Abstract
Objective
In-phase (IP) and out-of-phase (OOP) chemical shift imaging (CSI) is an established technique for clarifying the nature of indeterminate bone marrow lesions, a signal intensity (SI) drop of > 20% at 1.5 tesla (T) or > 25% on 3 T on the OOP sequence being consistent with a non-neoplastic process. Occasionally, SI increase is seen on OOP sequences. The aim of this study is to determine if this is related to marrow sclerosis or matrix mineralisation.
Materials and methods
In 184 cases, the SI change on OOP was calculated. For patients in whom the SI on OOP increased compared with the IP sequence, available CT studies and radiographs were reviewed to look for marrow sclerosis and/or matrix mineralisation.
Results
Forty out of 184 patients (34.25%) showed an anomalous increase in SI on the OOP sequence. CT studies were available in 27 cases (67.5%), of which medullary sclerosis was seen in 20 (74.1%) while matrix mineralisation was seen in a further 2 cases. Review of radiographs demonstrated matrix mineralisation in 6 cases, while punctate signal void consistent with chondral calcification was seen on MRI in 2 more cases. Based on either typical imaging features (n = 22) or histology (n = 18), 7 lesions (17.5%) were classed as non-neoplastic, 18 (45%) as benign neoplasms and 15 (37.5%) as malignant neoplasms.
Conclusion
When assessing focal marrow lesions with CSI, anomalous SI increase may be seen on the OOP sequence in approximately one-third of cases. In over 75% of such cases, CT or radiographs demonstrate either diffuse marrow sclerosis or matrix mineralisation.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The Dixon sequence was first described in 1984, and its applications in the assessment of bone marrow and musculoskeletal soft tissue disorders have recently been reviewed [1,2,3]. A particular value of in-phase (IP) and out-of-phase (OOP) T1-weighted (T1W) Dixon sequences (also termed chemical shift imaging CSI) is in the differentiation between fat containing marrow lesions such as focal nodular marrow hyperplasia (FNMH), reactive oedema-like marrow signal from any cause and atypical haemangiomas, and malignant marrow infiltration. This is based on a signal intensity (SI) drop of > 20% on the OOP sequence compared with the IP sequence at 1.5 T [4, 5] or > 25% at 3 T [6]. However, anomalous increase in SI is occasionally observed on the OOP sequence as illustrated by van Vucht et al., which may be related to marrow sclerosis [3]. The purpose of the current study is to determine the cause(s) of SI increase on OOP compared with the IP sequence on T1W Dixon MRI.
Materials and methods
The study was approved by the local Research and Innovation Centre of The Institute of Orthopaedics, with no requirement for informed patient consent.
The T1W gradient echo Dixon sequence was introduced into our routine bone tumour MR imaging protocol in May 2018 as an adjunct to conventional spin echo imaging for the assessment of suspected bone tumours in patients referred from the Musculoskeletal and Spinal Oncology Services. The duration of the study was 13 months. The MRI parameters for the Dixon sequence are presented in Table 1. From the resulting IP and OOP images, a region of interest (ROI) was drawn within the lesion and SI measurements were made. The % SI drop was calculated as follows:
Additional data collected for each case included patient age and gender, lesion location and final diagnosis. The latter was based on either histological confirmation from image-guided core needle biopsy or characteristic imaging appearances, following which lesions were classified as either non-neoplastic, neoplastic benign or neoplastic malignant. For cases where the IP and OOP sequence demonstrated an anomalous increase in SI on the OOP sequence, available CT studies were assessed for the presence of marrow sclerosis or matrix mineralisation. Marrow sclerosis was initially assessed visually, and when not obvious visually by drawing ROIs in the location of the lesion and by measuring the mean Hounsfield units (HU), then this measurement was compared with adjacent marrow. If CT studies were not available, then any radiographs were reviewed.
The IP and OOP measurements for each case were initially made independently by 2 consultant musculoskeletal radiologists with 24 and 4 years’ experience of bone tumour imaging. Any cases of discrepancy were resolved by consensus review.
Results
At the termination of the study, there were a total of 184 cases that had been imaged with CSI. Of these, 40 (34.25%) showed an anomalous increase in SI on the OOP sequence. This included 23 males and 17 females with mean age 48.25 years (range 9–81 years). Of these, 5 (12.5%) had been imaged at 1.5 T and 35 (87.5%) at 3 T. Lesion locations were as follows: acetabulum (n = 3), femur (n = 7), humerus (n = 3), ilium (n = 6), sacrum (n = 4), tibia (n = 5), vertebra (n = 7), pubis (n = 3), ischium (n = 1) and fibula (n = 1). Based on either typical imaging features (n = 22) or histology (n = 18), 7 lesions (17.5%) were classed as non-neoplastic, 18 (45%) as benign neoplasms and 15 (37.5%) as malignant neoplasms. The final diagnoses are presented in Table 2.
