Abstract
Hibernoma is a benign adipose tumour that contains foetal brown fat cells. We report a case of hibernoma arising in the left ischium of a 65-year-old female with a past history of ovarian carcinoma. The patient presented with a relatively short history of left sacral/hip pain. Radiologically, the lesion, which was large (5 cm) and sclerotic, had been stable for a number of years. Histologically, it was composed mainly of plump cells with foamy, multivacuolated cytoplasm. These cells showed no reaction for epithelial, melanoma or leucocyte markers but expressed FABP4/aP2 and S100, indicating that they were brown fat cells. There was no mitotic activity or nuclear pleomorphism and the lesion was diagnosed as a benign intraosseous hibernoma (IOH). IOH is a recently identified benign adipocytic lesion that presents typically as a sclerotic bone lesion. It has characteristic morphological and immunophenotypic features and should be regarded as a discrete primary bone tumour that needs to be distinguished from metastatic carcinoma/melanoma, chondrosarcoma and metabolic storage diseases containing numerous foamy macrophages.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
White and brown fat are the two main types of adipose tissue and both contain morphologically distinct fat cells (adipocytes), which are derived from primitive mesenchyme [1]. In soft tissue, a lipoma is defined as a benign tumour composed mainly of white adipocytes and a hibernoma as a tumour composed of variable proportions of brown fat cells mixed with white adipose tissue [2, 3]. Although there is abundant white fat within the marrow of bones of the appendicular and axial skeleton, primary adipocytic intraosseous tumours are uncommon. Lipoma and liposarcoma are included in the most recent WHO classification of primary bone tumours with lipoma accounting for fewer than 0.1 % of primary bone tumours and liposarcoma described as extraordinarily rare with only a handful of cases being reported [4, 5].
It is only recently (2008) that brown fat was first identified in bone [6–8]. Subsequently there has been corroboration of this finding with the brown fat being considered to represent part of an intraosseous hibernoma (IOH) in most cases [9–14]. As in soft tissue, IOH has been reported to contain mainly foetal brown fat cells with a variable admixture of mature white fat cells. Some authors have disputed whether IOH represents a discrete tumour and consider that this lesion represents a physiological overgrowth of brown fat cells. We report the clinical, radiological and pathological features of a case of IOH that presented as an ischial tumour. Diagnostic radiological, morphological and immunohistochemical features of this tumour, which radiologically mimics more common sclerotic lesions of bone, are described and the case for this rare lesion being included in future classifications of primary bone tumours discussed.
Case history
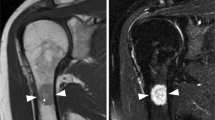
A 65-year-old female presented with an 8-week history of left sacral pain that radiated down her left leg and was worse at night. On examination, left hip movements were painful and straight leg-raising was limited to 70°. Six years earlier she had undergone a salpingo-oophorectomy and omentectomy, at which time she had also had a CT scan. She was otherwise well and haematology/biochemistry laboratory investigations were normal. A radiograph of her left hip showed moderate osteoarthritis. In view of her past history she went on to have a bone scan, which showed an abnormality in her left ischium. A subsequent MRI scan of the pelvis revealed a 5 × 2 × 3-cm well-circumscribed lesion along the posterior column of the left acetabulum. The typical bright signal of an intraosseous fatty lesion was not seen on T1-weighted MRI sequences. The lesion returned heterogeneous high signal on STIR-weighted images and intermediate signal with multiple low-signal foci on T1-weighted images (Fig. 1a, b, c). There was no evidence of a soft tissue mass or cortical destruction. Retrospective review of the CT scan performed 6 years earlier showed a heterogeneous sclerotic lesion at the posterior column of the acetabulum without periosteal reaction (Fig. 2). The lesion was thus thought to be relatively stable with respect to size and location. This was confirmed by a whole-body PET-CT scan, which showed that the lesion in the left posterior acetabulum/left ischial tuberosity was only mildly fluorodeoxyglucose avid (SUVmax 4.1), i.e. just above background marrow uptake (Fig. 1d).
