Abstract
Leiomyomas are benign tumors of smooth muscle cells. Leiomyomas of somatic soft tissue are a specific class of leiomyoma believed to arise from the smooth muscle cells found in the walls of blood vessels and represent less than 4% of benign, somatic soft tissue tumors. Of the somatic soft tissue tumors, approximately one-third will become calcified. We report an intramuscular, calcified leiomyoma arising from the left deltoid of a 47-year-old Caucasian male. To the best of the authors’ knowledge, this is the first reported case of a calcified, intramuscular leiomyoma of the deltoid. Imaging studies and patient presentation were initially suggestive of tumoral calcinosis or myositis ossificans. It was not until pathologic examination that the correct diagnosis of the calcified leiomyoma was made. Leiomyoma should be included in the differential diagnosis of calcified soft tissue masses. If excised with adequate margins, recurrence is unlikely.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
According to the World Health Organization, there are two classifications of smooth muscle proliferations of soft tissue; benign leiomyomas and malignant leiomyosarcomas [1]. The mean age of occurrence of somatic soft tissue leiomyomas is 37 years of age, with both sexes affected equally [2]. Previously, benign leiomyomas of soft tissue were reported sporadically to the extent that their existence was in question [2]. However, in a study of 1331 cases of benign soft tissue tumors, 3.8% were found to be leiomyomas with 4.2% of these leiomyomas arising in the arms and shoulders [3]. In 2001, Billings identified 13 cases of leiomyomas of somatic soft tissue with only two reported in the upper extremities [2]. Smooth muscle is located throughout the entire body with the majority located in the genitourinary, gastrointestinal, and respiratory tracts with a much lesser extent present in the walls of blood vessels. This distribution helps to explain the relative commonness of leiomyomas in the uterine wall with the comparative rarity of leiomyomas in somatic soft tissue [4]. Leiomyomas of somatic deep soft tissue may also calcify [5]. Calcification without alterations of calcium or phosphate metabolism is referred to as dystrophic and is related to alterations of local tissue [6]. In addition to developing sporadically, benign leiomyomas may arise due to a genetic predisposition or in an immunocompromised state. In hereditary leiomyomatosis and renal cell carcinoma (HLRCC), previously known as Reed syndrome, affected individuals are at an increased risk of developing leiomyomas and type 2 papillary renal cell carcinoma.
Case presentation
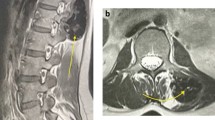
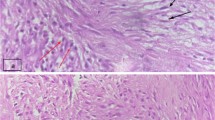
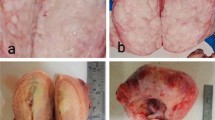
A 47-year-old Caucasian male presented to his primary care provider for evaluation of pain in his left upper arm for 2 years. Radiography of the left humerus revealed a calcified mass in the distal portion of the left deltoid muscle measuring 7 cm in length and 3 cm in width. The mass was comprised of dense, small mineral deposits in aggregate with a well-defined periphery and dense central portion (Fig. 1). This pattern was suggestive of a benign deposition of hydroxyapatite crystals. The underlying bone appeared unaffected and no other calcific deposits were seen. On T1-weighted MRI (Fig. 2), there was predominately low signal. T(T2 NOT T1)1-weighted images without fat suppression showed the mass to be isointense to the surrounding muscle fibers, without evidence for invasion (Fig. 3). The axial post-contrast image showed no enhancement of the mass. The patient then underwent surgical resection (Figs. 4 and 5). On gross pathologic examination, the mass measured 6.2 × 2.5 × 1.7 cm and was described as a hard, pink-grey mass with pale rays and a smooth, glistening external surface. Serial sections showed a yellow solid calcified surface. Pathologic analysis (Fig. 6) identified the mass as a calcified and sclerotic leiomyoma without evidence of malignant transformation. Six-month follow-up examination and imaging assessment by X-ray and ultrasound demonstrated no evidence of residual or recurrent calcification.
Discussion
There are no specific imaging characteristics of deep soft tissue leiomyomas that differentiate this tumor from other soft tissue masses [7]. Deep soft tissue leiomyomas are therefore difficult to diagnose prior to surgical resection and subsequent pathologic evaluation. Pathologic evaluation may include immunohistochemical stains to aid in the diagnosis of this lesion. Leiomyomas are proliferations of smooth muscle cells and are therefore desmin-positive [8].
