by fused magnetic resonance and transrectal ultrasound images and traditional 12-core systematic biopsy. Eur Urol. 2016;69(1):149–56. https://doi.org/10.1016/j.eururo.2015.03.041.
Le JD, Tan N, Shkolyar E, Lu DY, Kwan L, Marks LS, et al. Multifocality and prostate cancer detection by multiparametric magnetic resonance imaging: correlation with whole-mount histopathology. Eur Urol. 2015;67(3):569–76. https://doi.org/10.1016/j.eururo.2014.08.079.
Moore CM, Robertson NL, Arsanious N, Middleton T, Villers A, Klotz L, et al. Image-guided prostate biopsy using magnetic resonance imaging-derived targets: a systematic review. Eur Urol. 2013;63(1):125–40. https://doi.org/10.1016/j.eururo.2012.06.004.
Radtke JP, Schwab C, Wolf MB, Freitag MT, Alt CD, Kesch C, et al. Multiparametric magnetic resonance imaging (MRI) and MRI–transrectal ultrasound fusion biopsy for index tumor detection: correlation with radical prostatectomy specimen. Eur Urol. 2016;70(5):846–53. https://doi.org/10.1016/j.eururo.2015.12.052.
Borofsky S, George AK, Gaur S, Bernardo M, Greer MD, Mertan FV, et al. What are we missing? False-negative cancers at multiparametric MR imaging of the prostate. Radiology. 2018;286(1):186–95. https://doi.org/10.1148/radiol.2017152877.
Dianat SS, Carter HB, Macura KJ. Performance of multiparametric magnetic resonance imaging in the evaluation and management of clinically low-risk prostate cancer. Urol Oncol: Seminars Orig Investig. 2014;32(1):39.e1–e10. https://doi.org/10.1016/j.urolonc.2013.04.002.
Priester A, Natarajan S, Khoshnoodi P, Margolis DJ, Raman SS, Reiter RE, et al. Magnetic resonance imaging underestimation of prostate cancer geometry: use of patient specific molds to correlate images with whole mount pathology. J Urol. 2017;197(2):320–6. https://doi.org/10.1016/j.juro.2016.07.084.
Perera M, Papa N, Roberts M, Williams M, Udovicich C, Vela I, et al. Gallium-68 prostate-specific membrane antigen positron emission tomography in advanced prostate cancer-updated diagnostic utility, sensitivity, specificity, and distribution of prostate-specific membrane antigen-avid lesions: a systematic review and meta-analysis. Eur Urol. 2019. https://doi.org/10.1016/j.eururo.2019.01.049.
Afshar-Oromieh A, Haberkorn U, Schlemmer HP, Fenchel M, Eder M, Eisenhut M, et al. Comparison of PET/CT and PET/MRI hybrid systems using a 68Ga-labelled PSMA ligand for the diagnosis of recurrent prostate cancer: initial experience. Eur J Nucl Med Mol Imaging. 2014;41(5):887–97. https://doi.org/10.1007/s00259-013-2660-z.
Meredith G, Wong D, Yaxley J, Coughlin G, Thompson L, Kua B, et al. The use of 68 Ga-PSMA PET CT in men with biochemical recurrence after definitive treatment of acinar prostate cancer. BJU Int. 2016;118(S3):49–55. https://doi.org/10.1111/bju.13616.
Rauscher I, Duwel C, Haller B, Rischpler C, Heck MM, Gschwend J, et al. Efficacy, predictive factors, and prediction nomograms for Ga-68-labeled prostate-specific membrane antigen-ligand positron-emission tomography/computed tomography in early biochemical recurrent prostate cancer after radical prostatectomy. Eur Urol. 2018;73(5):656–61. https://doi.org/10.1016/j.eururo.2018.01.006.
Yaxley JW, Raveenthiran S, Nouhaud F-X, Samaratunga H, Yaxley WJ, Coughlin G, et al. Risk of metastatic disease on 68gallium-prostate-specific membrane antigen positron emission tomography/computed tomography scan for primary staging of 1253 men at the diagnosis of prostate cancer. BJU Int. https://doi.org/10.1111/bju.14828.
Yaxley JW, Raveenthiran S, Nouhaud FX, Samartunga H, Yaxley AJ, Coughlin G, et al. Outcomes of primary lymph node staging of intermediate and high risk prostate cancer with (68)Ga-PSMA positron emission tomography/computerized tomography compared to histological correlation of pelvic lymph node pathology. J Urol. 2019;201(4):815–20. https://doi.org/10.1097/ju.0000000000000053.