a Coronal T1-weighted MR image shows a lesion (arrow) that returns intermediate signal with multiple low-signal foci in the left ischium. b Coronal short-tau inversion recovery MR image shows the above lesion (arrow) returning heterogeneous high signal. There is no evidence of a soft tissue component. c Axial T1-weighted MR image through the lesion (arrows) confirms the presence of an atypical heterogeneous signal. d PET/CT demonstrates that the lesion in the left ischium (arrow) is only mildly fluorodeoxyglucose avid (SUVmax 4.1)
In view of the slow growth and location of the lesion (which made biopsy difficult), it was thought most likely to be a low-grade chondroid tumour although the possibility of metastasis could not entirely be excluded, particularly given the previous history of ovarian carcinoma. Other lesions considered in the differential diagnosis included osteoma and haemangioma. After discussion, it was decided not to biopsy the lesion as it was felt that there was a possibility that this would lead to collapse of the acetabulum and surgical excision of the tumour with limited left-sided hemipelvectomy and total hip replacement was undertaken.
Gross pathological examination of the resection specimen did not identify a discrete lesion but only a poorly defined area of pallor within the bone medulla. Histological examination showed a lesion composed mainly of cells with small round nuclei and plump cytoplasm containing small fat vacuoles (Fig. 3a). These cells had the morphological characteristics of brown fat cells; there was no cellular or nuclear pleomorphism and no mitotic activity. Brown fat cells were focally admixed with mature white fat cells within the lesion, which was not encapsulated but relatively well defined; bone trabeculae around brown fat cells were thickened. Immunohistochemistry showed that the lesional cells strongly expressed the adipocyte markers FABP4/ap2 and S100 [15] (Fig. 3b). There was no expression of epithelial (epithelial membrane antigen, cytokeratin), melanoma (Melan A), histiocyte (CD45, CD68) or proliferation (Ki-67) markers. The histological features were consistent with an IOH. The lesion was fully excised. The patient had relief of pain after operation; there has been no recurrence of the tumour after 12 months follow-up.
Discussion
There are 12 previously reported cases of hibernoma or hibernoma-like lesions in bone; in a number of these reports the tumour involved the pelvis and the diagnosis of IOH was, as in our case, not considered [8, 10, 12]. The mean age of patients with IOH (including the present case) is 60 years old with range 40 to 85 years. IOH has been reported in both sexes, but there is a slight female predominance (8 females and 5 males). Most IOH lesions present as an incidental radiological finding but, as in our case, it can be associated with clinical symptoms requiring treatment. IOH arises most commonly in the axial skeleton, particularly the pelvic bones (10/13), spinal vertebrae (2/13) and sternum (1/13) [10]. In most cases of IOH, the size is approximately 2 cm but there are two cases, including our case, where the tumour measured 5 cm maximum.
IOH has a sclerotic heterogeneous appearance in plain radiograph and CT images. On MRI, the lesion has a hypointense signal compared to subcutaneous fat and a hyperintense signal compared to skeletal muscle in T1-weighted sequence; a heterogeneous hyperintense signal with high signal rim in fluid-sensitive sequences with fat suppression and peripheral or homogeneous rim enhancement post-gadolinium administration can be noted. We noted non-specific heterogeneous high signal on STIR in our case, which could indicate peritumoural oedema. The lesion is rarely diagnosed with confidence before biopsy, and both conservative management and radiofrequency ablation have been used therapeutically in symptomatic cases [12, 13].
The diagnosis of IOH is problematic as it is a rare lesion that occurs in bones where other more common primary bone tumours are typically found [10, 12]. Distinction of a pelvic IOH from a low-grade chondroid lesion is particularly difficult because both tumours are relatively common in the pelvis and share some imaging features, including heterogeneous sclerosis and a relatively well-circumscribed margin on plain films and CT images, as well as similar signal characteristics in MRI studies. In addition, the growth of a low-grade cartilaginous tumour is usually slow and PET/CT may only indicate mild metabolic activity suggestive of a low-grade malignancy [10]. IOH also, as in our case, needs to be distinguished from a skeletal metastasis. There is a previous report of a female patient with a history of breast carcinoma and an osseous symptomatic lesion of the ilium that histologically proved to be an IOH [9]. Intraosseous lipomatous lesions can be identified in abdominal and spinal studies; these lesions most likely represent intraosseous lipomas that contain mature fat and, unlike IOH, typically have a bright signal in T1-weighted MR sequences. Radiologically, IOH enters into the differential diagnosis of other relatively well-defined sclerotic bone lesions of the axial skeleton such as intraosseous haemangioma, bone island and notochordal rest [10, 12].
