Abstract
Purpose
The purpose of this study was to compare the prognostic value of 11C-methionine (MET) and 18F-fluorodeoxyglucose (FDG) positron emission tomography (PET) in glioma patients.
Methods
The study population comprised 47 patients with gliomas (19 glioblastoma, 28 others). Pretreatment magnetic resonance imaging, MET PET and FDG PET were performed within a time interval of 2 weeks in all patients. The uptake ratio and standard uptake values were calculated. Univariate and multivariate analyses were done to determine significant prognostic factors. Ki-67 index was measured by immunohistochemical staining, and compared with FDG and MET uptake in glioma.
Results
Among the several clinicopathological prognostic factors, tumour pathology (glioblastoma or not), age (≥60 or <60 years), Karnofsky performance status (KPS) (≥70 or <70) and MET PET (higher uptake or not compared with normal cortex) were found to be significant predictors by univariate analysis. In multivariate analysis, tumour pathology, KPS and MET PET were identified as significant independent predictors. The Ki-67 proliferation index was significantly correlated with MET uptake (r=0.64), but not with FDG uptake.
Conclusion
Compared with FDG PET in glioma, MET PET was an independent significant prognostic factor and MET uptake was correlated with cellular proliferation. MET PET may be a useful biological prognostic marker in glioma patients.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Glioma is the most common primary neoplasm of the brain, and it is highly dependent on various clinical or biological prognostic factors [1–3]. The outcome of patients with glioma is quite variable, and there is no consensus regarding the best treatment. In some patients, the risk of treatment toxicity outweighs the potential benefits of early treatment. Other patients with progressive disease may benefit from an aggressive therapy modality [4, 5]. In addition, a significant number of low-grade gliomas undergo transformation to a high grade [6].
Currently, it is difficult to select patients with an unfavourable prognosis using established prognostic factors. Age, performance status, focal neurological deficits, mental changes, seizure, symptoms of expansion and contrast enhancement on CT have all been reported to be possible prognostic factors in cerebral gliomas in addition to pathological grade. However, some of these factors are still debated [7]. Clinicians need reliable new predictors to help decide upon initial therapy, evaluate patients after treatment and make decisions regarding second-line therapies.
Some clinicians have applied 18F-fluorodeoxyglucose (FDG) positron emission tomography (PET), which is frequently used for brain tumours, in the hope that the glucose metabolic status of the tumour might provide new prognostic information, and some investigators have suggested FDG PET as a metabolic prognostic marker for brain tumour patients [6–8]. However, the patient populations in these studies were too small to allow multivariate analysis, and some investigators have debated the prognostic value of FDG PET [9]. In addition, FDG uptake is known to be non-specific: high FDG accumulation has been observed in inflammatory cells and granulation tissue as well as in viable cancer cells [10, 11]. Therefore, other more specific metabolic tracers, such as positron emitter-labelled amino acids, have been proposed as new predictors.
Methionine (MET) is a natural essential amino acid and enters tumour cells via the l-amino acid transporter to meet the demands of accelerated protein and RNA synthesis in malignant tumours [12]. 11C-MET has been used in the diagnosis and follow-up of glioma patients [13–15]. Some investigators have found 11C-MET PET to be a prognostic marker for glioma patients [13, 16]. However, to our knowledge, no study has been published regarding FDG and MET PET and multivariate survival analysis for glioma patients. In this study, we applied both tracers, MET and FDG, to the same glioma patients and performed multivariate analysis to evaluate their prognostic value. This is the first study to compare the prognostic significance of FDG and MET in the same patients with glioma.
Materials and methods
Study population
A retrospective review of medical records was performed on all patients who had undergone both MET PET and FDG PET at Seoul National University Hospital from 1998 to 2002. Forty-seven patients with histologically verified glioma were included in this study. Pretreatment magnetic resonance imaging (MRI), MET PET and FDG PET were performed within 2 weeks in all patients.