The mean increase in SI on OOP sequences was 17.5% (range 1.4–114%) (Figs. 1 and 2). CT studies were available in 27 cases (67.5%), of which medullary sclerosis was seen at the site of the lesion in 20 (74.1%), 18 based on direct visual assessment (Figs. 2 and 3) and 2 based on an increase in the HU of the lesion compared with adjacent marrow. Matrix mineralisation within an otherwise lytic lesion was seen in a further 2 cases (Fig. 4). In 5 cases, CT showed a purely lytic lesion, these include 2 cases of plasmacytoma/myeloma: 1 patient whose biopsy demonstrated a non-specific lymphoid infiltrate and 1 case each of fibrous dysplasia and simple bone cyst. For patients without CT studies, review of 7 available radiographs demonstrated matrix mineralisation in a further 6 cases (15%) (Fig. 5). In 7 cases, no CT study or radiographs were available. In 2 of these, punctate signal void consistent with chondral-type matrix mineralisation was seen on MRI (Fig. 6). In 3 cases, MRI was diagnostic of an insufficiency fracture (2 sacral and one vertebral), in 1 case an osteoma and in another case an indeterminate cystic lesion.
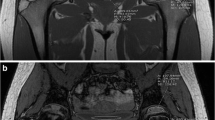
A 39-year-old female with left buttock pain. a IP and b OOP coronal T1W Dixon MR images showing a marrow lesion in the left sacral ala (arrows). IP SI measurement was 652 and OOP SI measurement 710 resulting in 8.9% SI increase on the OOP sequence. c Coronal STIR MR image shows extensive oedema-like marrow SI (arrow) containing a hypointense fracture line (arrowhead). d Coronal STIR MR image 3 months later demonstrates almost complete resolution of the marrow lesion and healing of the insufficiency fracture
A 50-year-old female being investigated for pelvic pain. a IP and b OOP axial T1W Dixon MR images showing a marrow lesion in the right ischial tuberosity (arrows). IP SI measurement was 175 and OOP SI measurement 312 resulting in 78.3% SI increase on the OOP sequence. c Axial CT study demonstrates a sclerotic lesion (arrow) with Hounsfield unit measurements of 1173 consistent with an enostosis
A 36-year-old male with neurofibromatosis and previous malignant peripheral nerve sheath tumour being investigated for a possible left acetabular metastasis. a IP and b OOP axial T1W Dixon MR images show a marrow lesion in the left acetabular roof (arrows). IP SI measurement was 558 and OOP SI measurement 572 resulting in 2.5% SI increase on the OOP sequence. c Axial CT study demonstrates mild medullary sclerosis in the acetabular roof (arrow). Biopsy proven metastasis
A 51-year-old male with pain in the left groin. a IP and b OOP coronal T1W Dixon MR images showing an expansile marrow lesion in the left superior pubic ramus (arrows). IP SI measurement was 587 and OOP SI measurement 600 resulting in 2.2% SI increase on the OOP sequence. c Coronal CT MPR demonstrates a pathological fracture through a lytic lesion with osteoblastic matrix mineralisation (arrow). Biopsy proven osteosarcoma
A 39-year-old female with known Ollier’s disease and right shin pain. a IP and b OOP coronal T1W Dixon MR images showing a lobular marrow lesion in the right upper tibia (arrows). IP SI measurement was 745 and OOP SI measurement 766 resulting in 2.8% SI increase on the OOP sequence. c AP radiograph shows a lytic lesion with chondral matrix mineralisation (arrows). Biopsy proven grade 1 chondrosarcoma
A 26-year-old male being investigated for a possible low-grade chondral tumour in the left distal femur. a IP and b OOP coronal T1W Dixon MR images showing a lobular marrow lesion (arrows). IP SI measurement was 1071 and OOP SI measurement 1115 resulting in 4.1% SI increase on the OOP sequence. c Coronal T1W TSE and d sagittal T2W FSE MR images demonstrate typical appearances of a low-grade chondral tumour containing punctate signal void due to matrix mineralisation
Discussion
CSI plays a major role in the characterisation of indeterminate bone marrow lesions, being able to identify marrow abnormalities that contain residual fat which is not evident on conventional T1W TSE sequences due to a > 20% SI drop on the OOP sequence compared with the IP sequence at 1.5 T [4, 5] and > 25% at 3 T. Examples of such lesions include FNMH which can mimic metastatic disease in the setting of known malignancy [7,8,9], reactive oedema-like marrow signal due to conditions such as trauma [10, 11] or tumour [12, 13], and atypical (lipid poor) haemangiomata [14]. Conversely, marrow infiltration due to conditions such as metastatic disease results in replacement of marrow fat and OOP imaging will show < 20–25% SI drop, allowing a diagnosis of malignant disease to be made with a very high degree of specificity [4, 5]. However, Dreizin et al. [1] pointed out that any marrow replacing pathology will result in SI drop < 20% including benign bone tumours, and therefore, such a level of SI drop on OOP sequences should be considered useful for differentiating non-neoplastic from neoplastic lesions, rather than benign from malignant.