Eiber M, Weirich G, Holzapfel K, Souvatzoglou M, Haller B, Rauscher I, et al. Simultaneous (68)Ga-PSMA HBED-CC PET/MRI improves the localization of primary prostate cancer. Eur Urol. 2016;70(5):829–36. https://doi.org/10.1016/j.eururo.2015.12.053.
Rahbar K, Weckesser M, Huss S, Semjonow A, Breyholz HJ, Schrader AJ, et al. Correlation of intraprostatic tumor extent with (6)(8)Ga-PSMA distribution in patients with prostate cancer. J Nucl Med. 2016;57(4):563–7. https://doi.org/10.2967/jnumed.115.169243.
Woythal N, Arsenic R, Kempkensteffen C, Miller K, Janssen J-C, Huang K, et al. Immunohistochemical validation of PSMA expression measured by 68Ga-PSMA PET/CT in primary prostate cancer. J Nuclear Med. 2018;59(2):238. https://doi.org/10.2967/jnumed.117.195172.
Donato P, Roberts MJ, Morton A, Kyle S, Coughlin G, Esler R, et al. Improved specificity with 68 Ga PSMA PET/CT to detect clinically significant lesions “invisible” on multiparametric MRI of the prostate: a single institution comparative analysis with radical prostatectomy histology. Eur J Nucl Med Mol Imaging. 2018:<xocs:firstpage xmlns:xocs=""/>. https://doi.org/10.1007/s00259-018-4160-7.
Pokorny MR, de Rooij M, Duncan E, Schroder FH, Parkinson R, Barentsz JO, et al. Prospective study of diagnostic accuracy comparing prostate cancer detection by transrectal ultrasound-guided biopsy versus magnetic resonance (MR) imaging with subsequent MR-guided biopsy in men without previous prostate biopsies. Eur Urol. 2014;66(1):22–9. https://doi.org/10.1016/j.eururo.2014.03.002.
Epstein JI, Egevad L, Amin MB, Delahunt B, Srigley JR, Humphrey PA. The 2014 International Society of Urological Pathology (ISUP) consensus conference on Gleason grading of prostatic carcinoma: definition of grading patterns and proposal for a new grading system. Am J Surg Pathol. 2016;40(2):244–52. https://doi.org/10.1097/pas.0000000000000530.
Futterer JJ, Briganti A, De Visschere P, Emberton M, Giannarini G, Kirkham A, et al. Can clinically significant prostate cancer be detected with multiparametric magnetic resonance imaging? A systematic review of the literature. Eur Urol. 2015;68(6):1045–53. https://doi.org/10.1016/j.eururo.2015.01.013.
van der Kwast TH, Amin MB, Billis A, Epstein JI, Griffiths D, Humphrey PA, et al. International Society of Urological Pathology (ISUP) consensus conference on handling and staging of radical prostatectomy specimens. Working group 2: T2 substaging and prostate cancer volume. Mod Pathol. 2010;24:16. https://doi.org/10.1038/modpathol.2010.156.
Chen M, Zhang Q, Zhang C, Zhao X, Marra G, Gao J, et al. Combination of 68Ga-PSMA PET/CT and multiparameter MRI improves the detection of clinically significant prostate cancer: a lesion by lesion analysis. J Nucl Med. 2018. https://doi.org/10.2967/jnumed.118.221010.
Rhee H, Thomas P, Shepherd B, Gustafson S, Vela I, Russell PJ, et al. Prostate specific membrane antigen positron emission tomography may improve the diagnostic accuracy of multiparametric magnetic resonance imaging in localized prostate cancer. J Urol. 2016;196(4):1261–7. https://doi.org/10.1016/j.juro.2016.02.3000.
Kalapara AA, Nzenza T, Pan HY, Ballok Z, Ramdave S, O'Sullivan R, et al. Detection and localisation of primary prostate cancer using 68Ga-PSMA PET/CT compared with mpMRI and radical prostatectomy specimens. BJU Int. https://doi.org/10.1111/bju.14858.