A summary of the patients’ characteristics is shown in Table 1. There were 27 males (57%) and 20 females (43%) with a median age of 46.5 years (SD 14 years). The older age group (≥60 years) comprised 26 patients and the younger age group (<60 years), 21 patients. Pathological diagnosis was obtained by biopsy or open surgery in all patients. There were 19 glioblastomas, 10 grade II gliomas (astrocytoma and oligodendroglioma) and 18 grade III anaplastic gliomas (anaplastic astrocytoma, anaplastic oligodendroglioma, anaplastic oligoastrocytoma, gliomatosis cerebris). The median Karnofsky performance status (KPS) for these patients was 70 (range 50–100). Fifteen patients died during follow-up. The median survival time after pretreatment PET for all glioma patients was 18 months (range 4–99 months).
Synthesis of 18F-FDG and 11C-MET
18F-FDG was synthesized by the fluorination of 1,3,4,6-tetra-O-acetyl-2-trifluoromethanesulfonyl-mannose with the produced 18F in the presence of phase-transfer catalyst, and subsequently hydrolysed with 1 N HCl. The synthesized 18F-FDG was neutralized with 1 N NaOH and filtered through 0.22-μm filters. The radiochemical purity of the prepared 18F-FDG was confirmed to be greater than 95% by thin-layer chromatography (TLC)/95% acetonitrile (Rf=0.5).
11C-MET was synthesized by using a minor modification of a reported method [17]. 11C-CO2 was produced by irradiating N2 gas with 13-MeV protons accelerated in a cyclotron (Ebco Technologies, Canada). 11C-CO2 was reduced to 11C-methanol using lithium aluminium hydride and subsequently converted to 11C-methyl iodide by adding hydrogen iodide. 11C-methyl iodide was distilled into a vial containing l-homocysteine thiolactone solution in water/acetone. 11C-MET was synthesized by the S-methylation of l-homocysteine in the presence of potassium hydroxide as a base. After neutralizing with 1 N HCl, 11C-MET was filtered through 0.22-μm filters. The radiochemical purity of the prepared 11C-MET was confirmed to be higher than 95% by TLC/80% acetonitrile (Rf=0.2).
PET methods
PET scanning was performed using an ECAT EXACT 47 scanner (Siemens-CTI, Knoxville, TN, USA). Patients were fasted for at least 6 h before PET scanning, and images were acquired with patients in the supine position, resting, with their eyes closed. Using a 68Ge source, transmission images of the brain were obtained for 7 min to correct for photon attenuation. A dose of 370–555 MBq of 18F-FDG or 555–740 MBq of 11C-MET was injected intravenously. Regional emission images of the brain were obtained for 20 min, beginning 40 min after the 18F-FDG injection and 10 min after the 11C-MET injection.
Data analysis
The pretreatment MR images were reviewed and classified as showing enhancement of tumour mass or not. Two experienced nuclear physicians visually interpreted the PET images and reached a consensus. During PET examination, brain MR images were available for all cases. Visually, on both 11C-MET and 18F-FDG PET, lesions were classified as hypermetabolic or iso-/hypometabolic based on comparison of lesion tracer uptake with tracer uptake by the grey matter contralateral to the lesion without any morphological abnormalities on MRI. Lesions classified as hypermetabolic showed definitely higher uptake than normal cortex, with delineation of the margin of the mass, while those classified as iso-/hypometabolic showed equal or lower uptake.
The MET and FDG uptake ratios were calculated by dividing the maximum standard uptake values (SUV) for the tumour by the SUV of the contralateral normal grey matter. A region of interest (ROI) was drawn manually by an observer around the hottest area of each lesion or its centre located by MRI, if increased MET and FDG uptake was absent. A mirror region of the same size was drawn for reference purposes in the contralateral tissue.
Proliferative activity
Proliferative activity was measured by obtaining the Ki-67 proliferation index by histochemical staining of the pathological specimens. Five-micrometre tissue sections were obtained from paraffin-embedded tissues. After deparaffinization, the sections were incubated with anti-Ki-67 nuclear antigen antibody from ZYMED Laboratories (San Francisco, CA, USA) for 1 h at room temperature. Sections were stained for Ki-67 nuclear antigen using an avidin/biotin conjugate immunoperoxidase kit (Vector Laboratories, Burlingame, CA, USA). Sections were then incubated with the second antibody for 30 min at 25°C, followed by incubation with streptavidin peroxidase, and washed with phosphate-buffered saline. 3,3-Diaminobenzidine tetrahydrochloride was used as a substitute substrate-chromogen solution, and sections were counterstained with Meyer’s haematoxylin. For negative controls, tissue sections were incubated with mouse IgG instead of anti-Ki-67 nuclear antigen antibody. After cell staining, fields were selected randomly for cell counting. A minimum of 1,000 cells were counted per tissue section. The Ki-67 score (%) was defined as the percentage of cells which stained positively for Ki-67 nuclear antigen.
