Abstract.
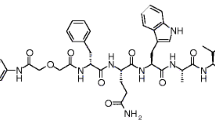
Demobesin 1 is a potent new GRP-R-selective bombesin (BN) analogue containing an open chain tetraamine chelator for stable technetium-99m binding. Following a convenient labelling protocol, the radiopeptide, [99mTc]Demobesin 1, formed in nearly quantitative yields and with high specific activities. Both unlabelled and labelled peptide demonstrated high-affinity binding in membrane preparations of the human androgen-independent prostate adenocarcinoma PC-3 cell line. The IC50 values determined for Demobesin 1 and [Tyr4]BN were 0.70±0.08 nM and 1.5±0.20 nM, respectively, while the K d defined for [99mTc/99gTc]Demobesin 1 was 0.67±0.10 nM. [99mTc]Demobesin 1 was rather stable in murine plasma, whereas it degraded rapidly in kidney and liver homogenates. After injection in healthy Swiss albino mice, [99mTc]Demobesin 1 accumulated very efficiently in the target organs (pancreas, intestinal tract) via a GRP-R-mediated process, as shown by in vivo receptor blocking experiments. An equally high and GRP-R-mediated uptake was exhibited by [99mTc]Demobesin 1 after injection in PC-3 tumour-bearing athymic mice. The initial high radioligand uptake of 16.2±3.1%ID/g in the PC-3 xenografts at 1 h p.i. remained at a similar level (15.61±1.19%ID/g) at 4 h p.i. Even after 24 h p.i., when the radioactivity had cleared from all other tissues, a value of 5.24±0.67%ID/g was still observed in the tumour. The high and prolonged localization of [99mTc]Demobesin 1 at the tumour site and its rapid background clearance are very promising qualities for GRP-R-targeted tumour imaging in man.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Author information
Authors and Affiliations
Additional information
Electronic Publication
Rights and permissions
About this article
Cite this article
Nock, B., Nikolopoulou, A., Chiotellis, E. et al. [99mTc]Demobesin 1, a novel potent bombesin analogue for GRP receptor-targeted tumour imaging. Eur J Nucl Med 30, 247–258 (2003). https://doi.org/10.1007/s00259-002-1040-x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s00259-002-1040-x