CSI has been used in a variety of clinical situations, predominantly the differentiation of malignant from non-malignant marrow lesions [4, 5, 15, 16] and the differentiation between benign and malignant vertebral compression fractures [17]. These studies used a similar methodology for calculating percentage SI drop on OOP compared with IP sequences as utilised in the present study. Based on the method of reporting of results, only the study by Zajick et al. [16] described SI increase on the OOP sequence in metastases, a mean increase of 3.0% being seen with mixed lytic/sclerotic lesions while osteoblastic lesions demonstrated a mean decrease of 2.4%. No benign vertebral fractures demonstrated SI increase on OOP sequences. The remaining studies only describe the difference between benign and malignant pathologies based on > or < 20% SI drop. Therefore, it is possible that some lesions which showed < 20% SI drop could have demonstrated SI increase on the OOP sequence. In the current study, 40 of 184 (34.25%) consecutive cases imaged using CSI showed an increase in SI on the OOP sequence. Since this also equates to a < 20% SI drop compared with the IP sequence, these cases should have represented neoplastic lesions based on the very high sensitivity of SI drop of < 20% for neoplastic lesions as previously reported [5, 15]. However, 7 (17.5%) were non-neoplastic and 18 (45%) were benign neoplasms. Review of available CT studies, radiographs and MRI studies showed that over 75% of cases showed either marrow sclerosis or matrix mineralisation. Therefore, these results suggest that CSI may not be reliable in differentiating mineralised benign and malignant marrow pathologies. Van Vucht et al. [3] in their recent review of the Dixon technique for assessing bone marrow illustrated cases of benign vertebral compression fracture and Paget’s disease which both demonstrated < 20% SI drop on the OOP sequence, which would have indicated that they were neoplastic lesions. Both cases showed marrow sclerosis on CT. Therefore, it would appear that the % SI change on the OOP sequence is not simply a function of the presence of residual microscopic fat, but is also affected by the presence of calcification which may manifest as trabecular thickening as seen with atypical haemangiomas, marrow sclerosis due to healing fractures or matrix mineralisation in various bone tumours such as osteosarcoma or chondrosarcoma. This may be related to susceptibility effects in lesions with a mineralised matrix as suggested by Dreizin et al. [1]. A further problem that may be encountered in this situation is that non-malignant lesions such as atypical haemangiomata, healing benign vertebral compression fractures and FNMH which contain not only residual fat but also trabecular sclerosis may not show > 20–25% SI drop on the OOP sequence and therefore be mistaken as neoplastic pathologies.
The study has several limitations. Histological confirmation of lesion diagnosis was only available in 18 of 40 cases (45%). However, in 22 cases, a combination of radiographic, CT and MRI features allowed a very confident imaging diagnosis in the remaining patients as listed in Table 2. Of the total 184 cases, 144 showed no SI increase on the OOP sequence. These were not reviewed further to determine whether they also included cases that showed marrow sclerosis or matrix mineralisation, and therefore, it is not known if increased SI on OOP sequences is specific to mineralising lesions.
In conclusion, we have described 40 cases in which CSI demonstrated anomalous increase in SI on OOP compared with IP sequences which may be related to the presence of reactive medullary sclerosis or matrix mineralisation. Such cases showed < 20% SI drop on OOP CSI which should have classified them as neoplastic lesions based on previous reports, while almost 20% were in fact non-neoplastic. In the situation of SI increase on the OOP, radiographs or preferably CT should be undertaken to look for evidence of marrow sclerosis or matrix mineralisation as a potential cause for the anomalous SI change.