Statistical analysis
The SPSS statistical package (Release 11.0.1, SPSS Inc., Chicago, IL, USA) and Medcalc software (version 6.0) were used to evaluate and present data. Survival time was defined as the time between the pretreatment PET study and death. For univariate analysis, survival curves were constructed using the Kaplan-Meier method. The prognostic importance of the clinicopathological variables, such as age, gender, KPS score, MRI enhancement, tumour pathology (glioblastoma or not), FDG uptake and MET uptake, was analysed using the log-rank probability test.
For multivariate analysis, the Cox proportional hazards model was used. Natural logarithm cumulative hazard plots were performed to assess proportional hazard function. Crude and adjusted rate ratios and their 95% confidence intervals (CIs) were calculated. Variables associated with a higher rate ratio by multivariate analysis were given a greater relative score. Linear regression analysis, including the calculation of Spearman’s rho, was performed to determine whether MET or FDG uptake was related to the proliferation index. A p value of <0.05 was considered statistically significant.
Results
MET PET showed hypermetabolism in 29 of the 47 gliomas (62%) and iso-/hypometabolism in the remaining 18 (38%). FDG PET clearly showed hypermetabolism in 20 cases (43%) and iso-/hypometabolism in 27 (57%). Mean uptake ratios and maximum SUVs in the lesions judged to be hypermetabolic or iso-/hypometabolic by visual analysis are shown in Table 2. Overall, a significant correlation was not found between MET uptake and FDG uptake.
The survival data are presented in Tables 3 and 4. Among the various prognostic factors, age, KPS score, tumour pathology and MET uptake showed significant differences, but gender, MRI enhancement and FDG uptake showed no significant differences.
The Kaplan-Meier plot showed shorter survival in patients with high-grade (III, IV) glioma than in those with low-grade (II) glioma (p<0.001, log-rank). Older patients (age ≥60 years) showed shorter survival than younger patients (age <60 years) (p=0.037, log-rank), and survival was also shorter in patients with a lower KPS score (<70) than in those with a higher KPS score (p=0.0285, log-rank) [(Fig. 1)]. The Kaplan-Meier plot of MET PET showed shorter survival in patients with hypermetabolism than in patients with iso-/hypometabolism (p=0.0416, log-rank); however, FDG PET results did not appear to be significantly related to survival (p>0.05, log-rank) [(Fig. 2)].
Kaplan-Meier survival curves for PET. a Graph showing survival probability of glioma patients with hypermetabolism versus those with iso-/hypometabolism on MET PET (p=0.0416). b Graph showing survival probability of glioma patients with hypermetabolism versus those with iso-/hypometabolism on FDG PET (p=0.6739)
By multivariate Cox analysis, WHO grade, age, KPS score and MET uptake were shown to be significant independent predictors of a shortened survival. The hazard ratio (95% CI) was 7.545 (1.283–44.358) in grade III (p=0.025) and 11.105 (1.905–64.752) in grade IV glioma (p=0.007). The hazard ratio (95% CI) for 11C-MET uptake was 3.682 (1.404–9.654) [(p=0.038)]. The hazard ratios (95% CI) for age and KPS score were 3.000 (1.100–8.180) [(p=0.027)] and 7.048 (2.183–22.751) [(p=0.001)]. However, FDG uptake, MRI enhancement and gender did not affect survival in this study (Fig. 3, Table 4).