The first reported observations of brown fat in the bone marrow were in 2008 [6, 7]. Thorns et al. [6] noted the presence of (S100+) brown fat cells in a trephine bone marrow biopsy of a female with essential thrombocythaemia. It was not clear whether the brown fat cells in this case represented an incidental finding or early development of a hibernoma. The possibility that inactive brown fat cells were present in other marrow locations was postulated. Subsequently there have been several reports of brown fat lesions in bone, most of these cases being considered examples of IOH [8–14]. In most of these reports, the lesion appeared as a discrete mass within bone, but in a few cases it was noted that brown fat cells were disposed in small groups separated by white fat; on this basis it was suggested that the lesion could represent physiological overgrowth of brown fat cells or possibly ectopic or residual brown fat within white fatty marrow [10]. Some hibernomas of soft tissue exhibit a cytogenetic translocation involving chromosomes bands 11q and 13, which results in the deletion of genes that play a role in brown fat differentiation; the identification of this translocation in IOH should help in clarifying the neoplastic status of this lesion.
Brown fat is a specialised form of adipose tissue found in newborn mammals and hibernating animals; it plays an important role in body thermoregulation [1]. It is generally believed that in humans brown fat is lost in the first few years postnatally with only small amounts persisting in adults in specific anatomical locations such as the interscapular region, neck, mediastinum and retroperitoneum [1]. Heaton et al., however, in necropsy studies, identified the presence of brown fat throughout life up to the 8th decade [16]. Active brown fat in adult humans has also been identified, particularly in the upper parts of the body (where it is predominantly located in neonates) in FDG-PET scans [17, 18].
It is a remarkable fact that neither in humans nor other mammals does brown fat appear to have been identified in bone prior to 2008. It is now recognised that brown fat progenitors are present in white fat and that bone marrow adipose tissue expresses a gene profile consistent with brown fat cells [19, 20]. Brown fat cells are metabolically active and share a common mesenchymal stem cell precursor with osteoblasts [21, 22]. Inducible brown fat cells (beige fat) in adipose tissue are anabolic for the skeleton [23], and it is possible that factors that result in the generation of brown fat cells in bone marrow may also locally promote osteoblastic development and in this way account for the osteosclerosis characteristically seen in IOH.
Our case illustrates that IOH should be considered in the differential diagnosis of a bone lesion that has a sclerotic heterogeneous appearance, particularly in the pelvis where this lesion has most commonly been reported. Although in our case a biopsy was not undertaken as it was feared that it would lead to collapse of the acetabulum, this is clearly recommended to identify this rare primary bone lesion. IOH is often asymptomatic and presents in most cases as an incidental radiological finding that needs to be distinguished from other more common bone tumours. On the basis that IOH can give rise to a sizable tumour with characteristic radiological, morphological and immunohistochemical findings, it merits consideration to be included as a discrete benign adipocytic lesion in future primary bone tumour classifications.
References
Brooks JJ, Perosio PM. Adipose tissue. In: Sternberg S, editor. Histology for pathologists. NY: Raven; 1992. p. 33–60.
Neil GP, Mansahl N. Lipoma. In: Fletcher CDM, Bridge JA, Hogendoorn PCW, Mertens F, editors. WHO classification of tumours of soft tissue and bone. Lyon: IARC; 2013. p. 20–1.
Miettinen MMF-S, Mandahl JC, Hibernoma N. In: Fletcher CDM, Bridge JA, Hogendoorn PCW, Mertens F, editors. WHO classification of tumours of soft tissue and bone. Lyon: IARC; 2013. p. 31–3.
Rosenberg AE, Bridge JA. Lipoma. In: Fletcher CDM, Bridge JA, Hogendoorn PCW, Mertens F, editors. WHO classification of tumours of soft tissue and bone. Lyon: IARC; 2013. p. 341.