References
Dreizin D, Ahlawat S, Del Grande F, Fayad LM. Gradient-echo in-phase and opposed-phase chemical shift imaging: role in evaluating bone marrow. Clin Radiol. 2014;69(6):648–57.
Pezeshk P, Alian A, Chhabra A. Role of chemical shift and Dixon based techniques in musculoskeletal MR imaging. Eur J Radiol. 2017;94(September):93–100.
van Vucht N, Santiago R, Lottmann B, Pressney I, Harder D, Sheikh A, et al. The Dixon technique for MRI of the bone marrow. Skelet Radiol. 2019;48(12):1861–74.
Disler DG, Mccauley TR, Ratner LM, Kesack CD, Cooper JA. In-phase and out-of-phase MR imaging of bone marrow: prediction of neoplasia based on the detection of coexistent fat and water. Am J Roentgenol. 1997;169(5):1439–47.
Douis H, Davies AM, Jeys L, Sian P. Chemical shift MRI can aid in the diagnosis of indeterminate skeletal lesions of the spine. Eur Radiol. 2016;26(4):932–40.
Kumar NM, Ahlawat S, Fayad LM. Chemical shift imaging with in-phase and opposed-phase sequences at 3T: what is the optimal threshold, measurement method, and diagnostic accuracy for characterizing marrow signal abnormalities? Skelet Radiol. 2018;47(12):1661–71.
Pui MH, Tan MH, Kuan JH, Pho RW. Haematopoietic marrow hyperplasia simulating transarticular skip metastasis in osteosarcoma. Australas Radiol. 1995;39(3):303–5.
Chow LTC, Ng AWH, Wong SKC. Focal nodular and diffuse haematopoietic marrow hyperplasia in patients with underlying malignancies: a radiological mimic of malignancy in need of recognition. Clin Radiol. 2017;72(3):265.e7–265.e23.
Shigematsu Y, Hirai T, Kawanaka K, Shiraishi S, Yoshida M, Kitajima M, et al. Distinguishing imaging features between spinal hyperplastic hematopoietic bone marrow and bone metastasis. Am J Neuroradiol. 2014;35(10):2013–20.
Cicala D, Briganti F, Casale L, Rossi C, Cagini L, Cesarano E, et al. Atraumatic vertebral compression fractures: differential diagnosis between benign osteoporotic and malignant fractures by MRI. Musculoskelet Surg. 2013;97(2):S169–79.
Mauch JT, Carr CM, Cloft H, Diehn FE. Review of the imaging features of benign osteoporotic and malignant vertebral compression fractures. Am J Neuroradiol. 2018;39(9):1584–92.
James SLJ, Hughes RJ, Ali KE, Saifuddin A. MRI of bone marrow oedema associated with focal bone lesions. Clin Radiol. 2006;61(12):1003–9.
James SLJ, Panicek DM, Davies AM. Bone marrow oedema associated with benign and malignant bone tumours. Eur J Radiol. 2008;67(1):11–21.
Shi YJ, Li XT, Zhang XY, Liu YL, Tang L, Sun YS. Differential diagnosis of hemangiomas from spinal osteolytic metastases using 3.0 T MRI: comparison of T1-weighted imaging, chemical-shift imaging, diffusion-weighted and contrast-enhanced imaging. Oncotarget. 2017;8(41):71095–104.
Kohl CA, Chivers FS, Lorans R, Roberts CC, Kransdorf MJ. Accuracy of chemical shift MR imaging in diagnosing indeterminate bone marrow lesions in the pelvis: review of a single institution’s experience. Skelet Radiol. 2014;43(8):1079–84.
Zajick DC, Morrison WB, Schweitzer ME, Parellada JA, Carrino JA. Benign and malignant processes: normal values and differentiation with chemical shift MR imaging in vertebral marrow. Radiology. 2005;237(2):590–6.
Ragab Y, Emad Y, Gheita T, Mansour M, Abou-Zeid A, Ferrari S, et al. Differentiation of osteoporotic and neoplastic vertebral fractures by chemical shift {in-phase and out-of phase} MR imaging. Eur J Radiol. 2009;72(1):125–33.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
van Vucht, N., Santiago, R., Pressney, I. et al. Anomalous signal intensity increase on out-of-phase chemical shift imaging: a manifestation of marrow mineralisation?. Skeletal Radiol 49, 1269–1275 (2020). https://doi.org/10.1007/s00256-020-03420-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00256-020-03420-0