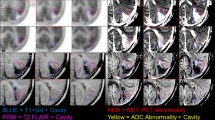
MRI, MET PET and FDG PET images of patients with glioma. a MET PET shows hypermetabolism (uptake ratio =4.98), but FDG PET shows iso-/hypometabolism (uptake ratio =0.94). Pathology was glioblastoma, and the Ki-67 index of this tumour was 40%. The patient died 14 months later, a short survival time. b Sixty-one-year-old man with a history of low-grade glioma treated by local resection and chemotherapy. MRI demonstrates residual non-enhancing tumour in the right insular region. FDG PET demonstrates hypometabolism (uptake ratio=0.58) similar to that of white matter, consistent with low-grade glioma. MET PET demonstrates a focal area of hypermetabolism (uptake ratio =3.97). This area was confirmed as anaplastic transformation with a relatively higher proliferative index (Ki-67 index=20%) at stereotactic biopsy
The mean uptake ratio on MET PET was higher in high-grade (III, IV) than in low-grade (II) gliomas (1.27±0.40, range 1.01–1.527), and among the high-grade gliomas a significant difference was seen between grade III (1.67±0.64, range 1.35–1.99) and grade IV (2.16±0.90, range 1.69–2.62). However, FDG uptake did not show any difference between low-grade (II: 1.03±0.63, range 0.63–1.43) and high-grade gliomas (III: 1.19–0.43, range 0.97–1.41; IV: 1.06±0.41, range 0.86–1.28) [(Fig. 4)]. Finally, the Ki-67 proliferation index was significantly correlated with MET uptake (r=0.64, p<0.001) but not with FDG uptake (r=0.23) [(Fig. 5)].
Discussion
The most important prognostic factor in malignant gliomas is the histopathology of the tumour. The survival of patients with anaplastic astrocytoma is much longer than that of patients with glioblastoma. The median survival of the former has been improved to almost 4 years by the recent progress in multidisciplinary treatment, whereas that of the latter has remained less than 1.5 years [18]. Other important factors that have been proven to be associated with survival are age, KPS on admission, surgery, radiotherapy and chemotherapy. There is substantial evidence suggesting that patients with a better KPS on admission live longer after treatment [19]. Gross total resection of supratentorial anaplastic astrocytoma is directly associated with longer and better survival compared with subtotal or partial resection [19]. For glioblastoma, however, gross total resection has not been proved to have a significant survival advantage over subtotal or partial removal. Radiotherapy has been proved to offer significant survival advantages over subtotal or partial tumour resection in patients with supratentorial anaplastic astrocytoma and glioblastoma [20]. Chemotherapy has not proved effective in prolonging survival of patients with glioblastoma. However, in patients with anaplastic astrocytoma, multidrug chemotherapy with CCNU, procarbazine and vincristine has been proved to offer a significant survival advantage over BCNU alone, suggesting that chemotherapy is also a prognostic factor in these patients [21].
18F-FDG PET has been used to investigate the malignancy, extent of spread, effectiveness of therapy and prognosis of brain tumours. MET PET can provide additional information when used in combination with FDG PET. The major clinical application of MET PET may be for the detection and differentiation of brain lesions in which FDG uptake is absent or lower than normal brain uptake [15]. Some investigators have found that MET is superior to FDG for delineating tumour margins and for differential diagnosis of recurrence from radiation necrosis [13, 14, 16]. In this study, we compared the prognostic value of FDG PET and MET PET in the same patients with glioma.
Tumour uptake of MET was first described more than 20 years ago, but its mechanism and biological significance in gliomas is not yet completely understood. At the blood–brain barrier, the sodium-independent L-transporter system, in the luminal membrane of endothelial cells, is the main mode of methionine transport into brain tissue [22]. MET uptake does not directly reflect protein synthesis, but it represents cell avidity for amino acids, which is related to the activity of the transport system and protein incorporation [12]. Because this transport is believed to be dependent on the proliferative activity of cells [23], it is appropriate to address the prognostic utility of MET PET.
We found that, among several clinical and metabolic factors, age and MET uptake as well as tumour pathology had prognostic value in glioma. Clearly higher MET uptake was a significant factor by multivariate and univariate analyses, and it was found to be an independent prognostic factor for survival, with a hazard ratio of 2.02 in glioma. Previous reports support these results. De Witte et al. [16] found that high MET uptake is statistically associated with poor survival in patients with WHO grade II and III gliomas, and Derlon et al. [14] reported that MET uptake is significantly correlated with histological grade in grade 2/3 and 2/4 glioma. In this study, MET uptake ratio and SUV were higher in glioblastomas than in other grade II or anaplastic gliomas. When visual analysis is combined with uptake ratio or SUV, MET PET may be a good prognostic indicator in glioma.