Kasivisvanathan V, Dufour R, Moore CM, Ahmed HU, Abd-Alazeez M, Charman SC, et al. Transperineal magnetic resonance image targeted prostate biopsy versus transperineal template prostate biopsy in the detection of clinically significant prostate cancer. J Urol. 2013;189(3):860–6. https://doi.org/10.1016/j.juro.2012.10.009.
Kasivisvanathan V, Stabile A, Neves JB, Giganti F, Valerio M, Shanmugabavan Y, et al. Magnetic resonance imaging-targeted biopsy versus systematic biopsy in the detection of prostate cancer: a systematic review and meta-analysis. Eur Urol. 2019. https://doi.org/10.1016/j.eururo.2019.04.043.
Coker MA, Glaser ZA, Gordetsky JB, Thomas JV, Rais-Bahrami S. Targets missed: predictors of MRI-targeted biopsy failing to accurately localize prostate cancer found on systematic biopsy. Prostate Cancer Prostatic Dis. 2018. https://doi.org/10.1038/s41391-018-0062-9.
Johnson DC, Raman SS, Mirak SA, Kwan L, Bajgiran AM, Hsu W, et al. Detection of individual prostate cancer foci via multiparametric magnetic resonance imaging. Eur Urol. 2019;75(5):712–20. https://doi.org/10.1016/j.eururo.2018.11.031.
Houlahan KE, Salmasi A, Sadun TY, Pooli A, Felker ER, Livingstone J, et al. Molecular hallmarks of multiparametric magnetic resonance imaging visibility in prostate cancer. Eur Urol. 2019;76(1):18–23. https://doi.org/10.1016/j.eururo.2018.12.036.
Eiber M, Weirich G, Holzapfel K, Souvatzoglou M, Haller B, Rauscher I, et al. Simultaneous 68Ga-PSMA HBED-CC PET/MRI improves the localization of primary prostate cancer. Eur Urol. 2016;70(5):829–36. https://doi.org/10.1016/j.eururo.2015.12.053.
Scheltema MJ, Chang JI, Stricker PD, van Leeuwen PJ, Nguyen QA, Ho B, et al. Diagnostic accuracy of (68) Ga-prostate-specific membrane antigen (PSMA) positron-emission tomography (PET) and multiparametric (mp) MRI to detect intermediate-grade intra-prostatic prostate cancer using whole-mount pathology: impact of the addition of (68) Ga-PSMA PET to mpMRI. BJU Int. 2019. https://doi.org/10.1111/bju.14794.
Roberts MJ, Bennett HY, Harris PN, Holmes M, Grummet J, Naber K, et al. Prostate biopsy-related infection: a systematic review of risk factors, prevention strategies, and management approaches. Urology. 2017;104:11–21. https://doi.org/10.1016/j.urology.2016.12.011.
Hupe MC, Philippi C, Roth D, Kümpers C, Ribbat-Idel J, Becker F, et al. Expression of prostate-specific membrane antigen (PSMA) on biopsies is an independent risk stratifier of prostate cancer patients at time of initial diagnosis. Front Oncol. 2018;8:623. https://doi.org/10.3389/fonc.2018.00623.
van Leeuwen PJ, Donswijk M, Nandurkar R, Stricker P, Ho B, Heijmink S, et al. Gallium-68-prostate-specific membrane antigen ((68) Ga-PSMA) positron emission tomography (PET)/computed tomography (CT) predicts complete biochemical response from radical prostatectomy and lymph node dissection in intermediate- and high-risk prostate cancer. BJU Int. 2018. https://doi.org/10.1111/bju.14506.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
The study was approved by the Ethics Committee of the Royal Brisbane and Women’s Hospital (RBWH) Human Research Ethics Committee (approval number HREC/17/QRBW/644).
Informed consent
Informed consent was not deemed necessary by the Ethics Committee of the Royal Brisbane and Women’s Hospital (RBWH) Human Research Ethics Committee.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Oncology – Genitourinary
Electronic supplementary material
ESM 1
(DOCX 14 kb)
Rights and permissions
About this article
Cite this article
Donato, P., Morton, A., Yaxley, J. et al. 68Ga-PSMA PET/CT better characterises localised prostate cancer after MRI and transperineal prostate biopsy: Is 68Ga-PSMA PET/CT guided biopsy the future?. Eur J Nucl Med Mol Imaging 47, 1843–1851 (2020). https://doi.org/10.1007/s00259-019-04620-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-019-04620-0