The differential diagnosis for this patient following imaging studies mainly included tumoral calcinosis of an idiopathic cause and myositis ossificans. Myositis ossificans is a benign, ossifying intramuscular mass of traumatic origin [9]. The patient denied history of trauma to the region of the left deltoid that contained the mass making a diagnosis of myositis ossificans less likely. Radiographically, myositis ossificans has a zoning pattern that is considered the defining diagnostic feature [10]. First, at approximately 6 weeks, new bone with a well-defined cortex and lacy appearance develops. Then, at 10 weeks the central area of the mass will enlarge until approximately 5–6 months when the mass may shrink, and a radiolucent region of soft tissue appears. Over time, the mass will become dense and ossified with possible trabecular bone formation [10]. Tumoral calcinosis, in contrast, is a process in which calcium deposits occur bilaterally and often near a joint. Tumoral calcinosis can occur primarily with or without hyperphosphatemia or secondarily as a result of chronic renal failure [11]. The patient did not have bilateral masses, the mass was not in close proximity to a joint, nor was the patient in renal failure. These factors in combination made a diagnosis of tumoral calcinosis less likely. Tumoral calcinosis on imaging may appear large and cystic or has been reported to have multiple lobules separated by radiolucent bands [12]. Tumoral calcinosis may appear similar to a somatic deep soft tissue leiomyoma, as is seen in the case of a 34-year-old female with tumoral calcinosis that on radiographic imaging showed a well-defined calcified mass of the posterior elbow.
The X-ray imaging studies of the mass of this patient showed dense, small mineral deposits in aggregates with distinct borders and a dense central portion (Fig. 1). On T2-weighted MRI, fat caps were seen which in a study of 50 intramuscular masses was reported in one other leiomyoma [13]. The imaging characteristics of calcified leiomyomas have also been described as having calcifications with a mottled mulberry appearance similar to uterine leiomyomas. This pattern was described in a calcified leiomyoma in the axilla of two separate pediatric cases [14].
In our reported case, while imaging studies were needed to initially guide the diagnosis of the soft tissue mass, gross and microscopic evaluation of the tumor led to the final diagnosis of a calcified, intramuscular leiomyoma of the left deltoid.
Conclusions
We report the first documented case of an intramuscular leiomyoma of the deltoid. Somatic soft tissue leiomyoma should be included in the differential diagnosis of a calcified soft tissue mass of the extremities. Calcified intramuscular masses that do not show typical clinical presentation and radiologic characteristics of benign etiologies should be correlated with tissue sampling to confirm both diagnosis and appropriate treatment plan.
References
Smooth-muscle tumours. In: Fletcher CDM, Bridge JA, Pancras CW, Hogendoorn FM, editors. WHO classification of tumours of soft tissue and bone. 4th ed. Lyon: International Agency for Research on Cancer; 2013. p. 109.
Billings SD, Folpe AL, Weiss SW. Do leiomyomas of deep soft tissue exist? An analysis of highly differentiated smooth muscle tumors of deep soft tissue supporting two distinct subtypes. Am J Surg Pathol. 2001;25(9):1134.
Myhre-Jensen OA. Consecutive 7-year series of 1331 benign soft tissue tumours: clinicopathologic data. Comparison with sarcomas. Acta Orthop Scand. 1981;52(3):287.
Kilpatrick SE, Metzel F. C.D. Leiomyoma of deep soft tissue. Clinicopathologic analysis of a series. Am J Surg Pathol. 1994;18(6):576.
Weiss S, Goldblum J. Enzinger and Weiss’s soft tissue tumors. 4th ed. St. Louis: Mosby; 2001.
Battistella M, Cribier B. Calcification and ossification. In: Smoller B, Rongioletti F, editors. Clinical and pathological aspects of skin diseases in endocrine, metabolic, nutritional and deposition disease. New York: Springer; 2010. p. 161–9.
Ramachandran R, Rangaswami R, Raja D, Shanmugasundaram G. Deep soft-tissue leiomyoma of the forearm mimicking a primary bone tumor of the ulna. Radiol Case Rep [Internet]. 2014;9(3)
Weiss SW. Smooth muscle tumors of soft tissue. Adv Anat Pathol. 2002;9(6):351.
Walczak BE, Johnson CN, Howe BM. Myositis ossificans. J Am Acad Orthop Surg. 2015;23(10):612.
Parikh J, Hyare H, Saifuddin A. The imaging features of post-traumatic myositis ossificans with emphasis on MRI. Clin Radiol. 2002;57:1058.
Fathi I, Mahmoud S. Review of tumoral calcinosis: a rare clinico-pathological entity. World J Clin Cases. 2014;2(9):409.
Martinez SM, Vogler JB, Harrelson JM, Kenneth WL. Imaging of tumoral calcinosis: new observations. Radiology. 1990;174(1):215.
Sung J, Kim JY. Fatty rind of intramuscular soft-tissue tumors of the extremity: is it different from the split fat sign? Skelet Radiol. 2017;46(5):665–73.
Ledesma-Medina J, Oh KS, Girdany BR. Calcification in childhood leiomyoma. Radiology. 1980;135(2):339.
Acknowledgements
We would like to acknowledge Dr. Patrick Lin for his contribution to the case.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Rights and permissions
About this article
Cite this article
Rowland, F., Call, C., Mujtaba, B. et al. Calcified leiomyoma of the deltoid: pathophysiology and imaging review. Skeletal Radiol 48, 625–628 (2019). https://doi.org/10.1007/s00256-018-3053-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00256-018-3053-y