Some authors claim that FDG uptake is a prognostic factor in patients with glioma [8, 9, 23], although the patient populations in these studies were too small to allow multivariate analysis. Other reports have suggested a prognostic value for FDG PET in non-small cell lung cancer, breast cancer and head and neck cancer [24–26]. In contrast to these findings, we found that univariate and multivariate analyses revealed MET uptake, rather than FDG uptake, to be a significant prognostic factor (Tables 3, 4). Kaschten et al. [13] also reported that MET PET was superior to FDG PET for predicting prognosis of glioma. In addition, Kubota et al. reported findings that support our own results. They observed high FDG uptake in activated macrophages and young granulation tissue as well as in cancer cells [11]. Compared with FDG, MET distribution in tumour tissue was found to be more specific for viable cancer cells.
Our study also showed that MET uptake correlated significantly with the proliferative Ki-67 index. Previously, Miyazawa et al. [27] found that the MET uptake rate in non-small cell lung cancer correlated well with proliferation indices measured by DNA flow cytometry. In glioma, Sato et al. [28] evaluated the relationship between MET PET and proliferating nuclear cell antigen (PCNA) indexes. They found a strong positive correlation (r=0.677, p<0.005) between MET uptake and PCNA indexes, and mentioned that MET PET is a potentially useful preoperative method to discriminate the malignancy of glioma. In the present study, FDG uptake, unlike MET uptake, failed to show a relationship with the Ki-67 index when assessed both by visual analysis and by uptake ratio, . Kubota et al. also found that MET uptake correlated more directly with proliferation than did FDG uptake [29]. Cancer cells showed the highest MET uptake in the early exponential growth phase, with low uptake in the plateau phase. We believe that these findings adequately support the concept that MET PET has a better prognostic value than FDG PET.
In conclusion, in contrast to FDG PET, MET PET was found to be an independently significant prognostic factor, and MET uptake was correlated with cellular proliferation in glioma. Therefore, MET PET may be used as a biological prognostic marker in combination with established clinical prognostic factors as a guide to optimal treatment in glioma.
References
Winger MJ, Madonald DR, Cairncross JG. Supratentorial anaplastic glioma in adults. J Neurosurg 1989;71:487–93.
Lacroix M, Abi-Said D, Fourney DR, Gokaslan ZL, Shi W, DeMonte F, et al. A multivariate analysis of 416 patients with glioblastoma multiforme: prognosis, extent of resection, and survival. J Neurosurg 2001;95:190–8.
Russell D, Rubinstein L. Tumours of central neuroepithelial origin. In: Rubinstein LJ, editor. Pathology of tumours of the central nervous system. Baltimore: Williams & Wilkins; 1989. p. 83–350.
Bampoe JO, Bauman G, Cairncross JG. Adult low-grade glioma: natural history, prognostic factors, and timing of treatment. In: Rock JP, Rosenblum ML, Shaw EG, Cairncross JG, editors. The practical management of low-grade primary brain tumors. Philadelphia: Williams & Wilkins; 1999. p. 135–48.
Olson JD, Riedel E, DeAngelis LM. Long-term outcome of low-grade oligodendroglioma and mixed glioma. Neurology 2000;54:1442–8.
Piepmeier J, Christopher S, Spencer D, et al. Variations in the natural history and survival of patients with supratentorial low-grade astrocytoma. Neurosurgery 1996;38:872–9.
Lote K, Egeland T, Hager B, et al. Survival, prognostic factors, and therapeutic efficacy in low-grade glioma: a retrospective study in 379 patients. J Clin Oncol 1997;15:3129–40.
Alavi JB, Alavi A, Chawluk J, et al. Positron emission tomography in patients with glioma. A predictor of prognosis. Cancer 1988;62:1074–8.
De Witte O, Lefranc F, Levivier M, Salmon I, Brotchi J, Goldman S. FDG-PET as a prognostic factor in high-grade astrocytoma. J Neurooncol 2000;49:157–63.
Strauss LG, Conti PS. The applications of PET in clinical oncology. J Nucl Med 1991;32:623–48.
Kubota R, Yamada S, Kubota K, Ishiwata K, Tamahashi N, Ido T. Intratumoral distribution of fluorodeoxyglucose in vivo: high accumulation in macrophages and granulation tissues studied by microautoradiography. J Nucl Med 1992;33:1972–80.
Schober O, Duden C, Meyer GJ, Muller JA, Hundeshagen H. Non-selective transport of [11C-methyl]-l-and d-methionine into a malignant glioma. Eur J Nucl Med 1987;13:103–5.