Rosenberg AE, Szuhai K. Liposarcoma. In: Fletcher CDM, Bridges JA, Hogendoorn PCW, Mertens F, editors. WHO classification of tumours of soft tissue and bone. Lyon: IARC; 2013. p. 342.
Thorns C, Schardt C, Katenkamp D, Kahler C, Merz H, Feller AC. Hibernoma-like fat in the bone marrow; report of a unique case. Virchows Arch. 2008;452:343–5.
Reyes AR IR, Wilson JD, Desai HS. Intraosseous hibernoma of the femur; an unusual case with a review of the literature (poster #20). USCAP 2008; 132: 3.
Kumar R, Deaver MT, Czerniak BA, Madewell JE. Intraosseous hibernoma. Skeletal Radiol. 2011;40:641–5.
Bai S, Mies C, Stephenson J, Zhang PJ. Intraosseous hibernoma: a potential mimic of metastatic carcinoma. Diagn Pathol. 2013;17:204–6.
Bonar SF, Watson G, Gragnaniello C, Seex K, Magnussen J, Earwaker J. Intraosseous hibernoma: characterization of five cases and literature review. Skeletal Radiol. 2014;43:939–46.
Hafeez I, Shankman S, Michnovicz J, Vigorita VJ. Intraosseous hibernoma: a case report and review of the literature. Spine. 2015;40:E558–61.
Jerman A, Snoj Ž, Kuzmanov BG, Limpel Novak AK. Intraosseous hibernoma: case report and tumour characterization. BJR Case Reports 2015; 1 (20150204).
Ringe KI, Rosenthal H, Langer F, Callies T, Wacker F, Raatschen HJ. Radiofrequency ablation of a rare case of an intraosseous hibernoma causing therapy-refractory pain. J Vasc Interv Radiol. 2013;24:1754–6.
Lynch DT, Dabney RS, Andrews JM. Intraosseous hibernoma or unusual location of brown fat? J Hematop. 2013;6:151–3.
Kashima TG, Turley H, Dongre A, Pezzella F, Athanasou NA. Diagnostic utility of aP2/FABP4 expression in soft tissue tumours. Virchows Arch. 2013;462:465–72.
Heaton JM. The distribution of brown adipose tissue in the human. J Anat. 1972; 35–9.
Nedergaard J, Bergtsson T, Cannon B. Unexpected evidence for active brown adipose tissue in adult humans. Am J Physiol Endocrinol Metab. 2007;293:E444–52.
Maeda T, Tateishi U, Terauchi T, Hamashima C, Moriyama N, Arai Y, et al. Unsuspected bone and soft tissue lesions identified at cancer screening using position emission tomography. Jpn J Clin Oncol. 2007;37:207–15.
Schulz TJ, Huang TL, Tran TT, Zhang H, Townsend KL, et al. Identification of inducible brown adipocyte progenitors residing in skeletal muscle and white fat. Proc Natl Acad Sci U S A. 2011;108:143–8.
Krings A, Rahman S, Huang S, Lu Y, Czernik PJ, Lecka-Czernik B. Bone marrow fat has brown adipose tissue characteristics, which are attenuated with aging and diabetes. Bone. 2012;50:546–52.
Takada I, Suzawa M, Matsumoto K, Kato S. Suppression of PPAR transactivation switched cell fate of bone marrow stem cells from adipocytes into osteoblasts. Ann N Y Acad Sci. 2007;1116:182–98.
Berendsen AD, Olsen BR. Osteoblast-adipocyte lineage plasticity in tissue development, maintenance and pathology. Cell Mol Life Sci. 2014;71:493–7.
Rahman S, Lu Y, Czernik PJ, Rosen CJ, Enderback S, Lecka-Czernik B. Inducible brown adipose tissue, or beige fat, is anabolic for the skeleton. Endocrinology. 2013;154:2687–701.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Informed consent for this study was obtained from the patient. This study was approved by the Central Oxford Research Ethics Committee (C01.070 and C01.071).
Funding
MV was an EU-funded Erasmus plus visitor.
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Vlychou, M., Teh, J., Whitwell, D. et al. Intraosseous hibernoma: a rare adipocytic bone tumour. Skeletal Radiol 45, 1565–1569 (2016). https://doi.org/10.1007/s00256-016-2460-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00256-016-2460-1