Kaschten B, Stevenaert A, Sadzot B, et al. Preoperative evaluation of 54 gliomas by PET with fluorine-18-fluorodeoxyglucose and/or carbon-11-methionine. J Nucl Med 1998;39:778–85.
Derlon JM, Bourdet C, Bustany P, Chatel M, Theron J, Darcel F, Syrota A. [11C]l-methionine uptake in gliomas. Neurosurgery 1989;25:720–8.
Chung JK, Kim YK, Kim SK, et al. Usefulness of 11C-methionine PET in the evaluation of brain lesions that are hypo- or isometabolic on 18F-FDG PET. Eur J Nucl Med Mol Imaging 2002;29:176–82.
De Witte O, Goldberg I, Wilker D, et al. Positron emission tomography with injection of methionine as a prognostic factor in glioma. J Neurosurg 2001;95:746–50.
Ishiwata K, Ido T, Vaalburg W. Increased amounts of d-enantiomer dependent on alkaline concentration in the synthesis of l-[methyl-11C]methionine. Appl Radiat Isot 1988;39:310–4.
Vecht C, Acesaat CJJ, Van Putten WLJ, et al. The influence of the extent of surgery on the neurological function and survival in malignant glioma. A retrospective analysis in 243 patients. J Neurol Neurosurg Psychiatry 1990;53:466–71.
Albert FK, Forsting M, Sator K, et al. Early postoperative magnetic resonance imaging after resection of malignant glioma: objective evaluation of residual tumor and its influence on regrowth prognosis. Neurosurgery 1994;34(6):45–61.
Walker MD, Green SB, Byar DP, et al. Randomized comparison of radiotherapy and nitrosureas for the treatment of malignant glioma after surgery. N Engl J Med 1980;303(7):1323–9.
Levin VA, Wara WM, Davis RL, et al. Phase III comparison of BCNU and the combination of procarbazine, CCNU, and vincristine administered after radiotherapy with hydroxyurea for malignant gliomas. J Neurosurg 1985;63:218–23.
Langen KJ, Muhlensiepen H, Holschbach M, Hautzel H, Jansen P, Coenen HH. Transport mechanisms of 3-[123I]iodo-alpha-methyl-l-tyrosine in a human glioma cell line: comparison with [3H]methyl-l-methionine. J Nucl Med 2000;41:1250–5.
Delbeke D, Meyerowitz C, Lapidus RL, et al. Optimal cutoff levels of F-18 fluorodeoxyglucose uptake in the differentiation of low-grade from high-grade brain tumors with PET. Radiology 1995;195:47–52.
Jeong HJ, Min JJ, Park JM, et al. Determination of the prognostic value of [18F]fluorodeoxyglucose uptake by using positron emission tomography in patients with non-small cell lung cancer. Nucl Med Commun 2002;23:865–70.
Oshida M, Uno K, Suzuki M, et al. Predicting the prognosis of breast carcinoma patients with positron emission tomography using 2-deoxy-2-fluoro[18F]-d-glucose. Cancer 1998;82:2227–34.
Minn H, Lapela M, Klemi PJ, et al. Prediction of survival with fluorine-18-fluorodeoxyglucose and PET in head and neck cancer. J Nucl Med 1997;38:1907–11.
Miyazawa H, Arai T, Ito M, Hara T. PET imaging of non-small-cell lung carcinoma with carbon-11-methionine: relationship between radioactivity uptake and flow-cytometric parameters. J Nucl Med 1993;34:1886–91.
Sato N, Suzuki M, Kuwata N, Kuroda K, Wada T, Beppu T, et al. Evaluation of the malignancy of glioma using 11C-methionine positron emission tomography and proliferating cell nuclear antigen staining. Neurosurg Rev 1999;22(4):210–4.
Kubota K. From tumor biology to clinical PET: a review of positron emission tomography (PET) in oncology. Ann Nucl Med 2001;15:471–86.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kim, S., Chung, JK., Im, SH. et al. 11C-methionine PET as a prognostic marker in patients with glioma: comparison with 18F-FDG PET. Eur J Nucl Med Mol Imaging 32, 52–59 (2005). https://doi.org/10.1007/s00259-004-1598-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-004-1598-6